

The Chronic Pain Connection
From “hand-warmers” to ion channel blockers to magnetic field generators more and more different approaches to get at the basis of chronic pain are being tried. Now comes an entirely different approach.
First, studies indicated that in rodents, chronic pain neurons thought to be associated with negative emotions showed increased activity after the onset of the pain. Those neuron’s connections to other parts of the brain associated with depression, also increased. The neuronal activity was associated with a drop in dopamine levels.
Combining a Parkinson’s drug called L-dopa with a non-steroidal anti-inflammatory drug completely eliminated any signs that an animal model was feeling pain. A clinical trial in humans is underway.
L-dopa isn’t the first Parkinson’s disease drug found to reduce pain. Pramipexole, another dopamine enhancer, had a similar effect on rats with chronic pain. Pramipexole did pretty well in a 2005 controlled, double-blinded fibromyalgia study. FM patients reported a 35% decrease in pain and about 40% had a 50% or greater reduction in pain. Fatigue dropped by an average of 30%.
A Parkinson’s – ME/CFS/Fibromyalgia Connection?
Parkinson’s has never been explicitly connected to chronic fatigue syndrome or FM, but many of the secondary symptoms (muscle rigidity, increased muscle tone, contracted muscles, autonomic nervous system problems, gait problems, cognitive issues, sensory issues and mood disorders) can be found in both. Small fiber neuropathy – commonly found in FM – appears to be the cause of the sensory issues in Parkinson’s disease.
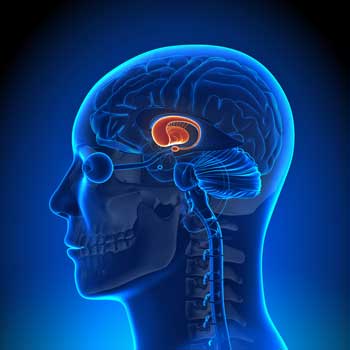
In Parkinson’s disease dopamine-producing neurons in the nucleus accumbens in the basal ganglia in the brain are destroyed. The neuron destruction sets off a variety of symptoms, the most prominent of which is the inability to move. First Parkinson’s disease patients experience shaking, rigidity, slowness of movement, difficulty walking and gait problems. Cognitive and behavioral problems arise later, and depression is common.
The Basal Ganglia Connection
Basal ganglia abnormalities have been implicated in all three diseases. Miller found that reduced activation of the basal ganglia was associated with reduced reward and fatigue in ME/CFS. Reduced blood flows to the basal ganglia were also recently associated with increased pain disability and the overall impact of fibromyalgia in FM patients.
- Unrewarding Reward: The Basal Ganglia, Inflammation and Fatigue In Chronic Fatigue Syndrome
- Dopamine, the Basal Ganglia and Chronic Fatigue Syndrome #II – Treatments
The basal ganglia is an intriguing organ which regulates not just movement and the autonomic nervous system, but cognition and emotions such as reward.
First, a look at the role basal ganglia problems may play in producing chronic pain and reward.
Chronic Pain, Emotions, Addiction and the Basal Ganglia
The neurons in the nucleus accumbens in the basal ganglia are activated by drugs such as opiates that produce euphoria or by participating in rewarding experiences such as sex, exercise, etc. The connection to the mesolimbic pathway – which is highly activated in addiction suggested to the researchers that the brain can become hardwired or addicted to producing pain. One asserted that the results indicate that chronic pain cannot be viewed as a purely sensory phenomenon but instead is closely related to emotions.
That’s no surprise. It’s clear that the anticipation of pain, for instance, results in increased pain. Anticipating pain could be viewed as the opposite of anticipating reward.
Miller’s ME/CFS study found that basal ganglia inactivity and reduced reward was associated with increased fatigue in ME/CFS. Reward is clearly a complex phenomenon that integrates emotions and motor activity. High reward states may be synonymous with increased motor activation and vice versa.
The Basal Ganglia and Movement
The basal ganglia are not just about emotions and reward, though. The primary function of the basal ganglia is to ensure that voluntary movements are carried out smoothly. It does this by regulating the activities of the motor and premotor cortical areas.
Excitatory neurotransmitters such as glutamate cause the basal ganglia to impair movement. Glutamate activates a pathway involving the basal ganglia which inhibits signals coming for the motor cortex. Dopamine-producing neurons in the basal ganglia, on the other hand, stimulate movement.
In Parkinson’s disease destruction of the dopamine-producing neurons puts the movement inhibiting portion of the brain on overdrive. Their muscles, for instance, remain in a state of contraction with a high muscle tone. This does not appear to be the result of muscle problems; their brains have simply lost the ability to tell them to move.
Autonomic Nervous System Breakdown
Recent findings suggest that autonomic nervous system problems run rampant in Parkinson’s. As in ME/CFS and FM, parasympathetic nervous system functioning is impaired. Problems with orthostatic intolerance are being found as well.
The autonomic nervous system findings in Parkinson’s strongly correlate with findings of small nerve fiber neuropathy. These findings are particularly intriguing since they suggest that, just as in fibromyalgia, Parkinson’s is more than a central nervous system disorder; the peripheral nervous system is also affected.
The Gait Studies
The first ME/CFS gait study in 1995 was prompted by Benjamin Natelson’s observations that people with ME/CFS walked differently. It proposed the gait problems it found were due to “balance problems, muscle weakness, or central nervous system dysfunction”.
Another gait study found that because the abnormalities seen began at the beginning of the ME/CFS patients walks they were not likely due to fatigue. Instead, they appeared to have a central nervous system explanation.
A 2008 study found that ME/CFS patients self-selected walking speed was much slower than average. In fact, ME/CFS patients were walking so slowly that their walking velocity was similar to that of above the knee amputees (!). The researchers attributed the reduced walking speed to the smaller slower steps the ME/CFS patients were taking.
A follow-up study using an oxygen uptake test found that ME/CFS patients used much more energy during walking than did the healthy controls. The ME/CFS patients, then, appeared to be attempting to reduce their energy expenditures by walking more slowly.
The researchers proposed several possible reasons for this including kinesophobia (a fear of movement leading to increased muscle activation and energy use), metabolic/mitochondrial issues and/or deconditioning. They noted that a similar issue is found in multiple sclerosis, stroke, and other disorders.
