Two million dollars for chronic fatigue syndrome (ME/CFS) research…You don’t hear those words very often. The NIH just gave Dr. Shungu and associates a big two million dollar grant to peer into chronic fatigue syndrome patients brains.
This is the kind of study that makes you love the NIH; it’s expensive, it’s big and if it works it could really change things.
Oxidative Stress and Glutathione Get Their Day…
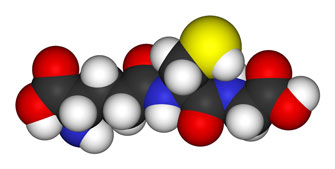
RIch Von Konynenburg glutathione depletion theory got a boost when Shungu found significantly reduced glutathione levles in ME/CFS patients brain.
Perhaps no finding has been more consistent in ME/CFS than increased levels of oxidative stress but it’s always been hard to know what to do with these findings. High free radical levels, after all, occur in many chronic disorders. You certainly don’t want a bunch of free radicals smacking your cells and then releasing more free radicals that simply feed a free radical firestorm but the finding was too general to base a biomarker on.
Rich Van Konynenburg’s glutathione depletion theory and his corresponding methylation protocol has been used successfully by patients and even been picked up by doctors but study evidence for the theory was weak. Both oxidative stress and glutathione, at least, at the research level appeared to be at something of a dead end…. until Shungu came along and fixed both problems.
The Brain – On Fire
Shungu’s 2012 study found an an alarming (36%) decrease in the body’s chief antioxidant, glutathione (GSH), in the cortical regions of the brain. (Rich Von Konynenburg, a longtime proponent of the glutathione reduction theory in ME/CFS, must have been smiling broadly.)
Shungu’sstudies suggest damaged mitochondria in the brain’s of ME/CFS patients are producing lactate
The NIH was no doubt highly impressed with the fact that the low GSH levels were associated with increased levels of free radicals and Shungu’s other findings of increased levels of cerebrospinal fluid lactate and reduced blood flows. Shungu believed free radicals whipped up by pro-inflammatory cytokines in the brain whacked the mitochondria hard enough to shut largely shut them down, causing lactate levels in the brain to rise. Dr. Teitelbaum pointed out that high lactate levels could contribute to anxiety/hyperventilation (think sympathetic nervous system activation/’arousal’).
Shungu hit a snag, though, when he did not find ‘significant’ differences between ME/CFS patients and patients with major depressive disorder but he did find a trend; ie a finding that almost reached the major number (p<.05) at which findings suddenly become ‘significant’.
Shungu’s small sample size (n=15; CFS, MDD and controls) suggested he didn’t have much differentiating power anyway, suggesting that simply increasing the sample size could very well turn the differences between ME/CFS and MDD patients into something ‘significant’ . Shungu would clearly love to differentiate MDD from ME/CFS patients but he’s actually up to much more than that, and it’s at that point this study really takes off.
Shungu knows he has a big problem with subsets in ME/CFS; that all you probably need to do is scratch the surface of this disorder to get a Pandora’s box full of subsets laughing at researchers attempts to get ahold of this disorder. This subset problem is almost certainly the problem holding this field back; it’s holding it back from getting recognition, it’s holding it back from getting funding and it’s probably holding back the biomarker hunt big-time.
Shungu is taking the bull by the horns; he’s going to try to break up both chronic fatigue syndrome and depressed patients into subgroups first and then scan the heck out of them. The NIH brief doesn’t say what tests he’s using to stratify patients but if he can do this successfully we could end up with a range of subsets; ‘pure’ CFS patients who don’t have a hint of depression, CFS patients with a bit of depression, MDD patients with some CFS characteristics ( post-exertional malaise?), and ‘pure’ MDD patients you’d never mistake for ME/CFS patients – all with different brain signatures..
Why Depression?
Why depression? Because many of the symptoms in the two disorders are similar and that makes depression, particularly if you’re doing brain studies, a good proving ground for a biomarker. If researchers can distinguish ME/CFS patients brains from depressed patients brains they’ve probably got a pretty darn good biomarker.
