



“These results reinforce the proposal of fibromyalgia as a neuropathic pain syndrome. Our results also show that fibromyalgia patients have multiple autonomic dysfunction symptoms.” Authors
The destruction of small nerve fibers in the skin of fibromyalgia patients (small fiber neuropathy – SFN) is one the most intriguing developments to show up in fibromyalgia in years. Not only does it indicate in striking fashion that fibromyalgia is a real, physical disease but it provides opportunities to rethink it and thus possibly open up new options for treatment. Studies suggest that 40-50% of fibromyalgia patients have SFN.
Small fiber neuropathy in women with fibromyalgia: an in vivo assessment using corneal
confocal bio-microscopy. Manuel Ramírez MD, Laura-Aline Martínez-Martínez, Everardo Hernández-Quintela MD, Jorge Velazco-Casapía MD, Angélica Vargas MD, Manuel Martínez-Lavín MD. Seminars in Arthritis and Rheumatism, http://dx.doi.org/10.1016/j. semarthrit.2015.03.003
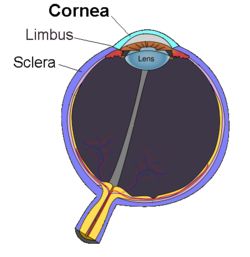
Given that, Dr. Martinez-Lavin figured, that if SFN was going to show up anywhere it was going to show up there. The added bonus was that new microscopic techniques allowed him to assess small fiber density quickly and non-invasively – no skin biopsy required.
In an email exchange he stated:
“The cornea is the place to go when studying small nerve fibers. Corneal confocal microscopy is a rapidly evolving technique. Normative values for corneal nerve fibers distribution are being developed. So, it is likely that with corneal confocal microscopy we will soon have a non invasive objective technique to confirm fibromyalgia diagnosis.”
The Study
In this small study (17 FM patients and 17 healthy controls), an expert in corneal pathology performed blinded (to diagnostic category) analyses of nerve density in two places in the cornea: the stromal nerves and the sub-basal plexus nerves. The study participants also assessed their level of neuropathic pain and autonomic nervous system symptoms using questionnaires.
Results
The questionnaires indicated almost every FM patient experienced high levels of neuropathic pain and autonomic nervous system dysfunction (sympathetic nervous system activation). (Neuropathic pain is often described as burning, stabbing, tingling, numb, hot, cold or itching pain, that (unfortunately) often worsens at night. Allodynia (sensitivity to touch) and altered perceptions of heat and cold can be present as well.)
The FM patients displayed about a 15% reduction of corneal stroma nerve fiber density (p<.01) and a 20% reduction in nerve fiber density in the sub-basal plexus area (p<.o2) relative to the controls. The reductions were not associated with disease duration or symptom severity. When the healthy controls and FM patients were lumped together, however, patients with more slender corneal nerves were found to have increased levels of neuropathic pain. The “ceiling effect”; i.e. too many FM patients clustered at the top of the neuropathic pain scale may have prevented the researchers from finding an association there.
Wrong Channel
Voltage-gated sodium (Na+) channels….have a critical role in the development and maintenance of several pain syndromes, including inflammatory pain, neuropathic pain, and central pain associated with spinal cord injury. The increasing understanding of the molecular organization, function, and regulation of voltage-gated Na+ channels, their plasticity of expression following injury…provide insight into the important role of these channels in the pathophysiology of pain and rationale for development of channel subtype-specific drug therapy. Sodium Channels and Pain – Bennaroch
The authors proposed that sodium channel problems in the dorsal root ganglia may be behind the declining numbers of small nerve fiber problems in FM patients and the pain they experience. Voltage-gated sodium channels are the venues through which the electrical impulses that run along the nerves are transmitted. The more active those sodium channels are – the more active the nerves are.

Damaged sodium channels in the nerves transmitting pain and autonomic nervous system signals could contribute to the pain in fibromyalgia.
Sodium channel problems or channelopathies are a relatively new and exciting finding in chronic pain research. Dr. Sommer recently called the findings on the voltage-gated sodium and other ion channels “eye-openers” that could explain much about pain sensitization. Of the nine sodium channels found, four are known to play important roles in the production of pain.
