

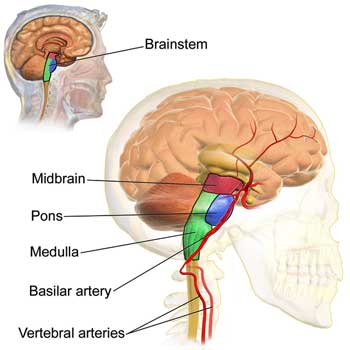
The Brainstem
The brainstem – a small area at the base of the brain that contacts the spinal cord – has become of ever more interest in chronic fatigue syndrome (ME/CFS). Barnden’s studies suggest brainstem involvement is present. VanElzakker believes brain scans focused on upper areas of the brain have missed a crucial aspect of ME/CFS.
Then there’s craniocervical instability (CCI). Surgical procedures that stopped the skull from impinging on the brainstem in some people with ME/CFS have led to recoveries – which have made it clear that brainstem issues have the potential to produce every symptom found in ME/CFS.
We know the brainstem is heavily involved in the regulation of basic processes that we need to survive, including autonomic nervous system functions such as heart rate, breathing, sleep and in conveying sensory and other signals to the brain. It turns out, though, that the a small area of the brainstem called the parabrachial nucleus also plays a role in chronic pain.
The finding that the brainstem matters in chronic pain illuminates two things: (a) how complex a process pain production is; and (b) how much researchers are learning about it. Several years ago, Alan Light, PhD noted that he aimed to do for the production of fatigue what we already knew about the production of pain. We know quite a bit about how pain in fibromyalgia (FM) is produced.
We know, for instance, that two kinds of small nerve fibers in the body that transmit pain signals to the spine, and ultimately the brain, have become hyperactivated in FM. We know that another process called “windup” results in other nerve fibers becoming hypersensitive to pain. We know that several neurotransmitters that upregulate pain production are increased in FM, that the pain inhibitory pathways are underperforming in the disease, and that areas of the brain that produce pain have become hyperactive.
So much is going on that it seemed as if every aspect of the pain producing process has gone sideways in FM, but researchers are not done learning about how pain is produced – not all.
Parabrachial Nucleus (PBN)
“This tells us that chronic pain is manufactured by the brain. It’s not a one-way process driven by something coming up from the periphery; the brain is not a passive recipient. To me that’s what’s exciting: The brain is actively constructing a chronic pain state in part by this recurring circuit.” Mary Heinricher
Another part of the brain that has not been assessed in fibromyalgia may play a key role in the pain found in the disease.
Pain and other signals get passed up the spinal cord to a part of the brainstem called the parabrachial nucleus.
Recent animal studies indicate that the brainstem also plays a role in pain production. Pain starts off as a sensory signal that gets interpreted by the brain. Because pain usually denotes some sort of injury, the brain has given pain signals a special emotional resonance to do something about the situation. So while a pain signal starts off as a kind of sensory signal, over time it becomes more than that. The first place the process of turning a pain signal into a complex response starts is at the parabrachial nucleusin the brainstem.
Studies have shown that PBN has a windup problem of its own: i.e. in chronic pain, nerves in the PBN remain active and ready to punch out pain signals long after they’ve calmed down in non-pain states. That suggested that some sort of inhibitory pain control had been lost.
The main excitatory neurotransmitters in the brain – glutamate – were lacking in the part of the PBN involved in pain production. Careful studies indicated that projections from the amygdala – generally known as the fear center of the brain – were responsible. They weren’t the projections that the researchers expected, though. Through its interactions with PBN, the amygdala was turning down pain – not ramping it up.
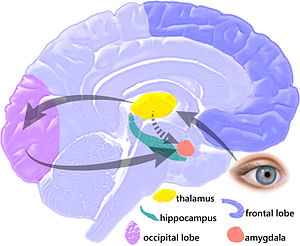
In a switch the amygdala – long known as the fear center of the brain – was actually sending calming signal to the brainstem.
While it’s mostly known for producing sensations of fear, the amygdala was shooting calming signals straight to the PBN in the brainstem. It was also doing it using different neurotransmitters such as dynorphin and somatostatin. Surprisingly, it was the loss of amygdala signaling that was leaving the PBN in a hyperactive state – and the subject in more pain.
