

RESEARCH AVENUES/POTENTIAL TREATMENTS
(Please note I have no medical background am in no way qualified to recommend any particular course of treatment for ME/CFS. The following is a theoretical discussion that logically flows from the proposed neuroinflammatory hypothesis and is intended to outline potential avenues for further investigation. Please see your physician before altering any component of your treatment plan).
“Why So Resistant to Treatment?” Plus “Is a Cure Possible?
The previous discussions propose that ME/CFS may be caused by an imbalance in GABA/Glutamate (and in the case of the ‘Wired and Tired’ type a hyperglutamatergic or high extracellular glutamate state). This suggests that anything that either boosts GABA or blocks glutamate (or assists with the other parts of the vicious cycle – oxidative stress and mitochondrial dysfunction) could be helpful.
This may indeed be the case however, many proposed ME/CFS ‘protocols’ are specifically aimed at addressing oxidative stress or mitochondrial dysfunction, and many of us do use either drugs (e.g. benzodiazepines), supplements or compounds that affect GABA/glutamate in this manner and we have not been ‘cured’.
‘Time The Healer’

A process called ‘kindling’ could cause the fatigue and pain producing pathways in the brain in ME/CFS/FM to become hyper-aroused.
If, we accept for the sake of discussion, that a GABA/glutamate imbalance underlies ME/CFS, then (referring back to Part II) it’s possible that the process of ‘kindling’ causes neuroplastic changes that result in the brain becoming easily hyper-aroused. It may help to think of this as an aberrant network or hub of interconnected neurons that have been primed to respond to little sensory input
The problem then (as I see it) is not just to break the vicious neuroinflammatory cycle but to do so completely and consistently, and for a sufficient period of time that the aberrant neural connections are themselves extinguished through the process of neuroplasticity. (You may recall that the repeated use and withdrawal of GABAergic benzodiazepines and alcohol can itself induce kindling).
Baclofen
The GABA-B receptor agonist (activator) Baclofen has been featured throughout this series, not only as a ‘cure’ for alcoholism, but also as a treatment for stiff person syndrome and tinnitus – all conditions in which a glutamate/GABA imbalance is likely to play a key role. Baclofen was a preferred med in Dr Jay Goldstein’s protocol for ME/CFS (who’s book ‘Betrayal by the Brain was discussed in Part I).
A similar situation may occur with Baclofen in the treatment of alcohol dependence/withdrawal symptoms where a high dose Baclofen regime (above the ‘threshold dose’ where alcohol cravings apparently cease) may be needed not just to break the cycle of addiction but to also prevent severe withdrawal symptoms due to ‘rebound’ induced glutamate excitotoxicity. Similarly an extended period of Baclofen treatment is also required to break the addiction and avoid serious withdrawal symptom. In fact, Baclofen treatment for alcohol dependency may require continuing (indefinitely) on a maintenance dose (see The Little Pill That Could Cure Alcoholism, in press).
Of interest is the fact that GABA B receptor agonists (like Baclofen) or modulators (that are only activated in the presence of endogenous GABA giving less undesirable side effects), block the rewarding effects of addictive drugs such as alcohol, nicotine or cocaine (Slattery et al, 2005). You may recall that the CDC (Unger et al 2012, abstract presented at conference) found decreased activation of the basal ganglia (a key part of the brain associated with reward, motivation and motor activity) in ME/CFS patients,and that this under-activation was correlated with fatigue levels.
Unger et. al.suspect dopamine dysregulation is occurring in ME/CFS but GABA-B receptors are also widespread in the basal ganglia and are increasingly seen to play a role in reward mechanisms. (Cohen et al, 2012). These receptors enable the brain to compute the balance between the expectation of reward and the actual reward received. (Note that the COMT gene associated with ME/CFS modulates both dopamine and GABA. See Part II).
Of course, as has been mentioned repeatedly, extreme caution is needed when any attempts are made to modulate the glutamate/GABA balance due to issues of tolerance and dependency and the danger of inducing neurological kindling (or exacerbating existing kindling). (In the case of stiff person syndrome baclofen pump failure in rare cases can be fatal (Mohammed, Hussein, 2004).)
It is also uncertain what adaptions might take place over time if attempts are made to modulate neurotransmitters e.g. it’s possible that exogenous boosting of GABA levels could result in reduced receptor sensitivity that potentially exacerbates the problem once supplementation ceases.
In short, this is a complex problem and any pharmaceutical interventions directly aimed at altering the glutamate/GABA balance should done only be under strict medical supervision.
‘Alternative’ Approaches
Alternative approaches may have some benefits. Regular meditation has be shown to increase endogenous GABA levels (Guglietti et al, 2012) and a recent study has shown that following a low glutamate diet may be effective in reducing the symptoms of fibromyalgia and IBS (Holton et al, 2012).
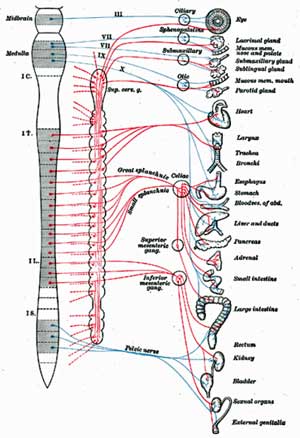
Addressing Autonomic Dysfunction
As discussed previously, autonomic dysfunction is a common finding in ME/CFS with a a pattern of ‘sympathetic dominance’ which may be responsible for some of the more disabling symptoms such as exercise intolerance, POTS, cognitive and sleep problems. Glutamate could be responsible for long term alterations in autonomic function or indeed may induce autonomic neuropathy of the type seen in diabetic autonomic neuropathy (DAN).
Regardless of the cause of autonomic dysfunction, even autonomic neuropathy can be treated (if not reversed), and the earlier the treatment begins better as autonomic dysfunction can result in eventual neuropathy. In fact, in type 2 diabetes it is recommended that autonomic testing begin as soon as the condition is diagnosed as autonomic dysfunction can precede clinical symptoms. Strikingly, it is reported (Vinik and Murray, 2008) that (bolding added) :
“medical leadership including the American Diabetes Association (ADA), the American Heart Association, the American Academy of Neurology, the American Academy of Family Physicians, the Juvenile Diabetes Research Foundation International, and the National Institutes of Health have published recommendations for autonomic testing as part of the standard of care for chronic diseases.”