The dorsal root ganglia (DRG) are cell bodies located just outside of the spinal cord that provide a relay station and filter for the sensory stimuli coming from the body. The axons or “limbs” of the DRG neurons extend into the tissues of the body. Sensory information from the body travels along nerve axons to the DRG and ultimately the brain. Herpesviruses are known to take up residence in these critical relayers of both pain and autonomic nervous system stimuli.
Turning the sensory and pain transmission lines to the brain on full blast may be all that’s needed to trip the brain into producing its own hyperexcitable, sensitized state.
Waxman proposed that sodium channel problems in these nerves are responsible for many cases of peripheral neuropathy and autonomic dysfunction. Studies indicate that genetic variants of one type of sodium channel (Nav1.7) found on the dorsal root ganglia (DRG), trigeminal ganglia and sympathetic ganglion neurons are able to throw the DRG into a hyperexcitable state. Martinez-Lavin’s study indicating severely ill fibromyalgia have these genetic variants, suggests this sodium channel could play a hefty role in FM.
Dr. Martinez-Lavin believes abnormal sodium-calcium exchange caused by issues in the DRG may be causing the small fiber degeneration in the skin and corneas of FM patients. When I asked him about the possibility that other areas in the body may be affected he suggested that the skin and the eyes are the most likely candidates, but that the mucous membranes (mouth (burning mouth syndrome), vaginal areas (vulvodynia), (gut?)) could be affected as well. It would be difficult to study SFN in those areas, however.
SFN Causing Autonomic Nervous System Dysfunction But Not Pain?
People with fibromyalgia often experience symptoms associated with both nerve (tingling, pain, numbness) and autonomic nervous system (gut, heart and standing issues) dysfunction. Many people with ME/CFS who don’t experience significant pain, however, do experience ANS symptoms (problems standing, gut issues, heat/cold intolerance). Could sodium channel problems affect only the autonomic nervous system nerves in some people? Dr. Martinez-Lavin thought that was possible.
Eye Damage?
Because the nerves involved are pain sensing nerves and do not affect sight the reductions in nerve density found will not affect eyesight. One wonders, though, if the nerve issues Dr. Martinez-Lavin found could play a role in some cases of eye-pain that are not connected with dryness or other factors.
Exercise
Since exercise enhances SNS activity I asked him if exercise induced SNS activity might be causing the symptom flares seen in ME/CFS and FM? Martinez-Lavin noted that in FM at least, gradually increased exercise may actually enhance parasympathetic nervous function and restore ANS homeostasis. Acute exercise on the other hand is pretty much guaranteed to throw the ANS out of balance – once again favoring the SNS – and increasing the symptoms associated with SFN such as burning pain, tingling, etc.
Treatment
With regards treatment Dr. Martinez-Lavin reported that the structure of the sodium channel he believes is producing the pain in FM (Nav1.7) is known, and that “specific blockers can be developed as potent and refined analgesics for small fiber neuropathy and for fibromyalgia.”
The growing incidence of diabetes and the painful peripheral neuropathies often accompanying it are surely driving much research. Similar reductions in corneal nerve density have been found in diabetes.
Pain researchers are paying increasing interest to the Nav1.7 and other ion channels and treatment options are being explored. This is a growth field.

Some ion channel drugs are being tested or used in neuropathic pain but better drugs are probably in the future.
Some studies suggest that drugs may need to target specific genetic mutations on these ion channels. That said, phenytoin (anti-epileptic drug), lamotrigine (anti-convulsant/anti-epileptic), ranolazine (angina) and lacosamide (anticonvulsant) target forms of the Nav1.7 channel, that are implicated in neuropathic pain. A blood-depressing substance from sea anenomes (BDS-I) may be able to help tone down hyperexcitable sodium channels. An antiarrythmic, mexiletine, is able to normalize sodium channel activity in sodium channels with a specific mutation. Xenon402, a novel Nav1.7 blocker, is being tested.
