Pain Is Not All the Same
Brain imaging studies like functional magnetic resonance imagery (FMRI) have provided tantalizing insights into the operation of the human brain. In this blog I’m going to review and highlight research and novel approaches by one of the leading researchers in the rapidly evolving area of chronic pain and the brain, Dr. A. Vania Apkarian, and his colleagues at Northwestern University, Chicago, Illinois.

Apkarian suggests that trying to study chronic pain using acute stimuli is like trying to pound a circular peg into a square hole … It doesn’t work
In the last 20 years an immense number of reductionist research papers exploring rodent models of chronic pain have been published but few new pharmaceuticals have been developed. Dr. Apkarian argues that some of the failings of pain research in the last 20 years have come from experimental designs focused on nociception and acute pain instead of the chronicity of chronic pain. He asserts that the pain processes in acute and chronic pain are very different.
The participants in most animal and human studies are subjected to a painful (acute) short term physical stimulant and their brain response is measured. Dr. Apkarian, though, questions if rodent models of acute pain can tell us about long-lived chronic pain in humans. He also notes that in these studies pain is rarely triggered where chronic pain is typically felt in humans, such as the tender points or allodynia locations in fibromyalgia patients.
Many pain researchers, then, are applying an acute pain stimulus to a site that is not correlated with chronic pain in humans. What are these results telling us? Not much about chronic pain, says Dr. Apkarian.
Perhaps the most successful concept animal models have taught us about pain is the idea of Central Sensitization. Central sensitization occurs when pain sensation is amplified (hypersensitivity) and when normally non-painful sensations such as touch, sound, light, and even smell are translated into pain sensations. The central sensitization concept, which involves pain amplification at the brain level, expanded the focus of chronic pain research from peripheral pain processing to the spinal cord circuitry and into the brain.
Our understanding of the critical elements that induce and maintain central sensitization, however, is still very limited. We don’t know why the sensitization starts or how it turns into chronic pain. Those questions are the focus of Dr. Apkarian’s research.
A Unique Approach to a Chronic Pain Therapy
Research indicates that different brain regions are involved in producing chronic pain and acute pain. Acute painful stimuli in healthy people generally activate the somatosensory, insular, and cingulate cortical regions, while painful stimuli in people with chronic pain activate prefrontal cortex and limbic regions. This brain region difference implies that the processes for acute pain and chronic pain are different.
Research indicates that the brain activity patterns of each chronic pain condition (fibromyalgia, chronic low back pain, Chronic Regional Pain Syndrome (CRPS), etc.) appear to be unique to that condition. Chronic low back pain (CBP) patients show different brain wave patterns from Chronic Regional Pain Syndrome (CRPS) patients which, in turn, are different from the brain wave patterns in fibromyalgia patients. More importantly, he says, the implication that distinct chronic pain conditions have unique brain signatures opens the possibility of developing novel, specific therapies for each pain condition.
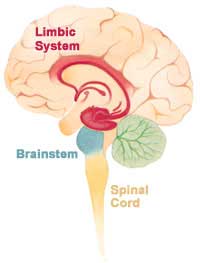
Instead of the somatosensory and other cortices that handle sensory signals, Apkarian’s studies suggest the prefrontal cortex and limbic system (pictured here) are involved in chronic pain.
Rather than the reductionist approach identifying the nerve fiber types, ion channels, etc. involved in pain the Apkarian lab took a top down approach by looking at the process of chronic pain from the brain’s perspective. Instead of building surrogate animal models of chronic pain in humans, the Apkarian lab is applying brain imaging information gathered in humans to rodent models of chronic pain, with the objective of developing new drug therapies.
Brain imaging of spontaneous pain (spontaneous pain is the ever present and ever fluctuating pain that all chronic pain patients experience) in humans showed the medial prefrontal cortex (PFC) and amygdala were deeply involved. The Apkarian lab interpreted this as implying that lateral atrophy of the PFC may underlie the enhanced activity in medial PFC.
In addition, rodent studies suggest NMDA transmission in the cingulate cortex may play a critical role in neuropathic pain. The Apkarian lab put these two facts together and wondered if D-cycloserine (DCS), an antibiotic, might be a potential drug for neuropathic chronic pain patients.