Treatment (usually using ANS modulating medications) needs to be individualized based on the overall pattern of dysfunction (sympathetic, parasympathetic dominant or mixed). With its chronic nature and frequently confirmed evidence of autonomic dysfunction, surely ME/CFS must be included in the list of chronic diseases that require early autonomic testing and treatment as part of ‘standard care’ to prevent autonomic dysfunction progressing to life threatening (sudden cardiac death) neuropathy.
In the absence of this, alternative and various ‘self-help’ approaches such as vagal nerve stimulation, biofeedback, diaphragmatic breathing exercises and meditation may all be helpful in restoring ANS balance. Reporting on the link between heart rate variability and cognitive dysfunction in CFS, Australian researchers (Beaumont et al, 2012) have proposed various methods including biofeedback and transcranial direct current stimulation for modulating vagal tone.
Addressing Neuroinflammation
Recalling Rönnbäck and Hansson’s model of mental fatigue discussed in Part II :
“If our hypothesis is correct, it may be possible to further improve the symptoms by suppressing the production of proinflammatory cytokines and, thereby, restoring the normal astroglial glutamate uptake.”
Addressing neuroinflammation by other means may also be helpful. Various antioxidants, vitamin B12 (Zoccolella et al, 2009), magnesium (Poleszak et al, 2008), N-acetylcysteine (Dean et al, 2011) and CoQ10 (Dumont and Beal, 2011) may all help to reduce neuroinflammation.
Pharmaceuticals or natural compounds that inhibit pro-inflammatory TNF alpha or COX-2 may similarly help to protect against glutamate neurotoxicity and break the vicious cycle (Article – McIntyre and Cha, 2011) :
“Cyclooxygenase-2 (COX-2) inhibitors have been shown to protect against glutamate-induced neurotoxicity; to prevent normal aging-related increases of cytokines, prostaglandins, and TNF in neurons; and to modulate the hypothalamic adrenal axis”
COX-2 inhibitors may also help restore NK cell activity and counteract dysregulation of the HPA axis through also inhibiting pro-inflammatory PGE2 (Lin and Nelson, 2003; Scemes and Spray, 2011 -in print).
Fluge and Mella are currently trialing the TNF-a antagonist Etanercept in their ME/CFS patients including some who did not respond to Rituximab.
Drugs Worth Investigating
Stablon (Tianeptine) is a fast acting ‘atypical’ antidepressant that, as seen in Part II, appears to be able to protect the brain (particularly the amygdala and hippocampus) against stress induced glutamate excitotoxicity.
Memantine (or other drugs that block glutamate NMDA receptors) have been shown to help with anxiety and sleep (Schwartz et al, 2012) and with neuropathic pain (Jamero et al, 2011).
Lyrica (pregabalin) an anticonvulsant, is one of only three FDA approved drugs for the treatment of fibromyalgia, and has been found in several clinical trials to significantly reduce pain (and fatigue, anxiety and sleep problems). Aside from fibromyalgia, Lyrica is also used to treat diabetic peripheral neuropathies, generalized anxiety disorder (GAD) and social anxiety disorder (peripheral neuropathies and anxiety have been discussed previously in the context of this neuroinflammatory model). Although a GABA analogue, Lyrica does not bind to GABA receptors like benzodiazepines. Instead it appears to block certain neurotransmitters including glutamate (Kim et al, 2009). A Lyrica trial in ME/CFS under the direction of the Lights is underway in Utah.
Doxycycline (and other tetracycline antibiotics such as Minocycline) has been shown to reduce glutamate excitotoxicity and may be effective in treating pain via its inhibitory effects on TNF-alpha (Cazalis et al, 2008) which may also contribute to neurotoxicity (Takeuchi et al, 2006). One theory (yet to be tested) proposes that Doxycycline treatment may help improve the mitochondrial content of skeletal muscle in metabolic syndrome (White, 2010 – research proposal).
Nuvigil (armodafinil) a recent stimulant medication commonly used to treat narcolepsy (also used off-label for ADHD) may help with improving cognitive issues and other sensory problems by increasing available dopamine levels (McIntyre and Cha, 2011). You may recall from Part II that dopamine also improves sensory gating by acting as a ‘gatekeeper’ of glutamate transmission by boosting strong (salient) signals and suppressing weak signals.
Similarly, Citicholine (CDP-choline), a psychostimulant, which increases dopamine receptor densities may help with focus and mental energy and has shown some efficacy in treating ADHD. CDP-Choline also modulates the HPA, reduces oxidative stress and lowers excess glutamate by increasing the re-uptake of glutamate and increasing the expression of the EAAT2 glutamate transporter (Wikipedia). Also notable is that under-activity of the EAAT2 gene may be implicated in common migraine (Antilla et al, 2010), while recent research suggests that some 80% of ME/CFS patients experience migraines (Ravindran et al, 2011).
Investigations into triptan migraine drugs suggest that their effects are due to reducing CSF extracellular glutamate (Vieira et al, 2007) and one case study reports complete remission of migraine after administration of the NMDA receptor blocker memantine discussed above (Spengos et al, 2008).
As discussed in Part II, Tecfidera (dimethyl fumarate) which has been recently approved by the FDA for the treatment of relapsing/remitting MS also appears to owe its neuroprotective effects to stimulation of the EAAT2 glutamate transporter.
Antibiotics – Interestingly, the EAAT2 glutamate transporter has also been implicated in neurodegenerative diseases such as Alzheimer’s disease, Huntington’s disease, and amyotrophic lateral sclerosis (ALS). Screening of over 1,000 FDA approved compounds showed that common beta lactam type antibiotics (which include penicillin derived antibiotics) activate the EAAT2 protein and in an animal model one such antibiotic, ceftriaxone, increased glutamate re-uptake thereby reducing excitotoxicity and providing neuroprotection (Kim et al, 2011).
GABA and the Immune System
Several GABAergic drugs are currently used for epilepsy and migraines associated with MS and may have additional protective effects in MS due to the proven inhibitory effects of GABA on immune mediated inflammation.
While GABA is usually considered solely as a neurotransmitter it now appears that certain immune components contain GABA receptors and that GABA actively influences immune activity (and GABA is also produced by the immune system). Multiple sclerosis patients have a low level of serum GABA and the GABA synthesising enzyme GAD. In an animal model of MS (experimental autoimmune encephalitis) GABA agonists can reduce inflammation (acting directly on antigen presenting cells, and indirectly on T cells leading to diminished production of inflammatory cytokines including IL1β, IL6 and TNF-alpha) thereby slowing or even reversing the neuronal damage (Bhat et al, 2010).