Plus many people outside the community still view ME/CFS as a kind of ‘depression’ and the only way to prove it’s not, is to do studies that prove it isn’t. This isn’t to say that the two don’t share some relationships; Hornig and Maes and others believe that inflammatory processes underlie both ‘depression’ and ME/CFS. Some otherwise healthy people, after all, wake up one morning with ‘depression’; no trauma needed, just some sort of mysterious system reset – just as occurs with ME/CFS. Some forms of depression do respond to counseling ..others don’t.
Dig Deeper – Marco’s Neuroinflammatory blogs propose that a neuroinflammatory condition characertized by, yes, high rates of oxidative stress, underlies ME/CFS and many other conditions
The important part of this study is NOT proving that chronic fatigue syndrome is not depression; we know that…it’s identifying brain abnormalities (biomarkers) unique to this disorder. Every possible biomarker is going to have to get tested against other ‘similar’ disorders to ensure that its real. To Shungu’s credit he’s way ahead of the game here; he ticked off anxiety with a 2009 study showing lactate levels in were higher in ME/CFS than in generalized anxiety. If Shungu can now snag depression as well as anxiety he’s got a solid basis for a biomarker.
The lactate levels weren’t just ‘higher’ in ME/CFS patients, they were 300% higher. Anxiety/arousal has been a touchy topic in ME/CFS but Shungu’s work lays out a clean biological basis for those problems. Teitelbaum’s assertion that hyperventilation is a natural outcome of increased brain lactate levels is doubly intriguing given the propensity some patients have for ‘shortened breath’ patterns.
The Natelson Connection
The Natelson tie-in is fascinating. One of the studies co-investigators is trying to do much the same thing, with different brain tests, inside the chronic fatigue syndrome community. Expanding on his earlier work Natelson is exploring whether ME/CFS patients with depression have less brain dysfunction than ME/CFS patients without depression.
Conclusions

The CFIDS Association, which provided funding for Shungu’s pilot studies, must be happy to see its pilot study program working out so well as well.







Great article . Love your writing style. Makes it easy to understand. Your new website is great. Hardly every go to PR these days. Good job.
Thanks for the boost Marci…I appreciate it 🙂
Cort, do you know when the award was announced? It would be instructive to know why the award happened-President Obama’s “committment” to CFS, or perhaps the large and loud voices heard at the FDA Ampligen meeting, or perhaps another reason. My point is that if we could find out the political reason as to why the award happened, we would know how to advocate for further awards more effectively.
This is an interesting point, I wonder this as well……
Way to go Cort. Great article and review of the literature as always.
Just wanted to note-THIS IS WHAT CHENEY HAS BEEN SAYING FOR THE LAST 2 YEARS. ANYONE
LISTENING???????
SAN DIEGO
They might listen if Cheney had a better track record, or even a decent track record, when it comes to patient improvement. Doctors who charge a fraction of what he does, even patients who can’t afford doctors and have been implementing Rich’s methylation protocol seem to be doing a lot better than typical Cheney patients.
Someone could also ask how did his grand, and costly (in more ways that one) stem cell therapy turn out?
Cheney gave his patients gluthathione 15 years ago & tried different types.
The NIH did a wonderful thing by giving two million dollars split between three medical research companies.
Now common sense should dictate not to start from the beginning, but incorporate the research done and the drug created by Hemispherex Ampligen. The darn thing works. Maybe not on everyone with ME/CFS but a good percentage of people. I am afraid this could become a political public relations battle and the losers are the patients. Let’s be honest here. Hemispherex and Dr. Carter and company do not have a pristine reputation. Rightly or wrongly they have been accused of many nefarious actions. But through it all they did produce a drug that is supported by a vast number of people. Will the FDA inconjunction with NIH postpone, in anyway, getting Ampligen approved? How will they justify giving two million dollars to research a disease and then come up with another drug. When one is already here? If one listened to and read about the Dec 20, 2012 FDA meeting there was a built in bias against the approval of Ampligen. Will this bias be pushed aside to be replaced by “let’s see what the other do now.” And how many years will that take to accomplish? The taxpayer’s money that was given away by the NIH should have a condition attached to this particular research. Use what has worked. Incorporate into your research that which has shown positive results. Get into a room and solve this TOGETHER AND FAST.
