



The Solve ME/CFS Initiative’s webinar with Dr. Avindra Nath revealed again what an extraordinary study the NIH intramural study at its Clinical Center is. The Webinar started off with Dr. Nath stating that it was “such a pleasure” to talk to the chronic fatigue syndrome community, and that he was “absolutely thrilled” to be leading the study.
His interest in ME/CFS derives from the patients he’s seen over the years (he’s an MD) as well as patients he’s seen in the multiple sclerosis clinic. Fatigue, he noted, is the most disabling part of MS. Given the immune nature of that illness as well as the effects MS drugs can have on fatigue, he thought it was likely there was an immune basis to ME/CFS. (He also mentioned the Rituximab studies.)
On that note be sure to check out, if you haven’t, one ME/CFS patient’s rather amazing response to an MS drug: A Chronic Fatigue Syndrome/POTS Patient Responds to a Multiple Sclerosis Drug – What Does It Mean?
Note that this is Nath’s study. He’s the one Collins came to and he’s the one putting the study together. Because he’s a neuroimmune infectious specialist the study is heavy on nervous system and immune studies but because he also studied the literature (and talked to a lot of people) there are also exercise, autonomic nervous system and other components.
Given Nath’s background it’s no surprise that the study focuses on people with infectious onset. He believes an infection triggered the immune system to whack the brains of ME/CFS patients.
Nath didn’t put together just a study, though; he put together a three phase series of studies that he hopes will end up in treatments for ME/CFS. In its duration and scope the NIH study resembles the Open Medicine Foundation’s End ME/CFS project.
Both first deeply examine a limited number of patients, then test their results in a larger number of patients, and finally both envision finding/developing immunomodulatory or other treatments based on those results.
Both, furthermore, are employing top researchers and cutting-edge equipment at two of the top research centers in the world. Nath repeatedly pointed out in his presentation that several of the techniques being used were developed by the researchers who are actually participating in the study. Ron Davis’s lab at Stanford has developed many new technologies over time. During the course of his interest in ME/CFS he’s created a new, more accurate (and cheaper) way to study HLA genes.
The Nath Study
Note that the NIH could have played “small- ball” as they reinvigorated ME/CFS research. They could have brought in a junior investigator, had him do a couple of tests and pronounced themselves done. Instead they brought in one of their top investigators who put together not one study but a series of studies that will take probably five years to finish and could culminate in a treatment or treatments.
Metabolic and Brain Studies
Each patient will stay overnight in one of the NIH Clinical Center’s “unique” metabolic chambers. These chambers, which were developed at the NIH decades ago, have been refined over time and allow for extremely accurate measurements of metabolism and energy production. Nath said the overnight chamber stay will provide an enormous amount of information on energy use in ME/CFS patients.
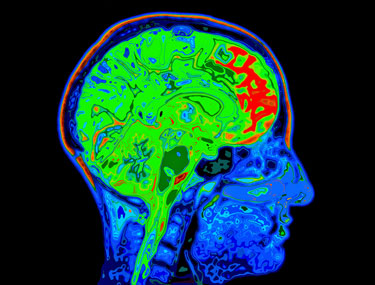
Two different MRI’s will be employed in the study; one looking at brain structure and one looking at brain functioning
The functional MR’s will measure blood/oxygen and glucose uptake (metabolism) flow during tasks. These will be able to tell what happens to ME/CFS patients brains when they’re stressed. Will their blood flows be adequate? Will different parts of their brains be activated? Will they produce normal metabolic activity?
Then comes another brain scan called transcranial magnetic stimulation. TMS is mostly used to evaluate a key topic in ME/CFS – the ability of the primary motor cortex in the brain to move muscles. It’s widely used to evaluate brain damage occurring in multiple sclerosis, stroke, ALS, movement disorders, etc.. Nath said he was excited that world class experts who developed this technology were participating in the study.
Autonomic nervous system testing is, of course, of huge interest in ME/CFS, and it was another area in which Nath reported that “world class experts” would be working.
Immune System
The study will compare immune factors in the spinal fluid and the blood – something that’s rarely been done in the same patients. First, they will look at 1500 cytokine and immune factors, and then they will do flow cytometry.
Those two studies will set up what comes next as they double down on abnormalities found. If B-cells pop up they’ll dig deep into the B-cells. If NK cells pop up they’ll do the same with the NK cells. Any unusual immune pattern Nath finds, he’s pretty sure he can find an expert to study it given the size of the immune intramural team at the Clinical Center.
They will look at antibodies (autoimmune factors) in his lab and cerebral spinal fluid proteins, where they have, yes, more “world class” experts who will do the analyses. Nath will later say his lab has developed a unique way of examining autoantibodies.
Nath said the immunology program at the Clinical Center is the largest and best in the world and they have a wealth of information from other diseases they can compare to ME/CFS.
