

The Fixer
Often when I communicate with the Solve ME/CFS Initiative (SMCI), I end up with Allison. She seems to have her fingers in everything. She seemed to know the nuts and bolts of just about everything. I knew that Sadie Whittaker, the SMCI’s Chief Scientific Officer, was making the high-level research decisions, but I concluded that Allison was probably the glue that held everything together.
The Director of Research Programs for the Solve ME/CFS Initiative, Ramiller formerly worked at the Brennan Center for Justice, a New York social justice think tank, and as a civilian employee at the Oxnard Crime Analysis Unit was apparently doing systematic analyses to identify and analyze patterns and trends in crime.
That sounds rather fascinating, but with her Masters degree in Public Health (UC Berkeley), and her Bachelors degree in Psychology with a minor in neuroscience (UC Davis), she wanted to get back into the health field. In preparation for her interview, she said she watched a Tony Komaroff interview and got hooked. She came on board as a scientific administrator in 2017, and since 2018 has been the Director of Research Programs.
She described herself as a fixer and a jack of all trades. First she took on managing the Ramsay program, budgets, peer reviews. She helped start Solve M.E.’s pilot program at the Iris Kantor Women’s Center at UCLA, which seeks to enroll UCLA faculty in ME/CFS research. With the SMCI just a short half an hour away from one of the top universities in the country, it makes sense for the SMCI to forge connections there.
The first question Iris Kantor researchers asked was: “where can we get patient samples?”. Ramiller certainly knew patient samples. One of her first jobs was overseeing the Biobank, which is still distributing samples – most recently to Bindu Paul at Johns Hopkins University. That brings us to the You + M.E. Patient Registry – the first ME/CFS patient registry ever.
SMCI Goes All In on the You + M.E. Patient Registry
“I firmly believe that a global registry and biobank will be the single most impactful driver of progress in ME/CFS, deepening our understanding, helping us pick apart the heterogeneity, and driving us towards treatments.” Sadie Whittaker – SMCI
There’s no doubt that Sadie Whittaker, Allison Ramiller and the SCMI are going all in on the You+M.E. Patient Registry. How far in is the SMCI going with the Registry? After spending 4 years developing it, they’ve hired a full-time staff member, Elle Seibert, a former researcher at a USC neuroendocrinology lab, to manage it.
This is a Registry, it should be noted, that is designed to grow – and that’s a good thing. The larger a patient registry is, the more effective it can be.
When COVID-19 showed up just as the registry was about to be launched, Ramiller, Whittaker and the SCMI pivoted, added a section to chart how people with ME/CFS do with the virus, contacted Body Politic – the largest post COVID-19 recovery group – gave them a tour of the platform, and got in touch with UCLA to see if the registry could figure in with their COVID-19 tracking efforts.
They’ve worked with the UK Biobank to begin collecting core sets of data that would allow the two databases to merge – in effect creating a U.S.-U.K. registry. They’re working with Emerge in Australia to in effect create a U.S.-U.K.- Australian registry.
They’ve started conversations with Tony Komaroff and the ME/CFS research center at Columbia University to adapt the app they’re creating for use in the registry.
Research and Clinical Trial Accelerators
ME Action’s Jamie Seltzer is certainly high on patient registries. In her Oct 2019 post, “Patient Registry is the Key to Achieving Big Data For M.E.“, Seltzer reported that patient registries have been significant research accelerators. Studies on Rett Syndrome, a rare neurological illness, for instance, increased tremendously in the years following the creation of the registry.
Particularly in rare/underserved diseases, even patient registries which do not contain biological samples (SMCI’s Patient Registry/Biobank contains them) have been shown to increase the number of clinical trials, research studies and published papers. It’s no wonder the SMCI has put so many resources into the Registry.
Help for ME/CFS Better on the Individual Level
Data from the mobile app could help each of us tease out what works and what doesn’t and how we’re doing over time. Illuminating patterns can show up when you track your symptoms. Rachel Carder used symptom tracking to identify drugs that made her worse – three days later – and to identify dietary changes that helped her sleep better.
