“If we are successful, we may not only produce an objective test for fibromyalgia and chronic fatigue syndrome, but we may also be able to develop more effective treatments for those disorders.” – Dr. Jarred Younger
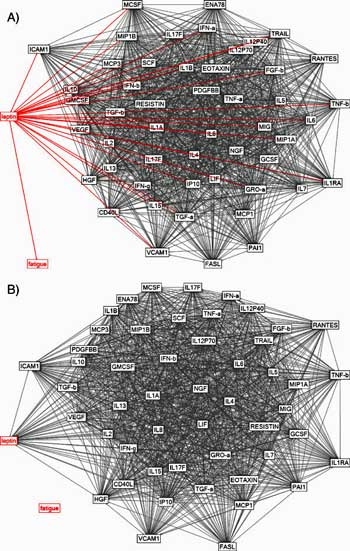
Jarred Younger dazzled attendees with his leptin and chronic fatigue syndrome presentation at the Stanford Symposium earlier this year. His pilot study that measured dozens of inflammatory markers every day in chronic fatigue syndrome patients produced one of the most visually startling graphics I’ve ever seen – an “all roads lead to Rome” type of plot that showed an array of immune factors converging on one… the putative trigger of them all: leptin. (See the video here.)
Younger has shown he’s willing to go “outside the box” of traditional treatments like pharmaceutical drugs to find relief for people with chronic pain and fatigue. His embrace of low dose naltrexone (LDN), for instance, has added a valuable treatment for many people with fibromyalgia and chronic fatigue syndrome.
Younger recently moved from Stanford University to the University of Alabama at Birmingham to open a Neuroinflammation, Pain and Fatigue Lab. Health Rising took the occasion of this move to review a recent video interview he did with Deborah Waroff of ME/CFS Alert, and then to ask him some questions.
Getting Started
When Deborah Waroff of the ME/CFS Alert asked Younger how he got started in ME/CFS research, Younger noted that while pain was the most “salient” part of fibromyalgia many patients told him that fatigue was the most debilitating part of their illness. [This fits with a recent ME/CFS and FM survey on Health Rising suggesting that post-exertional malaise was the biggest problem for ME/CFS and FM patients.] Even if the FM patients got rid of or reduced their pain, Younger said, it was the fatigue that was keeping them from going back to their jobs. That was what got him interested in the kind of pathological fatigue found in chronic fatigue syndrome.
Now, he’s traded the intellectual ferment (and pricey) environs of Silicon Valley to direct the new Neuroinflammation, Pain and Fatigue Lab at the University of Alabama at Birmingham. A step down, you might think? Maybe not. The University of Alabama at Birmingham contains a significant medical research center that enjoys robust NIH funding.
Deborah Waroff Talks to Jarred Younger
Younger said the decision to leave one of the top centers of medical research in the country – Stanford – was not an easy one, but he believes he’ll be able to move faster at UAB. Plus, it’s a return home in a way. Younger grew up in a small, small town (population 200) just a couple of hundred miles across the border in Tennessee.
Younger earned a Ph.D in Experimental Psychophysiology at the University of Tennessee, completed postdoctoral work at the University of Arizona in pain psychology, and then a post-doc in pain neuroscience at Stanford where he became an assistant professor. Now he’s back in the south.
“I now have more space, more people, and more resources – so now I can answer more questions.” – Younger
UAB made a good move. They snatched up a younger researcher focused on a medical topic that’s on the rise. They apparently lured Younger with the substantial commitment they’re making to this field. Younger brought two researchers, Joanne Lin, Ph.D., a pharmacist and neuroimaging specialist, and Luke Parkitny, Ph.D., a researcher in chronic pain immunology, as well as a lab manager from Stanford with him. Younger stated he will have access to the same leading-edge instruments he had as Director of the Adult and Pediatric Pain lab at Stanford.
“Prof. Younger’s goal is to end the chronic pain and fatigue that is caused by inflammation in the brain.” – Univ. of Alabama at Birmingham
Getting academic research centers dedicated to ME/CFS and FM at prominent universities is high on anyone’s list of “things needed to move these fields forward”, and now we have another one. Younger’s Neuroinflammation, Pain and Fatigue Lab is one of the few university labs dedicated to chronic fatigue syndrome and fibromyalgia research.