Medical marijuana can be helpful in reducing neuropathic pain and something called ajulemic acid, a synthetic cannabanoid, may help explain why. It inhibited the activity of all sodium channels associated with pain in a recent study.
Lamotrigine is a good example of incredibly varied uses modern day drugs can achieve. Lamotrigine is used to treat epilepsy, bi-polar disorder, several types of seizures on label. Off-label it’s used in peripheral neuropathy, trigeminal neuralgia, cluster headaches, migraines, depersonalization disorder, schizoaffective disorder, borderline personality disorder, and post-traumatic stress disorder.
Our data indicate that spider venoms are a rich natural source of Nav1.7 inhibitors that might be useful leads for the development of novel analgesics. Klint et. al. 2015
Perhaps the most interesting substances, though, may come from studies of spider venom. Forty percent of 205 spider venoms were found to have at least one Nav1.7 inhibiting substance. Seven novel Nav1.7 modulators were found during sequencing of spider venom.
Some of the drugs listed have issues that may preclude their use, and it’s not clear that any breakthroughs have been found yet. Interest in understanding the roles sodium channels play in producing pain, however, is high. With new techniques allowing researchers to be able to better understand the structure of these minute ion channels, the better drugs are clearly in the future.
Wrapup
The results of this small study using a new technique need to be validated, but they fit with prior findings in FM. The small nerve pathology found is probably restricted to the skin and eyes and perhaps the mucous membranes as well. The association of reduced nerve width with neuropathic pain suggested that the SFN found is contributing to the pain in FM patients.
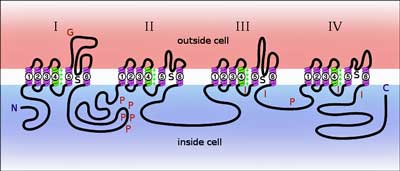
Voltage-gated sodium channels relay electrical signals: too much current equals too much nerve activity – and increased pain.
These findings offer a possible explanation for one of the most puzzling aspects of FM and the other so-called “functional disorders”: the inability to detect trauma or inflammation at the site of the pain. They suggest the trauma is there in the form of damage to very small, unmyelinated nerves and, digging deeper, in the ion channels found in those nerves.
It’s not clear if the sodium channel blockers available now would be effective in FM or not. Given the recent advances in understanding sodium ion channel structure, however, Dr. Martinez-Lavin expects the the development of effective drugs to occur.
Inquiry of the Day
How effectively is your neuropathic pain being treated? Let us know in the Neuropathic Pain Treatment Poll on the Health Rising Forums.






I admire folks who let anyone near their corneas.
I’m not sure about those meds for me. If you add up various meds proposed for each of the FM theories’ symptoms you’d make your pharmacist start planning his around the world trip, not to mention your having to cope with a basket of side effects.
I took the first Vicodin of my life in the dark, an error, was actually reaching for my first Tramadol LOL. When I woke up for the first of my several nocturnal bathroom sojourns I wondered why my fibro pains were wtf low. Now I see why people in pain take Vicodin and why House was addicted. Will put away for real emergencies. Today I tried Tramadol, also apparently quite addictive, and found it made me not be quite as troubled by the pain as much. I don’t think I would drive on it though, and it, also unfortunately, will need to be saved for killer days. I have an MJ card, pleased to hear it might help SNF pain, but unfortunately no dispensaries open yet here in MA. Will be attending an MJ conference to see if caregivers ie. growers have any options other than weed. A low level edible and a salve I got bootleg to try in CA were useless but will try others when available. Thanks for report, Cort.
Yeah, I don’t think I have fibro but I have lots of joint pain from ME/CFS and hydrocodone can actually make it worse for me. If I take less than 15mg hydro it actually makes the pain worse, possibly the result of some sort of histamine reaction. The thing is that the amount of pain should be readily controlled by as little as 5mg so 15mg is way too much. Luckily I found out that nightshade foods and foods that contain solanine (potato, tomato, bell pepper, eggplant, paprika/cayenne/jalapeno, okra, blueberries/blackberries, and artichokes) contribute greatly to my pain and by avoiding those I don’t have to take any pain meds at all. I also avoid soybean oil with success as well.