D-cycloserine is not your normal antibiotic. Not only is it able to cross the blood-brain barrier and penetrate into the central nervous system, it also enhances the activity of the N-methyl-D-aspartic acid (NMDA) glutamatergic receptors in the nucleus of the amygdala. Administered systemically or centrally, DCS has been shown to enhance cognitive processes, improve attention and memory, and facilitate fear extinction.
To me, enhancing the activity of NMDA seems counterintuitive. I’d learned that NMDA was one of the bad boys you tried to quiet in pain, but here the Apkarian lab was trying to enhance its activity!
Human trials using an antibiotic that penetrates the brain which will hopefully will extinguish pain memories is under way.
The Apkarian lab theorized that DCS-induced reinforcement of NMDA-receptor mediated transmission in the prefrontal cortex enabled the prefrontal cortex to disengage spontaneous pain from its previously learned associations which it then placed into memory (similar to fear extinction).
They hypothesized that DCS should enhance the extinction of pain-related memories and thus exhibit anti-nociceptive properties for neuropathic pain. (If your pain is associated with, say, walking, then walking will bring up pain memories and you will feel pain.)
So they tested the effects of DCS on chronic neuropathic pain behavior in lab rats. [1]The main finding was that repeated treatment with DCS reduced two signs of neuropathic pain in rats: tactile sensitivity and protective paw posturing.
This anti-pain effect was dose-dependent, and the effect increased in efficacy for up to three weeks. Upon cessation of treatment, DCS effects on pain behavior persisted for a duration proportional to the length of treatment.
A human clinical trial of DCS and chronic back pain by Northwestern University in association with the Apkarian lab is slated to be completed this spring, 2014. Dr. Apkarian states that this is the first clinical drug therapy study directly derived from human brain imaging results.
A NEW THEORY OF CHRONIC PAIN
In 2012 Dr. Apakarian proposed a new theory of chronic pain that incorporates the emotional suffering and behavioral/cognitive modifications caused by the reorganization of the circuits in the brain. He emphasized that sensory processing issues – long thought to play a key role in chronic pain – probably do not.
… chronic pain can no longer be viewed as a pure perceptual state, that is, persistence of pain.
… Overall, the brain activity patterns and the changes in morphology and connectivity show a general picture that more closely resembles the addicted brain and provides no evidence for increased sensory processing of pain.
Reorganization and representation primarily involve limbic and prefrontal brain circuitry and minimally impinge on sensory properties of pain. We presume and hypothesize that these changes are a reflection of both the suffering and coping strategies that impinge on learning and memory and on hedonics of everyday experience.
Emotional and Motivational Circuitry Targeted
Apkarian asserts the state of the brain’s emotional and motivational circuitry is more important in causing chronic pain than the brain’s sensory circuits. The anxiety, depression, and dramatically reduced quality of life found in people with chronic pain reflects the intense emotional component present. (Apkarian’s most recent study, however, did not find that anxiety or depression were associated with altered connections in the brains of people with chronic pain.)

Apkarian’s work suggests that in chronic pain the emotional circuits of the brain start processing pain signals.
The fact that drugs (aspirin and opiates) that are highly effective in treating acute pain often have little effect in chronic pain suggests the two conditions are quite different. This suggests to Apkarian that therapies targeting emotional suffering (the cortical limbic circuitry) may be more effective.
Therefore, we theorize that identifying and manipulating processes underlying the emotional suffering (cortical-limbic circuitry) should be more successful in treating chronic pain than the standard approaches … that have concentrated on the source of nociceptive signals in the skin and spinal cord.
Dr. Apkarian has some brain imaging research results to back up his proposed theory. Some of his lab’s other recent results were highlighted in recent blogs by Cort Johnson:
- Bad Wiring: Were You Set Up For A Life of Chronic Pain? A Source of Chronic Pain
- Shape Shifting Pain: When the Brain Becomes Emotional
The Apkarian Lab was the first to examine brain activity in a common pain condition as it transforms over time. They scanned the brains of 46 people who had low back pain for about three months before coming to the hospital but who had not had any pain for at least a year before. The researchers scanned the subjects’ brains and evaluated their pain with doctor’s examinations and questionnaires four times over a period of one year. About half of the subjects recovered at some time during the year, while the other half had pain throughout which the researchers categorized as “persistent” (think “chronic”).