I haven’t used the app yet but I’ve been told that it provides the capability to assess whatever factor that you wish.
The Big Picture – Identifying Disease Processes
That’s great on an individual level, but participating in the Patient Registry also has the potential to us better understand the disease. Your secured, anonymized, and encrypted data that will be fed to the Registry will allow researchers to do deep “phenotyping” they’ve never done before.
That could include uncovering symptom subsets, identifying different kinds of post-exertional malaise, understanding disease progression (and figuring out who is likely to progress to what), perhaps even matching up symptom profiles with treatments that work or clinical trials. One of the biggest questions, of course, is whether ME/CFS is a single disease or is made up of several diseases? The You+M.E. Patient Registry could help answer that.
What role does type of onset, gender, age of onset, comorbid illness, family history, or treatment history make? Does having a gradual vs an infectious vs a toxic onset effect what kind of ME/CFS you have? Do people who get this disease when they are younger do better than people who are older? And what happens to ME/CFS patients as they age – a big question for many of us. Is ME/CFS a pathway to autoimmune illness for some? Is ME/CFS different in women than men and how? Just how bad are the sleep problems in ME/CFS? The list goes on and on…
Size Matters
“The larger the data set available, the higher the probability that meaningful discoveries can be made.” SMCI
The You + M.E. Patient Registry has the potential – if it’s embraced by the ME/CFS community – to tell us much. The more people that use the Registry, the more effective it will be. The powerful machine learning tools that are now available present the possibility of learning more and more as the Registry grows.
The Registry adds a vital piece of medical infrastructure to ME/CFS. So far, about 1,100 people have signed up. Ideally we want tens of thousands. Besides ME/CFS patients, the SMCI is particularly looking for age and sex matched healthy controls. At the moment. the Registry is only open to U.S. residents, but other countries will be added. The idea is to go global.
Sign up for the You+M.E. Patient Registry here
Watch the Webinar
Thursday, July 30th | 10:00am PT / 1:00pm ET : You + M.E.: A Registry for the Whole ME/CFS Community
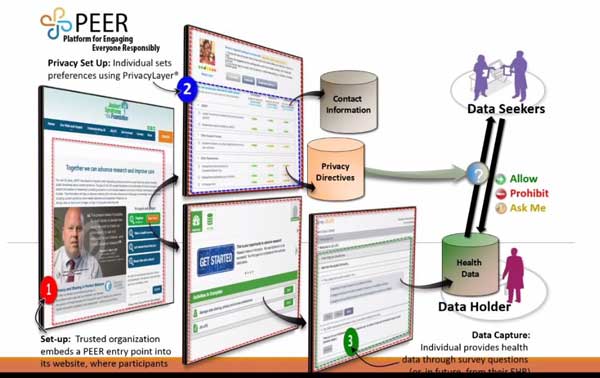
The You + M.E. Registry and Biobank is a collection of patient-reported data and biosamples (e.g. blood) from people living with myalgic encephalomyelitis (also known as chronic fatigue syndrome, or ME/CFS) and healthy controls.
The goal of the You + M.E. Registry is to register individuals with ME/CFS and non-ME/CFS control participants and compile demographic and health information into a data bank that will further our understanding of ME/CFS.
In this upcoming webinar, we will introduce the newly launched You + M.E. Registry and Biobank. Our Chief Scientific Officer, Dr. Sadie Whittaker, and our new Solve M.E. CEO, Oved Amitay, will take a closer look at what the Registry has to offer, and how to make the most of your You + M.E. experience!
Register here.





 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



I thought Body Politica was the biggest Post Covid support group until I found this Facebook page ‘Survivor Corps’ with 84,724 members and growing every day with people who aren’t recovering.
https://www.facebook.com/groups/COVID19survivorcorps/?ref=share
The ones who’ve had it the longest over 100 days seem to have less of the weird symptoms and are now experiencing mostly ME/CFS symptoms. However the earlier people are describing symptoms like rashes and hair falling out, ‘Covid toes’ with blisters, that I haven’t seen in ME/CFS. So maybe the end result once many of the weird symptoms go is ME/CFS
The interesting thing I’ve spotted is many but not all have ‘Post Exertional Malaise’ a condition unique to ME/CFS and it’s precursor Post Viral Fatigue Syndrome (PVFS)
And this post Covid group with 14,300 members
https://www.facebook.com/groups/longcovid/?ref=share
Both are worth going to and giving them advice, but not those without our ME/CFS symptoms, because it’s a big spectrum of people with various unusual symptoms
I’ve been giving the ones I’m sure have ‘Post Exertional Malaise’ this good information from the ME Association on how to manage it.
