



Engineering Better Health With Low Dose Naltrexone
One Size Fits All Approach Fading
“ We envision that the concepts presented in this paper will lead to novel individualized treatments where treatment dosages can be assigned by a mobile device at the disposal of the participant.”
Dr. Jarred Younger’s earlier work on Chronic Fatigue Syndrome that matched daily symptom and cytokine levels suggested, for the first time that leptin levels were playing a major role in ME/CFS. The kind of intensive analysis of daily shifts in symptoms and health had never been done before in ME/CFS, but it could be what future medicine looks like.
In a recent paper focusing on Low Dose Naltrexone and Fibromyalgia, Sunil Deshpande PhD, Younger and colleagues argue that this kind of intensive, individualized approach should guide not just a doctors decision regarding treatments, but a patients decision how to administer them. If it takes off it could revolutionarize how disorders are treated.
Medicine has taken a pretty standardized approach to illness. A doctor diagnoses something, looks up in his/her reference tables for the right drug and the standard dose of that drug and we’re given a prescription that says something like take x drug once in the morning every day for a month.
These researchers think people are far too complex for that to work really well particularly for people with chronic illnesses. It’s time to get personal – really personal – when treating patients.
In this system the one-dose fits all approach is replaced by a dynamic process that takes note of the daily fluctuations in a persons health and adjusts the dose of a drug or other treatment to them. This approach is simply one more aspect of an increasing trend towards more personalized health approaches.
Engineering
Engineering models are getting around. We sew Gordon Broderick use engineering methods to produce endocrine and immune models of Chronic Fatigue Syndrome and Gulf War Syndrome. Now, Deshpande and colleagues including Dr. Jarred Younger have used engineering methods to build a personalized approach to Fibromyalgia using low dose naltrexone.
They gathered an “intensive” amount of self-report data on things like general pain symptoms, sleep quality, stress, anxiety, mood, etc. during an earlier trial of low dose naltrexone in Fibromyalgia.