Previously, the Apkarian laboratory showed that the volume of grey matter in the brains of the same subjects who had persistent pain decreased over the same year. Grey matter describes the area of the brain where the central bodies and branched antennae, or dendrites, of nerve cells reside. In this study, at the beginning of the experiment the researchers measured the structure of brain white matter, which connects brain cells in different parts of the brain. They found a consistent difference in white matter between the subjects who recovered and the subjects who experienced “persistent” pain.
The researchers also found that the white matter of subjects who had “persistent” pain were similar to people with long term chronic pain. In contrast, the white matter of the subjects with acute pain who recovered looked similar to that of healthy control subjects. Next the researchers asked whether the white matter differences they saw during the initial brain scans predicted who would develop chronic back pain. They found differences in brain white matter found at the beginning of the pain process predicted at least 80 percent of the outcomes!
They also looked at the brain activity signatures of some of these patients. The acute back pain patients who had high functional connectivity between the medial prefrontal cortex and nucleus accumbens (areas more related to emotions) at the outset of the study were more likely to experience persisting pain.
“Brain signatures of pain became more emotional in nature, although the description by the patients of their pain remained constant. … The idea that certain perceptions, like pain, are only encoded by certain brain regions has left its mark on psychology and neuroscience. This is the first unequivocal evidence that that cannot be tenable. ”
It’s important to note that the studies were done on patients with chronic low back pain. The Apkarian lab results, among others, have made it apparent that the brain signatures for different chronic pain states are different, so it’s not clear that these findings apply to other chronic pain states such as fibromyalgia, complex regional pain syndrome (CRPS), the various forms of chronic neuropathic pain, etc., as Apkarian theorizes. One area that has not been explored is how chronic pain brain signatures change over time. Time and more research will hopefully tell the story.
Conclusions
The Dr. Apkarian’s lab reverse translational approach to chronic pain has resulted in clinical trials of an antibiotic for chronic neuropathic pain. Their research suggests people with “chronic” back pain had unique white matter brain signatures prior to the development of their chronic pain, and upon developing the chronic back pain have very different brain activity signatures from patients who have acute low back pain.
The brain signatures of the people who develop chronic back pain show increased activity in the areas of the brain that are connected to emotions. This increased activity may be due to atrophy in the prefrontal cortex, the part of the brain that controls executive functioning.
Brain imaging studies have shown no evidence of increased nociceptive representation in the brain but rather point to enhanced activity in the emotional cortical-limbic circuitry. This has led Dr. Apkarian to theorize that identifying and manipulating the processes underlying emotional suffering may be much more successful than the current methodologies which have been largely unsuccessful.
On a personal note, I spent the two days before this final edit of this piece, crashed and mostly in bed, humbled by the severity of the pain I was experiencing. So I want to emphasize that these researchers are not saying that your chronic pain is “all in your head” or “chronic pain patients are faking it”, but that some chronic pain conditions apparently result in a very complex dance between emotions and chemistry being performed and, like many of you reading this, I am simply exploring the moves to this dance in a quest to quiet the pain.
Check out more of Tim’s blogs here








Why are they still investigating new drugs for the condition instead for the cause? Pain is there for a reason for God’s sake!
When diabetics lose feeling in their feet it is detremental yes?
I am not saying I want to be in pain but so much research into drugs seems to be of paramount importance for some reason.
I fully appreciate painkillers and I am not against new ways of killing pain. However messing around with the nervous system with drugs seems quite dangerous. All drugs are toxic after all.
Oh how I wish they would investigate the cause. Without a cause there will never be a cure.
Is it not clear yet that drug companies are the only ones that can get the FDA et al to do anything through massive amounts of money and pressure. They, of course, aren’t much interested in cures but adding long term revenue sources to their bottom line.
well said Dawn money is in the treatment not in finding the causation or in it’s prevention
– unless they already know
Your point is well taken for many disorders, but I think this work applies to conditions where the pain response is the problem – not something that’s gone wrong with the body.
