

Researchers in both fibromyalgia (FM) and chronic fatigue syndrome (ME/CFS) are doing deeper dives into the genetics of these illnesses than have ever been done before and they’re coming up with some surprises.
This study produced the most comprehensive look at the genetics of mtDNA (as opposed to nuclear DNA) in chronic pain yet.
Travis Craddock of the Institute for Neuroimmune Medicine (INIM) at Nova Southeastern University (NSU) recently reported finding high rates of mutations in mucosal genes that produce the protective layers in our respiratory system, nose and eyes in ME/CFS (unpublished). Earlier, Liz Worthy took advantage of a Solve ME/CFS Initiative (SMCI) Ramsay Award to find evidence of genes associated with inborn errors of metabolism in ME/CFS.
Now, after a deep dive into the mitochondrial DNA of people in pain, a team of US and Canadian researchers has come up with a startling finding in FM.
A genetic polymorphism that is associated with mitochondrial energy metabolism increases risk of fibromyalgia. Miranda Al van Tilburg 1 2 3, Marc Parisien 4, Richard G Boles 5, Gillian L Drury 4, Julian Smith-Voudouris 4, Vivek Verma 4, Samar Khoury 4, Anne-Julie Chabot-Doré 4, Andrea G Nackley 6 7, Shad B Smith 6, William E Whitehead 8, Denniz A Zolnoun 9, Gary D Slade 10 11 12, Inna Tchivileva 10, William Maixner 6, Luda Diatchenko 4 Pain. 2020 Jul 10. doi: 10.1097/j.pain.0000000000001996.
Chronic pain often runs in families, but most genetic pain studies have not found evidence of a strong genetic association. Those studies, though, have mostly focused on nuclear DNA – the DNA found in the nucleus of the cell. Our cells carry another kind of DNA, though – mitochondrial DNA that is contributed through the mother – and that’s the DNA this group tested.
We mostly associate the mitochondria with energy production – and they are the main energy producers in the body but they have other functions as well. Mitochondria are important for nervous system functioning and help to regulate the immune system. Mitochondrial issues also typically show up in tissues associated with high – and fluctuating – energy requirements such as the nerves and the muscles – two areas of concern in chronic pain.
This study was unusual in its large size and its attempt to assess the fully range of genetic polymorphisms (small changes in the genetic makeup) found in the 37 mitochondrial genes.
Each of the 609 participants were diagnosed with a “complex persistent pain condition” (CPCC): episodic migraine (n=263), irritable bowel syndrome (n=223), fibromyalgia (n=96) vulvar vestibulitis (n=100) and temporomandibular disorders (n=172 subjects). Plus, 237 healthy controls were included as well. Almost 70% were Caucasian and 23% were African American.
Eighty-five percent of the people with a CPCC were female. Interestingly, the highest rate of female dominance was in fibromyalgia (7.8 x’s more females.)
Results
“Our results suggest that the m.2352T>C polymorphism has a strong clinical effect on the risk of fibromyalgia and possibly other chronic pain conditions.” the authors
They extracted the genomic DNA and then did deep sequencing and found that the FM patients stood out in another way: they and the vulvar vestibulitis patients were the only pain groups to have a significantly increased frequency of a specific genetic polymorphism; i.e. small change in one of the mtDNA genes.
(Vulvar vestibulitis refers to a condition characterized by severe pain in the vagina during intercourse, when using tampons or even when sitting on something like a bicycle seat. The pain can also be constant.)
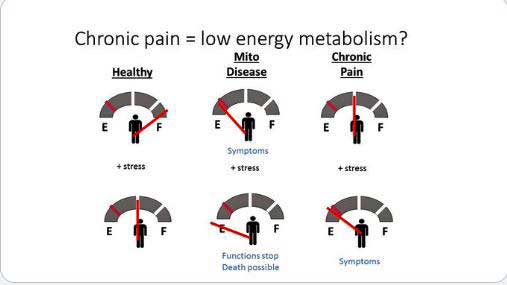
Tilburg hypothesized genetic problems with energy production – plus stress in FM – could result in more pain.
