Multiple sclerosis (MS) is not ME/CFS and ME/CFS is not MS – so why a blog on a promising drug for multiple sclerosis? Because even if the disorders are different – even dramatically different in some ways – enough similarities exist to suggest that common pathways could be operating in both.
In multiple sclerosis, a breakdown of the myelin sheaths covering the axons of the nerves in the brain and spinal cord causes impaired nerve signal transmission, difficulty with movement, strange sensations, and even paralysis. Fatigue, interestingly, is often the first symptom seen in the disorder.
Some evidence of impaired nervous system transmission is present in ME/CFS, but it’s not clear what is causing it, and it’s not on the scale seen in MS. The distinctive lesions that signal the loss of the myelin covers in the brains of MS patients are not present in ME/CFS patients. Key aspects of the fatigue found in each disorder may be different as well.
More women than men get both disorders, however, and infectious agents have been implicated in both. Infectious mononucleosis causes a 2-3 fold increased risk of getting multiple sclerosis and is a common trigger for chronic fatigue syndrome. The ME/CFS BDNF study was a very small one, but it did suggest that, just as in multiple sclerosis, neuron repair mechanisms were not up to snuff. Some studies suggest that neuroinflammation – in lower amounts than MS – may be present in ME/CFS. Similar age-related patterns of incidence suggest that hormones, autoimmune processes, and/or viruses may play a role in both.

People in both disorders often experience relief during pregnancy. Could an estrogen affecting drug benefit ME/CFS patients as well?
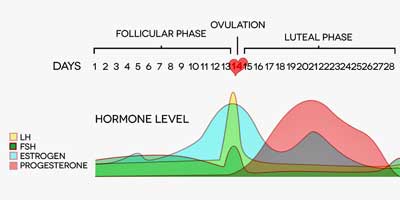
For the purposes of the compound this blog is about, though, the key factor is the often dramatic reduction in MS symptoms seen during the third trimester of pregnancy – when estrogen levels peak. Dramatic reductions in ME/CFS symptoms occur in some patients during pregnancy as well. (Women with fibromyalgia appear to get worse, not better during pregnancy.) That suggests female hormones play a role in both disorders.
Hormones
That wouldn’t surprise Gordon Broderick of Dr. Klimas’s Institute for Neuroimmune Studies of Nova Southeastern University. Broderick’s models suggest that estrogen in combination with HPA axis dysregulation may set the stage for ME/CFS in women. The female hormone connection probably wouldn’t surprise Dr. Unger at the CDC either. A CDC study found high rates of gynecological disorders in the CDC’s CFS patients.
The neurons in MS appear to be damaged by inflammatory processes produced by activated T-cells, macrophages, and glial cells. Estrogen has long been known to have neuroprotective effects, and low estrogen levels or signaling could play a role. The fact that ME/CFS incidence spikes in women in adolescence and middle age also suggests hormones do play a role.
Estrogen may be implicated in ME/CFS and MS, but estrogen treatment is not suggested in either disease and has been linked to an increased incidence of breast and uterine cancers.
Estrogen-Affecting Drug Created
In hopes of creating remissions similar to those seen during pregnancy, researchers from UC Riverside and the Rockefeller University in New York began experimenting with a compound called indazole chloride which can stimulate specific estrogen receptors (ER β ) without estrogen being present. This receptor is present in cells found in the peripheral immune system and the central nervous system (neurons, astrocytes, microglia), and the compound is believed to affect immune functioning in both.
The researchers focused on mouse models of experimental autoimmune encephalomyelitis (EAE) that have been developed to help understand brain inflammation and demyelination.
The results, thus far, have been fantastic. The drug has been able not just to reverse symptoms but to actually help rebuild the myelin sheaths surrounding the nerves, and it’s been able to do this long after the damage first occurred. It also increased levels of the nerve repair agent BDNF. It apparently can do all this, not just in mouse models of MS, but in traumatic brain and spinal cord injury. As good as the drug has been thus far, researchers believe they can tweak it to make it more effective.