In fact, one animal study, which painstakingly stimulated nerves known to carry inhibitory pain and hunger signals from the hypothalamus to other parts of the brain, found that only the activation of nerves leading to the PBN reduced pain behaviors.
Once again, but in a new way, we can see how vitally important pain inhibition process is to our experience. The researchers had found a new way to heighten pain signals – and create a chronic pain state.
Going Defensive
The twitchy PBN wasn’t producing just pain, though. Researchers have thought that the brain does most of the emotional processing, but it turns out that the PBN – the main processing center of pain signals coming from the body and up the spine – is doing some emotional processing of its own. The PBN determines whether brain regions involved in escape or aversive behaviors (yelling, hitting, getting angry) are alerted or not. Above all, the PBN is involved in defensive reactions; e.g. aversion.
Nerve projections to other parts of the brain indicate that the PBN plays a key role in our emotional responses to pain. In fact, it and the amygdala appear to be the two general alarm centers of the brain. A recent review called the PBN “a ‘hub’ for pain and aversion” that responds to any potentially dangerous situation. Not surprisingly, PBN neurons have been associated with the “freeze response”. One researcher likened the PBN to a home alarm:
“The alarm goes off while you’re away, and you don’t know if it’s a broken window, an intruder, or a fire—you just know that something bad has happened.”
Interestingly, given the possibility produced by Bob Naviaux that people with chronic fatigue may exist in a kind of hibernation-like metabolic state, the vast convergence of different sensory inputs (cardiovascular, respiratory, metabolic, and pain) at the PBN appears to give it a key role in determining whether an organism enters into hibernation. Inhibiting the activity of some PBN neurons prevented anorexia (e.g. the starvation phenotype) and malaise in laboratory animals.
It’s connected with several brain areas associated with FM, including the hypothalamus, insula and the periaqueductal gray area. The PBN also regulates autonomic nervous system functioning (respiration, blood pressure, heart rate, water balance).
Conclusion
Once again we see a part of the brain that has been largely ignored possibly playing a major role. The PBN used to be thought of as a passive relay center for sensory signals, but it’s clearly not. Not only does it determine to some extent how the brain will react to pain signals – with aversion or escape – but it’s also at the tail end of an inhibitory pain process.

The PBN and the amygdala are the two alarm centers of the brain. (Image by Sarah-Richter from Pixabay)
We don’t know if the parabrachial nucleus in the brainstem plays a role in the pain in fibromyalgia, but it’s hard to imagine that it doesn’t. As the PBN discovery fleshes out our understanding of how pain is produced, it also illuminates how complex the process of pain production is – and helps us understand why producing a good pain drug has been so difficult. (I was unable to find any drug associated with the PBN.)
We know that the pain in FM is associated with numerous issues in pain processing, neurotransmitter production and brain activation but we still don’t know what starts the process off. Even so, it’s good to know that researchers are continuing to produce new insights into how pathological pain (i.e. inappropriate pain) is being produced. Given that FM occurs in some people after an infectious event, it’ll be interesting to see if the research into the COVID-19 long haulers provides some insights in chronic pain.






I have always believed that the lack of inhibition and the leaky brain filters are intentional in order to bring our attention to the fact that the overactive sympathetic nervous system has caused all our muscles to become contractured and that we need to do something about it as the trigger points need to be manually deactivated. That would be the reason why the amygdala is not calming down the PBN.
Your idea makes sense to me. Learning why my muscles get so contracted and tight so easily would be such a breakthrough.
I’ve suffered for many years from pain and multiple system breakdowns from FM/CFS/ME. I’ve also researched potential causes and effects but I, for one, don’t support using animals in these research efforts. Intentionally causing pain and suffering to these creatures isn’t humane. It’s simply wrong. We all want answers to the puzzle that afflicts many of us worldwide but these studies are not the path we should take. Researchers from both private and public funded institutions have told me they readily acquire funding for these animal projects over pursuing other research avenues; unfortunately, many admit it isn’t worth it in the long run and consider it “junk science.” For example, cardiologists admit they don’t trust results using dogs in heart research valid. Why? Simply that dogs hearts aren’t like human hearts. The extreme suffering the dogs went through is well documented and nothing short of torture. Let’s invest in research that doesn’t involve harming other living beings.