Low dose naltrexone appears to enhance glutamate reuptake, thus removing glutamate from the extracellular spaces where it can prove toxic
Low dose Naltrexone (LDN) is an off-label treatment with a substantial following amongst those with ME/CFS, fibromyalgia and multiple sclerosis. Naltrexone is a powerful opioid antagonist used in the treatment of opioid and alcohol dependence. Anecdotal reports have attributed LDN’s effectiveness to partial blockage of the opioid receptors that upregulate endogenous opioid production, a reduction in proinflammatory cytokines impacting on microglia and certain unspecified ‘immune boosting’ effects.
One hypothesis posits LDN increases glutamate reuptake and prevents glutamate induced neurotoxicity in MS (Agrawal, 2005). While the effectiveness of LDN in MS has yet to be confirmed in clinical trials a small LDN trial has shown some promise in treating pain in fibromyalgia (Younger et al, 2013).
- Dig Deeper – Successful Low Dose Naltrexone Fibromyalgia Trial Points to Safe, Low Cost Therapy; Implications for Chronic Fatigue Syndrome
- Dig Deeper – Low Dose Naltrexone
N- acetylcysteine (NAC) is another commonly used treatment that may have potential in treating ME/CFS. Originally used as a mucolytic (relieving congestion in the common cold), this powerful antioxidant is the first line treatment for paracetamol overdose. As previously seen, NAC has also shown promise in the treatment of certain ‘neuropsychiatric’ disorders including OCD and bipolar disorder.
Being a glutathione precursor, NAC may support mitochondrial function peripherally and in the brain. Trials have shown NAC pretreatment can enhance endurance by reducing oxidative stress in exercised muscles (Medved et al, 2004). In addition to its antioxidant properties NAC has other properties that might address neuroinflammation.
A review of NAC’s potential use in psychiatry (Dean et al, 2011) indicated NAC attenuates neurotoxicity by reducing the levels of the pro-inflammatory cytokines IL1-b, TNF-a and IL6 and by modulating glutamate and dopamine levels. NAC has also been found to reduce levels of the pro-inflammatory factor C-reactive protein (CRP) in type II diabetes (Jeremias et al, 2009) (as discussed last time IL1-b, TNF-a, IL6 and high-sensitivity CRP are the inflammatory markers found to be raised in ME/CFS.) NAC has been shown to attenuate disease progression in experimental autoimmune encephalitis (the animal model for MS – Stanislaus et al, 2005). NAC has the added advantages of being cheap, readily available and with demonstrated safety in normal clinical doses.
Heat Shock Proteins
One final avenue worth exploring is whether it’s possible to enhance the protective role of heat shock proteins which appears to be attenuated in ME/CFS patients.
As was seen earlier, enhancing heat shock protein activity has the potential to break the ‘vicious-glutatmate production circle’ at each stage via HSPs’ ability to reduce oxidative stress, prevent glutamate induced excitotoxicity, and induce the expression of mitochondrial regulating PGC1a. It appears that in low energy states (due to poorly functioning mitochondria) even minimal levels of extracellular glutatmate can make the brain highly vulnerable to glutamate induced excitotoxicity (Blaylock, Maroon, 2012).
HSP’s may be induced by exercise (if tolerated), heat or cold stress exposure (an effect that may be enhanced by pre-treatment with common aspirin – Fawcett et al, 1997), certain ‘nutraceuticals’ and glutamine (Wischmeyer, 2006).
Drug ‘Repurposing’
The CFIDS Association of America/Biovista drug repurposing project (now completed although details of the compounds identified as potentially useful have not been released) searched a veritable mountain of data to find novel connections between the mechanisms of action of existing drugs and the key symptoms of CFS using an ‘unbiased’ computational approach (CFIDS association, Partnering for Cures conference, 2012).
I hope I have shown, in this and previous blogs that there is converging evidence to link ME/CFS to a range of other neuroinflammatory diseases and that (currently licensed and often cheap) medications used in these disorders may be useful in treating ME/CFS. This suggests, of course, that drug repurposing efforts that examine symptoms found both in ME/CFS and other neuroinflammatory disorders could be fruitful.
However, such associations are only likely to be made if all the overlapping symptoms (such as sensory processing disorders) have been noted in the ME/CFS literature. While laudable, there is no certainty that such connections are likely to surface using an ‘unguided’ sift of the existing literature.
‘Nutraceuticals’
There is growing evidence that a number of ‘natural’ compounds (including curcumin, resveratrol, quercetin) may prevent neurodegeneration due to inflammation/excitotoxicity and can induce production of neuroprotective HSPs (including curcumin – Blaylock, Maroon, 2012). If it can be shown that the core pathology in ME/CFS is a neuroinflammatory state, there may be scope for improvement through simple supplementation or dietary changes.
As seen previously, a glutamate/GABA imbalance may underpin peripheral and autonomic neuropathies which may be exacerbated by a thiamine (vitamin B1) deficiency. As discussed earlier, thiamine deficiency results in peripheral and autonomic neuropathy. Benfotiamine, a synthetic form of thiamine is a popular dietary supplement treatment for diabetic peripheral neuropathy in Germany. Thiamine may have additional benefits in a neuroinflammatory model by supporting the functioning of the GLAST glutamate transporter. In addition CoQ10 supplementation reduces neuron loss in the dorsal root ganglia in a mouse model of diabetes (Shi et al 2013) while a meta-analysis supported the efficacy of alpha-lipoic acid in reducing peripheral neuropathic pain in diabetics (Mijnhout et al, 2012).
Is a Cure Possible?
My gut feeling is that there is the potential for many of our symptoms to be resolved with appropriate treatment. It may be likely though, that the neuroinflammatory state, over a long period of time, results in long-lasting damage including peripheral and/or autonomic neuropathies that may not resolve easily.
I also feel that an underlying predisposition (perhaps genetic) may be present for the neuroinflammatory state to develop when we are faced with stressors which could result in any of the previously mentioned conditions developing at some stage including the typical ailments of ageing
Interestingly, given her key role in the upcoming Pathogen Discovery and Pathogenesis Program at the Chronic Fatigue Initiative (as blogged lately by Cort), a similar ‘multi-hit’ model appears to be very much in the mind of Dr Mady Hornig.