So It would be good if other ME/CFS sufferers did the same. As the more ‘Post Covid people’ that know what’s wrong with them the more likely we will get research funding for all of us.
https://www.meassociation.org.uk/wp-content/uploads/MEA-PVF-and-PVFS-Following-Coronavirus-Infection-30.04.20.pdf
Thanks Brendan – this “The ones who’ve had it the longest over 100 days seem to have less of the weird symptoms and are now experiencing mostly ME/CFS symptoms.” is actually the pattern seen in the early M.E. outbreaks. Lots of weird symptoms in the beginning – resolving to a familiar ME/CFS-like condition after some time.
Thanks for pointing that out. 🙂
i got over 3 decades the weirdest symptoms. Just the oposit. And so many of them.
I think there is also a long term trend where you gather more symptoms over time.
For me I had the most symptoms early on – quite a few them receded leaving the PEM, fatigue, muscle pain etc. for a long time
Over the longer period of time, though, some new ones like chemical sensitivities have, however, cropped up, however.
This is exactly the kind of thing the Registry could uncover…:)
This is most informative, Brendan – thank you for your insights!
hi cort,
do you know when other countrys wil be involved like belgium? i often find solve me cfs only for US citisans, even with for excample asking politicans for more money. I can do that not through them because it is only US based. And then you write colaboration with the uk and australia. So no belgium or even europe (uk is no more in europenian union-brexit), thanks!!!
It’s imagine it’s more complicated than we think with different countries requiring different degrees of security and having differing legal requirements (???)
The U.K. and Australia are in the mix early as they’re also creating their own patient registry type systems. Maybe we will find out in the webinar tomorrow about when people from other countries can join.
The SMCI knows that the more people that get in the registry – the more effective it will be at helping us understand ME/CFS. That’s why they’ve been working to get the UK and Emerge efforts aligned. I imagine they will do their best to get as many countries as possible in there – hopefully starting with Canada (for personal reasons) and then moving on to Belgium 🙂
What do we do if we experience registration problems? My registration can’t be verified, can’t be restarted and there seems to be no technical be help…
You could try emailing them at solvecfs@solvecfs.org
Yes, I had problems too – it just didn’t seem to acknowledge I’d registered. I’ve just tried again via the link, at the bottom of Cort’s blog, and it’s now worked.
I had issues registering for the webinar – I can’t register for the You + ME Registry as I’m in Ireland.
I am working through my registration and it is good that I can stop and start again another day. I was able to encourage another lady to register who was worried about the tracking program and thought she would be unable to keep up with that. She is very pleased with the experience and knows others to ask to register. I will ask one of my sisters to become a control. Please everyone….participate. We need to prove to the medical community how many of us there are.
I have to laugh about You + M. E. as I was the very first person to register. Funny thing is, what I got was actually the program to design the registry. I was quite confused trying to fill out the questions, but they soon realized what the problem was, fixed it and so now I’m set up to contribute! Bless their hearts for being so on top of it!
Regarding my comments about attempting to get other medical people more interested in ME/CFS research, I got commentary accepted on MedPage Today regarding the similarities between Covid long-haulers and those of us with chronic fatigue. I made an appeal for more research and so I hope a lot of doctors got to read it. Plant the seed!
As for symptoms, I got my weird symptoms first (100 days) and later they got to be the now familiar ME/CFS which has lasted for decades.
Hope everybody signs up for the registry!!!