In both groups, but more so in FM, an alteration in a single nucleotide (single nucleotide polymorphism (SNP) (m.2352T>C (rs28358579)) showed up with increased frequency in the MT-RNR2 ribosomal gene in women who a particular form of the gene called the c (minor) allele. An allele refers to a variant form of a gene and minor alleles refer to the second most common form of a gene in the population.
This small genetic alteration was also more evident, but to much lesser degree, in the other CPCC’s.
This gene, remarkably, had been unassessed in pain or other conditions, yet the evidence indicated that a small alteration in the gene was contributing to an increased risk for fibromyalgia and vulvar vestibulitis and perhaps other chronic pain conditions. (The polymorphism also increased the risk of a woman having more than one complex chronic pain condition.)
(One reason the polymorphism hasn’t been assessed much is that it’s found on an allele (the c (minor) allele) that is only very rarely found in Caucasian women and most genetic analyses have been done on Caucasian women. It is not currently assessed in 23andMe.)
Then they tested their finding in another pain cohort – the Orofacial Pain: Prospective Evaluation and Risk Assessment (OPPERA) Study cohort – which contained 52 women with FM and over a 1,000 without it. The researchers must have been dancing a jig when they learned that their initial finding stood up: people with FM indeed had significantly higher rates of the polymorphism.
Their deep dive into the mtDNA appeared to have uncovered the first evidence of genetic weakness for coming down with a “complex persistent pain condition”.
The effect was strong; in fact, it appeared to be unusually strong. The authors reported:
“The replicated genetic effect size of the C allele on the disease risk (OR 5.1 and 4.3 in discovery and replication cohort, respectively) is impressive and has little precedence within the field of common diseases.”
An OR (odds ratio) of 5.1, if I have it right, means that having that particular SNP on that allele increased a woman’s odds of having fibromyalgia being present five-fold. That appears to be a huge number for a single gene polymorphism.
While having FM was significantly associated with having this polymorphism, the alternative c allele that the SNP was found in, was more much prominent in African American women and almost never found in women who identified themselves as Caucasian (1%). (Indeed, the odds ratio boomed in African American women to 7.6). Because the C allele had a stronger effect in a mixed population than in the African American population, it may have strongly affected other racial groups as well.
The SNP is located in the mitochondria’s 16S rRNA gene in the ribosome. Since this SNP has rarely been tested for, it hasn’t been associated with any other diseases or conditions. It’s not clear what it’s doing, but it may be hampering the ribosome’s ability to translate mRNA into proteins.
Mitochondrial functioning impacted
Next, they determined whether the polymorphism was actually impacting mitochondrial function. Obtaining ten cell lines from women with and without the polymorphism, they assessed the mitochondrial functioning of each one. Cells carrying the minor allele on which the SNP was found showed no difference in mitochondrial functioning (mitochondrial membrane potential) when put in glucose media (glycolysis), but when put in galactose media, their mitochondria membrane potential declined – indicating those cells were producing less ATP.
Mitochondrial Abnormalities in FM
It’s not clear how mitochondrial problems enhance pain, but several studies have implicated the mitochondria in FM. While this study illuminated a potential risk factor for African American women with FM, these other studies have broadly implicated the mitochondria in FM.
A gene expression study that highlighted reduced activity in a mitochondrial ribosomal gene suggested the mitochondria in FM may have taken a hit.
A recent FM animal model suggested the mitochondrial inhibition in the muscles and the spinal cord could play a large role in FM. Skin biopsies showed reduced mitochondrial activity and increased oxidative stress – two factors the authors suggested could be contributing to the high rate of small nerve fiber problems in FM.
A small family study suggested that a mutation in a mitochondrial gene – and the resulting increase in oxidative stress – could be playing a large role in some people with FM and suggested that future studies concentrate on people with family histories of the disease on the mother’s side.
Decreased ATP production in the muscles of FM patients has also been found. Reduced levels of coenzyme Q10 – a critical component in the electron transport chain in the mitochondria – have been found in FM and CoQ10 supplementation has been found beneficial in several studies.