Robert, drug development and approvals by the FDA and research study grants by the NIH are two completely separate programs with very different purposes and goals. The study by Dr. Shingu and associates funded by this $2M NIH award is not about finding a drug to help cure or treat ME/CFS, it’s about finding or ruling out a particular biomarker that might help distinguish ME/CFS from other disorders.
The results of the team’s study might or might not be useful for Ampligen or for development of another treatment drug, or it may only point to further research needs; we won’t know until the study is completed. But its basic purpose has nothing to do with Ampligen or any other drug approval or development.
I wonder what appropriate treatments might look like if the Shungu study works out. I imagine it will have to do with raising glutathione levels and/or finding ways to enhance mitochondria production. Then again, finding low GSH doesn’t tell us why its there; Shungu thought the low GSH was the result of pro-inflammatory cytokine activity…possibly as a result of an infection…(as I remember :)) I guess it could ultimately swing around to a antiviral or immune modulator but that must be several studies in the future.
I sincerely hope they figure a way to move forward. I know of too many people who’ve done very well on the drug…(I also know of quite a few who didn’t – but what else would you expect in this vast disorder? (illness :))
I am verry optimistic about this study. This make sense. Many patiënts have EBV before starting with this desease. The liver is one of the organs producing glutathione.
Surprised you didn’t mention Komaroff’s study that showed abnormal EEG between depression and ME/CFS folks. Will this guy include such a test so we can get something validated? Komaroff said he was doing a follow up. What’s the status on that?
Right, that was a very encouraging study (and very hard to read :)). I didn’t realize that he checked for depression as well. Thanks for the reminder…I don’t know about Komaroff. I checked the NIH reporter; there was no mention of it there but that doesn’t mean its not happening.
Cort,
I wonder why you use the term “disorder” rather than “illness” ?
Always have been curious about that.
Thanks
Libby
I don’t know….I actually never really thought about it….hmmm – dis-order and illness; illness fits as well as disorder in my book…
Hi Cort,
Where did you learn that Shungu will be studying these subsets?
“…[W]e could end up with a range of subsets; ‘pure’ CFS patients who don’t have a hint of depression,, CFS patients with a bit of depression, MDD patients with some CFS characteristics ( post-exertional malaise?), and ‘pure’ MDD patients you’d never mistake for ME/CFS patients…”
Thanks!
Wonder if the studies will explain why my MRI scan says I have a white matter T2 hyperintense focus. It says it could be a microvascular focus point. Thanks for the write-up.
“Expanding on his earlier work Natelson is exploring whether ME/CFS patients with depression have less brain dysfunction than ME/CFS patients without depression.”
Interesting point.
Anecdotally, depression was one of my first symptoms (and I mean severe depression – not just a touch of the blues) which lasted for around ten years until all my other symptoms took a major downturn and new ones appeared including, for the first time, cognitive problems. I haven’t had to deal with depression since this downturn. Increased anxiety but not depression.
I wonder could duration of illness be another factor?
Hi Cort, I have been a reader & followed you for a long time now. I just have to say that this news/study excites me as I have been treated with glutathione for 13 years. The pain & malaise improved on the first day. I have tried to reduce it due to the cost , but the symptoms just come back. I was assessed for depression, & other than some sadness of not being able to do what I could do before being hit with this disease, I was not depressed. I wish I could be one of the people being studied.
Hey Cort, thanks for the great work you do for us. The problem I have with this study is that they are once again trying to tie this to a mental illness, if they don’t find any difinitive markers between ME/CFS patients, we could once again be thrown into the “Depressed” group. I know any research should be good research, but I have lost faith that they (FDA) are truly trying to find a answer. I also question these “Studies”, that will be, and has been done. They all seem to crunch the numbers to there favor some how, especially, the ones that are recieving these grants. The NIH clearly has favored this study, so they want it to work.