You’re like an ME/CFS Johnny Appleseed 🙂 Great to hear. (If you have a link to that Medpage commentary that would be great. )
O.K. here’s the link; https://www.medpagetoday.com/patientcenteredmedicalhome/patientcenteredmedicalhome/87764
I’ll double check it in a moment just to confirm.
My ‘seed’ is in the comment section after the article under Nancy Bolduan. One has to wait for each comment to be vetted so I am pleased it got through. There are some other people ‘on our side’ in that section as well!
I’m waiting for orchards of apple trees!
Decades !! ✊?✊
Thank you for working out the bugs!!! Surprised you weren’t the first one commenting here too! ?
Our organization, Birth Defect Research for Children, sponsors a National Birth Defect Registry which has 10,000 cases. Since the registry was launched in 1990, it has gone through multiple revisions. We are preparing to launch our newest cell-phone friendly version in mid-August. I will be interested to register in the ME/CFS registry and compare the structure and questions to our registry which was designed by seven scientists working in fields related to birth defects and the environment. Can you register now or do you need to wait for the seminar?
OMG – I forgot to put a signup link! Just added it – https://youandmeregistry.com/ You can enroll now.
Cort — or anyone else with direct knowledge…!!—should folks like me with “mild” and/or undiagnosed/hastily unofficially diagnosed illness, or folks with fibro or dual diagnoses, also register?
Given how unlikely (IMO reading this site a lot) it is that the two conditions are truly discrete, biologically separate entities—and how frequently were mis- and under-diagnosed—what is the Registry doing to account for that? Is it worth registering in case some studies might be symptom focused, or include complementary specialist evaluations…?? Or am I just dreaming? ?
That is a great question – particularly since many people diagnosed with FM or ME/CFS might have been diagnosed with ME/CFS or FM if they’d seen a different practitioner.
I will ask Allison.
I watched the webinar yesterday and enjoyed listening to the SMCI team chatting about the You + ME Registry and Biobank.
They seem to be a very talented team ?
I’ve just read the piece by Rachel Carder, on tracking her symptoms, and I can add that I did the same – not on anything as sophisticated as an app – just bits of paper/notebooks/calendars etc.
I have a blood pressure monitor, a blood sugar monitor, a peak flow meter (asthma), a pulse oximeter now and a thermometer.
I figured out the fructose issue I have by closely monitoring my blood sugar through the day. I couldn’t work out why I had a problem, when my blood sugar didn’t really rise very high. Eventually (and it took me a very long time) I came to the realisation that it was when I ate too much fructose, that I developed particular symptoms.
I actually bought the blood pressure monitor because I was concerned that my blood pressure dropped too low – that was connected to the fructose – particularly corn/maize products. But later on, I developed problems with high blood pressure. My BP is still up and down.
My blood sugar also seemed to plummet, again due to the after effects of high fructose levels. Basically now, I don’t eat much fructose.
My current brain monitoring system consists of a multi-coloured BIC biro and my calendar. If my brain is functioning well, I write the time in green biro on the calendar. If my brain is struggling, I write the time in red biro. Mornings are good, afternoons are not. I could do with an orange biro for middling but have had to make do with the blue instead.
Years ago I wrote out a plan of what I needed to do, if I developed particular symptoms – because generally my brain function diminished and I couldn’t think/remember what I needed to do next. I kept this on the fridge door.
There was also a time a few years ago, when my symptoms got so bad I had to stop monitoring them because it just made them worse – especially the high blood pressure. However I’m much more under control now.
Am a newbie to this forum.
Am having a 1000% pain day, but have been reading through one of the forums concerning Fibro and Dopamine. Am absolutely done in, physically and mentally.
I think found this bio bank thingy, and was interested in joining. Had to read paragraph after paragraph…..only to find at the very end, that it’s only in the US!
Please could you put that info at the very beginning.
Thanks
Sorry about that Janet. The Patient Registry should be opening up globally soon. That will probably be the next blog on it.
Hi Cort
Thanks.
Have registered for the CFS/ME forums.
Does that automatically register me for the Fibro one as well?
Sorry….not very familiar with how forums work. Haven’t been on one for years.
Yes, you have access to everything – welcome!