Conclusion
The first really deep dive into the mitochondrial DNA in chronic pain uncovered a surprise – a rarely assessed genetic alteration (single nucleotide polymorphism – SNP) which was highly associated with fibromyalgia. The finding was potentially a big win for Black women with FM, many of whom may carry the form of the gene the polymorphism or SNP was found in. Caucasian women, on the other hand, rarely carry that form of the gene. When it was present, the SNP appeared to have an unusually strong effect and it will surely become the object of further study.
Further analysis suggested the SNP may be reducing ATP production in women with that form of the gene. While the SNP appeared to be mostly found in Black women with FM, the finding underscores the role the mitochondria may play in FM, and other studies have highlighted possible mitochondrial problems in the broad FM population and other chronic pain states. Tilburg’s earlier study of Caucasian women with IBS, for instance, suggested that a subset may have an mtDNA-related mitochondrial dysfunction.
In short, different mitochondrial problems could be contributing to the pain in FM in different ways. Mitochondrial issues could impair muscle functioning, producing oxidative stress, damaging the small nerve fibers, or by affecting nerve transmission elsewhere.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



You’re on a roll Cort! Proud to support your work and I hope others will contribute too. I know of no other resource that aggregates recent research and advocacy developments for ME/CFS/FM/GWI in such a comprehensive and digestible way.
On the topic at hand, I’m sure the question on everyone’s mind is what types of interventions might correct these polymorphisms or their downstream effects?
Thanks Winston for your nice words and support. Given the strong finding the authors stated that it could open the door for new treatments although they didn’t say which. I assume more studies are needed.
The SNP was not found in increased frequency in men (they did have a few in there :)) by the way, but the authors stated that they didn’t have enough men in there to really tell.
Hi , I thought that Mt.DNA had been found that it can also be inherited from Males as well, in a recent study.
I looked it up and it appears that in rare cases men can pass it on too. Usually men destroy their mtDNA but in a few rare cases that process doesn’t work.
https://cosmosmagazine.com/biology/males-can-pass-on-mitochondrial-dna
https://medicalxpress.com/news/2018-11-rare-instances-male-mtdna-offspring.html
This sounds a good step forward. But what happens next. I’m 7o, and I have had M.E/CFS for 38 plus years. Then 5 years ago I had breast cancer. I now have fibromyalgia as well. I have serious gut problems. Sibo ,Candida and IBS. I have read so much about it all being closely related. Most Doctors are less than us less. I went private and was also told I have mitochondria dysfunction. No one knows how to help. And most Doctors look at you if your from another planet. Some one needs to hurry and get a drug to help us.
There are no FDA approved treatments for mitochondrial diseases. The main treatment is the mitochondrial cocktail, a customized blend of nutrients which encourage mitochondria to behave better and reduce progression of disease.
Where did you have your private testing done?
I have been Dx with FM at least 20 years ago by my amazing doctor. I am caucaian , now 65, We have tried many many things. It’s maddening. . Now last fall after being Rx moxifloxicin, , took for four days I am in agony. My tendons/ joints, hard to just live. Work full time and exhausted. It is migratory and I have tried alternative med like turmeric, vitamins, antiflamatory foods, exercise, ( suffer for days) after simple house cleaning. I will be seeing specialist soon. I would like to participate in any studies, will allow skin, muscle biopsies, blood work. I have 2 daughters with symptoms starting.
I have always suspected a Mitochondrial DNA dysfunction since a doctor yelled at me saying I couldn’t possibly have a problem with Citric Acid (which I do) because the Krebs Cycle runs on citric acid. I connected energy production with low energy and have figured there’s a mitochondrial DNA connection.
The doctors we have to put with….