- Dig Deeper: Infection, Autoimmunity and PANDAS: Dr. Hornig on Chronic Fatigue Syndrome at Dr. Klimas’ NSU Conference
Dr Hornig has co-authored a paper (along with Dr ian Lipkin – Miranda et al, 2010) which, using an animal model for schizophrenia and autism, demonstrated how maternal infection could increase the risk of the offspring developing ‘neuropsychiatric’ conditions. The mechanism suggested involved Toll-Like Receptors (TLRs). Intriguingly, the putative ME/CFS therapy Ampligen targets TLRs (Hemispherx Biopharma presentation to the FDA, 2012). In the context of this and previous discussions it is also worth noting that one of the neurological effects noted in the offspring in this animal model was a sensory gating deficit (the very neurological deficit that started this whole discussion). You may also recall from Part II that maternal infection with the H1N1 virus was shown to cause changes to glutamatergic and GABAergic genes.
- Dig Deeper: Not Fatigue After All? New Model Suggests Other Symptoms Better Explain Chronic Fatigue Syndrome (ME/CFS)?
Dr. Hornig proposes that ‘Chronic Fatigue Syndrome’ results from a genetic predisposition, a ‘window of opportunity’ or vulnerability and an environmental insult. A maternal infection during the gestation period that produces disruptive genetic changes may be one such window of vulnerability. This model suggests that the development of ME/CFS (or other ‘neuropsychiatric’ conditions) may take years or even decades and occurs when a genetically vulnerable person encounters any of a wide range stressors (including psychological stress, viruses and environmental toxins).

Lifestyle adjustments, pacing and medications may all be helpful in glutatmate induced neuroinflammatory illnesses such as ME/CFS/FM
It may be that the condition of ME/CFS must be treated in a similar way to diabetes requiring a lifelong attention to ‘lifestyle’ and potentially ‘maintenance’ medication.
This may require some compromise in how we lead our lives including some moderation of previously cherished activities or ambitions in the knowledge that we may have a lifelong vulnerability. As Rönnbäck and Hansson said in respect of dealing with mental exhaustion ;
“Furthermore, it is important for the patient to imagine and learn how much sensory stimulation they can tolerate prior to feeling too exhausted.”
This may sound like pacing and indeed it is. But it is also valuable advice that might have been useful for many to have had prior to onset of the ‘state’ of ME/CFS.








I had the typical ME/CFS GABA/glutamate imbalance verified on my Nutreval test and I also had to take large amounts of either GABA or theanine for many years to keep it in check. Not too long after starting methylation supplements, I was able to discontinue the GABA, indicating that this came back in balance. This has remained balanced for five months and counting.
Good to hear Caledonia…How much improvement would you say that you’ve seen on the GABA enhancement/methylation approach?
Read more: Cooling the Flames: Possible Approaches to Reducing Neuroinflammation in Chronic Fatigue Syndrome (ME/CFS) and Fibromyalgia http://www.cortjohnson.org/blog/2013/07/11/cooling-the-flames-possible-approaches-reducing-neuroinflammation-chronic-fatigue-syndrome-fibromyalgi/
Hi where can I get this nutrevyl test ?
I have FMS
Great article, Marco. I recently did some research on this and here is some things that I found:
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2312368/#
Interesting article on doxycycoline helping suppress P25 which could cause atrophy of hypocampus and neuro dysfunction, affects genetics and inflammation with increased cytokines. Granted, it’s a mouse study —but, what if this would help us humans too. Interestingly, I’m taking a small/low dose of doxy for my protozoa problem. For sure my brain function has improved and so has my POTS. Maybe, this is the reason.
Here’s some really interesting articles that I ran across showing that even if there is a genetic defect and doxy is given – it could potentially help with the genetic dysfunction and prevent neurological dysfunction. (Epigenetics —maybe????)
http://www.holtorfmed.com/index.php?section=downloads&file_id=65
http://onlinelibrary.wiley.com/doi/10.1002/glia.22496/abstract
http://www.off-label.com/drug/resultsdrugus.cfm?drug=doxycycline
http://cds.ismrm.org/protected/09MProceedings/files/00542.pdf
Issie
Thanks Issie
Looking forward to following up the links.
I was excited to find these articles. I didn’t realize how powerful doxy could be. And for sure didn’t realize that it had neuro protective abilities. With the autonomic nervous system playing into this – it could very well be of great help to many.
Issie
I was impressed by all the different approaches that might be taken to tamp down glutamate excitoxicity….I think there are alot more treatment possibilities than we realize and certain mounds more than most docs are aware of.
I am curious if Gabapentin would have the same effect as some of the RX’s mention in this article?
Hi Penny
Short term yes. Gabapentin is frequently used for various ME/CFS symptoms.
The problem as previously discussed is that GABAergic compounds come with problems of dependence, tolerance and neurological kindling with intermittent use and we also don’t know how the brain’s GABA receptors might adapt over time to exogenous GABA.
Baclofen appears (comparatively) less problematic in this respect, perhaps because it works on GABA B rather than GABA A receptors but again like all drugs there are associated issues.
FWIW a new version of Baclofen (ARBaclofen) has been developed to have much greater effectiveness with fewer side effects and is currently being trialed for treating social avoidance in autism spectrum disorder.
Ideally it would be preferable to find some way of rectifying the natural balance between glutamate and GABA rather than GABA supplementation and addressing other aspects of neuroinflammation may have this effect.
Hey Marco and Cort,
I think the subject at hand is likely a good one, but I must say I could only understand about 1/10th of it. :o( Much complexity to the issue. Is it possible to break this report down into laymen’s terms for those of us who are not quite as educated in the field of medicine and the body?
I’ll even pay you $10 bucks, yea, that’s right, $10 bucks on my 70th birthday which is January 1st. You can then save it for New Year’s Eve that next year. LOL How many days from now until then?????? Ten years…3,650 days excluding Leap Years….This is your chuckle for the day guys!!
Hi Brenda
Lots of drugs have effects far beyond their intended target and a number of drugs and natural compounds have the potential to target various aspects of the proposed ‘neuroinflammatory cycle’ involving glutamate neurotoxicity, oxidative stress and mitochondrial dysfunction.
If it could be shown that this neuroinflammation underlies ME/CFS, there may be many drugs available ‘off the shelf’ that could help.
How’s that?
Can we have the $10 now? : )
Marco and Cort, the check is in the mail…………………hahahahahaha
Looking forward to it…:)
ummm..maybe that is why I always feel better when Im on antibx.?