Also as a side note about mistaking pain medications, when I got my wisdom teeth out I got a dry socket that hurt like hell so I had to take pain pills. The thing is that I had previously taken 5/500 and 7.5/750 hydrocodone in the past for cluster headaches, so when I saw the 10/350 hydros that I got from the dentist I thought they would be weaker due to the pill itself being physically smaller than the others. So I ended up taking a few too many to compensate and got nauseous as hell. Those things can be dangerous if you’re not familiar with them!
Thanks for the note about the berries. I have the same problems with nightshades – didn’t realize berries could contribute as well; it explains why my knee flared up recently
steve- have you tried hemp cbd? (cannabidiol) it’s legal and the first time i tried it a 5 day migraine just melted away in less than half an hour.i now use it to help me sleep better. most of the time it helps with that but it seems to work best for those tense painful muscles. at least for me…..
Wow – now that’s impressive…marijuana candy worked really well for me to relieve muscle tension and relax – REALLY effective
What about smoke in your lungs from cbd
Yes ! After 40 years of living with FM, I have discovered wonderful success for pain relief with low – dose CBD. Relieves allover pain and calms my overactive brain. A God-send.
Had chronic Lymes after a yr of antibiotics and vitamins I slowly improved but every MD I saw 20yrs ago said I was left with fibromyalgia. Ihave neuropathy but not not diabetic. Taking some meds tramadol if it’s real bad.stoped neurontin,can’t walk far feet and knee pain. Hope the research finds an ans. Go see a neurologist again. Maybe something new.
Could you please start including citations at the end of articles you write about specific papers please Cort? If there are multiple links in the article itself it can be hard to find the specific citation for the main paper and if there’s no link at all it can be pretty hard to actually find the paper that the article is written about. Thanks.
Sorry – sometimes, somehow, I forget to include the citation for the main study. (It’s in the piece now.) I certainly intend to do that. For this piece there is no link to a study because it hasn’t shown up on PubMed yet.
Uumm this could well explain why the corners of my eyes hurt so much sometimes! I have over the years put it down to medication side effects or even having a different type of headache. I’ve seen optometrist about it and they reassured me my cornea and everything in my eye looked ok. I didn’t quite understand what type of test is required. Maybe is too early to tell. Thank you for all your hard work!
I think this test is probably really hard to find. It may be available just in research settings – I don’t know…
Would this have anything to do with the pain bright sun or lights cause my eyes? I can rarely be out in sun without strong sunglasses and the typical shades don’t work, either. I have to use a brown tint to stop the hurting. Green and Grey tints do not work. Stumps my eye doctors every time.
MY sensitivity to light went away after getting big doses of vit A in the form of FCLO fermented cod liver oil + betacarotene. After 6 weeks the light sensitivity went away and then it came back after a month or so after stoping these supplements.
Cort, thank you so much for all you do.
Coincidentally, there have been 2 or 3 articles that I have come across in the last week. On Tuesday, nearly 2 weeks ago, I went to eye Dr. He looked real hard until he found some cells in back of my eye. He diagonsed me with Uveitis. A serious eye infection with several different causes, all internal. Also I have been recently diagnosed with reticularopathy and neuropathy, (non diabetic).
The research that involves the CNS and ANS is very encouraging. I really believed that an infection of the brain and spinal cord is largely responsibility. Hence, ME goes along with CFS/FMS. As a 30 year sufferer and an avid researcher, this has to end. They have to find a cause and a cure.
The Neuronontin the Neurologist gave me has helped the pain, the fog and the sleeplessness. I do recommend it, but know it comes with a lot of side effects, none for me, thank God.
Thanks to all of you who write in, one of us may hit on something big.
Thanks Maryann. Good luck with the Neurontin. I know it works very well for some people. Glad you’re one of them.
Fantastic work but i think its an assumption that the detected small fiber neuropathy is caused by sodium channel abnormalities. Fibro is an acquired illness. In many systemic autoimmune diseases where sfn manifests small nerve biopsy demonstrates elevated inflammatory cytokines like tnf alpha such as in Sarcoidosis.