Perhaps more interestingly with respect to potential neuroinflammatory disorders such as chronic fatigue syndrome, it also inhibits inflammation (microglia activation).
Thus far the drug has only been tested in laboratory animals, but clinical trials are reportedly starting in humans soon. Other very promising drugs have failed to make the leap from animal models to humans, but this drug, which appears to able to both decrease inflammation and effect neuron repair, has a lot going for it.
Conclusion
This potential blockbuster drug hasn’t been tested in humans in multiple sclerosis yet, let alone chronic fatigue syndrome, but its ability to repair damaged nerves and reduce inflammation is more than intriguing.
We should keep in mind that, given the slow, slow, slow pace of drug development in ME/CFS, it’s likely (unless Ampligen is approved) that people with ME/CFS will find help from drugs developed for other disorders long before they will get help from drugs developed specifically for ME/CFS. It’s good to keep an eye out for such possibilities.
Thanks to Carol for passing this study on.









Yet another reason why we need activist Doctors like back in the days of AIDS that forced the NIH to speed up the process of allowing patients to be placed in trials of drugs. There’s already a history that can be followed!
Last year 2013, the President of the U.S. sent an order to elevate research for ME/CFS and it should have also included FM. They do overlap and in some cases Drs. put both under the same umbrella of illness (meaning if you have FM you have ME/CFS as well and it is a matter of how fast FM gets worse which leads to further complications of having ME/CFS also). What ever happened to that? This seems like a good time to have a committee (more powerful I think) to send him a reminder about this. He is now looking at all of his promises and trying to fulfill them. Cort you may remember the name of the man who was working on this and was a part of maybe the FDC but a special division that works with the President? I can see his picture of his face in my mind right now. Can you find out anything or do you have any info in your archives on this?
My Heart breaks, reading this comment more than 10 yrs later
There is most likely an herb that stimulates the estrogen receptors and is also very safe and doesn’t require a prescription!
There is a problem with that though, not much money to be made on herbs! Damn. RP
Any herb that stimulates estrogen receptors has risks and side effects. The idea that a plant somehow can have a physiologic effect but no adverse reactions is just magical thinking.
I don’t know about estrogen receptors Cort and I wouldn’t mess with estrogen without having a good medical Dr. but I do agree 100% that herbs, oils, vitamins, supplements and diet play a huge role in helping us with FM and ME/CFS. I personally have a regimen that includes an array of vitamins and supplements and oils that I use for various reasons with very positive effects. I think it is important to mention this because I researched so many hundreds of hours before deciding on a regimen that I have shared on my support group and openly on an event page on FB as well. As I share the information I always advise everyone to talk to their Drs. and get the ok before starting any form of supplementation. Some of it is obvious when our Drs. do blood work and we are all low on D thus need to take the form D3 for our health. I finally got mine up to 60 and want it at 90-100 for optimal health. D3 also plays a significant role in MS. Potassium and Magnesium should also be checked by our Drs. and I ended up with a magnesium health problem that has a name hypomagnesemia. Never had issues before with that but since FM and ME/CFS hit me I do. Potassium as well can change in my body very quickly from normal to low. So these are all supplements that have made a difference for me and came from Drs. checking my blood properly. The only one I had to push for was the vit D level which is not a normal part of blood work ordered. Then from there some other supplements work well like fish oils and more and other oils to relax us or help with our skin that many suffer problems with from both FM and ME/CFS and with epsom salt baths for pain. There is a place for all this and much is natural and to be used with Dr. permission but if anything messes with our hormone levels a qualified Dr. need to be involved in that decision. I do take estrogen because I have to. Not sure how much longer my Drs. will allow it but I need it for now. Can’t wait to see the results of these tests when they complete them.
I would be concerned about long term effects, especially cancer, given that one is messing with estrogen receptors. Also powerful drugs can have powerful side effects. That said many with ME/CFS need relief NOW and hopefully this will help them. It will be interesting to see what comes of the ME/CFS studies of men to see how hormones are affecting them. I know at least a few men who are very ill with ME/CFS and I have to wonder if all the men who are ill are really being counted.
