Of all the disorders commonly allied with Chronic Fatigue Syndrome and Fibromyalgia, chemical sensitivity or Environmental Illness (EI) probably gets the least respect and certainly gets the least funding. It was astonishing to hear Dr. Clauw, an FM doctor and researcher who has battled disparaging attitudes toward Fibromyalgia for decades, make a belittling comment about environmental illness at a Pain Symposium.
That’s the way it often goes, though, for Environmental Illness, a condition that gets little research funding. EI is not just associated with ME/CFS and FM but with PTSD, depression, anxiety, and asthma as well. Unlike ME/CFS and FM, it’s not clear whether mood disorders precede or follow EI, but if EI follows its counterparts (and why wouldn’t it?), mood disorders will be found to be a result of rather than a contributor to EI. Right now EI with its poor funding is open to all sorts of interpretations.
Environmental Illness: A Short Overview
In the “Allergic To Life” story in the Nov 2013 issue of Discover magazine, Jill Neimark asked EI expert Claudia Miller what she thinks causes chemical sensitivities. Her answer indicated she believes EI is a sister disorder not just to Chronic Fatigue Syndrome and Fibromyalgia but to disorders like epilepsy and chronic regional pain syndrome (CRPS).
She presented two models of EI:
The One Trigger Model
In the one trigger model an intense exposure “tilts” the central nervous system into over-reacting not just to that substance but to other substances as well. This general process – a single stressor that triggers a hypersensitive response to future stimuli – is seen in Fibromyalgia, but is most dramatically demonstrated in CRPS, where a small injury triggers excruciating pain and immune changes that spread from the site of the original injury.
The Intermittent Trigger Model
The intermittent stimulation of the limbic region of the brain that explodes into an epileptic seizure may be another model for EI. In this model, toxicants traveling straight from the nose to the limbic regions of the brain may, in susceptible individuals, trigger a state of limbic system hyperactivity.
Interestingly, a study of genes involved in detoxification suggested that problems detoxifying the offensive chemicals was probably not the issue. Dead ends in immune and olfactory studies have pointed to activation of a highly sensitive and excitable limbic system as a prime suspect.
[We should note, though, that Dr. William Rea, an environmental illness specialist in Dallas, has successfully used a combination of desensitization (allergy shots), detoxification (sauna) and immune factors to treat EI as well as some people with Chronic Fatigue Syndrome.]
Amazingly electroshock therapy worked for one severe EI patient. Other practitioners such as Dr. Rea use less “shocking” methods..
The idea the EI may be a central nervous system disorder was buttressed in the dramatic fashion when a person with almost off the charts sensitivity (QEESI score of 95/100) returned to near normal levels after he participated in electroshock therapy (ECT). By the third treatment his EI score was 30/100. The shock therapy apparently allowed his central nervous system to reset itself.
The parts of the brain that odors activate in people with EI read like a short list of brain regions associated with ME/CFS and FM: the insula, hypothalamus, thalamus, prefrontal cortex, somatosensory cortices and the limbic system. That suggests perhaps chemical sensitivities may be to people with EI what pain is to people with Fibromyalgia: different stimuli, but a similar overactive brain response.
Are chemicals and pain tweaking the brain in similar ways in EI and FM? A Swedish study used a functional MRI (fMRI) to examine how the brains of women and healthy controls responded when presented with very low levels of odor in women.
Let’s see what they found out.
The Study
Brain responses to olfactory and trigeminal exposure in idiopathic environmental illness (IEI) attributed to smells — An fMRI study. Linus Andersson, Anna-Sara Claeson, Lars Nyberg, Berndt Stenberg, Steven Nordin. Journal of Psychosomatic Research, November 2014
The bar for entrance into this Swedish study was not set particularly high. It was a women-only study because, like FM and ME/CFS, women are much more likely to have EI. The selection criteria said participants had to report that smells that didn’t bother others tended to bother them, and they had to have two symptoms at least once a week for the past three months. Twenty-five odor-sensitive women and twenty-six healthy controls participated.
As they were given increasing doses of a banana smelling odorant, a functional MRI (fMRI) assessed their brain activity. An odorless trigeminal nerve stimulant was also applied. The doses were very low – lower than had been assessed in EI patients before.
The Findings
“The lower BOLD signal in the prefrontal cortex and higher signal in the thalamus in the IEI group may thus be interpreted in terms of an inability to inhibit salient, attention grabbing, and arousing stimuli.”