Janice I totally agree with you about the torturing of animals for our benefit – it is despicable.
I’ve had ME/CFS/FM for 32 years and am now completely homebound, bedbound and cared for by my 90 year old husband – I am now 84. I would willingly give the rest of my life right now if it would stop the cruelty to animals. This wretched illness destroyed my life when I was 51 years of age and no cure has resulted by torturing animals.
There are efforts to use sophisticated modeling approaches instead of animals. They will take time, though, to develop. I don’t know that there’s a good answer to this. I agree that it’s a terrible situation. I do know a ton of them exist. I don’t know how important these animal studies are. These studies used animals to uncover a new pain pathway which probably, given how fundamental it is, probably does exist in humans. Is it worth it? That’s a great question.
Cort, I appreciate you offering to post my comments as I know the dilemma it has with some research projects and institutions. I started a nonprofit in 2007 to educate and bring awareness to animal issues. In my efforts, I’ve received phone calls from people participating in these research projects to ask for my help to help these animals. One person worked at a major private research facility in Ohio. This person was prepared to deal with animals in these projects. Unfortunately, it was so bad this person quit and had to seek mental health therapy to deal with the horrific experience. It was that traumatic. No, we should review these projects. Donors who oppose animals in research need to know about where their donations are going. I managed an abuse prevention fund for 10 years and know this process. On a personal note, my son-in-law discovered he had Stage 4 melanoma cancer 2 weeks before their wedding and died just months after marrying my daughter. He was 27 years old. As a science teacher I ask him how he felt about research on animals if he knew it would help him. He flat out opposed it. In fact, he participated in stem cell research that helps cancer patients today.
Cory, have you heard of the International Animal Rights Conference 2020? Here’s one featured speaker you might find of interest.
https://m.youtube.com/watch?fbclid=IwAR1mEkIXCr3o-EgpNJlxCFKWbV-j8AnKO7FyYay_0BJ8MzGlWgOw9Cxw08w&v=q6MsabrIyrs
I agree completely!
Homeopathy has never needed animal torture or sacrifice to produce the excellent healing results this field has produced for at least 300 years. Homeopathy is nontoxic and does no harm in acute or chronic cases; it is an exemplar of the best in energy medicine whose successes have been smothered by Western corporate medicine propaganda in the US since the Flexnor Act of 1912, which also forbade much of women and people of color participating in medical school curricula. Well utilized in Germany, Britain (especially among the Royal Family) and many world cultures for centuries, Homeopathy continues to be attacked because it is so affordable, accessible to the self-treating patient, and baffling to those who understand only non holistic outdated, mechanistic healing methodologies and refuse to expand their practical knowledge base of energy and Eastern medical approaches.
While chronic cases can be hard to figure out for oneself and might require a degreed pro for best results, acute first aid remedies are easy to apply by anyone and are available everywhere.
Best of all, this nontoxic branch of medicine saves animal suffering through nontesting, and vets can produce brilliant recoveries in animal patients when conventional medical approaches have failed.
Persistence in learning and research is required for success with this, as with everything else, and plenty of free info and training are available online, as are databases of remedies and their characteristics and applications.
I’ve long thought that my ME was some neurological mechanism malfunctioning, probably a small portion of the brain. The findings for the PBN fits that. My (admittedly vague) theory for ME is that something triggers an immune response in the body, which then triggers an immune response in glial cells. Those glial cells affect the function of certain neurons which in turn affects the glial cells, locking us into a positive feedback loop. There was another paper ( https://newatlas.com/science/brain-circuit-stress-insomnia-immune-system/ ) recently showing the link between stress, a small cluster of cells in the hypothalamus, and sleep problems. I then found some information on NG2-glia and their role in proper neural functioning, and the other glial cells also affect neural functioning, in ways that we probably are still unaware of. It certainly seems plausible that glial cells are directly involved in ME, and that their effects could form a feedback loop.
Hi Nico,
Yes, I wonder something similar to you. I came across an article by Michael B VanElzakker, in which he discusses research methods in neuroinflammation. I thought it was a really interesting and level headed look at what was going on in that research field and a sort of round up of ME/CFS in general, which I found affirming.