People with FM may experience similar limitations. One study found they used hip flexors more than usual -another more energetically intensive mode of walking – instead of their ankle flexors when they walked.
Slowed movement is one of the first symptoms seen in Parkinson’s disease. It is, again, associated with reduced dopamine in the brain. Miller’s studies suggest dopamine reductions may be present in ME/CFS as well.
Conclusion
Some similar symptoms, the basal ganglia problems, the dopamine reduction and the autonomic nervous system problems suggest ME/CFS and FM could have connections to Parkinson’s and other basal ganglia diseases.
The basal ganglia is a major organ containing several different sub-organs. Depending on the type of basal ganglia dysfunction found, several different diseases can result. They include Parkinson’s disease, Huntington’s disease, Tourette syndrome, dystonia, and addiction.
Perhaps at some point fatigue and pain disorders such as ME/CFS and FM will be added to the list.



 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!





We walk slowly to avoid post exertional malaise! We walk slowly because pacing with a heat monitor taught us to walk as if through molasses to avoid the beeper going off! We walk slowly because we still have two more stores to get to and if we walk fast we’ll be in bed tomorrow!
The point was not that you’re not walking slowly because you don’t have energy problems. The authors, in fact, that it did take more energy for people to walk than normal. That’s a double whammy. They are already depleted and it’s harder for them to walk anyway….That’s hardly fair! 🙂
The point was that contrary to what you might think, that walking slowly is actually a more energetically intensive way to walk. That’s apparently because when you walk normally you walk in a more efficient manner.
If you’re walking more slowly to keep your heart rate down – that’s another matter entirely. In that case I imagine that it’s better to walk slowly. If you have the option, though, it takes less energy to walk at a more normal pace. (The authors did not suggest walking fast was better by the way – they referred to a more normal pace.)
Another study found that people with FM tend to walk using their hip flexors instead their ankle flexors. That means they’re using muscles that move the entire leg instead of using muscles that move ankle to walk. That takes much more energy.
I was told early on by a Rolfer that I was walking in a very energy intensive manner. I assume I was using big muscles to walk instead of smaller ones.
I wonder if the muscle contractions and muscle rigidity talked about play a role in at least my tending to use bigger muscles when smaller muscles would do the trick. I’ve thought for years that I’m using my major, energetically expensive muscles far too often.
I find myself describing my worst years as “3 weeks on and 3 weeks off”. Not coincidentally related to the 3 week flu that recurred during in the early months of what is now 9 years of ME/CFS. Would go from feeling normal, for weeks to days where I felt like I was walking in quicksand. The scary part was realizing that it was “all in my head.” This information is very interesting indeed.
I walk really slowly. Good to know that it’s not just me being lazy.
Forgive me, but what is a “Rolfer”?
Rolfer’s do a specific kind of deep tissue bodywork.
I definitely completely changed the way I walk since FM started 6 years ago, simply because of stiffness/pain in certain areas. To suggest to try to walk faster is thus absurd as I’m not able to. But I’ve always kept on walking and even doing little hikes occasionaly (with loads of rest afterwards). Right now I’m suffering from pain in both knees which I’ve never had before. Drs say it’s osteoarthritis. I can’t help but wonder if I’ve maybe put too much strain on them because I couldn’t use certain muscles.
It would be interesting to know how many of us and how many of Parkinson’s patients share a mutation in the COMT, and/or MTHFR genes both of which can impact dopamine production.
I didn’t know that but I know that the COMT gene mutations have been found in both fibromyalgia and ME/CFS.
In 2014 I found out I was compound heterozygous for the MTHFR gene mutation and my Aunt was diagnosed with Parkinson’s. Hmmm. I don’t know about the COMT.
No COMT issues, but MTHFR and others for me.
Issie
I did 23 and Me and found that I have a mutation in the COMT genes. I think this means that I break down catacholamines more slowly, but I’ve got to review this. A couple of things slow down the COMT enzyme even more – green tea (maybe tea in general) and quercetin. I’ve stopped both and notice no difference.
I became friends 10 years ago with a lady with severe Parkinson’s Disease and despite the severity of her symptoms, we were surprised at how similar our symptoms were. She had recently skyped with Michael J. Fox as he was then the youngest onset of Parkinson’s in the northern hemisphere and she was the youngest onset in the southern hemisphere – she developed symptoms at the age of 17. However, I shouldn’t have been that surprised that the similar symptoms we shared were caused by low dopamine as I had been self-medicating with cannabis for 10 years before meeting her. She was taking L-dopa hourly which kept her more mobile and helped with pain, motivation and cognition but she always got worse as the day wore on and was particularly affected around 3pm when cortisol levels drop. I find the same thing in that I am usually going quite well in the morning but deteriorate as the day progresses no matter how much cannabis I have. On the other hand, strong coffee can lift my energy levels in the late afternoon but has other consequences (bloated abdo and difficulty sleeping) so only do this in exceptional circumstances when I need more energy. As for the speed of walking, I find I walk much faster than my husband and my friend with Parkinson’s, though she used a walking frame, needed to walk fairly fast in order to sustain momentum.
Interesting. The slowness of movement, muscle rigidity, contracted muscles and autonomic nervous system problems really jumped out at me. Plus there’s cognitive problems and now small nerve fiber issues! Where the heck did the nerve fiber problems in the body from in this central nervous system disease? (They thought from L-dopa at first but that doesn’t appear to be the case.
I have the same reaction to caffeine by the way. A few sips and I’m usually flying – I love it! but get cramps and feel depleted the next day.
When talking about walking fast I was referring to walking outside of the home. Of course, in a severe flare (of Parkinson’s or FM) or in an untreated state one can hardly walk at all and if so very slowly and bent over. The person with Parkinson’s actually shuffles with very short steps. However, in a treated person feeling well enough to venture outside the house (and my friend was on so much L-dopa she could go on outings), the walking becomes fast as it takes a lot of force to initiate movement and then speed to maintain momentum. As for SFN, I have always blamed trigger points on that and from my experience believe that the reason increased dopamine levels moderate pain is for the effect it has on moderating the status of trigger points. My friend with Parkinson’s was covered in trigger points as am I. I helped relieve some of her pain by treating her trigger points.