I am one who was on Doxycycline for several years, and was only concerned for its’ effect of suppressing the Rosacea from my face. Now that I see it also has neurological benefit, I am wondering if I should go back on it. Since the inflammation of this disease is so multi-factorial, with different causes and outcomes, I am thinking that it may have been of greater benefit than I can remember. At onset, my C-protein was a ‘panic’ call from the lab to a surgeon who was running a few tests to help me understand this mindboggling condition. We remained quiet while he scrubbed, and I insisted that it was not muscular only, it had the be my brain, as well. Sadly he died about six years ago, and I never got to thank him for his listening, intelligent ear. I was waiting to see a specialist and I hid the disease on the workfront as well and as long as possible. I did not make any errors yet I was suffering incredibly, and felt like I was dying. I did not want to stop working, yet finally after a long night I stopped. The pain was overwhelming and I had a call to return two hours after I got home. I could not, and would not do it and soon was alone, trying to still have a fixed understanding of this condition. I never have had a fixed understanding as it grows, and one theory replaces another. Yet, I was determined to pin down the causes of the most disabling symptoms and once I got to that point, I didn’t care anymore. Neurological symptomatology is fleeting in my mind now, and I keep trying not to remember. Yet, I am intrigued with further explanations…many thanks. MC
This is all interesting, at least the parts I can understand! But what are the chances that any of this will filter down to clinicians, the people we have to engage with to get any treatment? It’s still a crapshoot to find a doctor who doesn’t roll his eyes when you tell him you have fibro or CFS, or both. 3 months ago I had a rheumatologist snap at me because I’d written on the health history that I suffer from both. His question “Well, which is it? You can’t have both!” Don’t I wish. But, back to the larger question. Is there any chance that any of this research will reach down to family practice doctors or the average rheumatologist? Please say yes!
Hi Agnes
You do have to wonder about the ‘bedside manner’ taught in medical school.
My ‘diagnosis’ of ME/CFS went like this.
After many years of various symptoms my doctor suggested that I had fibro. When I pointed out that I didn’t have the classic tender points (as were then required) he then said OK – probably ME.
Next visit when I mentioned the ME he told me I couldn’t possibly have that and logged me as CFS. I have both on my medical records.
Getting back to your question, I’d like to think that some of our researchers might come across these postings and find them worth investigating and I am considering contacting some researchers just to suggest that its an angle worth exploring.
Its also worth noting that a few ME/CFS researchers are proposing models of ME/CFS not too far from what I’ve suggested. Plus there’s a growing tendency in medicine to look beyond individual diagnostic labels to processes that may underlie several disorders. About time too.
Will this filter down to grass roots medicine. Realistically that’s got to be some time away unless there’s a clear and unambiguous breakthrough that represents a complete paradigm shift and even then treatments still have to go through the various trial stages before widespread approval.
I’m afraid we’re likely stuck with ‘self-help’ for a while yet.
Thankfully, there are a few “maverick” doctors out there. Pioneering into fields that few know about. I’m very thankful to have found my doctor and to be experiencing a different approach. The more I research it, the more I realize what he must “know”. Because, the treatments that I’m doing are proving to be very beneficial – the science is out there – it’s just finding the pieces and putting it all together.
I was really encouraged by the articles that were posted today. Some of them show a connection with the glutamate/GABA balance (the fact of there being an excitotoxin response, with an imbalance, and reinforced my beliefs that there could be a genetic component that is triggered and a faulty immune system with lots of inflammation due to cytokines. Something as simple and inexpensive as doxycycline could be, one of the answers.
I don’t want to hi-jack Marco’s post, so I’m trying to limit how much I comment on this. But, the articles speak for themselves. Doxy may be one of many drugs that may be of great help —for more than one reason.
Issie
Thanks Issie, the more info the better…I think there probably a good number of inquisitive minds out there that are making connections…and I wouldn’t be surprised if some are following this series which is laying out new ground….
Wouldn’t that be something!
Really interesting stuff on doxy Issie. Thanks for the links.
Someone on Phoenix Rising also flagged up another potential ‘wonder drug’ called ibudilast :
http://forums.phoenixrising.me/index.php?threads/a-neuroinflammatory-model-of-me-cfs-final-part.24176/#post-370542
If the in vitro results translate this one has the potential to hit all the right targets to reduce neuroinflammation!
http://www.courses.vcu.edu/MED300FP-gso/RTH145/rth145unit6/mechanism_of_action__the_methylx.htm
This med looks like it could potentially increase tachycardia according to one article I read about it. As it’s supposed to help with slowed heart rate – not a problem with us that have POTS. (Our hearts are beating like a hamster running on a wheel when we stand up. That would not be so good for us POTS people.) I’d like to hear of people’s response to it – if anyone has tried it. In the USA it’s called Theophylline. It also appears to be in the same class with caffeine. Maybe good for some – but, could ramp up the sympathetic system too much – unless there is a paradox reaction and other factors that might could unexpectedly come into play. This looks like it could ramp up the CNS.
One thing that could potentially be of help to others would be something that those of us with MCAS use. It’s called GastroCrom or cromolyn sodium. It helps to prevent mast cell degranulation and also helps with the immune system in the gut (where a good bit of our immune system is active). It helps me with my sympathetic system too, it’s very calming.
Issie
As far as I know Nuvigil has been put on the market only because Provigil (modafanil) is going off patent. I’d stick with the latter (generic) for economic reasons. Provigil is a big favorite of geek types for improving brain function, although I am not convinced it does. It was first produced as a cure for narcolepsy and, indeed, sleepiness is what it vanquishes best. Very useful for jet lag.
I would hardly describe Ampligen as putative. Seems to be pretty well proven based on reports from normal human beings. Only the FDA would think that 90 seconds more bicycle riding is nothing. Let them try pedaling a mile in my body….
I hope someone reads this..it is six months after the fact 🙂 There is a LOT to take in here.
On the subject of Nuvigil, I take it for my ME/CFS and a few other auto-immune type things. It works extremely well for me. Provigil did not work for me, at least at the dose I got. In 7 yrs. this was the only medication that gave me any relief. Muscular, joint pain, exhaustion. Now that I have gotten Medicare/Disability and have an RX card (from AARP) the RX will not “allow” me to be on it. Using many excuses. Like I have the energy or brain power to fight them 🙁 It’s very spendy, so I believe that is the reason. I am still trying to find a way to get it. I get not much money on MediCare. I did get a free coupon from the manufacturer for a months supply. That’s it.