Gut samples will be sent to Ian Lipkin and they will examine viruses as well.
The Solve ME/CFS Initiative’s Avindra Nath Webinar
The Woo- Woo Section: Getting at the Central Nervous System Problems.
The most cutting-edge, woo-woo type type of inquiry comes in the nervous system side of the study. Referring again to a technology developed at the NIH – in fact, in his laboratory – Nath will take blood cells and transplant them into pluripotent stem cells and then make neurons out of them.
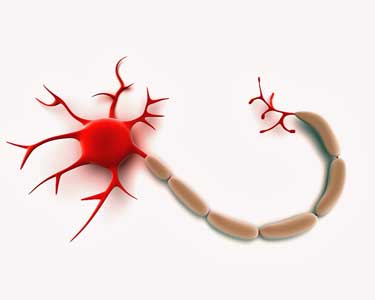
In one part of the study Nath will use stem cells to grow ME/CFS patient neurons outside of their bodies
That will allow him to grow ME/CFS neurons outside their body and test them. Interestingly, he referred to the possibility of uncovering mitochondrial dysfunction in ME/CFS patients neurons but he said many very sensitive kinds of tests can be done to determine what might be going wrong in these neurons. For instance, he can add blood samples to the neurons to see if some sort of factor in the blood is tweaking them. If a factor is present he’ll identify it and it could provide a prime target for a drug.
Nath considers this a critical component of the study.
They will also create “humanized ME/CFS mice” generated from immune cells that are added to them. They will test them in several ways including using cerebral spinal fluid and/or antibodies and determine if they become fatigued, etc. If they do they will try to determine what in those substances is making them fatigued, etc.
Study Population
Nath reiterated that one of the tests used for the Reeves criteria will be used to quantify symptoms but that the criteria itself will not be. Instead, the study participants will have to meet the Fukuda and Canadian Consensus Criteria, have a documented infectious onset and have the disease less than five years.
From his perspective there is no such thing as a perfect criteria for ME/CFS (or other diseases) but that’s not a problem. He expects to be adjusting the study as he goes along. If a phenotype (version of the disease) stands out, he’ll hone in on that, and noted that it can be helpful to identify outliers. Disease heterogeneity is something the intramural center deals with all the time, he said, and we’re very good at it.
(In truth, though, this study is set so that there won’t be many significant outliers – as least from outside the ME/CFS patient community. The participants will be coming from ME/CFS expert physicians and they’ll have to meet the criteria.)
Nath went over some of this research including studies on numerous viruses including Ebola and now the Zika virus. He said he comes from a virological background and that he’s an expert on the clinical side in neuro-infectious diseases and neuro-immunology, and in basic sciences as well. He said not many people have that kind of background. He believes that his team has a special kind of expertise that could be valuable for working on ME/CFS.
He noted, though, that his goal is to find something in ME/CFS and pass it on to other researchers. The study design he emphasized is quite flexible and the study ultimately will evolve depending on what they find.
Everyone except for a few full time people everyone else (about 150) are lending their time in addition to everything else they are doing.
Push Back
The scrutiny that some researchers have come under, however, has caused, he said, “a lot” of people to say that they don’t want their names associated with the study, and some have even said they don’t want to have anything to do with the study. He also said some researchers who haven’t said no, are apparently saying no by not answering his emails.
He suggested that patients “be a little careful” about how critical they become for fear of antagonizing people who might not end up helping. The criticism of Brian Walitt and perhaps Dr. Saligan, Gill and even Dr. Unger has apparently gotten around, and it’s had the unintended effect of making some of the researchers we do want working for us leery of doing so.
Nobody can be forced, Nath said, to study a disease – it doesn’t work that way at the NIH. Plus these researchers are already busy and are volunteering their time; i.e. they don’t have to participate in this study. .
. Someone recently related to me that some researchers she was interacting with were at first shocked at the intensity of the ME/CFS community. That rang true. We’re an unusually intense group. I don’t think we realize how intense we probably are relative to other groups. I imagine that critique’s of study members are almost unknown at the Clinical Center. I imagine that very few patient communities know about, let alone research the backgrounds of the investigators involved in studies done there
Of course, this is a huge moment for us. We’re hopefully on the cusp of finally making it in the research world and everything is under scrutiny. We don’t have the history of successes that other diseases have. This is our big moment. I hope Dr. Nath and the other researchers understand that, and we should understand that we are probably subjecting him and the NIH to stresses they’re not used to as well.
I remember Suzanne Vernon saying the researchers aren’t the most social of animals; they do, after all, spend their lives in labs. They’re probably not used to their comrades being questioned.
The NIH has been responsive. Dr. Nath has been quite gracious to talk to the ME/CFS community, now three times. (I wonder if he’s ever done that with a disease group before?) Lines of communication are being set up that I imagine are new to both him and the NIH as well. They didn’t get rid of Walitts but they did axe one problematic study.