I feel like my strange gait comes from my lower back which is also painful. It is most apparent when descending the stairs.
Ps I have always thought my symptoms were more like Parkinson’s when people compare ME to MS
We always think of MS – but could Parkinson’s be a better fit? (I wonder if they suffer from PEM?)
My mother and her sister have a movement disorder in the Parkenson family of disease. We have many on both side of the family with autoimmune diseases. I was just discussing with my Mom over Christmas that I thought some day she would be diagnosed with ME or that we would find a link to these diseases. A very timely article. I am going to show this article to her. Thanks for all your great articles Cort. I have appreciated your website for many years.
What a coincidence! I have had ME for 20 years–mostly unrelenting exhaustion and some brain fog with immune weakness. Two months ago I was diagnosed with Parkinson’s (based on gait, handwriting and vocal weakness. Now I have to wonder—is this another ME crash or . . .
Please continue with your valuable and very informative posts, Cort!
I have wondered about a connection like this for some time. I’ve struggled with CFS since the early 1970s, and when my father was diagnosed with Parkinson’s we talked about how similar our symptoms were. In a way it was a blessing, as it gave me a chance to be supportive of him and gave him a chance to understand some of what I’d been going through. He passed away recently at 91 after several heart attacks.
Fascinating article – thank you !
My first “wrong” DX was Parkinson. I got progressively worse on dopamine and SSRIs to balance it out. So bad, I was DXd with Multiple System Atrophy. That was also “wrong”. With lots of time, money and over 16 doctors at Mayo, I was finally DXd with POTS, and FMS along with MCAS and other things. This fit much better than the first two wrong DXs. We also have tremors with POTS. The autonomic nervous system just goes on over drive and they come. I have gait issues and neuropathy. My father had this too. Whatever causes some of these things can run in families. But we also had Parkinson in our family, but way far back. My sis has severe CFS. Hopefully we will find out more with Dr Bateman genetic study. Our family is supposed to be included in this. My sis is a long time patient of Dr Bateman and has been included in most of the ongoing research studies.
But, dopamine made me worse. I started improving when I got off things that directly affected that neurotransmitter. I do think that too high glutamate may play a role in overactive sympathetic nervous system issues. There is a fine balance between the neurotransmitters and it may be better to use precursors and possibly herbs to tweak if that is found to be an issue. But balance is the key.
Issie
Please be careful when self-medicating to raise dopamine levels. This is a potentially dangerous thing to do and could lead to psychosis.
Thank you for your concern. I am quite aware of this. It is all a matter of dose – all drugs can be dangerous at certain doses. Cannabis grown hydroponically and taken in large doses is much more likely to lead to psychosis than cannabis grown organically in soil and taken in small amounts, the latter of which I have used for the last 20 years and I can assure you I am definitely not psychotic!
I too self medicate and Tricia is right, so long as you keep doses low and organic you won’t suffer any psychoses.
I only use at night, so it won’t interfere with my day.
It is interesting that the particular polymorphism of the COMT gene that they found in people with FM actually helps us to make more efficient use of the little dopamine that we have which must be why we only need to supplement with very small amounts.
Right! Too much dopamine can cause psychosis. I took the antiviral drug amantadine for Epstein-Barr. It kills viruses, but it also increases dopamine. It gave me absolutely crazy, motion-filled dreams starting the minute I closed my eyes. And it caused insomnia. Incessant crazy dreaming isn’t quite psychosis, but I couldn’t tolerate the drug. However, I know someone who was prescribed amantadine by Nancy Klimas and it got her 80% better from ME/CFS. Maybe it was the increased dopamine rather than just the antiviral effect that helped her.
In the 80s when I fell ill along with other members of my family I looked at other illnesses trying to find out what we were suffering from. Parkinson’s disease was one that had some striking similarities,the shuffling gait and great difficulty in beginning to move out of a chair or get up from bed. I remember one of my sons teachers making disbelieving remarks about his illness but in the middle of it she mention “how odd his walking was”. If I remember correctly Betty Dowset said something about diseases that affect the brain “all having similarities as they all share the same symptoms”.
While on the subject of related conditions, has anyone ever noticed how similar ME is to the effects of high altitude, people can function, look normal but their brains are gone (inability to make decisions even remember their names and addresses etc.). Also the effect on physical function great difficulty putting one foot in front of the other, body feeling like an enormous weight, headaches etc.
Is the high altitude effect due to lack of oxygen and that was why many people with ME found benefit from high oxygen intake treatment?
Lack of blood flow to the brain can also have similar effects, causing orthostatic intolerance.
A friend has Parkinson’s and very similar symptoms to mine.
So, would this seem to indicate that FM and CFS/ME sufferers would benefit from taking L-dopa? Are some people already doing that?
I really don’t know. The basal ganglia are very complex.I think it’s a possibility but it’s always hard to tell.
Cort this is all really interesting and I hope these relations will be further investigated. In 1986 I believe in hindsight that my earliest symptoms of ME/CFS/FM were just beginning after suffering a few infections (one was herpes simplex). It was at this time that I met my husband and I can remember him making fun of me for the way I walked. He was teaching skiing at that time and has always been a natural athlete advanced in many sports so he innately understands movement. He said I initiated my steps from my hips and didn’t move my upper torso much at all. Since then I have noticed that I am always slower at walking than others. I used to think it was just that my husband walked much faster because he is taller, but even when I walk with others my own size and leg length I am slower. In my case I know this is not due to fear of PEM or completely due to increased heart rate or de-conditioning because fortunately I can cycle with much greater ease and much less energy output than walking. I know it is more kinesthetic. I know from weekly therapeutic massage and yoga that my hip flexors are extremely tight, so definitely overused. Interestingly I also beginning in those early years of my onset I also began to notice that my muscles developed at a much more rapid rate than others. Just one exercise session would lead to amazing results, but also greater amounts of pain than others experienced. I listened to a book about running (I think it was Born to Run) in which there is a section that provides the proper steps for walking (and therefore for running). It is extremely basic, takes the learner back to our original lessons for how to walk, what to do with every part of the body, rhythm, timing, etc. I found it so difficult to do these steps, but it seemed helpful. If I find it I’ll post it to Race to Solve ME/CFS. Thanks again!