My question is: How do I fight this? It is disgusting to me that after all the hardships, all the time, all the appointment-7 yrs worth, running here and there, with no relief…I am not allowed to have this (for me anyway) miracle drug 🙁 If anyone out there has any suggestions, please, help me.
p.s. my insurance before (blue cross/blue shield) never said a word and paid for it for two years, plus other meds I am on….we paid a lot of money for that service. It was nice to not worry about huge insurance payments. Life….it’s always something, eh?
Thanks in advance for any ideas, help.
Carol
Thanks Marco 🙂
As I was saying, I do find it quite fascinating that something like Baclofen – which I have been taking for 7 years I suppose and was primarily prescribed for muscle aches and pains and the involuntary spasming that is associated with ME; could possibly be having other useful effects. It is ironic that as a muscle-relaxant it only takes the edge off these symptoms most of the time and at others seems to have little effect at all. Maybe then it is having a more beneficial effect elsewhere. Just goes to show that there is possibly more to these drugs that meets the eye – or the accompanying leaflet.
Thanks for the feedback Firestormm.
Its always good to hear real life experiences after all the theorising.
another good piece Marco – I am a believer in the neuoroinflammation model for CFS.
Did you ever read this paper by my Aussie countrymen/women:
http://www.ncbi.nlm.nih.gov/pubmed/21474251
I downloaded it once but don’t have it anymore. I thought it was a very coherent hypothesis that made a lot of sense. I tried contacting the researchers a couple of times but never heard from them 🙁
they proposed using a TNFa inhibitor, and administering it by injection with the person tilted, to allow the drug to get into the brain area. There was another condition they referred to where this approach was effective.
To me its a pity I didn’t hear from them, or that nothing seems to have come from this, because I have to confess their
explanation for CFS is the most convincing I have ever heard!
Hi Thanks Matthias
Yes I have read it and have just re-read it to remind myself of the content.
Look out for a new paper from Jason et al that proposes a similar model of peripheral inflammation resulting in neuroinflammation (and cites the Annett et al paper’s proposal re the inflammatory effects of exercise in ME/CFS patients).
Two very similar models that aren’t a million miles away from what I’m proposing which I’ve summarised graphically. Unfortunately I can’t paste the graphic here but the associated notes are below :
A neuroinflammatory model of ME/CFS
Notes :
A neuroinflammatory cycle may be triggered by a variety of infective, physiological or psychological stressors in individuals with genetic of acquired predispositions.
Pro-inflammatory cytokines (potentially via attenuated clearance of extracellular glutamate by glial cells) trigger a ‘vicious cycle’ of neuroinflammation which in itself may be self-perpetuating via ‘feed- forward’ mechanisms / ‘kindling’. The neuroinflammatory cycle depletes antioxidant capacity which feeds back as a perpetuating ‘stressor’.
Neuroinflammtion may result in neural loss in the pre-frontal cortex compromising inhibitory mechanisms resulting in deficits in executive function, attentional mechanisms, working memory, sensory gating and vagal tone which in turn results in a state of autonomic nervous system ‘sympathetic dominance’. Neuroinflammation may also result in peripheral and autonomic neuropathies. Together these mechanisms result in increased pain and nociception, fatigue, exercise intolerance, postural hypotension etc. Again these effects feed back as perpetuating stressors.
Chronic sympathetic dominance is associated with reduced GABA, reduced stress resilience, reduced immune function (which may allow viral persistence) and is essentially a pro-inflammatory state increasing oxidative stress and the production of pro-inflammatory cytokines which in turn further stimulates the neuroinflammatory cycle.
Persistent sympathetic activity may attenuate the production of protective (against ROS and glutamate excitotoxicity) heat shock proteins (HSPs) – a degenerative deficit also found in ageing.
This model is potentially self sustaining at the neuroinflammation level and as a global system.
http://www.ncbi.nlm.nih.gov/pubmed/21906029
In conclusion, fatigue and CFS-defining symptoms are common in POTS patients. The majority of them met criteria for CFS. CFS-POTS patients have higher markers of sympathetic activation, but are part of the spectrum of POTS. Targeting this sympathetic activation should be considered in the treatment of these patients.
+++++++++++++++++++++++++
Interesting connection between CFS and POTS. The majority of these POTS people meet the criteria for CFS. Could this be a different presentation of the underlying same problem? How many have been tested for POTS and/or OI?
There is also a new study from Mayo on POTS and them finding an autoimmune component.
http://www.ncbi.nlm.nih.gov/pubmed/23562385
Could there be a genetic weakness that gets triggered leading to autoimmune dysfunction and then to inflammation?
Issie
http://www.ncbi.nlm.nih.gov/pubmed/18805903
BACKGROUND:
It has been suggested that postural orthostatic tachycardia syndrome (POTS) be considered in the differential diagnosis of those with chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME).
CONCLUSION:
POTS is a frequent finding in patients with CFS/ME. We suggest that clinical evaluation of patients with CFS/ME should include response to standing. Studies are needed to determine the optimum intervention strategy to manage POTS in those with CFS/ME.
Another article suggesting that CFS people may have POTS. Could it be that POTS is the problem and CFS is what those have been DX’d with – when it could be POTS? The above article shows that most POTS people meet the criteria for CFS.
My sister has CFS severely but has OI and not POTS. But, it appears that 2 of her kids have POTS and one of them OI. So, there must be a connection and also – in our family – some sort of genetic factor.
Issie
Hi Issie
I agree with the Newton team that testing for POTs should be the norm for ME/CFS patients as many of the causes are treatable but I don’t see why the occurrence of POTS as a symptom should suggest a diagnosis other than ME/CFS as POTS, OI and other manifestations of autonomic dysregulation are key findings in ME/CFS.
Is POTS a more meaningful ‘label’ than ME/CFS given that POTS is also a ‘heterogeneous’ condition?
http://www.ncbi.nlm.nih.gov/pubmed/23122672
Marco,
Good question! 🙂 Since, POTS is in the rare disease category and there are so many possible explanations —which label would be better? One thing about POTS is it can be shown and proven with science. A tilt table test can define it as being there. The dysfunction can be seen and sometimes —the appropriate bandaid – medicines – can be applied. Same as with OI – that can be found with a tilt table test and be well documented. With CFS – there are few things other than symptoms that can be shown with science. There are some markers like adenosine levels, (Here’s a thread with a link of a really cool slide show on this that has a link you can go to on adenosine. It’s a bit of a round about way to get to it – but, I think you will like it. http://forums.dinet.org/index.php?/topic/21517-cfs-fms-ms-studies-being-conducted-and-showing-genes-involved-in-connection-with-exercise-intolerance-maybe-this-would-apply-to-us-potsies-too/?hl=adenosine ) T-cell and B-cell abnormalities with CFS.