I imagine that everyone is being under counted and men are being under counted more than women. That said the ratio’s generally presented still suggest that 25-30% of people with ME/CFS are men – no small amount.
I wish more men would go to the Dr. when they don’t feel right because I think the #’s are at least at the level your stating Cort if not higher. Please men get checked for sudden pains, depression from no where and exhaustion. It will help everyone when the CDC and others start to see this is not a women’s illness. It will hopefully escalate the need for putting our illness in the category of those who had Aids and they got a cure rather fast. They spoke out and demanded help and we speak out but we need our men that are suffering to come forward also. Please take this from the thought that men are not as quick to go to the Dr. as woman are. I mean this with all the respect in my heart to men suffering and suffering alone because they don’t know what is going on and many have families to support and push through. Please get checked if you are suddenly not yourself as you once were.
Cort, here’s a link to a study finding that the estrogen Estriol is being used to help with this. There are 3 kinds of estrogen and this is the estrogen that is most prevalent during pregnancy. (It is also known to decrease breast cancer.) After my hysterectomy – it was the only hormone my body would tolerate as a replacement. I use it in a compounded transdermal form.
http://newsroom.ucla.edu/releases/preliminary-clinical-trial-shows-great-promise-for-new-multiple-sclerosis-treatment
I have been saying for years that we need to have the ratios of our hormones checked. It may be that the balance is off between them. This would also apply to guys as well.
There is an herb that I’m researching that I think may be helpful that works on the immune system, NK cells and T cells ——cistanche. Looks intriguing.
Issie
Interesting – thanks Issie
Issie thank you so much for sharing that link. I have been on HRT for over 20 years. I was very young when I had to have a hysterectomy. I was once taken off of HRT for 2 months and went back to the Dr. and said I have to get back on HRT right NOW or I am jumping off the bridge. I had to make it very clear that I simply couldn’t handle the effects without the HRT. I truly fell apart. I am always in the back of my mind wondering and worrying but can’t be without it either.
So I further checked this out and found this awesome link that is also extremely informative. Thanks again.
http://www.lef.org/Magazine/2008/8/Estriol-Its-Weakness-is-its-Strength/Page-01
Eva, I hear you about the going crazy part with no hormones. I was very young too and wasn’t allowed to go on replacement right away. With endometriosis if one spot is missed it is thought that estrogen will make it grow and spread. I was trying to stay off anything for 6 months to make sure everything was either burned off or died off. I only made it 5 months. My first hot flash was the day after surgery. But my hysterectomy was my 8th abdominal surgery and I didn’t want to have any more. I was determined to stay off the hormones. Then came the trial and error. Mostly error of hormone replacement. I was very glad to find the transdermal estriol and red clover – finally it worked and I settled out. Not all compounding pharmacies are the same. It took three different ones before I found the right fit for me.
I love Life Extension. That is a great article!
Issie
Love your work, that gives us hope. I had a brain scan 2 yrs ago, and my CF/ FM was gone for a week. But my Neuro Dr. wont coment on it!
This drug, and their concept of possible benefits?
No. No. No.
Terrible idea. Won’t work. Waste of time and money.
No.
Thanks for that link to the study on gynecological disorders in CFS.
I always wondered about that, I had heavy painful periods, always suspected there was a connection to CFS but had no way of knowing till I read that study.
I am surprised the MS patients and MS society aren’t pushing the drug more.
A little google research suggests that adding more soy to the diet may have similar effects.
In 5 or 10 years time this research may be exciting, for now it’s interesting (and deserves funding) but people are not mice and it’s a long route from promising research to worthwhile treatment
Unless the soy is fermented, this would not be a good alternative.
Unfermented soy interferes with the uptake of minerals like calcium, magnesium, iodine; it also interferes with the thyroid and the natural menstrual cycle of women.
Eating unfermented soy or drinking soy milk is one of the most unhealthy things one can do (not to mention the genetically modified nature of most soy).
I just have to say that soy is one of the few substances that can be really depleting for me. Soy milk, for instance, is huge energy sapper for me…I would like to know why!