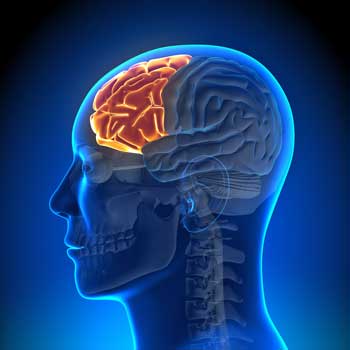
The very low doses of the banana smelling odor caused the thalamus to spring into action in the environmentally sensitive group and knock down activity in a portion of the prefrontal lobe (the superior frontal gyrus) compared to the healthy controls. Since the thalamus processes and relays sensory stimuli between the lower and upper parts of the brain, the increased activity there suggested the thalamus of the EI-sensitive participants may be hitting the prefrontal cortex with a barrage of stimuli.
The prefrontal cortex is known for its importance in planning complex actions and decision-making, but it also processes sensory stimuli. In a recent hypothesis paper Japanese researchers proposed that damage to the prefrontal cortex (DLPFC) played a major role in ME/CFS including difficulty processing sensory inputs (physical sensations, over-stimulation), causing high emotional lability and problems with attention span and working memory.
The reduced activation of the prefrontal cortex during the odor test could reflect a similar situation, and indeed, anybody with significant chemical sensitivities will recognize the emotional lability, difficulty concentrating, and short-term memory problems that occur after being exposed to too many chemical odors.
The two findings suggest people with EI may have a thalamus that pounds the prefrontal cortex with chemosensory signals when they’re exposed to chemicals, and a prefrontal cortex that lacks the resources to process them properly. Ever feel like you can’t get “outside” of or push aside chemical odors? That you can’t concentrate in the face of them? The kind of reduced prefrontal cortex activation seen in this study has been associated with an increased focus on external stimuli.
The PRC – the seat of executive functioning – should be mostly helping us decide, plan, and organize our day. But if your PFC is getting hijacked by “chemosensory” signals, it may not have the wherewithal to make decisions or plan your next moves.
Fibromyalgia, ME/CFS, and EI
Could a similar process be occurring in Fibromyalgia and Chronic Fatigue Syndrome? With regards the prefrontal cortex – possibly. FM imaging studies often show enhanced activation of pain processing regions such as the somatosensory cortices, ACC, and insula in conjunction with decreased activation of the prefrontal cortex. A study finding “weaker coupling” between the pain regions of the brain and the prefrontal cortex in FM suggested poor control of the pain circuits was present. Another study found that the prefrontal processes healthy people use to reduce pain were disrupted in FM.
No studies I was able to find suggested that increased activation of the thalamus plays a role in FM. However, increased levels of neuroinflammation in the thalamus of ME/CFS patients was found to be associated with increased pain levels in ME/CFS in the recent Japanese neuroinflammation study.
Is the PFC a weak link in FM, Environmental Illnesses and ME/CFS? Time will tell.
Klimas Group Opens Environmental Health Program

This is, to my knowledge, the first ME/CFS research center to explicitly focus on environmental illness and ME/CFS. It’s a welcome addition.








Thanks for writing about this topic, Cort.
Here is a new page on the Paradigm Change website, on the ability of mold toxins to cause psychiatric symptoms.
http://paradigmchange.me/psychiatric/
I have no doubt that other types of toxicity can have effects on mood too, but biotoxins (such as mold toxins and Lyme toxins) do seem to be particularly good at it.
Lisa Petrison, Ph.D.
Executive Director
Paradigm Change
http://www.paradigmchange.me
Lisa,
I have a friend with severe chronic Lyme. In addition to the challenges of depression and anxiety, she has had symptoms of psychosis as well as Austic-like repetitive behaviors such as rocking for hours.
Laura
oops — I meant “Autistic”
I am medically unfit for work for the last twelve years and one of the reasons is M.C.T.D, S.L.E and Fibromialgia. I suffer with the worst body aches,pains and discomfort. I find to help my skin to get rid of the “prickly pear,bobwire” feeling i get undressed. It help a great deal for my pain,is there anybody out there who got Fibro that tested this theory,I find this a lot better for me and to have a less painfree life. There is not a great deal people who really want to talk about this,but it works for me. Help please
I’ve found that it’s an allergic reaction to something being consumed. I’ve found since I have fibro, mayo facia, I’m now allergic to soy, gluten, dairy. It gives the pricky itching feeling wear no garments feel comfortable. I also eat organic due to GMO in the food. I found it decreased allot of pain & if I eat sugar it too effects my body with weird sensations and my body feels fatigue. I’d try elimination and also if you’re taking vitamins their is soy highly toxic one of the leading GMOs. Google GMOs too many pesticides in the food. Creates stimuli in the body and nervous system. This my own personal experience and not ink aged by doctors it’s change my illness in a positive direction.