With regard to bias, Nath reported as he has before, that the solution to potential bias is to design a study so that bias is not an issue. It’s not possible, he argued, for him to do a litmus test or reach inside everyone’s head and understand what they are thinking. He has produced a study, though, which he believes eliminates any potential problems with bias.
Nath handled, I thought, a touchy situation well. The push back is something to think about. The one potentially psychological aspect of the study – the functional disease subset – has been dropped at the patient communities request. You wouldn’t know it from some of the objections, but all the research being done in this study is pure pathophysiology with a strong neuro-immune thrust. Because no portions of the study are examining psychology or psychosomatism there’s no literally no way available to inject those into the study.
Ironically, none of the individuals that have been objected to are doing any research in the study, yet the criticism against them appears to be effecting the participation of researchers we do want in the study.
Questions
Bias – When asked about preventing the personal biases of the investigators effecting the kinds of patients entering the study Nath said he’s gotten this question many times. Again, he said that because you can’t eliminate bias you design a study so that bias is irrelevant. He did that by ensuring that the patients first come from ME/CFS experts, then by ensuring that they meet the specified criteria at the site.
He anticipates that if they get a patient or two that doesn’t fit, that that will become obvious quickly.
Somatization – When asked to clarify the problem of somatization (i.e. Walitt) Nath repeated that he’s only interested in neuroimmunology, that he doesn’t know anything about psychosomatic illnesses, is not interested in them or learning about them, and again, he’s designed a study that doesn’t pertain to them.
Exercise Testing – With regard to 2-day exercise testing and using the Stevens Protocol Nath said his goal was basically to get his patients fatigued and see what happens. He appears to regard exercise testing mostly as a means to an end.

Nath isn’t worried about a patient or two without ME/CFS slipping through. He believes he’ll be able to quickly identify them if they do
Consulting With Outside Experts – When asked whether the NIH would be open to consulting with experts in that area? Dr. Nath said he will begin a seminar series that will bring experts in. When asked if he had contacted Dr’s Fluge and Mella, Dr. Nath sounding a bit peeved now, stated that if he finds what they have found (or perhaps what the Rituximab study suggests they have found; i.e. problems with B-cells) he will do a much, much more extensive study than they have the capability of doing. He did say he would contact them about giving a presentation to the study team.
Autoantibodies – Will he test for the adrenergic and muscarinic autoantibodies recently found in POTS patients? Dr. Nath said he has developed a unique way of examining autoantibodies for the brain which involves mass spectroscopy. If there’s an autoantibody to anything in the brain he’s confident they’ll be able to find it.
Timeline – When does he believe the timeline for phase I will end? With 80 patients it could easily take two years.
Patient Participation – Dr. Nath said he was very eager to have patient participation but it’s turned out to be much more complicated than he thought (which patients, how many, etc.). He said they will use the participation structure the extramural (Working Group) structure is setting up.




 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!





A full transcript of the webinar is now available here:
http://www.meaction.net/2016/04/24/transcript-solve-mecfs-interviews-dr-avi-nath/
Thanks
I am perturbed by Dr. Nath’s response to patient concerns. Shhh, be quiet, or the good guys won’t participate in our study because we hired some bad guys to be in charge….” Say what? We are an intense community because we have had to deal with BS from the biased and ignorant so called “experts” for too many decades. Not years, decades. The choice of Brian Wallitt was a mistake and the solution is for the Dr. Nath to make a personnel change, not tell the patient community to “be quiet” about the mistake or the real experts won’t play. Dr. Nath needs to recognize that real experts don’t have to play with the ignorant and arrogant. Especially those who combine both qualities in one person. There are MANY, MANY persons who could be selected for the position who would not be anathema to the patient community and who would not contaminate the proceedings according to preconceived biases. As we all know, Brian Wallitt is a slap in the face and a choice that makes it difficult for patients for take anything happening seriously. It is a sign that NIH is not acting in good faith. If we can see it, certainly the medical experts in CFS can see it. Putting a man with those opinions of this terrible disease on the team is not just a slap in the face to the patients. It’s a clear signal to anyone who DOES know what they are doing that participating would be a waste of time. It is not patient outrage, Dr. Nath, that is the problem. It is the NIH staffing decisions that is the problem.
I understand your upset but I think it’s based on an assumption that is not accurate. I think there were probably only a few choices for that position because that position has to be filled by someone within the intramural Clinical Center. They also have to be on site in order to do that job which means they have to located very close to the NIH campus.
Since the Clinical Center has never done an ME/CFS study it’s no surprise that they didn’t have many people to chose from.
I really hope you do not believe that participating in a study that uses world-class experts, as Dr. Nath called them, to search for viruses, examine the autonomic nervous system in depth, do many immune tests, grow ME/CFS neurons and test them is a waste of time. Please consider that the most important parts of the study – the research that will make the difference – has nothing to do with Walitt; that he is a coordinator, nothing more – and that this study could actually launch new era of understanding of ME/CFS.