With Dr. Klimas’s and now Younger’s moves, we may be seeing an intriguing pattern emerging: ME/CFS and FM researchers being lured from higher ranked to lower ranked medical schools (#2 to #43 in Younger’s case). It suggests some Universities may be trying to get the early lead in what they envision to be a growth area in medicine. UAB’s creation of the Neuroinflammation, Pain and Fatigue lab may get other University directors thinking.
How the lab got created is unclear, but Younger and ME/CFS and fibromyalgia have some friends in the Alabama legislature. In 2013, an Alabama politician with Lyme disease, State Senator Slade Blackwell, helped pass a Senate Joint Resolution urging the state to establish a clinical care and research center for what Blackwell called “neuroinflammatory disorders”. Give some credit to Tina Tidmore and the folks at Pandora for preparing the ground in Alabama for this to happen.
Younger’s bringing a considerable portfolio of research projects with him. Here are some examples:
- He’s examining the effectiveness of low dose naltrexone (LDN) in FM.
- He’s doing daily immune monitoring and assessing the efficacy of anti-inflammatories in Gulf War syndrome.
- He’s doing neuroimaging to understand the effects opioid use has on the brain.
- He’s finding new and more accurate ways to test for low levels of neuroinflammation in the brain.
To top it off, he just got a nice, big NIH grant to study chronic fatigue syndrome.
Big Chronic Fatigue Syndrome Study
In his last ten-person study, Younger was able to accurately predict whether an ME/CFS patient was having a low or high fatigue day about 80% of the time simply by looking at their inflammatory markers. This new ME/CFS study, which will go much further, is a whopper.
The projected five-year study will test no less than fifty-one inflammatory immune markers every day for 25 days in 70 ME/CFS patients, 20 healthy controls and, significantly, 20 fatigue controls (people with hypothyroidism). The AHRQ report took the research community to task for not including fatigued patients without ME/CFS in their diagnostic biomarker studies. This study includes those patients. Everyone will assess their levels of fatigue regularly on handheld computers.
Younger appears to be measuring just about every inflammatory marker he can find — this is exactly the kind of exhaustive study ME/CFS needs and so rarely gets. It should reveal much about the inflammatory component of ME/CFS and is big enough that Younger expects to be able to identify subsets. Younger will also attempt to “develop a temporal pathway between immune factors and fatigue that identifies early drivers of fatigue.” I asked him what he meant by a “temporal pathway”:
“The temporal pathway is really about determining what comes first. We may identify 20 or so inflammatory agents that are associated with fatigue. But we really want to know where we can intervene in order to stop the process. So, we need to know what leads to what. We want to get as early in the causal chain as possible so we can stop the problem at the source. So, determining the temporal pathway is really about figuring out where the problem begins.” – Younger
It could start with leptin. They say all roads lead to Rome, and Younger’s last ME/CFS study suggested that all roads did in fact lead to leptin. Leptin appeared to be the ball that got the immune cascade rolling in ME/CFS.
Younger believes leptin sensitization of the microglia in the brain could be creating a low-level state of neuroinflammation in ME/CFS and FM
In the video interview with Deborah Waroff, Younger said that leptin, which is produced by the fat tissues in the body, is able to cross over and sensitize the microglia in the brain. Activating the microglia is one thing, but sensitization is a more serious issue since it could cause the microglia to respond to factors they shouldn’t be responding to. That would set the stage for the kind of low-level neuroinflammation Younger believes is present in FM and ME/CFS and which Japanese researchers produced evidence of last year in chronic fatigue syndrome. Leptin was correlated with fatigue levels in people with chronic hepatitis C infections.
Ironically, the home-based things one can do to reduce leptin levels are particularly difficult for people with ME/CFS and FM. The enforced sedentariness of the disorders makes it easier to gain weight – which increases leptin. Exercise can help reduce leptin levels, but significant exercise is, of course, pretty much out of the picture. Stress eating – which has got to be more common in any chronic illness – increases leptin levels.
Banking for the Future
Younger will also bank his samples and make them available to other researchers. Several biobanks have emerged recently, but it’s not clear that any of them, except Klimas’s at Nova Southeastern University, will have what Younger will have – thousands of samples categorized by the degree of fatigue present when the samples were taken. That’s an enormously useful data point to have.
The big ME/CFS study started in March of 2014 and it’s slated to end in February of 2019. I asked him – is it really going to take five long years?