Same here and when I took a UScript test for metabolic processing issues for drugs, suppliments and foods; citric acid showed up as a compound I couldn’t metabolize properly, in any form natrual or sythethic. My pain was exquisite. I did a FODMAP elimination diet and food/symptom journal and by eliminating corn derived products knocked the pain back about 70%. I had discovered that afloxotins are required for use on all commercially sold foods, usually citric acid as its inexpensive and subsidized by the government in foods, pharmaceuticals, and supplimemts. Even the organic produce and your ground beef are sprayed with it. Some foods are packed with citric acid soaker pads. Unless purchased from the farmer or farmers market directly, the EPA mandates an aflotoxin be used to prevent outbreaks of mold illness and wasted food, with the acknowlegement that it will make some people sick. For me, it triggers an increase in hypotonia as well.
Oh my gosh. It’s nearly impossible to avoid corn in the food markets of the US. May I as about the UScript test? I’ll google it as well. Thank you.
Many people have turned to a Lectin Free diet to remove the above mentioned toxins and distressing body results. Takes some time to get used to; most doctors were not taught about this stuff in medical school. Dr. Gundry has many books and online podcasts educating the public about these issues. He is a well known cardiovascular expert and heart surgeon.
GMO foods (almost everything, and especially corn, other grains, fruits, high oxalate foods, and legumes) are also implicated in this unholy mix of gut corrosion/ “leaky gut” and other genetically individualized symptom cascades. Of course, cell metabolism and the mitochondria are engaged in these gear wheel reactions.
Thank you for sharing Cort! Always like to read any research on fibromyalgia cfs/me
How do I get into one of these studies??
I know for sure that I have mitochondrial dysfunction and oxidative stress
I lost 2 infants from a rare genetic disease. Mitochondrial myopathy, cytochrome oxidase deficiency complex IV
It would be nice to get to the right doctors eventually not easy to diagnose some of these diseases
Wow…Georgette, it sure sounds like you should be in one of these studies. Here’s Van Tilburgs email address (from the paper) – vantilburg@campbell.edu – I would just drop her an email. She also shared the results of the paper on twitter and was very accomodating to questions.
If you have ME/CFS be sure to check out this blog. Fascinating stubborn on inborn errors of metabolism that are surely getting overlooked in ME/CFS.
https://www.healthrising.org/blog/2019/06/02/gene-mutations-energy-production-chronic-fatigue-syndrome/
You sound like you’re right up Camille Birch’s and Liz Worthey’s alleys. I found a Linked in contact for Camille Birch – https://www.linkedin.com/in/birchcamille/
I recently had a blood test, approved by the FDA, called FM/a ,which uses Luminex xMap bead array immunoassay to measure the production on cytokines by cultured blood. Based on these measurements, I scored an 82, out of 100 with the interpretation that I am strongly confirmable of having Fibromyalgia. It basically states that patients with Fibromyalgia have an immune system deficiency/dysfunction.In conclusion, the peripheral blood mononuclear cell, fails in patients with Fibromyalgia to be able to produce normal quantities of particular and critical proteins (chemokines and cytokines). BCG is a possible thought to be used to reverse the abnormal immune system. I also have interstitial cystitis, endometriosis and daily migraines. The hardest thing to deal with are my constant and chronic muscle spasms on my neck and entire back, bilaterally. These muscles pinch and burn constantly. In addition, my spine feels like someone is squeezing and twisting it. I think that this is a structural problem. I feel that if my spine was corrected, I may have less or none of the spasms altogether. The doctors I have spoke to though, do not agree with me. I wonder if it is because I am 66 yr. old. What do you think? LM
I recall Cort sharing info about some long term sufferers having a resolution of many debilitating symptoms after the realignment of their…?atlas? joint. I believe it was at the base of the skull-top of the neck. Maybe someone else recalls it more in detail.
Alignment is huge in many health issues. My migraines ceased after it was discovered I had one leg longer than the other …orthotics and a built up shoe….no more migraine! Also explains why my balance was bad and I would often lose balance to one side whilst standing still!
I suspect this is one of the things the Alexander Technique addresses as well.
Appreciate usual good reporting and analysis. Good study for white women, black women, …men? as usual.
We are getting left behind again…(lol( There were some men in the study – but not enough to really tell if the SNP applied to them.