This study just came in:
“These findings provide further support for a disruption of dopaminergic neurotransmission in FMS and implicate DA as important neurochemical moderator of differences in pain perception in FMS patients with and without co-morbid depression.”
Eur Neuropsychopharmacol. 2015 Dec 10. pii: S0924-977X(15)00390-9. doi: 10.1016/j.euroneuro.2015.12.007. [Epub ahead of print]
Relation of dopamine receptor 2 binding to pain perception in female fibromyalgia patients with and without depression – A [11C] raclopride PET-study.
Ledermann K1, Jenewein J2, Sprott H3, Hasler G4, Schnyder U2, Warnock G5, Johayem A5, Kollias S6, Buck A5, Martin-Soelch C7.
Dopamine D2/D3 receptor availability at rest and its association with individual pain perception was investigated using the [11C] raclopride PET-method in 24 female Fibromyalgia (FMS) participants with (FMS+, N=11) and without (FMS-, N=13) comorbid depression and in 17 healthy women. Thermal pain thresholds (TPT) and pain responses were assessed outside the scanner.
We compared the discriminative capacity, i.e. the individual׳s capacity to discriminate between lower and higher pain intensities and the response criterion, i.e. the subject׳s tendency to report pain during noxious stimulation due to psychological factors.
We found significant group effects of BPND in striatal regions (left ventral striatum, left caudate nucleus and left nucleus accumbens) between FMS+ and FMS- compared to healthy subjects. Correlational analysis showed negative associations between TPT and D2/D3 receptor availability in the left caudate nucleus in FMS-, between TPT and D2/D3 receptor availability in the right caudate nucleus in FMS + and positive associations between TPT and D2/D3 receptor availability in the left putamen and right caudate nucleus in healthy controls. The response criterion was positively associated with D2/D3 receptor availability in the right nucleus accumbens in FMS – and negatively with D2/D3 receptor availability in the left caudate nucleus in healthy controls. Finally, no significant associations between D2/D3 receptor availability and discriminative capacity in any of the groups or regions were determined.
These findings provide further support for a disruption of dopaminergic neurotransmission in FMS and implicate DA as important neurochemical moderator of differences in pain perception in FMS patients with and without co-morbid depression.
Many people have benefited from an antibiotic protocol e.g. Marshall protocol and/or cpnhelp.org. I used pulsed antibiotics for a period of 6.5years and no longer have chronic fatigue syndrome.Many people with Parkinsons have used the same protocols which much success
I use lots of fermented foods in my diet so my gut did not suffer
Low dose antibiotics and antimalarial herbs helped me too. Until I got pushed over my threshold with mold exposure. Then they quit working. On a path of detox at the moment, still addressing protozoa but herbaly at the moment. Starting to sense improvement.
Issie
Issie I rid myself of the fatigue but still have fibromyalgia as does my mother. I’ve done 23andme genetics but nothing really illuminating there but can’t really get anyone to comment on it that knows what they’re talking about.
Wishing everyone much auspicious prosperity in 2016 and always
Did you run your gentic data through Promethesis or one of the other detective/determinative sites? I have FMS, DXd at Mayo. But I also have EDS, also DXd at Mayo. I wonder if they may be one and the same with some subset types. EDS genetic data was indicated in my 23&me report.
Issie
Your recovery from using antibiotics is encouraging. I just took azithromycin for a cough that lasted a couple of months. I felt pretty horrible on the drug. But now that I’m done, I’ve had 3 pretty good functional days. I’ll look again a cpnhelp.org. Thanks for reminding me about it.
Rach, the most likely reason who felt awful whilst taking azithromycin would be that you were herxing. Zith is a major antibiotic in both Marshall Protocol and Chlamydia pneumoniae protocol along with covering up from the sun. All in all, the first 2 years were very challenging for me but I persisted as I understood what was happening and came out the other side.
So many people with chronic illnesses in one form or another, make great researchers and are persistent and determined. Onward ho……..
Jude, hi to you over in Australia! It took me a while to realize that maybe I was Herxing with the azithromycin. What I’m worried about now is whether there is any way to figure out whether 5 days on the antibiotic did the job or whether there are still chronic infections in me that need long term treatment. Years ago I had a test for chlamydia pneumonia – one doc said I had it, the other said no. How did you know what you had and how long you needed to treat it? I still have the cough that got my doctor interested in giving me azithromycin. It’s about 70% better. I don’t like the idea of killing off the weak bacteria and then letting the sturdy one go on to replicate inside me. Suggestions?
Rach, I can confidently state that after such a short dose of Zith, you have only just started on killing off bacteria. For me, it was 6.5 years of pulsed abx that did the trick for fatigue. You can read more on Bacteriality.com or chronicillnessrecovery.org or on cpnhelp.org. It’s necessary to cycle through quite a few abx and antifungals to cover as many bugs asas possible. I find tests quite useless for stealth infections. Lida Mattman has passed away since this video, but it’s worth watching
https://www.youtube.com/watch?v=WozrCFW0mRM
The links you listed are so different than anything I’ve ever read in regard to the immune system and Vit D. Since I haven’t researched this protocol , I was wondering what the idea is behind this? Since you’ve done this for over 6 years, I’m sure you may can explain why this protocol suggest this. Everything I’ve ever read says it’s so important to have your Vit D levels up to keep your immune system working optimally.
Issie
My mom and her aunt have Lewie Body Disease (2nd most prevalent dementia) and although led otherwise healthy lives did both get this disease AND it has Parkinsons like symptoms.
My mom’s sister (she has 5 and 1 brother) and her son have Fibro (I do too) and I also have CFS.
So, who knows. I do believe Fibro and CFS will turn out to be genetic.
Looking back on my mom’s life, we think she had Lewey Body dementia too. She started keeping meticulous notes in her early 50s. She also had MCAS and EDS. Hoping if this is genetic, and we can avoid with lifestyle changes – maybe genetic changes won’t flip on. I’m trying to tweak my body as best I can so that known illness and genetics stay at bay. Epigenetics is fascinating.
Issie
About 5 years ago I noticed that I was having a problem with my gait. At that time I was experiencing longer good patches of milder symptoms and had more energy for activity. I realized that I had a limp that seemed to appear from no where. If I concentrated on how I was walking I could improve my gait a bit. It has felt like I have forgotten how to walk normally at these times, yet by focusing on my walk I can override this problem. Then as soon as my concentration shifts the limp returns.