“I hope the project does not take 5 years. We will push to get it done as fast as possible. And once we have enough people (maybe around 30), we will run preliminary analyses and publish them. We don’t have to wait for the entire project to finish.” – Younger
Temperature Rising – Neuroinflammation, ME/CFS, and Fibromyalgia
Neuroinflammation is a hot topic in both ME/CFS and fibromyalgia. You can easily spot the inflammation in disorders like multiple sclerosis where the nerves are literally being destroyed, but finding the low levels of inflammation Younger believes are present in the brains of ME/CFS and FM patients has been difficult.
I asked him if any methods accurately assess microglial activation/neuroinflammation in the brain at this point.
“Regarding directly assessing microglia activation or neuroinflammation – no, there is nothing currently available that provides a direct measure. What we have now are proxy measurements, and the question is how closely they reflect true changes in microglia activation. The closest seems to be positron emission tomography (PET) assessment of the translocator protein (TSPO). It is a receptor that seems to be expressed more in activated microglia. So, if we inject someone with a ligand for that receptor that has a radio tag, we can measure the uptake of that chemical and get a rough idea of microglia activation. Unfortunately, the receptor is expressed on other cells, and there are individual differences in uptake. So, even the PET method is not perfect. Many people are working on this problem.” – Younger
Younger is trying to assess the amount of neuroinflammation present by taking the temperature of the brain. Just as when you’re sick the temperature of your body often goes up, Younger thinks low-level inflammation in the brain may be causing the temperature of ME/CFS and FM patients’ brains to increase slightly.
He’s using magnetic resonance spectroscopy (MRS) to determine the concentrations of certain chemicals in the brain and then creating temperature maps by running calculations on some of those chemicals. He’s working with several Institutes to produce point by point temperature maps of temperature in the brain. Ultimately these maps could produce a diagnostic biomarker for ME/CFS.
Low Dose Naltrexone… Plus
“All the research is about finding new treatments.” – Jarred Younger
A leader, make that the leader in the charge to validate the use of low dose naltrexone – a basically unmarketable substance (thus far) for drug companies – in fibromyalgia, Younger has shown that he’s willing to open up new ground in the treatment of the disease. LDN, when it works, appears to work on a variety of levels, reducing pain, improving energy, and reducing feelings of stress and negative moods.
One LDN study underway to determine if LDN reduces inflammatory markers in the blood may be able also to isolate which types of FM patients benefit from the drug. Younger also has several grant applications in the pipeline to run LDN studies in larger groups.
Younger is not stopping at LDN, though. In the Waroff interview, Younger stated, with some surprise in his voice, that his lab is finding that some Chinese botanicals, such as stinging nettle and curcumin, are very effective at suppressing the microglia as well. His lab is currently testing about 30 different substances. In the drug realm very low dose dextromethorphan (30 mg) and 3-hydroxymorphinan (not used in humans yet) are promising candidates.
Blogs on microglial inhibitors
- Microglial Inhibiting Drugs – Providing Hope for Fibromyalgia and Chronic Fatigue Syndrome (ME/CFS)?
- Marijuana, the Microglia and Reducing Nerve Pain
- Drug Repurposing I: Antibiotics to Reduce Microglial Activation in ME/CFS and Fibromyalgia?
- Palmitoylethanolamide (PEA) – A Medical Food for Fibromyalgia and (ME/CFS)?

Low levels of anti-inflammatory mediators during exercise could trigger the microglia to over-react – causing PEM
A recent fibromyalgia study found reductions in anti-inflammatory cytokines during and directly after exercise, but not in the days following the exercise – when the patients were still symptomatic. I asked him if a transient immune disruption like that might set the stage for microglial activation and post-exertional malaise in FM and ME/CFS. He thought yes.
“There are proteins designed to put the brakes on microglia activity. One that is mentioned frequently is fractalkine. Fractalkine allows neurons to exert control over microglia function, generally by maintaining microglia in their “resting” state. Anti-inflammatory cytokines such as IL-10 can control the expression of fractalkine from neurons.
“A hypothesized scenario would go like this: During exercise, there is a decrease of anti-inflammatory cytokines in patients, which in turn lowers fractalkine activity in the CNS, which results in microglia becoming sensitized and moving to an activated state. The microglia then remain in the activated state for a few days until fractalkine activity normalizes and the brakes are restored. There is no analysis examining that entire sequence of events, but the pieces are all established.”
A small study just recently found reduced fractalkine levels in people with fibromyalgia. A review of that study will be coming up in our next blog.
Conclusion
Younger’s and Dr. Klimas’s moves from higher to lower ranked academic centers willing to provide them with more resources may suggests some academic centers may be trying to get a head start in what they believe will be a growth field.