1. From what I’ve heard, Fluge and Mella are hypothesizing about antibodies to endothelial cells, I hope the NIH hypothesis about antibodies to mitochondria and/or brain cells will be able to sort all that out.
2. I wish they would include gradual onset cases as controls and can think of no reason whatsoever not to do so.
3.”Someone recently related to me that some researchers she was interacting were at first shocked at the intensity of the ME/CFS community”- So again since patients are furious at being neglected and abandoned for decades, if only patients weren’t so furious then there would be no limit to the amount of research being done? Circular lack of logic anyone? Would someone please buy these people a copy of ‘And the Band Played On’? There’s probably an audiobook version they could listen to on their way to work if they wanted to.
Nath is very confident if autoantibodies are there he can find them.
The reason infectious onset was chosen was because Nath is leading the study and he specializes in infectious onset diseases. He believes and infection triggers and immune response which then causes the problems. Having gradual onset patients as controls vs using healthy controls would make it difficult to legitimize the disease and tell how it differs from people who are healthy. Healthy controls are virtually always used as a control group in studies.
The researchers aren’t objecting because people are furious over not getting funded. I’m not sure where you got that from. That’s not the issue at all.
I meant using gradual onset cases in addition to healthy controls, of course! If researchers aren’t objecting because people are furious over not getting funded, then what are they objecting to? People verbalizing their anger?
With regard to this specific study they’re objecting to the presence of some of the people doing patient intake and to Walitt being the study coordinator. Some NIH researchers are apparently objecting to those objections.
My point was if the entire point of the study is finding subsets with different disease phenotypes, which IMO is what it should be, what better way is there then to use gradual onset cases as controls to the sudden-onset cases?
Agreed….I think logistics is getting in the way, though. Nath said the Clinical Center is set up to do intense analyses of small populations of patients – the larger studies will come in the extramural studies that the study will hopefully birth.
‘With regard to this specific study they’re objecting to the presence of some of the people doing patient intake and to Walitt being the study coordinator. Some NIH researchers are apparently objecting to those objections.”
I don’t understandwhat that means. If the patients are going to be coming from expert clinicians, there should be no issue with selecting gradual onset cases. Who are the ‘they’ who are ‘objecting o the presence of some of the people doing patient intake and to Walitt being the study coordinator.’ If Wallit’s job is to push papers and green light hand picked patients selected by expert clinicians, what is the problem? The only problem I can see is how the study site apparently lets people self-apply to be part if the study.
I guess in the long run, who gives a shit? The people not taking part in the study won’t have their names associated with the discovery of one or more new clinical entities. Tough shit.
1. “With the success of The Normal Heart, Kramer had found his measure of vindication, and another play was beginning to form itself in his imagination. He had even made his peace with Paul Popham, the staid GMHC president with whom he had had so many struggles in the early years. When they talked for the last time, only days before Paul died, Larry apologized for their fights, and Paul just said, “Keep fighting.””
http://www.amazon.com/And-Band-Played-On-20th-Anniversary/dp/0312374631
2. http://surviveaplague.com/
http://www.imdb.com/title/tt2124803/
https://en.wikipedia.org/wiki/How_to_Survive_a_Plague
3. https://wewereherefilm.com/
http://www.imdb.com/title/tt1787837/
https://en.wikipedia.org/wiki/We_Were_Here_%28film%29
If what is reported here is true, the largest problem with the N.I.H., intramural ME/CFS study, is that it is being done by volunteers donating their time. Once, again, the ME/CFS Community is being short-changed by the N.I.H. not putting its money where its mouth is. Once again, ME/CFS patients and research are second-class citizens. When will it end?
N.I.H. Institute Directors receive budgets which they divide among their programs and Laboratory Chiefs. I gather that not a cent of any budget of any Institute, or any laboratory within an institute is going for Intramural N.I.H. research.
And so, rather than complain, or dare to voice this disparity, the community is to say, “thank-you, we are so grateful for your charitable contribution to our pain and suffering? We accept our second class, possibly third-class, citizenship in this country, because prejudice and faulty science have prejudiced the research community, the medical community, and even the general public against us?
How many years of timidity will it take for us to learn that timidity receives little attention. Timidity did not solve AIDS, and it will not solve ME/CFS.
Yes, most of it does appear to be done by volunteers but I think that’s understandable given that all the funding had already been accounted for this year. We’ll know much more about the governments commitment sometime in May when the Working Group have a chance for the first time to apply for and get funding.
Volunteering will certainly not be enough but I think it’s really most due to more to timing than anything else. We’ll see soon.