Here is a study done by Lucinda Bateman in SLC shows that she is finding OI and POTS with her CFS patients. (This is my sister’s doctor. There is a genetic component in our family – as all three of her kids have what they think is either POTS or OI. My sister has OI/CFS/FMS and I have POTS/EDS/MCAS/FMS and some autoimmune problems.)
http://www.fcclinic.com/AboutCFSME.htm
I really think that we are just looking at the same thing – but different angles – with different presentations. I feel that genetics play a big role in the problem and then the autoimmune system becomes dysfunctional and inflammation is a big player. If we work on the immune system and get it to working properly and work on what may be causing inflammation —maybe, things will get better.
One thing that I’ve thought of —if this is a family thing —what is it in the family that is consistent – other than genes? What is the one thing that is passed down that could possibly make a difference in the presentations and the “turn-on” of these genes? Is there a habit that we do or learn that might have an impact? What can affect the body and it’s function? The one thing and the one answer that I get is —–FOOD. We pass along our eating habits to our children and they pass that along to their children and if there is a genetic weakness there that something we are doing may not be so good for —–what would happen if we totally changed the way we nourish ourselves? That is the approach that I’ve taken with myself. I’ve been on a low-fat, vegan diet for 7 months now. I can see some great improvements in my symptoms and in the function of my body. It addresses the immune system too. It also has greatly improved my inflammation and lessened pain. My doctor has me addressing my immune problems with low dose antibiotics (doxy) and herbals that are antibiotic like. I’m also on GastroCrom which address the immune system in the gut. I can’t say that I’m “well” —but, I feel better then I’ve felt in years. It takes a lot of dedication to make these lifestyle changes and I’ll be the first one to tell you —it’s not that easy. But, if it will improve the quality of my life —that’s what I’m after.
Here is another report from the same doctor. If you look under “Exercise and Physiology and Energy” you will see that she is finding a connection to genetics and also Mast Cell Activation disorders and pathogens. (I also have a protozoa that is like a malaria type one cell organism and a co-infection that is connected to Lyme disease. Working on those things with diet, low dose antibiotics, GastroCrom and immune moderating herbs —I’m finding many improvements —in pain, function, fatigue, POTS etc.)
http://orwh.od.nih.gov/research/me-cfs/pdfs/ORWH_SKW_Report.pdf
Labels and DX’s of certain illness, which is better? I can’t answer that. But, pursuing other possibilities will give you another piece of your puzzle. If there is something that can be done to improve your quality of life —isn’t that what we’re all after? It’s not the quantity or length of time we live that matters —-it’s what we do with the life we have while we’re alive that matters. Improve the “quality” and that will give a happier life.
Issie
Couldn’t agree more Issie
I’d say more but I’m suffering badly in the heat here at the moment.
I’ll check out the links later when (if?) its a little cooler.
hi Marco,any thoughts on use of minocycline -which inhibits neuroinflammation – in cfs?
one other thing…fluoxetine (prozac)has been by far the most helpful treatment for me over the years.I have read a couple of recent papers that conclude that fluoxetine calms neuroinflammatio,so maybe the positive results I have had with fluoxetine are more to do with neuroinflamation rather than depression.
Hi Matthias
I thought I’d mentioned minocycline – maybe not I’m losing track.
There seems to be some dispute though whether or not it is neuroprotective or deleterious and in which conditions.
Your experiences with Prozac don’t surprise me as I’m pretty convinced that the symptom of depression is of neuroinflammatory origin.
I quite like this blog which discusses the anti-inflammatory effects of ADs :
http://evolutionarypsychiatry.blogspot.fr/2012/11/inflammation-and-depression-cause-or.html
thanks Marco, good article. I believe neuroinflammation is the key in CFS.
Annett’s excellent paper provides a good explanation as to why prozac might be acting as a useful treatment for CFS people like me. how have other people found it? As I say it is has been by far the best thing for me.
what did you think about their ideas about using a TNFa inhibitor, if I recall correctly they proposed administration with the head below the feet (eg lying down in tilted position) so that the drug gets into the brain area more easily.
I understand the Norwegian researchers are testing a TNFa inhibitor in addition to rituximab. I don’t know if they will be administering in the manner suggested by Annett et al. I wouldn’t be surprised if they find it more effective than rituximab
To be honest, I’m surprised as to the lack of testing of TNFa inhibitors in CFS. I think Dr Kerr in the UK had planned to do it.
Thanks Matthias
I feel that there is potential to reduce neuroinflammation at various stages of the vicious circle including tackling those proinflammatory cytokines consistently found in ME/CFS (IL1-b, IL6 TNF-a) or those up or downstream including COX-2.
As Issie posted below for example, a flavonioid found in celery may reduce neuroinflammation in multiple sclerosis by downregulating microglial production if IL6. On the other hand, as Issie’s article also states, inflammation is also protective and is only detrimental in excess. I don’t have the reference to hand but I did read recently that inhibiting TNF-a in MS patients may actually increase disease progression. So much more research would be needed for ME/CFS even if the principle was sound.
I do look forward to hearing what Fluge/Mella find with etanercept and like yourself I wonder why ‘low level inflammation’ is a fairly well accepted finding in ME/CFS but little done to investigate how reducing it might impact on symptoms.
Anecdotally I had a one-off but striking reaction to one of my wife’s meds that had been lying in the cupboard for many years – Celebrex which is a COX-2 inhibitor.
I’d taken one tablet as an anti-inflammatory as I had a pulled tendon in my neck and without expecting it I found that my long standing general anxiety was gone. Not just reduced but gone as if it had never been there. Unfortunately I couldn’t repeat the effect.
So yes, I very much agree that drugs such as ADs may be very helpful far beyond any anti-depressant effects or their proposed impact on serotonin.
Nice article – inflammation plays a big part in many functions of the body.