You’re probably using your prefrontal cortex to do things that your brain used to be able to do without your thinking. Check out Doidge’s recent book and the Parkinson’s story for more. I will probably do a blog on this at some point.
My neurologist at Mayo, is of the opinion that many of us POTS patients have mitochondrial issues. He has tested some markers known to indicate mitro issues and they have been negative. But he still feels not all test can determine everything. There are some things that there is not YET testing for. There are times when I can barely get up out of a chair, my leg muscles are so weak. I may have tremors in my limbs and that causes weakness. One leg, at times, seems to drag. And if I’ve over walked my feet don’t want to pick up. He has us using what he calls a mitro cocktail. CQ10 is one of the things he suggest we use. Also, when it was thought I had Parkinson, my neuro doc then put me on high doses of CQ10. She said it would stop Parkinson and it should not progress if I kept taking the high doses. There must be a connection. I too swing from the hip when I walk. But I think it’s a stability issue. Having EDS, my kneecaps can displace. I think walking more stiff legged is a compensatory thing to be more steady. But this also happens in those with dementia. They get real unsure on their feet and walk with short shuffle steps. The latest findings is a possibility of mold issues with dementia. My having a recent exposure to mold, my legs are really weak and my tremors more pronounced. But my detox protocol should help all this and today I felt better and feel hopeful.
Issie
Issie, when you say high doses of CoQ10, how high did your doctor recommend? Years ago I discovered that 300 mg of ubiquinone got rid of my restless leg syndrome on the first day I took it. Lower doses didn’t work at all. And ubiquinol didn’t work either. For reasons unknown, the 300 mg CoQ10 also got rid of most of my food allergies.
Issie
thank you so much for sharing your medical info with us.
It was like a light bulb went off..reading
your post..
my husband has a tremor started in one arm and at times his head will shake some
He also is slowing down and I asked the internist 8 months ago could it be parkinsons?
He did not seem to think so but no answers..
so your doctor at mayo and your case has taught me something new today…
I have undiagnosed orthostatic intolerance for the past 30 years…and cfs and fibromyalgia and multiple chemical sensitivity illnesses as well as my husband has the same ones too.
I want to mention that I walk from the hips and I walk very fast and have for years..
because once I slow down walking or even stop to stand still I look for a bench or place to sit down
or go get in the car and sit down if not too hot of weather..
Same way in our house..
I have to look for a place to lean on a door or sit down if I do not move fast..
I would like to know if you can give us an idea of how much CQ 10 per day and times of day was suggested to you …
did CQ10 make you hyper or nervous at all?
Can you tell us what else the doctor told you for the cocktail you take with the CQ 10 and dosages?
Thanks,
Sheri and husband Larry
USA
I’m so glad you had a light bulb moment. I love when that happens, a piece of the puzzle comes to light.
For me, having HyperPOTS, I found to much CQ10 to be over stimulating at the 1200 dosage. Since I didn’t have Parkinson and did not have low dopamine. However, it may be very beneficial for those who do have those issues. Yet the latest research I’ve seen debates that. But I do find that CQ10 is a necessary part of my weekly supplements. Though I don’t take them daily.
The DXs that I have been dealing with over the last few years have been what has made the most difference for me with my addressing them. It was found that I have coinfections related to Lyme and also Protomyzoa Rehumatica – another malaria type protozoa that can be gotten from mosquitoes. In addition, I have autoimmune issues that play a part in this. If my autoimmune system were working correctly it would hold these things in check despite them being in my body. My doctor has found this protozoa in those who do not appear to have health issues, but for those that do – trying to tame them makes a difference. For 2 years I was on low dose doxycycoline and antimalarial herbs and enzymes to break down the biofilm they create to protect themselves. These biofilms attach to the walls of veins and not only can hinder blood flow, but interfere with expanding and contracting of the veins. (This is the feelings of my doctor and the bioscientist who discovered this protozoa. Dr Stephen Fry of Scottsdale, AZ. He also thinks this may play a part in POTS. He also released a paper in London showing this protozoa discovered in the plaque removed during heart surgery.) Based on my response to his protocol and changing my diet (I had to become a lower fat, whole food, Vegan) I got considerably better. Then I had a mold exposure of over a year and my system couldn’t handle that additional burden. My antibiotics quit working and 3 others we tried didn’t work either. Then I started addressing the mold issues and doing cleanses of varying types, addressing mold, yeast, parasites and heavy metals. I’m also addressing the protozoa with herbals now. What I’m doing is starting to make a difference. Things are turning around. I’m starting to feel better. One thing to note, when I had these protozoa under control, I still had POTS, but at times it was not detectable. My tremors were better, energy greatly improved, and I felt I finally had a measure of health back.
I also discovered that I MUST address MCAS or everything is worse.
Hoping this helps someone else find their puzzle pieces. We all can be similiar but may be very different with our subset types.
Issie
One thing I learned from Travell and Simons is that one should rule out trigger points as a cause or contributer to one’s symptoms first before one looks further for an alternative diagnosis. Altered body mechanics can definitely influence one’s gait. I have weak ankles and have always had trouble with dancing with my hips. Treating my hip muscles has helped a lot in that regard and also I have to do a lot of work on my calf muscles. My upper body is worse now and it seems like the disease started in the feet, worked its way up and may end up in my head,LOL!
You gotta be talking about severe or moderate to severe ME/CFS here, because I can’t relate to any of this… I have always walked faster than practically any of my friends, and last summer I found that after having done no exercise at all during all winter, I went on a mini-hike with a healthy friend and still walked considerably faster than her! (I was under the influence of bee venom, without which I wouldn’t be doing any exercise though). My CFS doc – a very clever one – says that people with CFS are naturally inclined to exercise more, because they need to compensate for the dysfunction of the sympathetic nervous system (meaning reduced blood circulation, etc.).
Probably…I’ve been the same way. I saw a video of me escorting my grandmother down the aisle at my sister’s wedding. I just blasted down the aisle dragging my grandmother with me!
I’ve always done everything fast, fast, fast – including walking even though I’ve always been tired, tired, tired.
it’s kind of bizarre!