You could look at it another way. though….Nath got, at least, at first, somewhere around thirty researchers and their labs to agree to work in ME/CFS . That’s not bad…
I think we should be excited and grateful that Dr. Collin has engaged a researcher like Nath to do a study like this even if its mostly being done at least right now on a voluntary basis. Even if the researchers are giving their time this is going to be an enormously expensive study given all the testing that’s going to be done.
We’ve gone from getting virtually no attention from the NIH to a large three part study using the latest technologies and as Nath repeatedly said world class experts intensively studying ME/CFS patients. That’s pretty good and let’s see more!
Given that most of the contributors to the study are going to be doing so on a part-time basis, it’s all the more annoying that the one full-timer employed upon it is Dr. Wallitt.
The NIH need to appreciate that from a PR point of view, this doesn’t play well with the people that they’re trying to build bridges with – i.e. the patient community.
It’s pretty basic stuff really.
Nice point: from a PR point of view it sucks :). He did say they are hiring a coordinator plus I’m sure that Nath is not a volunteer. I don’t know why they choose Walitt. I suspect that few researchers in the intramural center have any experience with FM or ME/CFS – it may be that they didn’t have many options. I don’t believe that any ME/CFS studies have ever been done in the intramural center. We are starting off from a very low point; as we all know the NIH has devoted very little funding to ME/CFS over the years.
I might as well point out again that Walitt, despite his psychosmatic views, believes ME/CFS is a physiological disease and aims to prove that – he has stated that in his papers – and his studies reflect that; he has also stated that he does not believe that CBT or exercise provides much help; i.e. while the psychosomatic thing is totally off-putting he is no Simon Wessely.
The opportunity of this and other studies is to build a clientele of interested researchers. Given all the work they’re doing I would be really surprised if it didn’t.
I think Wallit is simply a glorified water carrier in this study, filling out the forms, etc. I also think he simply was smart enough to know what to say in the conference call and his ridiculous views haven’t changed a single ounce. He even refers to Wessely approvingly in one of his papers!
Dr. Nath himself said at the CDC Grand Rounds that ACT UP and other patient forums “…had a great impact on the way disease was handled, treated and moved the federal government to make changes at every level.”
Good to hear. Nath did seem at the end of the webinar to really welcome patient input and he has shown up now three times. At the same time he was relaying some problems that had cropped up…I don’t know that there are any easy answers here.
I think Nath will speak at next year’s UK CMRC, so that’s a good one. It’s just the study cannot start soon enough for those of us with progressive disease courses. I wonder what the agenda will be for the next IACFS/ME conference? They should be having fluge/mella, ron davis, avi nath, david patrick, etc. It should be an absolutely furious agenda. I also wish Avi Nath would go to London for the IiME conference and pre-conference meetings.
What kind of research facility, that my taxes pay for, has researchers that only work on projects that they “volunteer” for? Ridiculous! If the head of a dept., the head of a project wants them to work on something and they say no, show them the door, there are plenty of other people ready to ride that gravy train! I’ve been told by a top administrator at the NIH about how “busy” their researcher are.
I think things are a little more collegial than they used to be!
The CDC has budget with billions more than before. Time to fund M.E. Congress and Feds, HHS need to think about this objectively.
AIDS is 75% males patients, 1.25M people ill total gets $3B NIH funding
ME/CFS is 75% females, 2 million ill and gets $6M NIH R&D in 2015
The is a 500X discrimination against women, and against M.E.
The $3B per year AIDS R&D has been a huge success and NIH is applauded for taking what was a death sentence, and converting to a treatable livable disease that still needs some R&D. But ME/CFS is a much worse disease now than AIDS, affecting more people. For a fair government, there is no longer any justification for spending 500X less on ME/CFS. Govt needs to fairly represent all people. ME/CFS deserves at least $250M/year ASAP, or $3B per year to be equitable, if the HHS want to stop this discrimination, WHICH IT MUST DO. We need to ask NOW, National Organization for Women, to campaign for funds and for all govt to fully fund this disease R&D. Approx 10% of women may get breast cancer and 1% may die of this. 1% or women may get ME/CFS and never recover, which is not dying of this, but are sentenced to a life imprisoned in their house or bed in many cases, never to recover. 250,000 are housebound or bedbound, having not even been accused of any misdeeds, and little chance of getting out of this Prison. This is social injustice by congress. Some do die from ME/CFS, and if approx 1% of people die per year vs 3% of MECFS, the added 2% deaths is approx 40,000 per year, more than die of car wrecks in US. This is a major disease, costs up to $50B/yr to US economy (CDC Feb 2016) to spending $250M/year in 2017 would end the gross discrimination of the past, and set us on a new, enlightened path together with NIH, CDC etc. This is an exciting year for all, but need more funds, and need buyin by 100% of 100 senators during current election cycle, or we need to vote any discriminators out of office in Nov or sooner. In May we need to educate 100 senators, and 100% should agree for major funding. Put them each to the litmus test: will they stop discriminating against Women and M.E.! I hope and pray we get 100%
The only “litmus” test we have for researchers are for people who are the ones actually dealing with the patients who have sided with the somatic/psych/get/pace Strauss crowd.
