



“The immune system has to think on its feet” Mark Davis

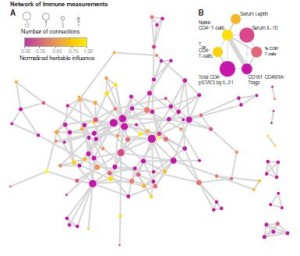
Davis and his team measured more than 200 immune-system components in 210 pairs of twins. They found that three-quarters of the time, nonheritable influences (previous pathogen or toxin exposure, vaccinations, diet, dental hygiene) determined more that 50% of the immune changes between the twins. Startlingly, almost 60% of the immune changes found between the twins were determined almost entirely by the different factors they’d bumped into in their environments. .
To put it simply, with regards to the immune system what you do and what you interact with matters more than who you are. In the nature vs nuture argument, the winner is nuture.
Age Matters
That pattern strengthens with time. The longer you’re on the planet, the more distance your immune system puts between its genetic roots and the way it functions. In retrospect, this only makes sense. No other system in the body places such a premium on adaptability. Not only are pathogens all around us but they can evolve at a tremendous rate. If our immune systems can’t counter that diversity by adapting we’re dead ducks.
The larger than expected role the environment plays in shaping our immune response was unexpected. What was really shocking, though, was how large a role one familiar group of viruses plays.
Herpesviruses Are Major Agents of Change
The biggest agents of immune system change were pathogens. One herpesvirus, in particular, demonstrated a remarkable capacity to re-engineer the functioning of much of our immune systems. Davis found that twins exposed to cytomegalovirus had vastly different immune systems than twins who had not been exposed to it.
Note that both twins were healthy; the twins with CMV were clearly successfully controlling the virus. Their immune systems, though, had shifted dramatically, in order to keep it under control. Almost 60% of the immune factors measured in the infected twins were different as a result of their exposure to CMV. The study demonstrates the power that even latent viral infections can have.
It also brings to mind the immense changes in immune networking Dr. Broderick and Klimas at the Center for Neuroimmune Medicine have found in ME/CFS patients. Broderick’s proposed that the more complex immune networks he’s found in ME/CFS patients could be driven by a infection. That jibes well with the more complex immune systems found in Davis’s CMV exposed twins as well.
“We suggest that repeated environmental influences like herpes viruses and other pathogens, vaccinations, and nutritional factors cause shifts in immune cell frequencies and other parameters and, with time, outweigh most heritable factors.”
The herpes virus most commonly associated with chronic fatigue syndrome, Epstein-Barr virus, wasn’t highlighted in the study. That’s not surprising given the fact that almost everybody has been exposed to it. Time of exposure may be a more relevant factor regarding EBV, however. Being exposed to EBV during childhood usually results in few or no symptoms, but being exposed to EBV during adolescence often causes a protracted case of infectious mononucleosis. The Davis findings suggest a major and perhaps prolonged shift in immune functioning could occur during that period.
That’s intriguing given the anecdotal reports suggesting high rates of past infectious mononucleosis episodes may be present in the ME/CFS community. If that’s true, it’s possible that the immune shifts occurring during that time increase the risk of ME/CFS either then or later in life. Some researchers believe late exposure to EBV sets the stage for the appearance of autoimmune disorders decades later.
Davis’s findings may also fit well with those of the recent Chronic Fatigue Initiative Lipkin/Hornig ME/CF study. That study found a strong immune signature in early duration but not later duration ME/CFS patients. It appeared that a common factor such as an infection produced profound and similar changes in ME/CFS patient’s immune system functioning early in the course of the disorder. Whatever the factor or factors were, they brought the immune systems of people with ME/CFS into alignment for a period.
As time went on, though, and new challenges showed up, that distinctive immune signature wanned. One person came down with the flu, another received a vaccination, another changed their diet. As each challenge produced new riffs in immune system functioning, the strong connections in the group faded.