“Combining a Parkinson’s drug called L-dopa with a non-steroidal anti-inflammatory drug completely eliminated any signs that an animal model was feeling pain.”
******************
Makes sense to me…for years I have been taking a small amount of tyrosine in the morning to raise dopamine levels and a ASA – aspirin in the afternoon to alleviate pain; rid myself of flu-like symptoms and improve my mood. You need a dopamine source in order for certain pain meds to work, otherwise it’s like trying to run a car without fuel.
Issie, when you say high doses of CoQ10, how high did your doctor recommend? Years ago I discovered that 300 mg of ubiquinone got rid of my restless leg syndrome on the first day I took it. Lower doses didn’t work at all. And ubiquinol didn’t work either. For reasons unknown, the 300 mg CoQ10 also got rid of most of my food allergies.
http://www.life-enhancement.com/magazine/article/782-coenzyme-q10-slows-parkinsons-disease
Here’s an article on how much CQ10.
Issie
Thanks for the article on CoQ10 dosage for Parkinson’s. I now remember reading that 1200 mg was used. I wonder if this means that that huge dose would be helpful for ME/CFS. Right now I’m taking 300 mg ubiquinone, 100 mg ubiquinol (both from Life Extension), and 10 mg of MitoQ, a new variation on CoQ10 that is supposed to be over 800 times stronger – it has been engineered to be able to pass through the membrane around the mitochondria. Maybe I’ll experiment with more CoQ10. I hate my new tremor, mild as it it.
Thanks Issie and Rach for these commebts about Q10. I read aboutthe benefits of it for healing the gut. Ive been inky taking the recommended daiky amount of 150mg. I will increase to 1200 today. I have terrible MCAS issues – was my first symtom of severe/acute ANS dysfuntion 1.5 years ago.
I imagine the decrease in food intoletances youre experiencing is directly related to its mitochindria healing functions . As the gut lining heals, less food can leak and wreack havoc in our bodies. I am experiencing a massive allergic reaction to Tramadol. Knew i was allergic to Codeine, but after taking this synthetic form a few times, the reaction is fully out of control inflammed and hot and itchy. Antihistamines dont seem to work and even maybe make it worse. I read something about that last night – so stopped taking the antihistamines.
I’m only getting the information confirmed recently that in addition to FMS/MCAS/ME, it appears that my symptoms are strongly indicative of PD. I am having a seriously difficult time coming to terms with this. I havent even fully absorbed or accepted the ME/CFS diagnosis.
But anyway im gkad to hear Q10 helpsPD as well.
Ive also recently come across a drug called Rasagiline, which pretty much halted the progression of motor related symtoms, including dyskinesias and gastro intestinal issues. I may not have intertreted this correctly so here is the reference to the online study (I couldnt copy tgr kink): Pistacchi, M., Martinello, F., Gioulis, M., & Zambito Marsala, S. (2013). Rasagiline and rapid symptomatic motor effect in Parkinson’s disease: review of literature. Neurology and therapy, 3(1), 41–66. https://doi.org/10.1007/s40120-013-0014-1
I took tyrosine for maybe 6 months a while ago. It put me in a great mood. Then it stopped working. When I tried it a long time later, it gave me chest pain. After doing some research I found that it can cause spasm of the coronary arteries. So no more tyrosine for me.
I have tied several pedometers but none of them will record my steps. The 1st three were $30 range. Then I bought am UP band (about $125) and it didn’t register even while stomping feet and swinging arms. It worked fine for my husband and daughter. Then I put it around my ankle and finally registers steps. Anyone had a problem with pedometers?
Interestingly, my father had Parkinsons. At 97, my mother had symptoms of Parkinsons.
Hmmmm
It seems to me that Parkinson’s might be part of a spectrum disorder that includes FM and ME/CFS (or at least some cases of it). The very first thing that I noticed when I was coming down with FM was that I couldn’t sign my name in a fluid way at the bottom of a check. I looked at the scrawl and I tried again… no dice. That night at home I really practiced, and I realized that no matter how hard I tried, I couldn’t get my fine motor skills to cooperate. At the time I just thought, how strange. Since then my handwriting has gotten worse and worse. Thank goodness for keyboards.
I didn’t start experiencing tremors until I got ME/CFS a couple of years later. And now I have tremors only if I overdo. Then I have them, and there’s nothing I can do to make my hands be still except complete rest for a good while (sometimes several days).
I take carbadopa/levodopa nightly for restless legs syndrome, and it has made the symptoms of RLS go completely away. I’m grateful for that and wonder, after this blog and your comments if I would be less able to walk if I weren’t taking the levodopa. I actually like to walk a little bit fast. I have orthostatic intolerance, so I think walking more quickly helps keep blood pumping to my brain. I can’t stand still for five minutes without feeling really ill, and many days I have trouble sitting up. I’m definitely most comfortable lying down.
We don’t have Parkinson’s in our family to my knowledge, but I’m pretty sure my mother and brother have FM.
Thanks for the blog, Cort, and to each of you for your comments. I always learn something here.
I’ve had ME/CFS for 8 years and only recently noticed a tremor and a deterioration in my handwriting. I’m a realist painter, so I’m not too pleased with the loss of fine motor coordination. And I’m not too pleased that ME/CFS might be related to Parkinson’s. The neurological symptoms feel kind of freaky to me.
I had a recurring tremor about ten years ago – which ended up going away completely. I don’t think people with ME/CFS will end up looking like Parkinson’s but I wonder if something similar is causing the fatigue and other problems.
I think it’s something to consider. You never know. I guess time will tell. Walking faster does help activate the muscle pump in the legs which keeps blood from pooling down there. I tend to walk faster as well and if I stand still I tend to move around.
Cort, I used to walk really fast with a long stride. My hubby and I could out walk nearly everyone. This was in my younger years. But now, I’m much slower.
