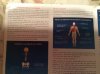
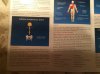
ABOVE: Scans of the brains of mice show a reduction in dopamine (colored areas) in the striatum of the Parkinson’s disease model that was injected with pathogenic α-synuclein (right; control mouse on left).
TED DAWSON ET AL. / NEURON, 2019
TED DAWSON ET AL. / NEURON, 2019
I
n 2003, Heiko Braak, then a neuroanatomist at the University of Frankfurt, suggested that Parkinson’s disease pathology may start in the gut and travel from there to the brain long before a patient shows symptoms. The idea, based on postmortem analyses of samples from parkinson’s patients, has been hotly debated ever since.In a study published today (June 26) in Neuron, Ted Dawson, a neurologist at Johns Hopkins School of Medicine, and his team created an animal model of the disease by injecting particular proteins into the stomachs of mice. About a month later, the animals showed symptoms of Parkinson’s disease. The model not only demonstrates how the disease protein can travel up from the gut to the brain, but also presents nonmotor symptoms rarely seen in other animal models.
The Scientist spoke with Dawson about the work.

Ted Dawson
JOHNS HOPKINS MEDICINE
Ted Dawson: Well, there is this idea that was building that was started by Dr. Braak that Parkinson’s disease could start in the gastrointestinal tract, and there was good human data that suggested that possibility. What was lacking was an animal model that could validate that hypothesis. [The model] validates [the hypothesis] by providing evidence that it’s possible that Parkinson’s disease could start in the gut.
There’s been this very detailed study by Dr. Braak and later by other pathologists that the pathologic alpha-synuclein seemed to progress from the gut up the neuroaxis into the brain, and what our work shows is that this is possible.
See “Can the Flu and Other Viruses Cause Neurodegeneration?”
TS: How did you do it?
TD: We took pathologic alpha-synuclein, . . . and then we injected those preformed fibrils into the pylorus [an area of the stomach close to the small intestine] into one of the mice near where the vagal nerve innervates those regions. Over time, the animals developed Parkinson’s disease.
[To clarify], we’re injecting exogenous pathologic alpha-synuclein. And that is causing the endogenous synuclein to misfold and transmit up the vagal nerve. We know that because when we injected the exogenous alpha-synuclein in the alpha-synuclein knock-out, nothing happened. So the exogenous alpha-synuclein is not the synuclein that’s transmitting. It’s causing the endogenous synuclein to misfold and transmit.
TS: What is the importance of the vagus nerve in the pathologic process?
TD: It’s essential for the pathologic alpha-synuclein to get up to the brain. In a subgroup of animals, we performed vagotomies and the pathologic alpha-synuclein did not ascend into the brain. And as a consequence of that, the animals did not develop Parkinson’s disease.
TS: What else makes this model unique?
TD: One of the other things that we think is really exciting about this model is that not only do the mice have the motor features of Parkinson’s disease, they also have the nonmotor features. They’ve got cognitive dysfunction, anxiety, depression, problems with smell. And so we now have an animal model to study those problems. We hope that it opens up a whole new set of investigations using those animal models.
This model shows that it’s possible for synuclein to ascend from the stomach via the vagal nerve to the brain. What we don’t know in humans with Parkinson’s disease is how that process starts. That would be the next step, to figure out how it actually starts in humans.
S. Kim et al., “Transneuronal propagation of pathologic α-synuclein from the gut to the brain models Parkinson’s disease,” Neuron, doi:10.1016/j.neuron.2019.05.035, 2019.
https://www.the-scientist.com/news-opinion/mouse-model-shows-how-parkinsons-disease-begins-in-the-gut-66048