Caveats
Some caveats are present. Davis studied the immune makeup and genetics of healthy twins. It’s possible that some of the adaptive machinery of the immune system has broken down in immune-mediated illnesses leaving genes to play more of a role than they would otherwise. We know that genes do play a significant role in autoimmune illnesses.
The few heritability studies done in chronic fatigue syndrome, and/or fibromyalgia suggest that higher degrees of heritability may be present in ME/CFS and FM than in most diseases.
Davis’s findings don’t suggest genetic makeup doesn’t play a role; it’s an important component – just not as important as once thought, and it’s importance wanes over time.
Innovator, Disrupter and ME/CFS Researcher
“For the mice in the audience, I have wonderful news!” Mark Davis
Davis is quite comfortable in his role as the disrupter of received wisdom. Mouse models have guided immunological research for decades but in a 2012 editorial Davis excoriated the immunological world for using out-dated models that poorly reflect human immune functioning. The technology is now available for human immune modeling to step forward.
It was just a couple of years ago that he was asked to create an Institute that would bring together three isolated communities. The immunology, infectious disease and transplant communities have a heck of a lot in common – but which rarely talk to each other.
Mark Davis Phd on the Center for Immunity and Infection
His ultimate goal at what became the Institute for Immunity, Transplantation and Infection is to be able to produce simple tests that portray the health of the immune system and guide doctors decisions. Davis hopes to develop tests that can pick people out before they get autoimmune disorders, or cancer or perhaps ME/CFS – and develop protocols to help stop them from getting those diseases.
First, though, he’s going to have to figure out what a healthy immune system looks like – a surprisingly difficult task.
“If a patient were to ask me, ‘How’s my immune system doing today?’ I would have no idea how to answer that, and I’m an immunologist. None of us can answer that. Right now we’re still doing the same tests I did when I was a medical student in the late 1960s” Gary Fathman, MD. Associate director of the Institute for Immunology, Transplantation and Infection at Stanford.
For all the money spent on it, the ever so complex immune system is still mostly a black box. Researchers aren’t even clear what impacts immune drugs are having on it.
“Despite billions spent on immune stimulants in supermarkets and drugstores last year, we don’t know what — if anything — those really do, or what “immune stimulant” even means.” Mark Davis PhD
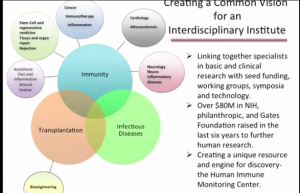
Davis’s center brings together immunologists, infectious disease specialists, transplant specialists and more.
Understanding what a healthy immune system looks like – in order to give researchers a baseline to understand what an unhealthy immune system does, is huge goal, but it’s ignited a lot of interest – and money. Since the Institute has has been launched Davis has received over $80 million dollars in public and private funding. That doesn’t include the $50 million the Gates Foundation gave him last January to create the Stanford Human Systems Immunology Center to produce better vaccines within five years.
He’s engaging one of the most sophisticated immune monitoring efforts in the world to do it. One of only five machines like it in the world (in 2012) shatters cells and tags the proteins found in it. Another can tweak a cell or any number of cells with a pathogen or other stressor and determine how it responds. All told he’s collecting about 40,000 data points on each blood sample.
As of 2012, the Center was running studies on biomarkers of aging, Alzheimer’s, autoimmune disease, cancer, chronic pain, rejection in organ transplantation and viral infection — both acute (influenza) and chronic (HIV). The biggest one running now, though, is a 600 person ME/CFS study. It will take them a year to run all the blood samples in the study.
It’s great having a heavy hitter like Mark Davis engaged in ME/CFS research, but he’s gone further. He’s one of eight distinguished researchers to sit on the Open Medicine Foundation’s scientific advisory board with Ron Davis. He’s a also major collaborator in the End ME/CFS Project.
Mark Davis and other members of the OMF’s scientific advisory board – including two Nobel laureates – are an example of the unique clout Ron Davis has brought to the ME/CFS field. That clout is opening and will continue to open doors that have not been open to us before. We’re very lucky to have them both.