Since this was noted by Marco above in his blog and I ran across it in my saved files, thought I’d post the link to the article by Blaylock. There are lots of suggestions on inflammation and alternative treatments for it.
http://www.surgicalneurologyint.com/article.asp?issn=2152-7806;year=2012;volume=3;issue=1;spage=19;epage=19;aulast=Blaylock
Issie
Anyone, like celery? Here’s a great article on Luteolin and it’s affect on inflammation.
http://www.science20.com/news_releases/luteolin_flavonoid_in_celery_may_impact_multiple_sclerosis
Issie
Hi Marco
I read an article in New Scientist today about evolving thoughts on the causes of depression, thinking seems to be that glutamate might have just as much of a role as serotonin. Interesting given the theories on glutamate and CFS.
Even more interesting was the mention that a drug called ketamine can positively affect glutamate imbalance.
Any thought on this and its potential use in CFS?
Thanks for the heads up Matthias
Major depressive disorder and bipolar disorder were two conditions I suggested might be linked in the first blog in the series and I discussed neuroinflammation and the ‘glutamate hypothesis’ of depression in the second blog :
http://www.cortjohnson.org/blog/2013/02/15/glutamate-one-more-piece-in-the-chronic-fatigue-syndrome-mecfs-puzzle-the-neuroinflammatory-series-pt-ii/
“Stablon (tianeptine) is an atypical antidepressant that has demonstrated remarkable clinical effectiveness in major depressive disorder that is often resistant to treatment with traditional SSRI’s. The interesting fact about tianeptine is that its efficacy does not conform to the monoamine hypothesis of depression as tianeptine may actually lower serotonin levels.
An investigation into the neurobiological properties of tianeptine (McEwen et al, 2010) concludes :
“Converging lines of evidences demonstrate actions of tianeptine on the glutamatergic system, and therefore offer new insights into how tianeptine may be useful in the treatment of depressive disorders.”
Intriguingly the antidepressant and anxiolytic effects (plus protection against cognitive defects) appear to be due to its ability to protect the brain (particularly in the amygdala and hippocampus) against stress induced glutamate excitotoxicity :
“A modification of glutamatergic mechanisms by tianeptine may therefore be implicated in its ability to oppose the negative influence of chronic stress upon hippocampal neurogenesis, cell proliferation, and dendritic remodeling, processes profoundly disrupted in depressive states”
Similarly ketamine has a rapid and sustained antidepressant effect through modulation of glutamate neurotransmission as does the novel compound GLYX-13 which has the same beneficial effects without inducing dissociative symptoms usually associated with schizophrenia (Burgdorf et al, 2012)”
thanks Marco.
So I want to see some of these drugs tested in CFS.
As people have said elsewhere, enough of the theory, time for action!
Yeah – me too Matthias.
This article might also be of interest :
http://www.uab.edu/news/uab-magazine/item/3237-mr-excitement-what-glutamate-can-teach-us-about-depression-schizophrenia-cancer-and-more
Really enjoyed that article Marco. I feel that so many might could benefit from looking into glutamate as a possible cause of their dysfunction Thanks for the link!
One thing of interest, Tramadol works on so many levels and also works on NMDA. I was not successful with any of the SSRI’s or SNRI’s – Tramadol works on so many things (NMDA, opiate receptors, NE, Serotonin, Dopamine). They are not sure exactly how it works. But, they are using it off label for some that those other meds did not work for. My main reason for using it was for pain due to Ehlers Danlos (EDS). But, found that it benefited so much more, it’s been one of my best POTS meds. In POTS one of the treatments is an SSRI – which many do not do well with. That’s one reason I started my research on glutamate – because I felt that they needed to look at things from a different angle. I do believe that this may be one of the parts that is missing.
Issie
Tramadol eh?
“Tramadol works on so many things (NMDA, opiate receptors, NE, Serotonin, Dopamine). They are not sure exactly how it works.”
Isn’t that the truth!
Can’t help but think you have more ideas about Tramadol. Have you tried it?
It shouldn’t work for me. Since it would make NE hang around longer and my NE levels are too high with standing. But, that makes me think that NE is a compensation and not the enemy. Maybe NE isn’t going where it needs to go or being uptaken correctly and something about Tramadol helps with that. Or is it needing to have NMDA tweaked or is it something to do with opiate receptors? That’s my one —mystery —that I can’t seem to figure out. So far, no one has been able to help me sort that one. But, I guess, I should just be glad I found something that helps – even if I can’t figure out WHY.
Issie
No Issie I haven’t tried it and don’t know much more about it than you’ve described.
I could try it but I’ve seen a few comments on the net that it can increase anxiety levels for a few hours before any ‘euphoric’ effects kick in. As I’m not looking for any ‘euphoric effects’ or even pain relief and the last thing I want if heightened anxiety it doesn’t sound ideal for me.
I’m glad its helping you. it would of course be nice to know why but with so many putative effects on various neurotransmitters its going to be a messy one to figure out.
PS – Cort has now added the graphic of my neuroinflammatory model to my profile page :
http://www.cortjohnson.org/authors/marco/
I don’t get anxiety –AT ALL. I take it and it calms down anxiety. That’s where the paradox thing comes into play. I don’t get euphoria from it either —-sigh —-a little of that would be nice. But, what it does is calms down my overactive sympathetic system that causes a type of anxiety feeling and tachycardia that goes along with too high levels of noriepi (NE) that is the result of hyperadrenergic POTS. All of that settles out.
Ha! You’re not kidding – I can’t figure it out and I’ve looked at it from every direction I can think of. Had one of the smartest people I know, look at it with me and he couldn’t figure it out either. So, I’m down to just being thankful and glad that I found something that helps and putting the need to know WHY on the back burner.
Nice graphics – I want to look closer at it and analyze what you’re thinking. Thanks for the heads-up.
Issie
Its such as you learn my mind! You appear to grasp so much approximately this,
such as you wrote the book in it or something. I feel that you just could do with some percent to pressure the message
house a little bit, however instead of that, that is wonderful
blog. A fantastic read. I’ll certainly be back.
Thanks
You’re right.
I don’t spend enough time summarising and thinking of presentation and making things more accessible.
Not really my thing I’m afraid.
Hello and thank you.
Yes, liver support is necessary for me to reduce irritability/overwhelmed symptoms.
Also a dental mouth guard for the clenching from stress overload.
Gut balance is an additional aspect for brain health.
So these things help reduce pain or anxiety, cutting back on carbs and sugar, magnesium, B vitamins.
Gaba/glutamate balance is important.
Therefore, I now seek balance not stimulating products, energy producers or overwhelming people or stressors.*