That could be due to the phytic acid that is present in unfermented soy. It inhibits the digestive enzymes, making unfermented soy highly indigestible. Anything that is hard on the gut is likely to sap people who are not well of their energy.
Cort, here is something to keep an eye on in this vein of thought; birth order. Not gobs of science here, but what I’m aware of seems to indicate some interplay in birth order, mothers’ immune systems and suppression of foreign body recognition especially when applied to multiple male births, and possible outcomes for mother, and offspring including vital human elements such as hormones and/or sexual preferences, amongst other stuff I can’t remember right now.
I can’t agree w/ yearsofmisery. Speculation is a tough place to be in science, yet informed postulation is it’s cornerstone. W/o asking ‘is this true?’, or ‘what if?’ in defiance of dictates and preconception, humanity and its science would still be subject to the politics of the dark ages. I just wish my doctors would get on board.
This sounds terrific! What is exciting about this research is that nerves show repair and lessened inflammation.
I had very low estrogen from the lead up to ME/CFS and on through it for 20 years now. Low, low estrogen. So I opted for some estrogen replacement, enough to keep me just clear of hot flashes, which had added to my insomnia miseries. Even on that low estrogen addition, I was able to go through menopause. I also have low body fat, which I have read generates extra estrogen in many women. Since about one third of women are now considered obese and more just “overweight”, this probably adds to their cancer risk. I have never been worried about cancer risk because still my estrogen level is low compared to other women, both pre and post menopause, and on or off HRT.
I wonder if the slightly increased risk for cancer which women have on HRT happens in women whose overall level of estrogen is higher than typical levels in ME/CFS? I doubt if that research project tested their actual levels. It probably just generalized over an average group. I would like to see a research study which showed what blood level of estrogen is associated with risk or no added risk.
“More women than men get both disorders,…”
I do not beg, ever. Now that is a lie, because beg is exactly what I am about to do.
I am begging you Cort, to stop using the above line/wording when discussing these disorders.
It is true that more women are DIAGNOSED with MS, ME/CFS, FM, and a host of other issues.
However, there are no truly quantifiable statistics to conclusively say that men are not equally affected. Times and medicine are changing, but it still quite often takes years to conclude that these disorders are what is affecting a woman, and a man has almost no hope of ever being properly diagnosed. My own case took over 30 years to “solve” for FM, when the plain truth was right in front of anyone actually looking. They weren’t, and it truly was that simple. Men are treated as less vulnerable or likely to have general malaise, etc. And, we are told to, and do, “suck it up” to a greater extent. We are our own worst enemy in this fight, and our champions (such as you) have got to stop perpetuating these myths, and I do believe them to be myths.
You would be doing a great service, and no disservice, by simply changing these statements to “More women than men are diagnosed [with (insert disorder of the day)]…”
I agree that it would be good to change the wording to indicate more women “are diagnosed with” this disorder or that because this is a correct statement about the facts.
@Blaine Bravo! Well put!
I fully agree with Blaine, Cort. I feel a huge sense of wrong being perpetuated upon men whenever I read those words “More Women than men get ME/CFS”. It is a cruel dismissal of all the extremely ill men in the CFS community, and I’d like to never, EVER read those words again. Perhaps, due to gender bias (like how men appear to be treated agressively for preventative heart health than women are. Perhaps because women are not seen as the breadwinners in American society,. Regardless, it’s a gender bias, and women die from heart disease at a higher rate than their male counterparts.).
Perhaps a more accurate statement would be “In Women diagnosed with ME/CFS studies find…” and “Women Receive Faster ME/CFS Diagnosis than Male ME/CFS Sufferers”. I just don’t believe more women than men have ME/CFS, only that they receive are more likely to receive a diagnosis of ME/CFS because of gender bias, or societal pressure on men to “take it like a man” and be “tough”, “suck it up”.
I hate the headlines declaring that we women are the ones suffering here. We need to be more careful of how we word things in this subject, as people new to ME/CFS will probably learn about it first from a website like this. It’s a big responsibility, but that’s what you take on when you chose to represent the voice of an entire community. Thanks for all your work. But let’s be fair to all patients.