- Coming up soon: The End ME/CFS Project’s First Project!
Health Rising Forums: Inquiry of the Day
Were you diagnosed with infectious mononucleosis/glandular fever? Let us know and tell us how it turned out in


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



Very interesting indeed. I do believe theres a third nobel laureate on board now. Paul berg
True. Stanford’s Paul Berg, Nobel Laureate, recently accepted Ron’s request to join him on OMF’s Scientific Advisory Board!
Never had much of a fever since the 60’s when I became ill. But a recent EBV test that I demanded showed a result 24 times higher than the normal range!!
As I don’t have Mono, they said “don’t worry about it”, it’s the new normal for you!
I postulate that the immune system is not infinite in how many battles it can wage at the same time.
For example: if it is dealing with a major fungal infection and also maybe a form of Lyme, an infection elsewhere (EBV) may not be defeated but at best kept under some control.
I wasnt aware that I had ever had a bout of glandular fever but my recent tests showed that I had. No idea when though.
I like very much this line of thought (and this freedom of thought) breaking away from a conception of our body as a pre-programmed closed system, and indeed, in order to help us think more clearly about the nature-nurture issue, identical twins are just perfect! It reminds me of yet another Nobel Prize who thought along the same lines, Gerald Edelman, equally at ease with immunology and neurosciences, and who shared the same interest with Mark Davis for… identical twins! Not only are their immune systems quite different depending on the environment each has evolved in, but there is also another “detail” about identical twins which is not identical at all: their BRAINS!! No less… And for the same reason as for the immune system: the neural connections will vary greatly and be shaped differently depending again on environmental influences or factors. – It seems that both the brain and the immune system, these tremendously complex systems, the most complex of our bodies (and perhaps of the universe entire…) have this same fundamental feature in common: they are constantly modifying themselves through interaction with their environment. That makes it hard for science to study them…
Interesting Christian. I am an identical twin- my twin, of course, has take a very different path. Interestingly I seemed to be the healthier one growing up – more athletic – but that availed nothing ultimately!
Regarding the possibility of glandular fever/infectious mono precipitating ME/CFS:
When I was 10, I had most of a year off school with what was diagnosed as ME, but recovered fully.
When I was 17, I had glandular fever and shingles at the same time, but recovered from both quickly.
When I was 47, two years ago, I and my two children aged 13 and 16 developed what has been diagnosed as post-viral syndrome/CFS, following a gastro-flu-like illness. This seems to be like what I had at age 10. My son has evidence of exposure to EBV but has not had diagnosed glandular fever.
At least in our cases then, glandular fever has probably not been a precipitating factor. In my case, the ME/CFS came first.
I became ill with glandular fever at age 14 which i recovered from, at age fifteen I developed severe gastritis and seemed to go down hill. At age 18 seemed to ‘get’ glandular fever again and never been the same since . That was 1984. Others I met back then that had got ill in the teenage years have also remained ill. I’ve always wondered why and what was it about teenage years that kept us ill. Ofcourse no one really believes my story 32 years on, but for me the sequence of events were as clear as day. ( disabled with ME 32yrs n countin)
There is sort of a common-sense component that should, does, and will continue to transpire throughout all of the interactions, studies, hypothesis, etc.: LATENT-RESIDING VIRUSES are 1) foreign, 2) parasites, 3) are DNA-based, and 4) take-over our host cells. Think about these basic, fundamental factors…
Thank you for a really interesting article!
Like Christian it got me thinking about Edelman. He’s theorizing about the brain as a selectionistic system – most of neuroscience still doesn’t really understand the implications of those aspects of the system, one being that any computer metaphor for brain functioning is fundamentally flawed. His Nobel prize was for discovering the selectionistic mechanisms of the immune system. Two extremely adaptive systems communicating with the environment, getting shaped by it, and strongly interconnected.
Amazing work by Mark Davis, amazing blog by Cort. Thank you.