Hope this helps.
There are a few small studies from almost 20 years ago that showed a decreased rCBF to the thalamus in FM, indicating reduced neural activity at a resting state compared to controls. One of the studies actually divided patients into two groups -traumatic onset and gradual onset. Both groups had reduced rCBF to the right and left thalamus but the gradual onset group also had reduced rCBF to right and left heads of the caudate nucleus.
Thanks Tricia – good to hear 🙂
Spot on Cort.
Regardless of anything else it’s clear to me that in MCS/Environmental illness and ME/CFS our brains are over-reacting to everyday what should be harmless stimuli (potentially also for FMS although small fibre polyneuropathy may suggest that at least as far as pain is concerned FMS patients pain sensations are not innocuous).
Either a big knock (viral onset) or a number of smaller nudges (by a variety of means) have sensitised the brain – potentially via the brain resident immune microglia. Frontal lobe dysfunction and ANS dysfunction follow on from this I believe.
Very interesting report. I got a fealing that one day researchers will find that PTSD,GULF WAR SYNDROM, FIBROMYALGIA,ME-CFS could all be tied together some how.Before becoming bedridden after contracting. Epstein Barr Virus at age 48 i had been suffering with fatigue,and joint and muscle pain and Sever Chronic Sinusitis since 1999.I was a professional auto painter and have been spaying cars,trucks, busses,military tanks and military vehicles and equipment for 33years. I often wondered if the heavy exposure to issocynate,laquers,synthetics, urathanes,polyurethanes and everything under the sun in automotive chemicals had anything to do with why i am suffering from sever Chronic Fatigue Syndrome and Fibromyalgia.Back in the 1970s when i started my 33 year career in Automotive Painting and Autobody and Frame alignment we did not wear any protection. We did not wear respiratory protection. and body shops did not have air supplied respirator and suits.I was exposed completely untill it was mandatory for shops n 1980.I really believe that their is something to this Environmental Illness. I know people from all walks of life in all different kinds of careers have Fibromyalgia and ME-CFS.I pray for the day that research will find a cure.
Boy were you exposed to a lot of chemicals. Paint is the one of the worst substances for me now. I remember Dr. Rea saying he got a lot of people from the paint industry in his practice.
I used to work at a vocational school and couldn’t stand the toxic smells those women worked with every day when I had to go to the cosmetology shop. I always wondered if they had more cancer or neuro illness than the mean.
Hi Cort. Thanks for mentioning my article. Seeing that you provide active links, it would be great if you could actively link to the Discover piece when mentioning it. I also covered the topic more recently in Nautilus, here:
nautil.us/issue/15/turbulence/is-the-world-making-you-sick
I personally don’t think fibro and EI are linked tho they may overlap in some people.
There is also good research by Beatrice Golumb, at UCSD, on the 1990-1991 gulf war vets, who were uniquely exposed to organophosphates–and they are the GWI sufferers. Check out her research and recent study on CoQ10 helping them.
My apologies Jill. I actually didn’t know it was online. I found it in a Discover magazine that I picked up in a laundromat believe it or not, and should have looked to see if it was online as well. It was nice to see EI covered in a such a empathetic way.
I put the link in and here it is as well – http://discovermagazine.com/2013/nov/13-allergic-life
Thanks. The Nautilus story gives further insight into Dr. Miller’s thought process and work, as well. The Nautilus story actually got more attention–and from a more intelligent audience, I think. The comments on the Nautilus story were fascinating.
I hope the copy of the laundromat magazine wasn’t covered in fabric softener! 😉
🙂
Very interesting reading the last posts.. I was diagnosed with PTSD about 5 years ago (though already had it for 5 before that!) along with fibromyalgia/CFS and chronic anxiety and depression. I believe I may have been suffering with EI also as have been very sensitive to pretty much all chemicals I would notice on exposure to them.. so much so that at my worst, just spraying a disinfectant for work surfaces near me could make my skin start burning.. Also seeming to be multiple food sensitivities.. Better now.. ? Not sure about sensitivities.. I eat as much organic as poss, and make very natural soaps shampoos etc myself, avoid E numbers obviously.. I seem to have to still watch it re muscle pains and soreness which can occur in my body if I over do it in the direction of coffee.. (more than a few a week)tea? too much work, too little good nutrition. too little good quality water, possibly too much wheat and sugar.. definitely inactivity.. best to exercise 3/4 times a week and something every day..