Maybe next time…
There is more to a mitochondrial disease than “just” impaired energy production. The symbiosis between humans and their mitochondria goes two ways: humans can’t survive without their mitochondria providing for them AND mitochondria can’t survive without the human body providing for them.
mitochondria lost key genes for stand alone survival even if well fed. For the mitochondria, being able to shed part of their genes and become genetically less complex does have clear advantageous. Extra genes do come at an expense, so less genes is less expensive in terms of energy and maintenance.
Several functions needed for the mitochondria’s survival have been taken over by their host cells. Things like providing mitochondria by many key enzymes, hormones, key proteins… is done by the host cells.
Such symbiotic providing of key chemicals by the host cell to the mitochondria makes most sense when the host cell needs and produces these chemicals already for themselves.
When we have genetic mutations of the mitochondria, we may well need to adapt amounts of key hormones, enzymes, proteins… in order to get correct amounts of energy produced or in order to for example produce correct amounts of oxidative stress: not too much but neither too few (we vitally need some amount of oxidative stress to survive).
That is where the symbiotic relationship becomes a burden. If for example the mitochondria need a double amount of hormone X in order to be able to function good enough, then the host cell and much of the body sees double the amount of hormone X too. That has a big chance to disrupt some processes within the body. Therefore a mutation or disability to produce enough energy or to control oxidative stress can have a whole slew of unexpected consequences rippling through much if not all of the body.
Cort
How can I contact Klimas’s group about educational information to help local doctors? The email addresses I was given don’t work. I am sending information to local doctors to try to improve care around Lawrence, KS.
Wow, Josh, so sorry to see how much you’ve been through.
You mentioned your antioxidant therapy (of sorts). I wanted to mention camel milk for this – it reduces oxidative stress (I think due to its high lactoferrin levels). But it also heals diabetes, including type 1. There’s loads of info on it. You might want to check it out.
Blessings for you for much improved health. ❤️
Cort, another great article, thank!
I had a hard time following a lot of this. Can you help me understand more about these findings? Would you say this polymorphism, or mutation to the DNA, is genetic or epigenetic? IE, Are we born with it (and it’s in hibernation, so to speak) or is it triggered by an expression in the proteins made by the genes?
Basically, epigenetic mechanisms do not involve a change to the DNA sequence per se, whereas genetic mechanisms involve the primary DNA sequence and changes or mutations to this sequence.
Thanks!
Thanks.
Polymorphisms refer to small changes in the DNA we are born with; i.e. they are fixed – genetic.
Epigenetics, on the other hand, refers to a process which can turn or turn off (put into hibernation) the expression of our genes. As you note epigenetics does not affect our basic genetic blueprint – instead it determines which parts of it are expressed. In this case my guess would be that it’s possible for the gene polymorphism to be there but not be expressed or not expressed fully.
According to Wikipedia many different processes ( paramutation, bookmarking, imprinting, gene silencing, X chromosome inactivation, position effect, DNA methylation reprogramming, transvection, maternal effects, regulation of histone modifications. etc.) can affect whether the genes we have are expressed or not.
Its so uplifting to see the progress in research in this area. Obviously, may people are suffering greatly from this imbalance and all steps in the right direction are a source of inspiration. But yes, I to feel that we want to know, what is the Epigenetic aspect of this. Certainly the massive ongoing exposure to the aflotoxin riddled preservative citric acid- can’t be helpful, as the one women mentioned. I’m sure there are a number of other things, and would love to see that explored. Research is expensive, yes. But I think that the we should, by now, admit that in general, with most chronic illnesses – when researched intimately, will have underlying DNA factors. And in most cases, normally, the genes do not express into chronic illness. For example, in the case of Mitochondrial related problems. I believe that there has been research that indicates the there is often a cell wall inflammation, that is directly related to toxicity (often heavy metal, biotoxin, endotoxin related) and when this is occurring, the cell wall not only is compromised in it’s ability to allow nutrients in to the cell, but also, to allow the cell waste to leave. In addition, the hormone receptors get blunted, and also don’t bring in the hormones to activate the cell optimally. THEN, after this goes on for a long time, the Mitochondria will begin to mimic the cell wall, getting inflamed with the toxicity of the cell, and thus progressively malfunctioning. AND, there is already a detox program designed to specifically address this mechanism. It’s certainly not a magic bullet, and for people with FM, it would probably take many months to see sustainable results. But a step in the direction of getting to a deeper, core aspect of the imbalance, a chance to, at the very least stop the progression of the illness, maybe see serious recovery even, for some that investment I’m sure, would be considered of great value.