Interesting point about how changes over time may explain why initial signal disappears in Hornig study.
I don’t think the cause for ME will be found in the immunesystem. Also there will never come a single immunemarker for this this disease. They look at the wrong place.
I at age 5, after always being sick with every cold and flu around they removed tonsils and adnoids for better school attendance. At age 9 I became a type 1 diabetic (insulin dependent). Within the first year of DM-1 I got very sick with a herpes sore at the back of my thought, though I never knew the type it was a whammy. At age 48 (2007) I had a formal DX of FMS. I see heredity playing with the FM since others on my paternal side have it, however was told the DM was most likely triggered by a virus as well of course the herpes.
I always wondered if having your tonsils and adenoids removed had any bearing on whether we contacted an autoimmune disease since they are part of the immune system. Does anybody know?
Hear Hear! This is what my sister and I have been saying for years. There are a variety of triggers for CFS, but they all cause an autoimmune reaction. In our cases, pathogen and toxin exposure, and vaccinations have had the greatest impact. We were diagnosed in the late 80s, but have had CFS/FM symptoms since we were little kids. Every virus we caught over the years gave us a severe case of whatever it was: varicella zoster virus (chicken pox, a herpesvirus), flu, roseola, mumps, rubella just to name a few. I got rubella twice in a nine year period, and had an autoimmune reaction afterwards, both times (liver, spleen and lymph glands blew up and I felt horrible). My sister got roseola twice, less than a year apart. Both these second cases should have been prevented, if our immune systems had been functioning correctly. Vaccines always hit both of us HARD. Unfortunately, doctors are just as bad at recognizing and treating autoimmune problems as they are at recognizing and treating CFS. I guess that’s not surprising if CFS is an autoimmune disease. In the caveat category: our father had a couple of autoimmune diseases, and our mother has at least five. There are some studies that show that your likelihood of having an autoimmune disease greatly increases when you have a parent with autoimmune disease, and it really jumps when both parents have it. (from the 2005 book Living Well with Autoimmune Disease by Mary J. Shomon)
Kim & Kelly D
I’ve long questioned the role of HHV as I was hit hard with mono at age 39 in 2003. I recovered to maybe 95% of my previous health but came down with CFS symptoms a couple of years later. The symptoms came and went to various degrees until 2011, when they became more severe and permanent. Previous to 2003 I had always been in very good health.
I’m sure interested to know if and to what extent there is a connection between HHV and CFS.
Wow Greg! Your path sounds exactly like mine and even the same years and same age! Nasty viral infection, EBV, got better and worked for 5 or 6 years, then crashed and that’s where I’ve been since. When I had EBV I also tested positive skin test for TB and was given Isoniazid. I have wondered if that helped me in some way to get better for those several years. So many question marks
I actually had glandular fever twice! Once at 8 and again at 13. The second time I remained weak and tired after the gf had subsided, for several months, but was not allowed to stay off school. I then had, what I at the time believed to be, two short flares of the gf in the long summer holidays, and continued to have less energy or stamina than my peers, though didn’t really think about it at the time. Of course, it’s possible that I had a mild case of ME at this time, but, if so, it was very mild, and had few of the accompanying symptoms other than fatigue and some postural intolerance.
It wasn’t until I was 29 that I developed full-blown ME, following a bad bout of flu, and 9 months later FMS after slipping and subluxing my hip (turns out I have EDS-Hypermobility too).