Therefore id suggest its just as likely the sfn in these patients is an acquired autoinflammatory etiology where abnormal b-cell autoimmunity is directing a cytokine-driven inflammatory response against either pain-receiving small fibers or autonomic small fibers. So basically like Crohns or RA only the target is not the stomach lining or joints but rather small fibers.
This could account for the higher female to male ratio, the sudden onset following immuno stressing event and the waxing and waning course.
Overdoing activity would also have a flare up in response.
Interesting thoughts, Gibbo. My son has had Crohn’s for twenty years and I’ve often wondered about an autoimmune similarity to FM. I also feel that we understand FM about the way they did CD twenty years ago.
Not sure if exercise always exacerbates the autoimmune response, at least for my son, as he went on to become a fitness expert and bodybuilder. Exercise also never affected my FM symptoms until the disorder generally started getting much worse for me two years ago. Only recently do I find my AM cardio/weight routine worsening my neuro-type leg pain, a nasty development for me.
To the woman who suggested CBD…bring it on, I’m waiting for the corrupt incompetents in our legislature to get a dispensary open!
you can find hemp cbd on line- just google it .i’m not sure there is any thc in it at all- because hemp has so very little thc ,that’s why it’s legal no matter what state you live in 😉
Puzzling, I just asked the top company in CO if they were going to sell in MA and they said there were too many regs to start a branch here and that they couldn’t even ship hemp CBD because the tiny amount of THC in it would get them in trouble. So…
i don’t know if this will help but you might want to check out johnhicksmd.com and hempmedspx. i didn’t realize that it might be a problem in other states.i live in OR and pot is going to be legal here starting soon.
Thanks for your response, Marta, will check it out. I’ll also be asking for info at the first MA MJ Convention, here this w/e in, of all places, Cape Cod. It looks to be a pretty big deal, with an odd assembly of stoners, growers, big time entrepreneurs, health nuts, and others.
I hope OR puts a better system in place for you that doesn’t take years to implement. I have often thought if everyone in Congress had fibro pain for only an hour, they’d vote wtf $ for research immediately.
Could be!
With that theory, Rituximab would work for Fibromyalgia the same way it does for ME/CFS wouldn’t it?
found that all the rising agents ie; yeast gluten sugar ect cause high pain in FM (also alcohol)
gave them up years ago and my fybro has gotten better and better
sugar definately the worst—causes shortness of breath
Thank you, so much for the last few articles on Neuropathy. I have struggled with this for years, and Doctors have tested and retested me for diabetes and brain injuries, as it has gotten worse. But it’s just so affirming, does that sound so crazy, that we at least see a cause, in a syndrome that’s a ghost at best. Having it since 1991, means I have a knee jerk reaction to Doctors at times who still dwell in the dark days that make Fibromyalgia a stigma. I am just thanking God for credibility! Because I read and can say, that’s me, look that’s me. This is best, most credible, site I have seen in all my years of looking for answers. As a person with Fibromyalgia, a lot of times, we need to bring the knowledge to our care givers and Doctors, your site is so professionally done, with current RESEARCH, with hard facts and case studies. I so appreciate the efforts, because many moons, failures and lots cash ago, I became tired of good hypothesis and warm wishes, and I have been waiting for you and here you are.
Thanks Deborah. I love digging into this stuff – I find it fascinating and I hope I’m getting most of it right :). Thanks for your nice words..
It’s weird that the ANS dysfunction they noted in the research is for an overactive SNS. I have fibro and was recently diagnosed with dysautonomia with an ANSAR test and was told I have parasympathetic dominance.
I have heard that as well. Sympathetic dominance is more common but some people have described the ANS problems as all over the place.
I am currently on Nuerontin 600 mg which has helped my pain a little bit but I am pretty foggy. I am supposed to start Elavil with a beta blocker for the ANS problems and if they work out I will then wean off of Nuerontin. Midodrine was another drug that may be added to the mix depending on my bp. I have low bp and the beta blocker may lower it even more so that is where Midodrine will come in to raise it back up. Such fun!