Just one opinion from one female ME/CFS and FMS sufferer.
I understand that saying that more women than men puts the focus on women and I think we should and I should impart more that men, are often effected by ME/CFS but I don’t saying that saying “more women than men” get ME/CFS or FM or IBS or whatever is necessarily dismissive of men with ME/CFS. It’s not saying men don’t ME/CFS – it’s simply stating, based on study results – more women than men get it. I do agree that ME/CFS is probably more undiagnosed in men than women, however. We don’t have any stats to indicate that women are diagnosed more quickly, however, so I couldn’t say that.
If women are more likely to come down with ME/CFS than men it does point to potential autoimmune processes being present. That’s a valuable finding if its true.
On the other hand we certainly don’t want males with possible ME/CFS or FM to think only women get this disorder and I will think about how to better present the fact that many males – studies suggest 25-30% of ME/CFS sufferers I believe – have ME/CFS as well.
Blaine: If ME/CFS is an autoimmune illness, like many people are beginning to believe it is. There may be hormonal reasons why women are diagnosed more often with the condition, than men. It has been known for decades that females are more susceptible to inflammatory autoimmune diseases including multiple sclerosis (MS), rheumatoid arthritis and lupus (SLE). One of the reasons may be a high ratio of estrogen, to that of progesterone in women. Progesterone is protective in women from excessive inflammatory responses, as is testosterone in men. Men who develop autoimmune illnesses, may do so after a decline in bioavailabe testosterone.
I’m not surprised that pregnant women with ME/CFS experience some relief, since the immune system eases up in order not to reject the fetus by considering it a foreign invader. This is consistent with the Rituximab findings, since its killing of B cells reduces immune system activity, providing temporary relief. ME/CFS is probably a CNS virus which creates inflammation by immune activating the microglia in the brain in fighting it. It has yet to be identified and targeted, however. I was hoping Michael Van Elzakker and Anthony Komaroff would tackle that at some point.
Hormones, particularly sex hormones, have always been a major player in my ME/CFS. I never knew whether progesterone or estrogen was the primary culprit. I now think estrogen dips that come with the cycle, was affecting my immune system. There is a connection or reaction of immune system to estrogen levels. Starting last year, I started continuous birth control pills, no week off, to stop the peaks and valleys. Has removed one cause of major crashes. Now, if we could remove the others.
“The strong association of MS with estrogen has led to an illogical, but popular and well-publicized medical conclusion that estrogen is protective against MS, and some have claimed that estrogen has beneficial therapeutic effects. This strange way of thinking has its equivalent in the idea that, since women are much more likely than men to develop Alzheimer’s disease, estrogen is protective against it; or that, since women have more fragile bones than men do, and their progressive bone loss occurs during the times of their greatest exposure to estrogen, estrogen prevents osteoporosis.”
“In this medical environment, close associations between estrogen and degenerative diseases are acknowledged, but they are given a meaning contrary to common sense by saying that the association occurs because there isn’t enough estrogen. The stove burns you because it isn’t hot enough.”
“Multiple sclerosis (MS), like other autoimmune diseases, affects women more often than men (about 2 to 1), has its onset during the reproductive years (especially after the age of 30, when estrogen is very high), is often exacerbated premenstrually, and is sometimes ALLEVIATED by PREGNANCY (Drew and Chavez, 2000), when PROGESTERONE is very high. Women with a high ratio of estrogen to progesterone have been found to have the most active brain lesions (Bansil, et al., 1999). Most of the mediators of inflammation that are involved in MS–mast cells, nitric oxide (NO), serotonin, prolactin, lipid peroxidation, free fatty acids, prostaglandins and isoprostanes, and the various cytokines (IL, TNF)–are closely associated with estrogen’s actions, and in animals, autoimmune diseases can be brought on by treatment with estrogen (Ahmed and Talal).”