Younger’s commitment to leave no stone unturned in his search for effective treatments and his embrace of new technologies that merge sophisticated data mining techniques with personalized data gathering tools is probably emblematic of a younger generation of researchers. His ability to turn a small pilot study into a $2 million dollar or so NIH grant is very gratifying as well. It’s going to be interesting.
The most difficult task Younger may have going forward is finding ME/CFS or FM patients in a state with few (if any) ME/CFS or FM experts. If you have ME/CFS or fibromyalgia and live in or near Alabama and might be interested in participating in one of Younger’s studies, email the lab at youngerlab@uab.edu or call them at 205-975-5907. You can sign up for the lab’s newsletter here.
Younger Webinar
Jarred Younger will be featured in Pandora’s first webinar on January 23rd
- Research Report: Leptin in the Role of Neuroinflammation, Pain and Fatigue
- Jan 23, 2015 at 1:30 PM EST.
- Register at https://attendee.gotowebinar.
com/register/ 5883680598116491522 After registering, you will receive a confirmation email containing information about joining the webinar.











Thanks Cort for this very interesting post. I am constantly impressed with the information that comes and that you do it with me/cfs brain is astounding. Please know that your blog is often one of the few bright lights for me. You are valued.
Ditto!!
Thanks Margo and Laurie – very glad to hear it. It’s personally exciting for me to keep up with this stuff. I love to see that the field is progressing 🙂 🙂
Ditto, Cort! Thank you!!!
Thank you Cort for all that you do, and keeping us up to date with what is happening in the research of ME/CFS, & FM. This all gives us hope knowing that great work is being done towards a cure in the future.
Thanks Anita 🙂
Go Dr. Younger!
:)….I hope he gets all the resources he needs.
You give us great articles about these researchers independently discovering these horrible issues in our body..Each finding is disastrous in itself but if those researchers saw all the articles and all the other disastrous discoveries/research you’ve written about over the years, they would be floored..imagine combining all your past research articles into one..we would have a really nice book on cfs and the hell it really is..:)))
I think the nay-sayers would be floored….Thanks Mike!
Very thorough, as usual, Cort. Wish I understood all you and Younger were saying in all the medical talk. Although,community pride and news reports of the groundbreaking research done at UAB would make me think UAB is “lower ranking” is more perception than reality. But, Birmingham’s in the ME/CFS and fibromyalgia game now, as they already were in other diseases. And if our state history in sports is any indication, we should have some research touchdowns coming soon. Roll Tide or War Eagle! Or, Go Younger!
Hey, UAB is ranked in the top fifty medical schools in the country – iti’s no spring chicken – they are doing very well and he landed at a very good school…
Hey the SEC is way over-ranked but go Alabama at Birmingham 🙂
Had already heard of another Pioneer in Birmingham. Dr. McClain.
This may be a distance I can travel.
Great Work Cort. I am excited.Is he seeing patients or still in research?
Sorry TINA- OREGON DUCKS ARE GOING TO TAKE IT ALL!!!!!!
🙂
I think Younger is all about research. I don’t know if he has an MD – I think but am not sure that he is a PHD.
Go Ducks! (Go Pac-12!)
SEC doesn’t have a team in the championship – ha!
Younger’s description of exercise response is very accurate. I find that vigorous exercise relieves my symptoms in the short run, but I cannot do too many days in a row, so I take every fourth day off. On my day off I generally have a splitting headache, but the next day I feel better upon starting the cycle again. It’s a tricky balancing act, but despite the penalty incurred, I need the short term relief to cope.
I am interested that you say “vigorous”.
While I was declining with undiagnosed fibro, I was a highly-active twice-daily training semi-athlete.
Not knowing what was happening to me, and the discovery of the importance of pacing being 20 years in the future for me, I spiraled downwards into worse and worse pain. I believe this was harmful.
Since then I have suffered constantly from “post exertion malaise”, which is the perfect term that has popped up recently. I feel pretty good while exercising, I am driven and competitive, and I always overdid it and caused myself worsened pain and hence increased inaction for some days. And my condition has declined and my weight ballooned. And I can relate to the headachy, stagnated feeling on all the days of enforced inactivity.