Anybody with a chronic illness suffer from some degree of depression.
I am very optimistic about this study and believe (praying) this study will eventually lead to discovery in new treatment to raise Glutathione (GSH) levels. I agree with Cort’s comment and believe this study will lead to further understanding and research in discovering ways to enhance mitochondria production. I also believe this study will arm the scientific community with better understanding that can lead to linking the cause of low ATP and NKC levels in ME/CFS patients.
This NIH grant study will also help lead to better understanding with many other health conditions. I am a firm believer that the Glutathione infusions I been taking are preventing me from further cognitive decline. MS patients have been taking Glutathione infusions up to three times a week for years to help from further brain related deterioration.
I look forward to the day there is complete understanding that depression is a symptom from having ME/CFS. The best of the best, smartest of smart, toughest of the tough would get depressed if their life were put on hold from ME/CFS.
Cort, thank you so very much for keeping us informed.
Cort and others,
An “illness” can be psychological or a matter of perception.
Whereas a “disease” has a biomedical causation
Meanwhile “sickness behavior” is not behavior at all in the sense normally associated with unruly four year olds. Rather, it is the set of symptoms resulting from the body fighting back at an invader; i.e. flu-like symptoms.
As to a “disorder”, my faithful giant Webster and Oxford abridged are 10 yards away and I’m not moving. But my bet is that it is anything contrary to the normal functioning of a being, such as Tourette’s, auto-immune diseases,clinical depression.I’m inclined to think that both illness and disease are disorders.
the confusing part is that many of us who try to supplement with glutathione get much sicker very fast. i have done oral and injectable and both made me much worse ME/CFS-wise.
Cort, thanks! But what’s up with stealing Susannah Cahalan’s title?? Her book Brain on Fire documents what appeared to be her own experience going stark raving mad, which was in fact a treatable autoimmune disorder. (The problem was with the NMDA receptors in her brain, which caused the inflammation and suggested the title. The brain has other receptors… maybe one of them has something to do with us?)
Interesting….I think I heard about her book but didn’t think about it….I always think of oxidative stress as a fire in the body since it can feed on itself. I’m going to check out that book 🙂
A fire is a fire. In the meantime, I think mitochondria fail to produce energy, signals are then messed up ie systems start to behave erratically, and wrong cues received all over the body with crazy-wild repercussions that mystify linear thought /scientific method…a storm being expressed uniquely in each person, or in one person, at different points in time. Synergistic, holistic model needed.
Interesting that they are doing this however, it seems they aren’t serious about it. For example, the NIH just recieved $1.5 million to study lesbian obesity as well.
Seems to me that CFS isn’t really being taken seriously.
Yes, I find it IS difficult to maintain any kind of objectivity, to be upbeat let alone excited about anyyy research these days. And while I remind myself that being skeptical is healthy, useful …and very normal… given our collective ongoing suffering… my new bent of cynical is, I fear, less useful, even damaging to myself and others. Still it is great to have a place like this to express my cynicism. Some days are worse than others. Whole lot of them recently.
Sorry to hear that Deborah. I would love to find a way to get rid of the negative stuff (the hopelessness and anger and frustration) and just be able to deal with the illness as it is…I think we all get bitter and angry at times….
did anyone else see this article which seems very relevant to CFS:
http://www.prohealth.com/library/showarticle.cfm?libid=17923
I’ve said before that I think the problem isn’t an ongoing virus (although a virus might kick things off), but this sounded quite a credible theory to me, and might explain why rituximab works.
Rituximab may “work” in some patients, but so far, only in a small subset…and at what cost?
In addition to CFS, patients with fibromyalgia, lupus, RA, and MS all have extremely low levels of glutathione. I believe this is due to the lack of the essential amino acid methionine. Here is an excerpt from my book on this.