Jessica, can I ask what beta blocker? Also, if you have started treatment with it and Elavil, how is it working out for you? Thanks.
Hi,
I believe it’s the generic for Coreg. I haven’t started it yet, as I am taking my time titrating up on the Elavil. So far at 10 mg, I have not had any weight gain, nor much pain relief. It definitely helps me sleep. I had a really hard time waking up in the morning (as if it was possible to be any more difficult!) at first but that got better. I am doubling up to 20 mg next week. Hopefully it will be well tolerated and start helping my pain more.
Might this explain why i seem to bruise so easily????
One of many things that I have noticed an improvement in as I experience gradual fibro remission, is that light no longer hurts my eyes.
So many things, I did not blame fibro for when it was really all interconnected – I went to the doctor about my eyes many years ago because I was so worried about how they were starting to hurt on sunny days. As usual the medical people found “nothing wrong”. I have been heavily dependent on sunglasses for years but this has now gone, along with many other irritating conditions.
I have posted at length on other threads how I am achieving a bounce-back from more than 20 years of fibro; this is still ongoing.
It amazing how this stuff goes away as you get healthier…
Phil, could I ask what you are doing to have a fibro remission? Or could you provide a link to your posts? Thanks!
Thank you Cort for both the Dr Sommer and this eye Pain connection to SFN.
I Have ME and my eyes and face have always had the worst pain and light sensitivity. I am waiting for a referral for SFN diagnosis. I was recently told by an ME Research doctor that he thinks I have an autoimmune disease.
My pain consultant believes my CNS is involved too.
Many years ago, living abroad, I went to see a doctor with what I described as a blister covering my iris. He laughed but admitted it did look like that. I guess it was something to do with the cornea but can’t remember what it was. Can anyone help out with the answer please??
I was also treated with Quetiapine last year and my pain disappeared – wonderful – but side effects of course and only a short term drug, as Dr Sommer said in her previous article.
This will be fascinating for my pain consultant and the neurologist to read.
Ann OConnell
I’ve had 3 flares which have effected my eyes greatly. Each has worsened in the intensity of pain and length of time until it clears.
This most recent flare caused not only the normal “gritty & sandiness” in my eyes, but also includes stinging in my eyes that worsens as evening falls. Right now I have down right pain in my left eye. I feel like I want to pop it out, it hurts that bad.
My eye doctor always says I have a good deal of inflammation. As I said, of the 3 times this is the worse. If I give this information to my doctor, will there be anything she could look for?
Thanks,
Lorraine
Great post.
Thanks
I wonder what effects b12 supplementation would have on this? I understand if its genetic than that might be an issue, but I can also see pain sensations from hyperexcitability and also “fractured” nerves, as in diabetic neuropathy, MS, etc… My guess is some would be helped with b12 and others would perhaps have symptoms overdriven, at least in the beginning.
Fluoroquinolone antibiotics – Cipro, Levaquin, Avelox, Floxin and their generic counterparts – cause peripheral neuropathy. It has been on the warning label for years and in 2013 the FDA changed the warning label to note that the peripheral neuropathy caused by fluoroquinolones could be permanent. http://floxiehope.com/2013/08/22/fda-permanent-peripheral-neuropathy-warning-label-fluoroquinolone-antibiotics/
Fluoroquinolones also cause dysautonomia. I believe that the mechanism is that they cause mast cell activation and histamine release. However, the mechanism may be the chelation of minerals from cells or mitochondrial toxicity or hormonal disregulation. http://floxiehope.com/2015/10/01/can-fluoroquinolones-activate-mast-cells/
Fluoroquinolone antibiotics contribute to fibromyalgia and ME/CFS for many.
My FM predates that class of meds by years but a course of levofluoxavin for sinusitis last year exacerbated my FM to such an excruciating degree that I had to stop med. Strongly advise caution for FM folks.
anyone here suffer from the mucous membrane- affected SFN such as burning mouth/lips/tongue? If so, have you found any relief?