These people have caused great harm to us over decades and are the reason we have became a joke. Klimas says this type of thinking spread by the clinicians we have demanded being taken out of the study helped to cause what Klimas said is a PTSD from doctors treating us like Walitt and the rest. If I hate that the patients who are in for a very difficult week are being taken care of by Walitt at the helm and 5 more of his thinking.
I have no worries about the 120 people running the tests. it about the ” CFS clinical experts” asking the questions and reporting patient answers. Somebody has to write down what the patients are saying and the people put in charge of the patient care are exactly who the IOM said to het away from. I see the peonlem being there are no real experts at the NIH, they have all been scared of ny the powers that be as reported by many, not the patients.
The fact that he calls our litmus test meaningless shows a lack of understanding and having these be the people who will be interviewing patients is so wrong on so many levels.
I’d like to point out that no other researchers who are looking for real answers gets grief from us. We don’t pick apart who is working with Klimas or Davis or Lipkin because they know better than to use these yahoos. I don’t appreciate being portrayed as just willy nilly complainers who don’t know enough about science to understand why no bias is possible.
the science itself does sound exciting but it will be at least 3 years before it’s even done so.. we do what we do best wait.
So, here we are, complaining about the study being laid out to help us. If they see some results of interest, Nath and co. will have something to base a bigger, better funded study. I say thanks.
Selecting a patient group with much similarity seems logical to me. Sudden onset is a likely characteristic to choose, as it seems, beforehand, to suggest a shared mechanism. 80 patients is way better than the handful so often studied. If this is a new start , why not say some kind words to encourage them? Yes I am disappointed with medical folks, 26 years with so little help of use to me. But this guy seems to have picked up on the main issues, has some notions of hypotheses to explore. Researchers risk being wrong (if you know the answer before you start, then research is not needed) and that will kick them in the pants on its own. We can as a group make useful comments, but the extra insults after a failure, those are hard to take. Thanks for the write-up of the third talk.
Totally agree. AIDS had tons of media types with money behind them ,supporting them and giving them good press. We do not have that. I wish those in the ME/CFS would stop the constant complaints.
It was the people who complained the loudest along with a few great doctors who were the impetus for the research starting to begin with. Ever hear of ACT UP? People with AIDS were left to die terrible deaths because no one cared except for most of the dying peoples friends and some of their family. The ‘tons of media types with money’ only came after after it was discovered HIV was infectious. Read ‘And the band played on’, please. Major newspapers didn’t cover the AIDS crisis for many many years until it was discovered that it was an infectious virus that anyone could get. Again, please read ‘And the band played on’.
“The criticism of Brian Walitt and perhaps Dr. Saligan, Gill and even Dr. Unger has apparently gotten around, and it’s had the unintended effect of making some of the researchers we do want working for us leery of doing so.”
Anyone made leery by the valid criticism going around is not someone that I would want working for us actually. If you can’t handle scrutiny then this is probably not the field to be in. People like Ian Lipkin have taken plenty of heat over the years, and he remains as dedicated to our cause as always. Probably because he realizes that the critical voices are but a small minority of the millions of sick patients out there that need his help.
We can’t control what other patients do or say. I’m frankly getting sick of this emotional blackmail we’re being subjected to lately. What are we supposed to do, track down all these critical people, and then what? Shut them up or else? This is just a part of life. If they want to work in a public health field then they are going to be exposed to the public, and there is nothing anybody else, especially us patients, can do about it. They can choose to ignore it or they can find something else to do.
Although slightly encouraged by Cort and his presentation of the content of this trial I’m still trying to figure out if the problems with it are intentional or just incidental. I have to say though that when the researchers bring out an old chestnut i.e. that researchers have been scared off by irrational and demanding patients it just makes me all the more suspicious as this is exactly the tactic used in the UK to demonise patients (by the Biopsychosocial group only). First you present something utterly outrageous to the ME community then you condemn them when they react with horror and indignation. Isn’t that the tactics of an agent provocateur
Slight encouragement is better than no encouragement 🙂 I recognize that we’ve had some stumbling blocks but I think you can tell that I’m totally jazzed by this study.
I didn’t interpret Nath as condemning anyone – I think he simply pointed out what has happened – that some researchers have taken pulled back a bit. I appreciate him for his candor; we should know what effects our actions have. Having researchers looking for a biological cause to ME/CFS pull back a bit was obviously not one of them! I certainly didn’t anticipate that would happen.
It just shows things a bit trickier than we thought.