A different perspective regarding estrogen’s neuroprotective effects from Dr Ray Peat PhD Biology
Read more: http://raypeat.com/articles/articles/ms.shtml
Thanks. One thing I learned about estrogen is how complex it is. There are no simple answers here and no one is suggesting that women with ME/CFS or MS take estrogen.
Also with MS, researchers believe that an infectious agen underlies MS…in particular EBV (EBAN1) and HERVs – MSRV, HERV-K (HML-2), HERV-K, HERV-Fc1.
Typical ME/CFS patients have shown across the board high IGA titers above the normal high range in Herpeviruses- HVS-1, EBV HHV-4 , parvovirus B19, HHV-5 CMV, HHV-6 Varicella Zoster, and coxsackie viruses than the regular population. Furthermore, activation of ME/CFS usually follows a period of post viral infection such as Mononucleosis.
The good news is that clinical trials in each category – Inspire for EBV and Chargot for HERVs – are being conducted in the UK to verify if patients with MS would benefit with antiviral and antiretroviral (HAART) medication.
http://multiple-sclerosis-research.blogspot.com/2014/12/canada-why-i-think-ms-is-caused-by-virus.html
HERVs in Multiple Sclerosis Charcot Project:
http://multiple-sclerosis-research.blogspot.com/p/charcot-project.html
http://forums.phoenixrising.me/index.php?threads/hiv-and-ms-could-a-link-lead-to-new-ms-treatment.32143/#post-495327
Inspire Trial:
http://multiple-sclerosis-research.blogspot.co.uk/2014/07/charcot-project-inspire-trial-fully.html?m=1
http://multiple-sclerosis-research.blogspot.com/2014/02/guest-post-professor-michael-pender.html
http://forums.phoenixrising.me/index.php?threads/frankenstein-mice-provides-insights-into-the-immunology-of-ebv-infection.32768/#post-505944
Why this is important to ME/CFSers? The the primary fact that if these trials are successful, their research (Charcot/Inspire Trial) could impact and give credence to current and previous ME/CFS research. It would also open the door to antiviral and art treatment protocols for ME/CFSers if similar research is validated in ME/CFS patients
Very interesting! I feel so much worse during my period(when I have a week off of birth control pills). It makes sense that the drop in hormone levels could greatly effect our health.
Wanted to point out that pregnancy also increases circulating blood volume. Perhaps that has an impact on why many women get symptom relief at that time. It may not be estrogen. Or it may be some combination of the two.
Something that needs to be looked at.
Interesting – that could be a major factor…
drlewisclarke
Estriol increases NO and vasodilates. Helps with endothelial function. Can improve blood flow. May help prevent breast cancer, osteoporosis and cognitive decline. Lots of other possibilities too.
Issie
drlewisclarke.com
Here’s that link again. Maybe it will work this time.
This is mostly a statement of my body’s reaction to progesterone and the more commonly given estrogen estridol. I had an estrogen dominant state before my hysterectomy. Having endometriosis and knowing estrogen dominance as one of the causes – the thought is to balance the dominence with progesterone. What I found with progesterone was it made all my symptoms worse. My hot flashes were unbearable and it caused a lot of anxiety. I was using compounded transdermal and also tried compounded troches. I also have EDS (Ehlers Danlos). There is some talk in that community that progesterone will make you more stretchy and cause this to be worse. I wasn’t able to find any studies to support this though. After my complete hysterectomy you’d think my body would want a replacement of all the estrogens and progesterone. But it didn’t tolerate them. The only sucessfull one for me was estriol. I also found benefit from Red Clover.
Issie
Just a side note. Endometriosis has been thought to be connected to MCAS (mast cell activation syndrome).
Issie
I have to disagree with low estrogen being an issue. When I was first diagnosed I had the estrogen level of a girl in puberty. I was put on Progesterone to try to get a balance. That was when I was feeling at my worst.
After reading that pregnant women feel less pain due to estrogen have you not done a study to see how many of the women that felt less pain was having a boy maybe its as the mother creates testosterone to determine a boy so as there body is awash with testosterone there pain is less and its not so much the estrogen ?