“The lack of methionine would lead to a deficiency in glutathione. In the
study entitled “Serum antioxidants and nitric oxide levels in fibromyalgia: a
controlled study” glutathione levels were found to be “significantly lower in
fibromyalgia patients than in controls” (Sendur, 2009).
Low glutathione levels are a common finding in people suffering from
autoimmune diseases. In the study “Correlation of lipid peroxidation and
glutathione levels with severity of systemic lupus erythematosus: a pilot
study from single center” the researchers concluded that, “A significant
correlation between plasma GSH (glutathione) and SLE severity exists that
may aid evaluation of the disease severity and usefulness of the treatment of
SLE” (Tewthanom, 2008).
Patients with rheumatoid arthritis are lacking glutathione as well. In
the study entitled “The glutathione defense system in the pathogenesis of
rheumatoid arthritis” the researchers concluded that, “Rheumatoid arthritis
was associated with significant depletion in glutathione levels compared
with normal control subjects” (Hassan, 2001).
Multiple sclerosis (MS) patients are also lacking glutathione. In the
abstract entitled “Adenylate kinase activity and glutathione concentration
of cerebrospinal fluid in different neurological disorders” researchers found
that patients with MS had literally no glutathione in their cerebrospinal
fluid (Ronquist, 1979).”
Here is a study that shows fibromyalgia patients lack the essential amino acid methionine.
Altered Amino Acid Homeostasis in
Subjects Affected by Fibromyalgia
Bazzichi, L., L. Palego, G. Giannaccini, A. Rossi, F. De Feo,
C. Giacomelli, L. Betti, L. Giusti, G. Mascia, S. Bombardieri, A. Lucacchini. 2009.
Clin Bioche, 42(10-11):1064-70.
Significant lower plasma taurine, alanine, tyrosine (Tyr), valine, methionine,
phenylalanine and threonine concentrations, and the sum of essential AAs were
observed in FM patients vs. healthy controls (P<0.05). Tyr CAA’ ratio and the sum
of AAs competing with tryptophan for brain uptake were significantly reduced in
FM (P<0.05). A significant correlation was found between FM clinical parameters
and certain AAs.
The following study shows that the lack of methionine will depress brain glutathione.
Nutr Neurosci. 2001;4(3):213-22.
Sulfur amino acid deficiency depresses brain glutathione concentration.
Paterson PG, Lyon AW, Kamencic H, Andersen LB, Juurlink BH.
SourceCollege of Pharmacy and Nutrition, Cameco Multiple Sclerosis and Neuroscience Research Center, University of Saskatchewan, Saskatoon, Canada. phyllis.paterson@usask.ca
Abstract
Dietary sulfur amino acid content is a major determinant of glutathione concentration in some tissues. We examined whether brain glutathione (GSH), a key component of antioxidant defense important for minimizing ischemic injury, was also responsive to short-term sulfur amino acid deficiency. Female Long-Evans adult rats were fed a sulfur-deficient L-amino acid defined diet for five days; the control diet was supplemented with L-cystine and L-methionine (n = 6). Sulfur amino acid deficiency was confirmed by a reduction in liver cysteine and GSH concentrations, marked decreases in food intake, and weight loss. GSH concentration analyzed by reverse-phase high performance liquid chromatography was significantly depressed in the neocortex and thalamus of deficient rats. Brain cysteine was not decreased in a parallel manner. Classical glutathione peroxidase activity was increased in the liver and brain of sulfur amino acid deficient rats. This suggests an upregulation of antioxidant defense but these findings may be complicated by alterations in tissue composition. The depletion of brain GSH by a reduced supply of dietary precursors may be important during brain ischemia when the rate of GSH utilization and the need for synthesis are increased.
$1.5 MILLION TO STUDY LESBIAN OBESITY????????????
We’re living in hel- with illnesses thats NOT our fault and their studying something thats not even NORMAL and obesity can be PREVENTED!!!???
please tell me this is only a nightmare!!!