I too have to keep moving. I fidget all the time. I’m moving my legs usually. Very necessary for blood flow. I think you said you are tall, like me. Us tall ones seem to have even more issues as there is a lot further that our blood has to flow with gravity pulling things downward. We tend to have more vascular issues. That’s one reason why most POTS people are better with their legs up. But as for standing in a line, let’s say – we learn to move around without breaking into dancing a jig and try to keep our legs moving. It does help with blood flow and may prevent a faint. Some find if they feel “one” coming if they can get a chair or sit on the floor before slamming to it – we can recover after a bit to keep going. But if there is a long line and having to stand still – forget it. Sadly, we look perfectly well on the outside. Yet we have disability plates. My hubby can see me beginning to have a “spell”, before I even know it’s coming. I lose all my color. At times I may have a hot flash and right before that is a nauseating headache with severe anxiety. All signs of MCAS and POTS.
Once a person knows the why of things, it takes the fear out of it. Then we just have to learn how best to cope with what we get handed. But I never give up looking for the best “purple bandaid” I can find. I think I’m close to having it. I feel encouraged.
Issie
Well said Issie!
I am just reading this info today–August 31, 2016. I was diagnosed with fibromyalgia in 1994, then in 2015 with Parkinson’s. I have been tired for years! It would be interesting to see some research on how many folks with FM end up with Parkinson’s.
I am very confused. I am fast walker but I also have extreme fatigue even when I am sitting down. My walk does a lot of left to right and back again along with forward. I am constantly bumping into walls, street signs etc. The only way that I can avoid it is by forcing myself to walk slower and really concenrate. I have a tremor so I asked my neurologist if I had a whole lot of tests if I had Parkinson’s or not. He just stared at me. He has also said that I am too complicated for him and I need to see someone at university hospital. Can’t because it is so far. so I know something is not right but not sure what it is.
Too complicated for him – ha – who has not heard that before? We just don’t fit anywhere really well…and puzzle just about everyone…
Each muscle movement is controlled by the central nervous system which decides in what order to recruit muscles to perform a particular movement. Myofascial trigger points affect the recruitment order of muscles and force the CNS to develop a new order so that the movement can be performed as smoothly as possible. I dare say that if trigger points are present in early life, we will learn movements incorrectly. Please refer to Devin J Starlanyl and John Sharkey’s book Healing through Trigger Point Therapy, A Guide to Fibromyalgia, Myofascial Pain and Dysfunction.
Cort, as you well know, I am a long term believer in the CFS problem lying in the brain / CNS.
I’ve been reading a lot in this area, and trying to reconcile my thoughts with some of the great research done by Hornig, Lipkin et al.
They found strong evidence of immune activation in the first 3 years of CFS, which then subsided.
My view is that this early onslaught primes activation of microglia, and neuroinflammation. The profound peripheral immune activation goes away, but the neuroinflammation is left behind.There is quite a lot of non-CFS research that supports this theory.
So, ultimately, researchers need to work more on neuroinflammation in CFS. There are real treatment options potentially available, so I am quite hopeful. And several researchers seem to be now focusing on this much neglected area. Here’s hoping.
Further to my last comment. There is reasonably good evidence that pregnant women with CFS often get better during pregnancy.
Perhaps it is the estrogen dampening microglia activation?
http://www.pnas.org/content/110/9/3543.full.pdf
Hi Cort
This is yet another intriguing post. Thank you.
I don’t have the tremor, small handwriting, delusions, difficulty swallowing or quiet speech. I hadn’t realised before that you don’t have to have the tremor to have Parkinson’s disease, so I had never considered it.
However I do have a weird gate, stiffness, shuffling, slowed movement and stooping which I mostly manage by using walking sticks, walkers or an electric mobility scooter. I have long had orthostatic intolerance, sleep and cognitive difficulties, restricted arm range of movement, loss of smell and alternating constipation and diarrhoea as well of course as fatigue. I recently developed stiff and painful muscles, a persistent cough, especially after I have drunk anything from anything, difficulty getting up from sitting, metallic taste and mouth dropping open day or night. I also suspect I’m developing the masked face. I also have the freezing when walking/trying to move my feet. This would seem to be a moderate case.
Some of these symptoms overlap between’s CFS and PD. It makes me wonder whether I now have both. My GP has given me a referral to a neurologist for an assessment.
I also have PEM and I haven’t heard anything about this being present in PD. Has anybody else?
I also have MCS and osteoarthritis and so was putting down some of the strange symptoms to those.
I have read Doidge’s chapter on Parkinson’s and wasn’t helped by the suggestion of extensive vigourous exercise in walking fast. However Amazon have a Kindle book by the proponent of the method, John Pepper: reverse Parkinson’s disease as well as another walk on using tai chi: by Daniel Loney and somebody. I will read these in the hope of finding something not too energetic that I can do to improve the movement issues.
I wonder if any readers have explored a Parkinson’s disease possibility as well as their CFS or FM?
Alexa
Good luck Alexa! yes, the exercise prescription for Parkinson’s disease is different in Doidge’s book but note that the Parkinson’s patient was able to stop the disease from progressing by focusing his attention on the muscles he was working. Doidge believes he’s using his prefrontal cortex to do what his brain can no longer do – move his muscles properly – that’s the key I think. I imagine the same thing may be happening in Tai Chi.
Good luck and please tell us how it went.
Hi
I saw my neurologist last week. After examination, he denies Parkinsons, which is a relief, blaming much on my two bad osteoarthritis hips.
My sense is that the diffuculty initiating movement, jaw dropping and now occasional trwmor when pouring may be very early signs, so I’ll keep an eye on it.
Thanks for posting on this.
Get checked for Lyme and coinfections, protomyzoa rehumatica, and mold. It can also be connected to POTS. My first wrong DX was Parkinson. My final DX was all the above. Tremor is still there, but improving with treatment. Still have muscle weakness.
Issie
I am curious, how many with FM/CFS had Mono when they were younger?
Based on anecodotal reports it appears that a substantial number probably did. I don’t think we have any hard facts on that, though.
I had mono when I was in college. Then a few years into CFS I tested positive for chronic active Epstein-Barr.
To answer your inquiry, I had mono about age 18, my first year at college.
I did.
I had mono Twice. About 20 years ago. And then chronic active virus, which i take famciclovir daiy for past 15 years. I read somewhere (dont remember where) that increasing medication may dampen down tge effecrs on the CNS and therefore ME. But my dr won’t go for it. He’s a less is more kind of guy. Maybe if i can find the science again add bring it in to show him…
Mono was the absolute start for me. I had severe Mono in 1984 that never went away. I was tested and show high, chronic reactivation of Epstein-Barr. I gave Mono to my ex boyfriend 3 months after coming down with it. Even weirder, I gave Mono to my then boyfriend a full year after I had it.