I and my fellow ME’er friends have long conjectured that contracting gf played a large part in our contracting ME. It’s good to have the science that backs up our theories.
http://www.bmj.sk/2010/11103-09.pdf
Very interesting article that talks about immune compromisation of both cellular and humoral immunity and the auto antibodies that they found in lyme patients. People near me have autoantibodies to mitochondria and anti parietal cells of stomach, which produces intrinsic factor necessary for absorption of B 12. … Also interesting is that anti parietal cell antibodies and Hashi are mentioned elsewhere. Anti parietal cell antibodies are also discussed vis a vis infection with H. pylori. Atrophic gastritis and lyme too. Daughter has anti parietal abs and I have Hughes Sydrome or APS and beta 2 glycoprotein 1 abs. High risk of PE and DVT
We hear that certain HLA types get autoimmune diseases when exposed to lyme disease. Also, there is a connection to A spondy.with HLA B 27. That said, all of these people in the above article have different HLA types and wound up with auto antibodies. I have seen many with compromised immune systems with lyme, and many have no infection with CMV but do have other herpes viruses. Compromisation of the immune system allows the herpes viruses to come out again and wreak havoc. We see this as an opportunistic infection in AIDS and transplant patients and also lyme patients. Sharing of info between all of these probs helps us all in the long run which is why we need to stay abreast of what is happening in MS, ALS, Parkinsons, CFS, ME, CFIDS etc. Thanks for all you do Cort.
[Cort: if these Qs belong better on different thread(s) (or 1 or 2 of them do), pls relocate. Also: Can responses to my Qs go straight to the email address I listed? If not — being mostly asleep day in and out — how can I best know if/when there are responses, and get right to them quickly? Thx.]
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
My deep gratitude, Cort, for this Health Rising website — its altogether excellence in “just BEing” / uniqueness / wordsmithing & visuals / wise judgment / expansion / timely-ness, & all the such-evident very hard & extremely helpful work you do — (while having ME yourself)!! My huge thank you, also, to all who participate here, and to all doctors who’ve chosen to focus on ME/CFS/SEID!!!
I’ve have severe ME for ~15 yrs, while living alone in the Washington DC USA area. My Qs.
(1) I want to be very comprehensive getting annual blood tests this time, hoping for clues re: what could be perpetuating the severity of (& even what caused) my ME, and re: what I could address (resolve) that’s contributing to such severe exhaustion (& also severe MPS trigger points). (1a) What blood test list would you recommend I get (names & abbreviations if possible)? (1b) Where would my (ME-inexperienced) doc send blood (hair etc.) samples, for the non-usual tests?
(2) Can anyone offer a possible explanation for why I had onset of ME of severe bedridden-level without any flu or other illness immediately or recently prior? There seemed to be no trigger. (see brief history below)
(3) Being in DC: (3a) What 2 or 3 DC area ME doctor(s) do you most recommend? (3b) Any idea also of a best doc who comes to homes (there are a few who have set up practices to do this…no idea if any know ME/CFS)?
(4) What do you recommend as best energy-limited contribution(s) to solving ME, by me, a DC area bedridden patient? ex’s: * Write-up about my ME-caused life devastation — to where/whom? * How could I — a bedridden patient — get my blood sample added to BioBank? * At (what) limited key time(s) to ask for (what) from my relative who’s senior US Senator (Dem) for Ohio (I forget his Senate Cttees, formerly was top Dem on House health cttee)?
(5) The most life devastation is surely wreaked on single adults with from severe bedridden-level ME — with no nearby or supportive or helping family or friends — no longer able to work thus no income — no (or insufficient) savings — thus evicted for unpayable rent — thus no place affordable to live — no transportation — no internet affordable to learn about resources to help them (if there are any) — and onward down a beyond-grueling-life spiral. (See “A Postcard From The Edge” by Gina Kerner, from the CFIDS Chronicle). Who could keep going in these circumstances? I have read (and heard Cheney once say) that suicide is the largest cause of death from ME. All ME patient descriptors seem to be of those who have enough life supports hobbled together to cope. (5a) Is anyone or entity trying to start, say, a national 800# — with links to phones/volunteers in every county or Congressional district — to provide such ME sufferers in dire straights with some emotional support and practical help? (5b) For eliciting awareness of the human and moral toll of ME (–> compassion –> fund raising), is anyone somehow accessing and compiling case descriptions like THESE? (If identified to every Congressional District (–> votes on a ME funding bill)?) (5c) Is there anywhere where a shelter/building just for alone homeless or without-money bedridden ME sufferers has been set up? (Especially if a medical entity set one up (or one in each region of country), this wonderful humanitarian act could also offer opportunity to study severest-case bedridden ME patients … the very patients who might yield the clearest data, and yet are hardest to find/contact, who can’t go anywhere for studies. Calls to inquire re: it/them could also give leads to these most vulnerable ME sufferers.)