Then when following the link in Cort’s first paragraph at the top of this article (VanElzakker in blue) I found that Cort had done a blog for Simmaron Research on the same article.
Here’s a link to the original article:
https://www.frontiersin.org/articles/10.3389/fneur.2018.01033/full
And it’s title is:
Neuroinflammation and Cytokines in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Critical Review of Research Methods
Great link, Tracey Anne! It really shows why there seems to be so much variation in cytokine studies in ME/CFS! Or other studies as well. Clearly we need more co-ordinated research!!!
If glial cells could be a target, I wonder which drugs could modulate them? Think I’ll go on a google safari! Never stop looking!
Michael is a daring author and a very smart guy 🙂
Your hypothesis could be right on. Thanks for the link to sleep/hypothalamus connection – that’s exciting stuff.
The association with craniocervical instability finally provides a good mechanism for the association between fibromyalgia and neck injuries reported by Chaitow in his book ” The Fibromyalgia syndrome”.
It also accounts for many features of the so called ” Affective Spectrum Disorder”.
Craniocervical syndrome is strongly associated with whiplash, TBI and connective tissue laxity disorders such as EDS.
Given the progression of disordered stress response and immune response in these conditions secondary (epigenetically based) ligamentous laxity is plausible.
However it has been known for some time ( See” Manual Therapy in Children” Ed H. Biedermann MD) that these neck instability conditions tend to persist.
I have a very unstable neck myself, which contributes to both fatigue, autonomic dysfunction and periods of irritability (loosely called Bipolar- another of the Affective Spectrum Disorders. It was greatly worsened by a whiplash injury in 1987, and has been additionally worsened by years of desk work, to the point where I have had to give up work.
What we do to help the severe cases is one matter, but it is clear to me that exercise that improves core strength and stability helps the situation- and that Yoga and Tai Chi are classic examples, and may have evolved to address these kinds of problems.
Ive spoken to my Tai Chi teacher about this ( Ive not seen him for a couple of years) as I was looking for a more comprehensive solution to my issues. His comment was that cases of my sort of severity would be dealt with by prolonged martial arts training in a monastery- maybe lasting for 3 years or more.
What interests me here ( and I realise I am coming from a very different angle here, one that not all will relate to) is that the Asian martial arts have been known to have particular “kill points” which, if they did not produce an immediate fatality, would precipitate the victim into a state of decline that could only be called fibromyalgia.
Now I do know some of these points and I am not going to repeat them online- but they would all produce a flipping of the head on the upper neck that would replicate a whiplash.
In any case the measures of working at good core tone, at avoiding high speed accidents and head injuries, and eating a diet that favours good production of collagen makes a great deal of sense.
However, from my own point of view, it is not at all easy to engage a program of Tai Chi or Yoga ( especially as the actual reasons for them are rather obscured by the mystical way in which they are presented.
Im mentioning this to query the whole question of core tone and what exercises everyone has found best to initiate better tone and strengthen the core muscles- because initiating that process is the hard part for any of us who has fallen in this hole.
I suspect we would all be much better off if we could build up our necks to look like “The Rock”——- but how to get started ?
More on “Affective Spectrum Disorder”. These conditions cluster strongly in families:
Affective spectrum disorder (ASD) rep- resents a group of psychiatric and medical conditions, each known to respond to several chemical families of antidepressant medications and hence possibly linked by common heritable abnormalities. Forms of ASD include major depressive disorder (MDD), attention-deficit/ hyperactivity disorder, bulimia nervosa, cataplexy, dysthymic disorder, fibromyalgia, generalized anxiety dis- order, irritable bowel syndrome, migraine, obsessive- compulsive disorder, panic disorder, posttraumatic stress disorder, premenstrual dysphoric disorder, and social phobia.
Excellent and practical contributions, Andrew. There are many portal approaches to healing; some are more effective than others at different times in the healing process. Integrative mind/body medicine, which includes forms of energy medicine, can produce great results when combined with conventional medicine.
This article reminds me of all the past articles about balancing the parasympathetic and sympathetic nervous systems through modulating vagus nerve responses. Some of this can be done through yoga, breathing techniques, etc.