For instance, some people with no discernible structural problems in their back nevertheless have strong pain. People with fibromyalgia and allodynia develop such exquisite sensitivity that even clothes touching their skin can cause pain. Their skin is not injured – the nervous system is.
Cort,
You have summed up the pain response issue so well. A good example of pain and the brain is CRPS (complex regional pain syndrome). The human pain response seems to be completely out of whack in this disease.
A group of German researchers, Pleger, et. al., recently published a FMRI brain imaging study in PLOS ONE on Complex Regional Pain Syndrome. Their main finding was that they found an increase (not decrease) in gray matter density in the dorsal medial prefrontal cortex and the involvement of emotion related portions of the brain. These researchers speculate, “A possible explanation for these differences between healthy individuals and chronic pain patients is, that the pain experience in patients suffering from chronic pain is mostly driven by the brain itself and that the afferent noxious input is no longer of great relevance for the pain experience.”
They found an increase in prefrontal cortex density (interesting) but similar findings in the rest of the brain. Very interesting…This – ” that the afferent noxious input is no longer of great relevance for the pain experience.” is so fascinating…:)
I have read that pain serves a purpose. I am struggling to correct the underlying causes of my fms, but I read that pain ages you, and there are times throughout the “healing process” that we need relief.
I also had nerves damaged while undergoing “minimally invasive surgery” (don’t buy into the baloney; it is risky). I underwent an operation 8 years later – after finally finding a doctor who would even work on me. He did relieve me of a lot of the pain, but still get shooting pains down hip, groin & leg. He was an excellent dr. but I wanted him to cut the nerve; I didn’t care if I lost all feeling – just didn’t want to feel the pain anymore. He wouldn’t do it. Told me to desensitize using TENS & massager.
I just want relief. The “pain” is not serving a purpose anymore; therefore, it needs to be resolved. Glad I found this site. Anybody know if I can sign up for a trial for this?
Have you tried Low Dose Naltrexone? It’s very affordable, has few side effects and really works for some people. The antibiotic trial is already underway, by the way.
Good luck!
Notification.
I will be interested to see how the results of the D-cycloserine trial turn out.
I get fed up with having to make a consistent effort to clean up my cognitive processes in order to minimize pain. It’s like an involuntary spiritual practise, and i’ve never been that interested in spirituality or mindfulness.
But getting upset, stressed or even bored can definitely be counterproductive for me pain -wise.
One sure fire trigger for me is meeting someone in the street who wants to chew my ear for 3 hours or so, especially while standing up. I start to stress and can actually start throbbing with pain if i cant make a quick exit.
I’m not as tolerant of epic story tellers as i used to be.
I don’t mind some chemical assistance for pain until they can unwind all the pathologies around me/cfs/lyme/etc
I love this
“It’s like an involuntary spiritual practice” (so true)
and this
“I’m not as tolerant of epic story tellers as i used to be”.
rings a bell as well.
They gave me a good laugh. Thanks 🙂
Very interesting Tim.
I came across a similar proposal for the treatment of post traumatic stress disorder where psychological treatment aimed at fear extinction and often only partly effective or ineffective.
It seems that glutamate plays a key role in the strength or the memories created following trauma and the proposal is to administer a glutamate agonist during fear extinction therapy in order to over-ride (or over-write) the negative memories.
Marco,
The D-cycloserine trial appears to be based on a similar theory to fear extinction. It would be great if some positive results came from that trial.
Thanks for explaining what why the Apkarian lab used a NMDA agonist.
I tried D-cycloserine for a week, but without any clinical help. Just ordered it over the net.
Basically i endeavored to take it easy and have as much fun as possible in that week. I meditated and thought of instances that bought up pain, often whilst eating chocolate (Lindt, preferably). Sound crazy ?.. I can be.
I got minimal results. The same as putting some effort into any mindfulness practise. The problem is that any gains can be quickly eroded by not sticking to a strict sleep, diet, no stress regime. Or even just by the up and down vagaries of illness.