I think the choice of Walitt was bound to elicit a strong response and it was appropriate that that happen. I personally think he’s not going to be a problem – no psychosomatic research is being done (and he’s only interested in biology anyway) plus he appears to be a coordinator basically and I’ve heard from a couple of patients who really enjoyed working with him (and one who didn’t).
It’s really too bad he popped up in this study though. If not for that I think a lot of people could get behind it more…
The study, though, will be fine, I believe.
from everything I’ve read Wallit has no interest in biology, only people thinking themselves ill- https://www.youtube.com/watch?v=FmSKgd-940s
How can everyone except you have a major problem with Wallit? It’s like when the CDC was sticking their thumb up their asses with the exception of a few good papers and you never caught on except for one time I remember you said something like ‘oh God, it’s as bad as everyone was saying’. I don’t have a problem with Wallit pushing papers, but to promote his research as having an interest in pathophysiology is just weird. Saying there’s no effective treatment, biomedical or psychological, isn’t saying that treatments are needed.
“The medical field does poorly with the treatment of fibromyalgia in general, chasing [a cure] with medicine doesn’t seem to work. The people who seem to me to do best sort of figure it out on their own by thinking about things, getting to know themselves, and making changes in their lives to accommodate who they’ve become.” Brian Walitt, M.D., M.P.H
http://www.pipain.com/uploads/6/5/3/8/6538150/fibromyalgia_drugs_only_effective_for_22_percent.pdf
You’re telling me that someone who talks about “chasing a cure with medicine” and prefers people “thinking about things, getting to know themselves, and making changes in their lives to accommodate who they’ve become” is interested in biology and not psychosomatisation? That’s so ridiculous I can’t stand it.
I am very confident that Nath wil find something significant. Finally somebody is looking at the right place! Only this pilot study wil take 2 years or longer if they are replicated these findings it takes much longer i guess. I am very positive about this study!
Yes I think this study has good promise too.
A great question to ask would be whether we’ll see any studies published or get any word of what they’re finding out before 2 years are up…I imagine if they go through say 20 patients and 20 controls and find some immune abnormality they could publish a short paper on that. I don’t know if they would – it would take some work – but I think they could…
I am also very excited about Michael van Elzakker, i think he has already found something important 🙂
I think some of the anger that CFS patients have lobbed the way of researchers has been a disgrace. Almost all of them are doing their best, with their resources.
Being downunder, I can say Andrew Lloyd has had a hard ride at times. For daring to say maybe CFS isn’t a chronic illness of the immune system, but of the brain (but initiated by an immune event/insult). Oh, that can’t be right, we are all 100% sure it’s caused by a chronic virus, that can be treated by ampligen…but it’s not.
It seems to me that Dr. Nath has a limited tool box (money, researchers, volunteers). He’ll have to make do with what’s available so, let’s see what he can do with it. I’m hopeful and optimistic.
If 2 years ago someone told me “… The government is going to crank up research for ME/CFS, but some of the researchers/volunteers have a questionable predetermined position”, I would still be ecstatic. I think the doubts and assumptions need to be questioned.
Stop grilling the people that are actually doing something for us. Give ’em a chance. That’s my stance.
It is a good strategy for everyone to remain overall positive. This supports the energy and forward momentum. When we act like a negative “mob”, however, as some of us have when we were sure of what ME/CFS was caused by or how it works (think XMRV) or the drug that ought to be designated to treat it, etc., we do start taking things down. But if we can instead take strong, positive stands about the progress we want, I think this will help get us out of the ditch and onto the road…
Obviously, no one pre-warned Dr. Nath of our community’s passionate need for a cure. The MECFS community has experienced years of being treated with disregard, so it should come as no surprise to the NIH that we would respond with such vibrato. However, since they have clearly heard and responded to most of our concerns, I think the MECFS community should show support for and trust that Dr. Nath will keep our best interest in mind.
Neuroinflammation, Pain, and Fatigue Laboratory at UAB
Sponsored ·
..
I just released a video on our new technique to measure brain temperature. I hope we can use this brain scan to identify when someone has neuroinflammation that is causing pain and fatigue. You can see the video by going to our youtube channel, or by clicking on the link below:
– Jarred Younger
https://www.youtube.com/watch?v=1p6UojKL010
Do you have a hot brain?
Cool! Thanks Jeanie. 🙂
As the mother and full-time caregiver of an ME/CFS patient, I have been overjoyed at the response of Dr Collins and the NIH to the need for research on this disease. My child is on the moderately severe/severe end of the spectrum, diagnosed via CCC by one of the recognized ME experts in our community.
Just as I understand the response of the patient community, I understand Dr Nath’s response to some of the behavior in the patient community. I have been reluctant to speak my mind or involve myself in conversations about research, treatments, doctors and protocols because of the tendency of some to “shout” at or tear down those who may not see the situation as they do. It is intimidating for someone like me and I stay quiet. There is no question that patients have been horribly mistreated and continue to be mistreated–particularly badly in the UK and Europe. The system has failed everyone.