It is only in the last few months that the discovery of correct pacing has reversed this destructive trend for me. Now, I am more like what you are describing, only my exercise absolutely must not be what I would call “vigorous”. I consciously limit myself to an intensity where my heart rate is roughly between 100 and 120, generally closer to 100 most of the time. I am careful to remain as free of tensing as possible in my muscles, giving my limbs an occasional good shake. But I am slowly building condition, able to go faster within these limits. Plus I can go for hours, properly pacing myself. I think in terms of having to substitute TIME for intensity of exercise.
One of the things I was doing before that was massively harmful, was trying to properly run for a longer time. One minute – two minutes – three minutes. What an achievement! But a whole week of pain, stiffness and enforced inaction was the result every time. A pulse rate of 140 for 3 minutes is a killer. Even though I can DO it, dammit! It is waking up the next morning and trying to move, that is the point of reckoning.
Now I am pacing and going long rather than hard, I am just getting better and better. It is as if all the endorphins and whatever else is meant to help when you exercise, actually ARE helping now.
If you have come to understand your condition in the early stages, you are lucky. How vigorous are you really going? Could you be more careful with your pacing? Are you really improving overall, or flat-lining or even declining and being reluctant to admit it?
I don’t think anyone who has broken through into significant recovery of condition and quality of life, has done anything other than be careful with their pacing. I don’t believe there are any true “post fibro” athletes. It would be quite possible, I think, to participate in an “all-comers” long distance event and finish well before the absolute last person. These events have a lot of ordinary people in them just having an outing for a bit of fun! I am seriously considering some challenge like this as an ultimate goal. But to be near the front of the field at any time mixing it with the eventual top place-getters is just an idle dream.
My activity:
Two 15 minute sessions of body weight exercises every day, morning and late afternoon (see http://www.mattfury.com, Combat Conditioning)
One of the following on three consecutive days, none on day four:
One hour run (steady pace)
40 minutes jump rope (weighted rope, speedy)
One hour swim, multiple strokes, steady pace.
I am a nerd, not an athlete. Hope this helps.
You must have diagnosed the problem early enough and adjusted your activity early enough, to have been able to maintain condition where you can run for a whole hour, even slowly.
This is massively important. Fibro victims need to get as early as possible diagnosis and then as early as possible good advice about pacing. I am blown away with your confirmation of just how active a fibro victim can actually remain using the right approach. Your story could be an inspiration to a lot of people. How many other fibro victims are that active?
It might also be to your advantage if you are one of those skinny geeks, this won’t strain your muscles and cardio-vascular nearly so much as if you have a weight problem (like me).
I am regretting all the years of not knowing about pacing and just running myself down. I wonder if I will ever build up to the point of being able to run much again. 3 minutes moderate running sets up a major post-exertion malaise with me that lasts days. So does any vigorous burst of any exercise, even including swimming. My rule is keep it gentle. I can go for literally hours, gently. Your routine is actually not a lot unlike mine, except that I do not do any running, it is walking for me. And at every opportunity I try to use TIME – my gentle bicycle ride today was 3 hours.
I am interested that Cort says he is building up fitness by pacing; I am too. It is far slower than what a well person can achieve because they can go hard without self-harming like we do. I am trying to see if devoting large amounts of time to it, will substitute for going hard, and help build up faster. So far I am optimistic, including that I seem to have got weight coming off again after months on a plateau.
I love this
“I am careful to remain as free of tensing as possible in my muscles, giving my limbs an occasional good shake.”
Because it’s tenseness in my muscles that is the first sign of problems for me I believe. This is the first time I’ve heard someone express something similar.
“But I am slowly building condition, able to go faster within these limits. Plus I can go for hours, properly pacing myself. I think in terms of having to substitute TIME for intensity of exercise.”
During my extended camping trip I also experienced this….I am going sooo much slower – resting more – avoiding tension in the muscles and increased heart rate – and I really am stronger – able to do more….
This happened inadvertently. I wanted to enjoy nature and I can’t do that with my heart pounding, my muscles tensing up etc. so I have to stop very early on….
I find that I can do Yoga everyday. Just different movements. It even helps me to get back to sleep -if I wake up during the night.
Increases the bloodflow.
I was a runner before I became ill years ago. Still do walking, but have to careful with dizziness.
Yoga you can do anywhere.
Really want to know if Younger is going to be seeing patients.
Thanks, Cort
Carole
I think only within the context of doing a research study but Tina in a comment below mentions a fibromyalgia doctor in the area and Younger hopes to bring in an ME/CFS doc at some point.
Good advice about using nocturnal wakeful episodes to do something gentle and beneficial, like Yoga. It seems obvious but I never thought of it. I have generally tried listening to relaxing music. Can’t say it works. I have much improved sleep patterns anyway compared to what I used to.