How have I got myself better.. Chanting Nam Myoho Renge Kyo (got rid of CFS) detoxing as much as possible.. Clearing out my colon (colonics), doing liver flushes (Andreas Moritz) (gave up on this one after 5-6 as very severe.. Juicing, doing juice feasts.. up to 3 days,, cutting down on acidic foods (more lately this is my tactic if I have overdone things) eating alkalising foods.. High vegetable content and plenty of sprouted food.. feels better almost immediately alongside juicing mostly veg.. Making sure I have enough Magnesium (use a transdermal spray if needed) the odd Epsom salts bath is also good if feeling toxic.. Other techniques I have used whilst detoxing are oil pulling.. to pull out toxins out of the bloodstream and above all one which really works on improving overall health and wellbeing is Tuning Fork Sound Therapy (Suara Sound Academy).. I qualified as a therapist this year.. after trying everything to get back my focus and attention span… It is the only thing that has worked (Clinical studies of Meditative states.. which you go into whilst using them/or being worked on by a therapist with them have been shown to turn off stress genes, and thereby improve overall health)… I do believe I am not yet fully at pre illness capability (though now well in my 40’s.. so that has some effect!), but believe it’s not that far off now.. I experienced all those things with the brain getting fatigued and getting worse if no rest was had… I sat an exam! last week which I had tried and was incapable of even studying effectively for in previous years… I felt my brain was a bit strained..but I did it.. and feel things are only improving now… My memory was appalling still even a year ago, (predominantly very short term memory).. This made my part time job difficult (Could only work part-time then.. building up now towards full time) Now it is great..have they odd difficulty, but it seems so do a few people my age!
I am now a Suara sound tuning fork therapist which is amazing.. the lady that taught my had regained 70% health from ME using EFT.. the rest she regained as a result of tuning fork therapy.. My percentages are the same.. except I would say up to 70 % regained from my own efforts (long slow process though.. predominantly using natural therapies.. various complementary therapies also.. plus other things I mentioned above. I was already trained in a few of them so had a lot of knowledge of how they can work..
I also worked with a lot of chemicals through work (in a microbiology lab.. used a fair amount of solvents without gloves, plus in a fingerprint lab,, exposed for a while to ninhydrin plus other chemicals…) Wonder if this did have any effect or merely coincidence! for many years have found it hard to even walk through a perfume department!…. Hope this might help someone… I believe our bodies have everything they need inside them to heal themselves… just need the right diet, and treatment (Mental, emotional, spiritual etc)
Wow I came across these post’s tonight by reading on proHealth web site.. I am not really Very good at been on computers yet!! Don’t know how to use them properly.. But learning a bit now and again.. I have server FM/ CF/Me chronic anxiety and depression and now pain in face, gums and jaw” and loads of other things that come with it all.. I was diagnosed with severe fibromyalgia 8 years ago.. But could of had it mildly 14 years and not realised there was some thing going on.. Well not as bad as I do now. I can remember going to my Gp all them years ago and telling him how tiered I was feeling and having to lay down and sleep during the day. Which is some thing I never did at that time.. He replied well now your 40 it’s all down hill for you.. You will get ache and pains now.. But like a few of you have said on the above post’s.. I also did a lot of paining and decorating for a living painting inside houses so I was breathing in Chemicals the same as some of you was.. It does make you think if that is some of the cause of my illnesses.. I am very sensitive to spray’s and smells now!!. I have found all your post very interesting I am so pleased we are getting more up to date research done now for our Horribly Painful illness and other problems that go hand in hand with it that we all Have to live with.. I hope I can find this research page’s again loved reading them and all your post’ I don’t like to say it but I have crap doctors that don’t care enough to learn any research or any thing about fibro so I am doing it myself and when I do find the energie and health to go visit them again will take it with me.. Take care everyone xx
For over five years I suffered from Chronic Fatigue and Fibromyalgia. I could not stand the smell of gasoline, perfume, finger nail polish, household cleaning agents, etc. I overcame chronic fatigue with an alkaline diet, but recently discovered that I have MTHFR. a genetic mutation that prevents the liver from detoxifying, It is corrected by taking methyl B-12 and methyl folate. Since then I have found that many people with MTHFR have chronic fatigue and fibromyalgia.
MTHFR explains why I had chronic fatigue for years, then liver cancer, why I am so sensitive to smells like perfume and gasoline, why when years ago I was depressed and my doctor gave me Prozac I felt suicidal. People with MTHFR cannot take some medications because the liver cannot detoxify them. It explains why I am allergic to all pain medicine and antibiotics.
I wish I had known this twenty years ago.
Here is a link to a doctor speaks on MTHFR.
http://www.methyl-life.com/a-doctor-explains.html
Patty Butts, Ph.D. Holistic Nutrition