However, when the people within the ME community attack, disparage and malign those who are attempting to help–particularly those who haven’t lived in this community and are unfamiliar with the abuse it has suffered–the community is validating the criticisms that people like Simon Wessely heap on it and proving those smug, egomaniac psychiatrists right. Is that what the community wants?
At some point, I have to be confident enough in the physicians who select patients for the study to get the ME cohort correct. If patients from my child’s doctor’s practice are selected, I know this doctor will not send anyone that fails any part of the CCC. Those researchers who may not have as much inclination toward the biomedical model will be able to witness for themselves how sick these patients are. Dr Nath and his team deserve a chance to prove themselves and I’m willing to cut them a wide berth. I don’t feel like I’m groveling at the feet of the NIH or settling for crumbs. Everything needs a beginning and this is what we’ve got. Never underestimate the power of a seed to grow into a massive oak tree.
What it comes down to is hope. I can choose doubt or hope and I’m choosing the later.
Nice analogy Chris – never underestimate the power of a seed to grow into a massive oak tree. All we need really is one breakthrough and this study provides the opportunity for multiple breakthroughs. Nobody knows, of course, how it will all turn out – but the possibility is there. As Ron Davis said, we (the research community) are digging too deeply into ME/CFS not find something.
Chris, I am in the same situation as you, my 25 year old son has severe ME and requires full time care. In the UK we are still a long way behind with GET and CBT the recommended treatments. There are small shoots of research interest in the biology of the illness but no coordinated push that I know of. The ME community has been ignored for so long but the Nath project is a tiny flame of hope and it has the potential to offer some real understanding and hopefully, in time, cures for this awful illness. I believe keeping good communication channels open between sufferers and scientists such that each can hear what the other is saying will speed this process.
“Help” from this and other research projects either planned or underway is probably not going to save my life, but I have appreciated the entire journey with this community, as we have been companionship on the way. We have been through twists and turns, thick and thin, all while trying to understand, to find and make alliances, to get through conflicts, pull back from unproductive avenues, and keep on going. I think if there is a testament to the fact that we are not essentially mentally ill, this has got to be evidence for it. When hope and help start to arrive after long deprivation and suffering, there is a temptation to lash out, to punish those who were not part of our reality, who ignored, denied and maybe even exploited it, and who did not reach out a hand when we so needed it. Imagine victims released from a prison where they have been held and tortured indefinitely. When suddenly released, they may not break out in sunny smiles and dance around their liberators. Instead, they may scowl and say, “Where the hell were you?” Trust was broken and that is a fact. Every phase of a challenging process we have to work through. Having companions on the way is what has made it rewarding and possible.
In all the discussion I have seen nothing of Dr. Petersen and Simmaron’s Efforts. Are they being totally ignored?
Simmaron is doing great stuff but I don’t know that there’s any reason for them or to be involved in this particular study. Simmaron’s lead research does have an NIH grant and Simmaron is using samples from the XMRV studies at the NIH for another study. Of course Simmaron is working closely with Dr. Lipkin who does have a role in this study. Hopefully when an RFA comes out (cross fingers) Simmaron will be able to take advantage of it.
Yes, some aspects of the study are world-class as Dr. Nath says. Many are third-world- the speed, funding, the (co-)Principle Investigator Brian Wallitt, the initial study design and some of the attitude. The criticism was very well-deserved. Don’t insult us with more talk of researchers being put off.
I don’t see it as insulting. I see it more as simply a statement of fact. I didn’t feel that Nath was deriding ME/CFS patients. I think he was kind of stating “well, I want to do this study and I’ve run into this problem”…
I don’t think anyone anticipated that other researchers would have this reaction. I certainly didn’t. I also don’t think we should get caught up in whether those researchers who have become leery are right or wrong…It’s simply something that happened – something to think about.
Thank you, Cort, for offering a re-framing point of view, from judgement to neutrality. I’ve been learning how this is actually more powerful and useful, though our culture has mostly taught us negative judgment and fighting modes as the way to change situations. But from a position of neutrality, observation and wider perspective, it is easiest to move and move forward. I have every sympathy for negative judgment, anger and despair as I have had them and know the hurt and suffering they come out of. But if we can find balance in ourselves and our vision, I think it will help get these projects onto a good track.
Really nicely said Cecelia…I think we really are drawn to finding out what is wrong and concentrating on that and we forget to look at what is driving the other side – where they are coming from. Striving to come from a place of neutrality – which I’ve been told is Buddhist concept – sounds like a great practice. Thanks for the idea.
Yeah, it (neutrality) seems weak, but it actually is strong.
As individuals and a community, our anger over the past and questions about the future studies are wise and justified. We can’t be surprised when the research community responds to our community’s criticism with criticism in return. It’s a relationship. And a better one now, when communication seems to be going in both directions.