I don’t think I have shared before, that Escitalopram (which my doctor tried on me) gave me exponentially worse nights and I soon realised that I was experiencing side-effect symptoms of the exact kind that drive some people to suicide.
http://www.drugwatch.com/ssri/suicide/
“…Antidepressants have also been linked to akathisia, which is extreme restlessness and an inability to sit still. The discomfort can be so great that suicide becomes a welcome alternative to feeling this type of agitation. Sometimes akathisia is misdiagnosed as worsening depression, so medication dosage is increased, causing the restlessness to persist…”
I like how they say “persist”. You mean, “get even worse”!
“Agitation” and “restlessness” is a major understatement – I suppose there isn’t any word that means those things “to the power of 10”. The apparent (hallucinatory) stimuli to the restlessness is a kind of deathly “presence” like an invisible cloud of the same pit-of-the-stomach sensation of depression itself, only descending on the entire body in waves, each of which causes restlessness almost as surely as if it was a physical wave of pressure on the parts of the body concerned.
The accompanying information with drugs that potentially have these side-effects needs to be a lot more specific about the kind of side-effects that are the drug itself and NOT worsening depression! The information was all about “if you feel suicidal”. It did not say anything about side-effect sensory causes of these suicidal feelings. I presumed that added causes of loss of hope or fresh unhappy happenings were required. It never occurred to me until quite a few weeks in to this horror, that what I was experiencing might be a drug side-effect, not something the drug was meant to be countering. Stopping the drug caused the episodes to end. QED.
I read somewhere recently that fibro victims seem to have an unfortunate tendency to experience all the bad side effects of almost any drug they are prescribed. That would be me. Amitriptylene gave me a dry mouth so bad that I got an inoperable stone in a saliva duct that is still there now well over a decade later. At least the public health system doctors SAID it is inoperable, but now I no longer trust them and their rationing and their death panels.
I would love to participate in a study of Younger’s. Thank you Cort for this article and contact info I would not have known about this opportunity to apply for a study without your website, which I now read, every article and often twice.
Thanks Judi – I hope you can get in there. I have a feeling it’s not going to be easy to find patients in the beginning. Birmingham is not that far from Tennessee so folks in Tennessee might want to check his lab out as well.
Cort, I am in Knoxville, TN, and have already offered to participate. I have contacts in Birmingham to help get the word out. Thanks so much for this amazing update on Dr. Younger’s work.
Excellent! Good luck Cindy 🙂
This is slightly off-topic, but it is a core study interest of mine and indeed where I am having increased success in getting my own writings published. It is a significant, observable phenomenon right now that all kinds of people (generally younger) and activities are relocating out of California and some other parts of the USA where the local housing costs are astronomical. Reading about a medical research project ending up in Alabama instead of Stanford is just typical of the kind of stuff I am reading regularly anyway! I just bought a new book on Amazon, “The New Geography of Jobs” by Enrico Moretti, to add to several books on urban economics and so on in my collection.
Joel Kotkin’s “New Geography” web site is an excellent source of regular information on this trend. There is a kind of ideological war going on, between those of the “creative urbanism” philosophy and those of the “opportunity urbanism” one.
Richard Florida has long been a prominent voice for the creative urbanism but he has very decently started admitting recently that there are major problems with the whole idea, that are similar to “trickle down capitalism” having proven flaws. Florida admits that “trickle down urbanism” also does not work. The underlying theory is this. You use urban planning to make your (often already pleasant) city “attractive to more creative people” and theoretically they gravitate to your city and make it a success. The problem is that the policies used always force up the cost of housing by a lot more than the actual “amenity value” created, because they effectively create monopoly-rent type distortions in the local property market.
This has a kind of vicious circle effect on the local economy. It is not just that high housing costs are such a burden that people end up further behind even if they earn more there; it is that these costs are taken out of discretionary spending of all kinds in the local economy. One of my favourite quotes of all time on this general point, is this from a New York Times article:
Mr. Olson, 42, said his family had not shed tears over leaving New York (for Oklahoma City). “There’s a little less to do, yeah,” he said. “But now we can afford to do it.”
http://www.nytimes.com/2014/08/04/business/affordable-housing-drives-middle-class-to-cities-inland.html?_r=0
While mortgage lending and property landlord institutions suck money out of the local economy (and that money ends up as incomes in Wall Street or somewhere else a long way away) there is simply less to go around on all the other stuff that makes a local economy work, including local tax revenue, which is ultimately dependent on local workforces discretionary incomes. So a kind of stagnation creeps up on the local economy and it seems to take forever for the economics and local policy “experts” to work out what the problem is. But of course there are powerful interests that don’t want this to be worked out.
http://www.newgeography.com/content/004031-density-unpacked-is-creative-class-theory-a-front-real-estate-greed
I don’t know if Younger and his colleagues might be able relate to this……..