After my Parkinsons Disease diagnosis, my neurologist was very open when looking at alternative medicines and procedures, with his guidance i started on natural PARKINSONS DISEASE TREATMENT from Rich Herbs Foundation (ww w. richherbsfoundation. c om). The treatment worked very effectively for my Parkinson’s, most of my severe symptoms simply vanished within the first 3 months on the treatment, i feel better now than I have felt in years and i can feel my strength again.
My doctor and I have discussed this very issue. FM/ME/CFS, MS, and PD are all related. There is also a link with ADHD. A gluten-free diet has been shown to benefit all of these diseases, and untreated Celiac Disease is possibly a factor.
The fact that the only FDA- approved medications for FM/ME/CFS are antidepressants and antiseizures is infuriating. There is NO clinical data to support their use, and in fact, can cause FM/ME/CFS to progress into Parkinson’s.
I have been conclusively diagnosed with Fibromyalgia. The FM/a blood test is now going through clinical trials, in which I was a participant, to get FDA approval for conclusively diagnosing FM with a simple blood test.
Here is the link: https://fmtest.com/
Anyone who tests positive is eligible to participate in a groundbreaking clinical trial that actually targets the biological cause of FM. This is not Big Pharma. But rather a company run by a doctor who was a pioneer in diagnosing FM, with the goal of curing the disease, not masking symptoms and causing others.
Antidepressants and anti-seizure meds cause Parkinsonism for me. Some of the effects have not gone away, and I am now taking Ropinirole, which is a dopamine agonist commonly used to treat PD. The similarities are just too great to ignore. My mom had the same problems with the meds as I do. She was diagnosed with FM/CFS before anyone took it seriously. She died at the age of 69, with her doctors suspecting MS and/or PD.
Everyone who tests positive for FM should also be tested for MS and PD. An MRI of the brain is necessary. If lesions are present, then a lumbar puncture is necessary for the differential diagnosis between MS and PD. If no lesions can be seen, then a lumbar puncture should still be performed as sometimes lesions are not visible on an MRI, and there is still evidence of the immune system attacking the myelin sheath in the cerebral spinal fluid.
In the Canadian study, 75% of people with multiple sclerosis had a previous diagnosis of fibromyalgia, and met the diagnostic criteria for both diseases. In another study in the United States, 53% of people with fibromyalgia received a conclusive diagnosis for multiple sclerosis within 7 to 10 years.
My doctor has multiple patients with fibromyalgia and multiple sclerosis and Parkinson’s. He says they are most definitely related, And it is not yet known exactly what factors make one progressing to another or coexist with another. I suspect there is a gluten intolerance component, Both my mother and I were/are gluten intolerant, and I am going to be tested for celiac disease. Changing to a gluten-free diet has noticeably improved my symptoms in just a month.
My dad was diagnosed with Parkinson’s at the age of 40. I started with fibromyalgia/chronic fatigue symptoms aged 50. I am waiting for a formal diagnosis but unfortunately I ‘tick all the boxes’ . My symptoms are different to my dads his were more shaking, movement problems and mine are chronic fatigue, headaches and aches and pains. I do wonder though if there is a genetic link especially if they are both neurological conditions?
C 25 years of illness. When I hope that I am winning, I fail again, I am the beginning. I can’t stand sitting, I have to help myself by lying on the bench at the doctor’s office, which upsets others, my husband and even the doctors. I have nothing to live for.omment *In the Czech Republic, a hopeless situation, if I have both legs and arms, I have no chance of help or medical aids. A wheelchair would help me, but I can’t ask, “because they say I can stay on my feet at home.”
If I don’t sleep even during the day, the brain falls asleep by itself and doesn’t work, then I have trouble thinking and speaking.
I’m a caregiver for my HWP. I wanted to inform PWP that there’s hope, the Uine healthcentre . com has been of great help. the PD-5 treatment programme they offer has completely help with reversing my husband Parkinson’s symptoms.
Really interesting, l’ve had fibro known since 1969 from an osteopath and Myalgice Encephalomyelitis since flu March 1971 and l never recovered, it was coxsackie B3 and B4 in blood tests , told it was a wild strain of Polio with flaccid paralysis. And multiple food and chemical food and sensitivities.
By 1984 finally diagnosed, bed ridden in The Nightingale Hospital, Marylebone, London, gravely ill, had fits, gone blind twice. Was a ward sister S.R.N. S.C.M, dip: I.I.H.T.T
Wished l was well enough to travel for the conference, but only out of bed nowadays in the mornings, age 86. Ran several M.E groups in different counties in the 1980/90’s
My partner has Parkinson’s disease, adding PD-5 to her nighttime Parkinson’s meds has completely changed his sleep issues. He slept all day and up all night, we had to hire care nurses. Now using this PD-5 medicine for the last six months and a normal routine he sleeps almost completely through the night, and may get up once to use the restroom. It’s improved so much we were able to let go of the night nurses. This medicine also helps a ton with memory. we got the treatment from Uineheath centre . c om
Wow!
In 2019, I began experiencing symptoms of Parkinson’s disease, which was diagnosed in 2021. Among my severe symptoms were tremors, difficulty walking, weakness throughout my body, and stiffness and rigidity. My meds only provided temporary relief, so I decided to try other approaches and started the PD-5 treatment program from uine healthcentre . n et. With the help of this therapy regimen, I have seen significant improvements in my walking, balance, tremors, and appetite, and I have resumed my guitar playing again and sign my name as I had before Parkinson’s. I am incredibly grateful.
For 7 years, I suffered from Parkinson’s, tremors, stiffness, sleepless nights, and countless failed treatments. Nothing from the medical field helped, and I was losing hope. Then I found EarthCureHerbalClinic.com. Skeptical but desperate, I began their holistic healing program. Within weeks, my symptoms started improving. Over time, they completely disappeared. Today, I’m symptom-free, no more tremors, no more fatigue. EarthCure didn’t just treat me, they healed me. I’m beyond grateful. Thank you, EarthCure Herbal Clinic, for giving me my life back.