MY BRIEF HISTORY for Q (2): I have had ME/CFS/SEID since Jan 2006 (age 49). ME began at severe bedridden level — but began without any flu or other illness immediately or recently prior. I have PEM, OI, & unrefreshing sleep; some but fewer cognitive symptoms than I’ve read can exist; pain but unsure whether ME is source.
Long-living parents: no remarkable illness in 2 parents, except father mild alcoholism, until old age: heart disease (2), prostate & vulvar cancers, Alzheimers (1).
Childhood: Normal-length & severity chicken pox and measles. Tonsils/adenoids out at 4yo. Athletic through early adulthood.
Very good to excellent adult health, blood tests, no smoking/no drinking/no drugs, etc. Very hard-working scholar, then professional consultant; nationwide travel. No mononucleosis. Very occasional cold-sore type (herpes I assume) on facial lips. Consistently high alkaline phosphatase blood test. In early 2000’s: ova/parasite test, long-term Lyme, & auto-immune tests all negative. EBV test positive. More recent (~ ’12) blood test pointed to anemia. Only other possibly relevant factors:
— age19-24: bulimia (no vomiting: exercise & sometimes juice fasts after binges)
— sensitivity to sugar (fuzzy-brain feeling next day) — used artificial sweeteners since 1980s (past year only Stevia).
— morning phase delayed circadian rhythm (confirmed via Johns Hopkins sleep study)
— sensitivity to insufficient sleep, but with enough, and starting mid-morning, energy was fine
— dependent on 12 oz daily coffee nursed through morning, but enough energy without for morning shower (once ME began: shower energy & going downstairs to get coffee: impossible). Possibly earlier onset of mild ME disguised by coffee and adrenaline
— 1983: 11 mos in basement apartment w/ frequent anti-insect spraying w/ no air circulation
— immediately post 2 wks Nicaragua 1985: severe diarrheal something.
— about 1985-2000, used hair spray about 50% of days; also hair highlighting ’85-’06
— starting about 1994-2005: noticed energy being variable, with a little less stamina starting about 1998 but also described as having more energy than most people.
— anxiety with occasional insomnia and anxiety attacks, rare by later 1990’s
— 1999 – Feb/Mar 2000: very stressful very intense 7-8-mo romance & engagement to public figure, the last 2/3 of which he was verbally and emotionally constantly critical/abusive, driving me to end it.
— Occasional massage therapist said I was developing some upper back knots in 1999-2000, felt by July 2000. Early Aug: post deep-water canoeing. Later Aug 2000, back pain that did not stop. Diagnosed with left mid-back sprain, non-contributing cervical disk bulge, straight cervical spine, and severe myofascial pain syndrome (MPS) “trigger points” in neck & upper right back (could not turn neck for several mos); these latter 3 continue, now with more cervical bulges & MPS in lower body also (still almost all right side).
— In the work-ups for MPS, was diagnosed with FM (2001), a shoulder tear (2002 – surgery for this did not change trigger points), hypothyroidism, and hypothesis of thoracic outlet syndrome (proven correct/severe in 2009).
— In 2005: I did have 2 or 3 one-two-week timeframes when I had noticeable energy dip affecting normal activities. That year also had: — just one-day sudden diarrhea after weekend in W VA (food poisoning I assumed), — no intercourse romance w/ guy I learned had genital herpes, which I have had no symptoms of & also in 2000’s no longer had previous cold-sore type.
— 2013 chest CT showed calcifications throughout left adrenal gland (unknown for how long).
THANK YOU FOR YOUR INSIGHTS.