Thank you Chandra.
What would you recommend as a source for this detox program you mention?
My understanding is that the mitochondrial dna are more prone to damage from environmental and medically prescribed toxins than the nuclear dna. Thanks to their lack of epigenetic protection. Therefore one wouldn’t necessarily have to be born with a genetic mutation to their mtdna to develop a mitochondrial disease. The so called adult onset, toxin induced, mitochondrial myopathy. Basically the heavy metals, environmental, and /or pharmachemical toxins get inside your mitochondrial membranes and screw up your production of ATP. Then the disease process starts in various places around your body. The mitochondria I was born with( probably based on my mother’s toxic exposures or lack there of) are different than the next person. So my reaction to toxic exposures will be different from the next person most likely. Thanks in part to the strength of my mitochondrial dna. Its tricky and still a work in progress for sure, but talk about a big loop hole for our poisoner’s to jump through. Those of you looking for a potential cause of your “fibromyalgia” don’t forget to look into mri contrast with gadolinium or fluoroquinolone antibiotics.
Gadoliniumtoxicity.com
https://journals.lww.com/investigativeradiology/Fulltext/2019/08000/Gadolinium_Based_MRI_Contrast_Agents_Induce.1.aspx
https://www.nature.com/articles/d41586-018-03267-5
https://youtu.be/xu6KR9APk2k
A MitoSwab test correlates pretty well with a muscle biopsy and is kess invasive. If us useful for testing whether nutrient interventions (the “mito cocktail”) are working. My doctor uses a Genova Diagnostics NutrEval to adjust my mito cocktail, and iteratively using these interventions has greatly helped my function.
How do you treat your symptoms, or do you?
How , when, & where can someone get genetic testing for these markers & deficiencies? Are Drs familiar with this research yet?
In reading this, there was a mention of future studies in genetics on the mother’s side.
I have 3 cousins with fm diagnosis on my father’s side.
I’m hoping the future studies will include the father’s side as well.
This is a great read and study. Anything to help move forward with FM, ME, CFS is huge!!
Thank you
Are there any commercial sources for the Mitochondrial genetic test, or an ongoing study that I could enroll in? I have longstanding ME/CFIDS
Can deficiencies affect gene mutations/polymorphisms?
Could the mitochondrial “issue” be a deficiency?
Carnitine connection with Acetylcholine, Muscarinic/Nicotinic Receptors, a7 involving MPS, CVS, FMS, RA, etc.
CoQ10 connection to Acetyl-CoA and the Kreb Cycle.
I don’t know if they can affect polymorphisms but I would think they could certainly effect the functioning of genes and the production of proteins.
Robin Rutan and Diana Arnold, this comment is for you. I also have fibromyalgia and I have found the cause to be citric acid consumption. It’s hard to avoid, but I have removed it from anything I consume and feel great. It’s a night and day difference and it took a long time to figure out because it’s a delayed systemic reaction. Don’t listen to doctors that don’t have any causative reason for Fibromyalgia–its Citric Acid, which is derived from mold.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6097542/
SelfDecode:
Although the specific tests above are not provided by SelfDecode, it is after years of searching online the best DNA analysis and amazingly detailed yet very user friendly service I have seen bar none. After uploading one’s genome from 23 or Ancestry, or using the SD DNA analysis, every SNP, symptom report, and recommended mitgators (including genetically appropriate supplements) is tailored to exactly the full individualized genome. Countless research articles are cited for each claim.