Knowing one’s tendencies to systemic mind/body genetic weaknesses through genetic data bases targeted to precise individual needs (SelfDecode and its sister site SelfHacked are awesome, the only ones I have ever seen so user-friendly) can help prioritize healing approaches; SD is that detailed. It even covers the status of people’s CBD/endocannabinoid pathways and what might work, all based on NCBI research which is posted at the end of every SD or SelHacked report.
Fascinating Andrew! HR has covered craniocervical instability quite a lot and we have more blogs coming up. Problems with the neck/spine area can certainly produce every symptom we see in FM and ME/CFS.
Cort, are you aware of the book “Craniocervical Syndrome and MRI”? there is very good evidence for both neurosurgery and precision chiropractic (see Scott Rosa).
Both upright MRI and other imaging techniques such as fluoroscopy ( Digital Motion X Ray) can demonstrate the degree of compromise to the upper spinal canal very clearly. I have a very unstable R C1-2 joint- and knowing that the damn thing is shifting that much off the safe range is concerning to say the least. Neurosurgery is more permanent but has more side effects, while the chiropractic techniques need to be repeated about every 3-6 months in many cases.
The neurosurgeon Joel Franck gives a good presentation both in the book and here:
https://www.youtube.com/watch?v=spMkX3XAWjs
His emphasis in the book on the psychiatric symptoms caused by these problems is well worth reading. The implications of the PBN dysfunction/dysafferentation go well beyond fibromyalgia.
Your comment in the post above – “he PBN determines whether brain regions involved in escape or aversive behaviors (yelling, hitting, getting angry) are alerted or not. Above all, the PBN is involved in defensive reactions; e.g. aversion.” is highly relevant to psychiatric presentations both in many of the affective spectrum disorders and the more recently described “intermittent explosive disorder”.
From what I can read these reactions are “bottom up” and relate to the failure of normal inputs from the body/spinal cord, rather than to faulty cognitions. The faulty cognitions- more the realm of traditional psychiatry/psychology, come in when we try to figure out why our behaviour was so irrational.
Ha! I actually know the monastery that maybe you are referring to…
I know of a child that was sent there for health issues
and they were brutal to him.
His parents switched him to the taiji village, with another master,
where they were maybe better to him.
I’m not sure that his health issues resolved.
My certainly got better – and that was just from one summer
It was were I first learned that I have soft joints – hypermobility.
People do, do that.
They told two kinds of people usually go to the village for year long stays – the unhealthy in body and the unhealthy in mind.
I started practicing taiji when I was a teenager, because of back injuries.
It did always make me feel better, and be able to stand, afterward.
I didn’t keep it as a daily practice and I do wonder if I would have, would I have had experienced the decline I did.
😉
There’s – there’s a (I don’t know his specialty – chiropractor?) in South Korea, he uses a mouth piece for a daily regimen to treat things like Parkinson’s, and dystonia, and I was able to discern a patient or two that may have hEDS. At first is like whoa! is this real?! Same principle, cervical and craniotomy cervical instability. His method is non-surgical.
Great, great great info. Thank you
Great article. I’m firmly convinced there is brain stem inflammation in LC . I’ve been experiencing bewildering episodes of intermittent shortness of breath in which my breathing rate would ramp up for no reason at all and it felt like I couldn’t get enough oxygen, yet my lungs were clear and my O2 sat was 100%. I was an extremely fit competitive athlete prior to contacting LC. Drs tried to brush it off as hyperventilation, anxiety, prescribing an inhaler- no, no and no. A recent study(2023) in the American Journal of Neurology compared brain PET scans of a control group against a matched sample of patients who had been infected with Covid. It showed “ RESULTS: Compared with the control group, patients with a history of COVID-19 infection exhibited focal areas of hypometabolism in the bilateral frontal, parietal, occipital, and posterior temporal lobes and cerebellum (P = .05 uncorrected at the voxel level, family-wise error–corrected at the cluster level) that peaked during the first 2 months, improved to near-complete recovery around 6 months, and disappeared at 12 months. Hypermetabolism involving the brainstem, cerebellum, limbic structures, frontal cortex, and periventricular white matter was observed only at 2-6 months after infection. Older age, neurologic symptoms, and worse disease severity scores positively correlated with the metabolic change”. Same study needs to be done with Long Covid and ME/CFS patients!