Actually Phil my interactions with him suggested it may have played something of a role – and how could it not? Stanford and the Bay Area are not just expensive – they’re astronomically expensive. The dot coms that have added so much to the economy there have also driven housing expenses out of sight.
I imagine that even Stanford can no longer compete. i.e. offer salaries high enough to keep many of their “younger: (pun intended) researchers in the game…The older faculty who bought houses before the boom are sitting pretty but how anyone starting out can buy a home I don’t know.
I imagine that they were not happy to lose Younger….He’s got a basket full of projects, he’s gives great presentations, he’s on the cutting-edge, and he sounds like he’s really easy to get along with….
Cort, what an exciting article! Not only are we now seeing more and more young, bright researchers working on this illness, they’re doing the type of research I feel will help us. I go to the Klimas Clinic and my proinflammatory cytokines are elevated at each annual testing as are those of other patients at the clinic. LDN , of course, has been recommended but two tries with minute doses were still too much for me, causing dizziness and increased confusion. Other friends were unable go tolorate it too. People whe can take it have reported feeling better, though I have no ides about their bloodwork. I’m glad he’s looking into alternatives that may lower those, too.
I can only do mat exercises so that’s not enough to make a difference. I follow the HR and exercise guidelines laid out at Klimas’ clinic.
Thanks again for keeping us up to date!
I’m just curious what your minute starting dose of LDN was? I tried to start at 0.5g 3 times and failed. I was successful when I started at 0.1mg. I got results at 0.3 and am now at double that dose.
I see Dr. Rey.
Typo – I tried to start at 0.5mg.
Thanks for such interesting articles. It will be very interesting to see how using hypothyroid controls works out – how people will be allocated to different groups. Some people I have come across were diagnosed with ME or CFS but they actually turned out to be hypothyroid. People with ME quite often develop autoimmune thyroid disease (Richardson and Spurr) and others have problems with blocked thyroid receptors (Meirleir). Since blood tests aren’t very reliable I think it would take an exceptionally good doctor to be able to accurately differentiate between people with pure ME or CFS with no thyroid involvement, ME patients with various degrees of thyroid problems, whether to do with autoimmunity or lack of conversion or uptake, and controls with thyroid disease alone. I certainly can’t tell, with the patients I care for, how much is acute onset infection associated ME – how much is inadequately treated thyroid disease – and how much is other co-morbid conditions such as POTS or EDS!
Indeed – a thyroid blog is planned.
My descent into Fibro started following removal of 2/3 of one lobe of my thyroid due to a cancer – this is 25 years ago.
But do you think I can get those health-care-rationing charlatans in the public health system in my country to do any genuine more intensive investigation and testing of the OBVIOUS possibilities?
I am a furious opponent of all those socialist types who assure us that public-monopoly systems are best. It has never done anything but betray and stiff-arm me. They do operations to remove cancers and a few obvious things like that to present an illusion of “providing health care”, but on every condition that is complex and debatable and especially on every condition that is chronic but not immediately life-threatening, we might as well not be paying taxes at all for all the “coverage” we get.
I remember when the media used to run occasional stories about people falling through the cracks of the public system, and dying while on waiting lists and so on; but seeing that proposed solutions raised ideological bogey-men like “public private partnerships” and the media are unashamed socialist propagandists, this sort of coverage has been totally absent in the media for probably 20 years now. Meanwhile everyone personally knows of multiple cases of waiting-list deaths and medical misadventures and denied treatment – it is a bit like Orwell’s 1984 for the cognitive dissonance. Just yesterday a personal friend had an operation for bowel cancer that had been undiagnosed during multiple doctor’s visits complaining of pain and blockages, for MONTHS – 2 days ago he finally got taken in to hospital in an ambulance writhing and gasping with pain and the emergency surgeons say “what a pity we didn’t get onto this months ago (you probably won’t survive now)”.