I guess the duration and quality of clinical care while taking D-cycloserine are crucial.
Nowadays i often take paracetamol (tylenol) if i’m anticipating stress, on account of its reputed physical and emotional pain relieving qualities. Just to roadblock any pain superhighways that might be forming.
http://www.sciencedaily.com/releases/2009/12/091222154742.htm
Tylenol eases emotonal pain.
http://www.medicalnewstoday.com/releases/227298.php
I would put Gupta practise or simply distracting myself with friends and fun as my most effective non-drug pain interventions.
Thank you, Tim, for a great review of Dr. Apkarian’s studies so far. I’m glad someone is taking a look at pain and brain activity signatures from a different perspective.
About Dr. Apkarian’s finding:
“The brain signatures of the people who develop chronic back pain show increased activity in the areas of the brain that are connected to emotions. This increased activity may be due to atrophy in the prefrontal cortex, the part of the brain that controls executive functioning.”
I’ve been concerned for years about grey matter disappearing in my prefrontal cortex–like there are big holes where my executive functioning used to be. I do wish some researcher was collecting and studying donated brains of people with fibromyalgia and ME/CFS who have died. I’d sign up today to donate my brain to a study like that.
Given the numerous studies showing reduced grey matter activity and volume in those of us who suffer with fibromyalgia & ME/CFS, I don’t understand why our illnesses are not taken more seriously. Parts of our brains are disappearing! Isn’t that in itself cause for alarm?
Amen! I’m donating all of me for CFIDS/FM research, just don’t know exactly who or where yet. Hopefully, by the time I need to name a place, this will be getting more attention and I’ll have some suggestions.
I have suffered from fms for over 20 years. I totally disagree with your statement that aspirin and opiates are highly effective on acute pain but often have little effect on chronic pain. After taking 800 mgs. of ibuprofen 3x a day for approx 2 yrs. my dr. switched me over to oxycodone/apap, 325/5 mg. 2x per day. This has, and IS my “lifesaver”. It not only takes away most of my pain but also clears my fibro fog. I have been taking this for over 10 years and, after all that time, I have not needed to increase this dose for relief. I also take klonopin at bedtime and 60 mgs of Cymbalta in a.m.
Glad to hear it Joan. (The statement was by the researcher by the way.) That’s great that it clears your fibro-fog as well. I’ve heard of other FM patients opiates work well for. Thanks for sharing your experience.
HOW CAN SOMEONE GET CRPS WITHOUT PREVIOUS INJURY?
This CRPS…It’s bacterial….She was taking Cephalodroxil, synthetic Keflex….her symptoms disappeared. CEPHALOSPORIN…STAPHYLOCCUS…IF THERE’S MERSA….then the next Where does the money in WORKER’s Comp Go??? PHYSICAL THERAPY.
ACCUPUNCTURE WORKED BETTER THAN PT.
MAYBE THAT BACTERIA IS IN SWEATY PT ROOMS???
THE WAX???THE TABLES, THE EQUIPMENT… MAYBE YOU ARE SUBLTY EXPOSED, DONT REALIZE IT, AND WHAM…THE PERFECT STORM…A DOCTOR ISNT GOING TO WORK HARD TO KILL HIS PRACTICE..
MAYBE WE HAVE A COMMON GENE…CIRCUMSTANCE OR SURGICAL SUPPLIER,
INSTEAD OF TALKING ABOUT EMOTIONS AND PAIN, WE SHOULD COMPARE MINOR DETAILS AMOUNG OURSELVES AND WE SHOULD UNIVERSALLY RECOUNT OUR PERSONAL TIMELINES…WE HAVE COMMON THREAD….THERE IS A CURE
Great post…Pain is an unpleasant conscious experience that emerges from the brain when the sum of all the available information suggests that you need to protect a particular part of your body. When a health illness or condition is chronic it means it is long-lasting. Chronic pain is pain that persists or progresses over a long period of time. Chronic pain typically has persisted for at least 3 months. visit our site for good tertments for Back Pain, Diabetes, Fibromyalgia, Thyroid, chronic pain, sciatica, whiplash, headaches & nutritional disorders. http://www.drbastomski.com/