For self help with all neuroimmune issues that might require special diets and health practices, this service has told me what to lean into or lean away from with nutrition, supplements, medications, environmental chemicals. and epigenetics in general. Eventually the entire bioterrain can become much more robust as the mitos etc. are given the stressor relief and appropriate boosts they need.
Great article to give to our receptive, open-minded healthcare providers. Thanks.
The Lectin Free protocol does not allow any corn products or derivatives, as far as I know (See Gundry et al).
Corn and grains in general have long been known to harbor mold not removed by processing. So grain-eating animals suffer from this too. Also, in the last 30 years untested Roundup is generally sprayed on harvestable crops of grains and legumes to make the job more profitable for corporations. The toxic Glyphosate is not removed from the grains and legumes as the plants die and dry out. We and out animal companions eat this stuff daily since Roundup snuck under the statute$$$$$ of food safety. Roundup punctures holes in the gut mucosa so that it becomes permeable; then come the cascades of food caused allergies and diseases for both humans, pets. See the research of Dr. Stephanie Seneff/MIT and others.
Ergot mold on damp-stored rye grain has been cited for incidents in the MIddle Ages in which the entire population of towns went on a collective hallucinating and dancing in the streets type madness. A derivative of said ergot is supposed to be one of the earliest forms of LSD.
Tara, OMG. I just read this document on citric acid!!! I take the vit c effervescent tabs, thinking it’s good for me. It is made of Aspergillus. I looked up my Mold Report from last year!!! Guess what was on there, yep. I had no idea!! Thank you so much!! I have done many counter intuitive things trying to get better. I only kept fruit juices for my children growing up; thinking it was healthy. Turns out we have a genetic mutation causing the histamine levels to reach extreme levels. Check any DNA results for AOC1 variant; diamine oxidase) Reduced Production, and the HNMT variant that eliminates it both cause Migraines & :Pruritus (itching especially of the skin, eyes, ears, and nose)
Urticaria (hives) (sometimes diagnosed as “idiopathic urticaria”)
Tissue swelling (angioedema) especially of facial and oral tissues and sometimes the throat, the latter causing the feeling of “throat tightening” (sometimes diagnosed as “idiopathic angioedema”)
Hypotension (drop in blood pressure) , tachycardia (increased pulse rate, “heart racing”) Symptoms resembling an anxiety or panic attack, Chest pain, Nasal congestion and runny nose
Conjunctivitis (irritated, watery, reddened eyes), Some types of headaches that differ from those of migraine, Fatigue, confusion, irritability, Very occasionally loss of consciousness usually lasting for only one or two seconds, Digestive tract upset, especially heartburn, “indigestion”, and reflux….. all of which I have, I used to drink ONLY OJ all day thinking I was being healthy!!
Citric Acid could also be related to HFI Hereditary Fructose Intolerance. I know one Woman sick 48 years her total illness was HFI she was told ME/CFS Fibro. HFI is a Born Genetic illness
Josh mentioned muscle biopsy. I was reading about Iron overload one of the 9 genetic mutations; rs855791 happens to occur mostly in men. hemochromatosis. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2777090
HFE C282Y rs1800562 A
HFE H63D rs1799945 G
Iron build up, (milder)
hemochromatosis risk
HFE S65C rs1800730 T Iron build up, (milder)
hemochromatosis risk
BMP2 rs235756 A — Increased ferritin levels with
HFE (above)
BTBD9 rs3923809 G
HBS1L-MYB rs4895441
SLC40A1 rs1439816 C — More liver damage with
HFE
TMPRSS6 rs855791 G Increased iron stores (men)
SLC40A1 rs11568350 A Higher ferritin levels
I am 73 and had just given up hoping the medical world would discover, in my time, the cause and cure for this nasty disease . I’ve suffered with pain and fatigue, IBS, brain fog, put up with insinuations that I was faking it etc., for over 40 years. The next generations of women (and some men) may be spared, which is a beautiful thought. Thank you for studying and helping to discover these results for current and future fibromyalgia sufferers .