But these public system rationing charlatans have statutory immunity to prosecution.
personally I have no doubt that neuroinflammation, pain and fatigue are going to be growth areas in research and that it’s reached the breakthrough point in America. There will be many setbacks along the way but each bit of scientific research paves the way for something better. An area where some young researchers will become the giants of their profession.
!!! 🙂 🙂
At one point Younger said lots of researchers are interesting in figuring out how to accurate measure neuroinflammation. .
Excellent research and excellent article.
Again – the brain, neuroinflammation. That’s where the answers lie!
We will get there soon!
At one point Younger said – lots of people are interested in this 🙂
Shoemaker has been talking of leptin for years in the context of mold exposure. That ties in with Brewers startling results in treating CFS patients for mold colonization.
I had no idea! Thanks…
Yes, for the first time in quite q few years, Alabama doesn’t have a team in the running for college national championship. I think those Ninja Turtle green and yellow ducks cause a vertigo effect on opponents. Whatever it takes, huh?
Younger is a PhD. We don’t have an expert MD (sees patients)in ME/CFS in Alabama, but maybe Younger’s activities will create one, maybe a young one just coming out of medical school (remember Younger also teaches). But, we do have Dr. David McCLain, known as an expert in fibromyalgia. He had already been part of Younger’s LDN in fibromyalgia research. And the two have connected again since Younger moved. Also doing research in Alabama and seeing some patients is Dr. Bill Pridgen in Tuscaloosa. But, he is a surgeon, so these type of illnesses is only part of his clinical practice. And he is most noted for his work in fibromyalgia, not ME/CFS.
As for property values, Alabama is great compared to other parts of the country. We don’t have the extreme boom and bust economy other major cities experience. It goes up and down, but not to the extremes of other metro areas. For the whole state, property values and property taxes are very low, utility costs are low also.
But, we also have subtropical weather. Only drawback, of course, is tornadoes. And, many tell me after they move here that they are surprised at how beautiful it is here. Traffic around Birmingham is not awful but not wonderful. lots of music, great museum of art, botanical gardens, wilderness park right in the city, theaters, symphony, and a lot of culture. Oh, and don’t forget southern food restaurants. And, t my knowledge, we have some of the most interesting festivals, such as Pinson’s butterbean festival and a boll weevil festival. Car racing, football, and many other sports. Golf is bog here also. State parks are also common. Heck, believe it or not, in our state, we even have skiing.
Hmmm – low property values, taxes and utility costs – and a very good medical research center….Intriguing…
Exactly!
There is a whole paradigm shift going on where the “opportunity urbanism”, get-the-basics-right, cities and regions are going to own the future of western civilisation, while the heavily-planned, elitist trickle-down “creative” urbanism is destroying local economies everywhere it is being imposed.
The flow of PhD’s and entrepreneurial people is becoming more and more clearly AWAY from the latter, much to the chagrin (and denial) of the advocates of the kind of policies that ultimately prices young people out of the local property market. A kind of momentum of its own is building up in despised places like Alabama that nevertheless focus on the basics of local governance – note that Airbus Industrie built its US factory there. Boeing opened one in North Carolina – Seattle takes them and their employees for granted. The lessons of Detroit are being learned by businesses (do NOT have all your eggs in one local economy basket) but not by certain local governments.
Texas is of course the stand-out on this. Rick Perry was right that if it wasn’t for Texas, the Federal administration would have almost no jobs recovery to boast about.
Thanks, Cort. Good piece.
Terrific article, Cort, and great news regarding Jarred Younger’s studies. Fingers crossed we might actually get a biomarker one of these days!
Cort, you’d fit right in. Come on down!
I got fibromyalgia after getting lyme disease and found that a stinging nettle urtication protocol has given me relief from fibromyalgia since 2008.
Thanks this was very helpful I wish I could participate in this study I AM so tired of suffering from fibromyalgia pain
am 55 year old male had 2 surgeries 2 years ago and got cfs have high epstien bar virus tried many doctors having a hard time any advice am on nexavir for 3 months used to be kutapression don’t know what to do hope someone can break this awful disease thank you for any advice
Isn’t the fatigue caused by mitochondrial dysfunction? I thought research was already revealing this. I hope you can find treatments for that problem!
Fatigue can certainly be caused by mitochrondrial dysfunction and it can be caused by many other things as well. Time will tell!
Dr. Younger,
Will you be taking any new patients for your clinics for trial?
You should be able to sign up for trials on his website here – http://www.psy.uab.edu/younger/index.html – finding patients for the trials is often the most difficult part of a study. I hope you sign up 🙂