Medicine is beginning to move to the place where people with chronic illnesses may be able to programs to daily determine the best treatment doses for them.
Every day the participants entered the answers to twenty questions such as “How did you sleep last night” or “Overall how severe have your FM symptoms been today” into a hand held computer. Other endpoints included fatigue, sadness, satisfaction with life, overall sleep quality, trouble with sleep, ability to think, headaches, average daily pain, highest pain, and gastric symptoms. They also assessed stress, anxiety and mood.
They then used a data-based modeling engineering technique called system identification to produce models that explained changes in pain and other symptoms.For example, one FM patients chart indicated a dramatic drop in their FM symptoms and a dramatic improvement in sleep, no change in anxiety levels (which were already low) and occasional large changes in mood, once they began taking the drug. Looking at this chart, which could be uploaded to the doctors office, a doctor could immediately see where and how effective a drug or treatment input is. .
They then used model predictive control (MPC) technology to produce algorhythms that suggested optimal doses of LDN to take based on a persons symptoms as well as outside events (such as stressors and anxiety)
“We illustrated how the controller can assign treatment dosages to reach a desired set point target and then maintain this goal under conditions that involve disturbance changes in symptoms (known and unknown).”
They found they were able to produce day by day alterations in LDN dosage to maximize it’s affects. All you apparently need to run this program is a hand-held computer containing the right program.
In this way, an optimal dosage profile for that individual could be rapidly determined without requiring office visits or substantial clinician involvement.
There was no intimation that it’s possible to purchase this system now. The fact that it was done using LDN data for Fibromyalgia, however, suggested LDN works very well indeed for some FM patients and provides hope that an optimal system for Low Dose Naltrexone may be one of the first to become available.
This is individualized treatment in a never before thought of scale. If this approach proves to be effective it could greatly increase treatment effectivess.
N=1 Trials
So called N=1 (one person) trials that track symptoms, medications and other factors using smartphones and other devices in order to determine more effective treatment options are expected to become an increasingly important way of managing one’s health.
Of course, good doctors have been doing their own N=1 trials for decades, as they switch from one medicine to another or one combination of medications to another in their patients.
Many doctors in our fast-paced medical system, however, simply don’t take the time to examine all the possibilities. A recent Discover article related an n=1 trial of on asthma patient which found out that one of the three asthma medications prescribed was actually harming the patient. Once they removed that drug the other two drugs worked. The drug they removed was a good drug – just not for that patient.
Getting a drug that may be bad at least for some people, is a predictable outcome of the traditional drug trials that determine drug effectiveness for wide groups of patients – not individuals.
Until recently, antiquated IRB rules had made N=1 trials virtually impossible to do in a research setting, but that’s changing. The first large-scale N=1 trial using an adaptive approach to determine the best treatments options for a patient is underway.
Free myLDN Treatment App Now Available!
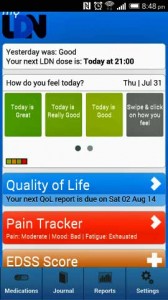
Talk about tracking your results for better health – a free LDN app has just been released by the LDN Research Trust that will assist you in doing your own N=1 LDN trial. The app, which works on all platforms, and has been under development for several years, is able to track the effects of LDN (plus other medications) on symptoms, diet, exercise, supplements, mood, quality of life, pain, etc.
Among other things the app will allow you to
- Set alarms to remind you when to take your LDN and other medications
- Track Diet items which might have affected you
- Keep track of any Exercise you have done
- Keep track of any Medical Aids you might have used
- Specific questionnaires for Multiple Sclerosis, Fibromyalgia, Chronic Fatigue Syndrome and Spinal Cord Diseases are presented to users with those conditions – more questionnaires for other conditions will be added over time.
- Keep a Journal of how you are getting on
- View Graphs of the data entered
- Create PDF reports of your data for printing and taking to your GP
- Receive Notifications from LDN Research Trust direct to the app
Download the free app below.
- Android download
- iPhone download
- Window Phones download
- PC’s and Laptops download
- Kindle download link:
LDN Research Trust Conference – Nov 7th to 8th, Las Vegas
This conference, which will be streamed live for $25, will feature experts in the LDN field presenting on a variety of topics. People who participate via live streaming will be able to ask questions during the interactive periods that dot the conference.
The LDN field, of course, has a strike against it; because LDN can be manufactured in any compounding pharmacy most drug companies aren’t interested in producing a drug formulation of it, and that means more difficulty getting clinical trials underway. The field, though, is picking itself by it’s bootstraps. Dr. Lev will be presenting a Medication Usage Evaluation project he will use to assess LDN’s effects in doctors practices worldwide. The LDN Research Trust has also just released an app that allows patients to fine-tune their use of LDN and document its effects.
We’ll also hear how the one drug company interested in LDN, TNI Biotech, is doing in its efforts to produce a clinical trial for a low-cost patentable version of LDN. The FDA’s designation of LDN as an “orphan drug” for the treatment of pediatric Crohn’s disease, is extremely helpful as it requirements for the approval of orphan drugs are less burdensome.
Last year TNI Biotech announced that it had acquired the patent rights from the several researchers for LDN treatment for ulcerative colitis and Crohn’s disease and stated it expected to begin talks with the FDA to begin Phase III trials in these disorders. (Phase III trials are the major last roadblock before FDA approval.) TNI Biotech also expects to test LDN’s effectiveness in other immune mediated disorders including several autoimmune disorders.
Conference presentations of special interest to the FM and ME/CFS communities will focus on LDN’s effects on pain, two talks by Dr. Kent Holtorf on Chronic Fatigue Syndrome and Fibromyalgia and thyroid disease, and LDN’s effectiveness in treating small intestinal bowel overgrowth (SIBO). Along the way we’ll also get talks on how LDN works and how to use it in clinical practice.
Check out the conference overview here.
Prescribing LDN
Need help getting your doctor to prescribe you LDN. The LDN Research Trust has helped over 15,000 people to obtain LDN from a General Practitioner or Neurologist, either through the National Health Service or by private prescription in the UK as well as in urope, as well as in the USA, Canada, West Indies, Australia, New Zealand, and others. Find out more here.
- Check out Health Rising’s Fibromyalgia and Chronic Fatigue Sydnrome Low Dose Naltrexone Resource Page for more blogs and information on LDN









several links on the LDn website don’t work. When I tried to contact one of the prescribing doctors previously (link now broken) they didn’t reply.
I’d like to try LDN but it’s not easy….
Hang in there Tatt. I encountered the same thing only to have the links work later. Something funky is going on with their website…