Dr. Barnden of the National Centre for NeuroImmnology and Emerging Diseases (NCNED) in Griffith University, Australia has been doing some fascinating brain imaging work in chronic fatigue syndrome (ME/CFS). Over the past six years Barnden has been publishing results from a multifaceted brain imaging study, which analyzed magnetic resonance imaging results in two ways.
ME/CFS produces a plethora of possible central nervous system (fatigue, pain, movement, gait, stimulus problems, etc.) and autonomic nervous system symptoms (fatigue, pain, sleep, gut issues, cognition. Barnden’s unique contribution has been to tie both together by identifying parts of the brain that may be causing the autonomic nervous system problems.
2011 Study
A brain MRI study of chronic fatigue syndrome:evidence of brainstem dysfunction and altered homeostasis Leighton R. Barndena,b*, Benjamin Croucha, Richard Kwiatekc, Richard Burnetd, Anacleto Mernonea, Steve Chryssidise, Garry Scroopf and Peter Del Fanteg. NMR in Biomedicine, 11 May 2011: DOI: 10.1002/nbm.1692
The Gist
- Increased gray matter loss associated with fatigue duration suggested that gray matter loss may be proceeding more rapidly in ME/CFS over time. (As we age we all lose gray matter.)
- Reduced white matter levels in the midbrain suggested it may be involved in the autonomic nervous system problems found in ME/CFS
- Other clusters in the hypothalamus and brainstem suggested that the central autonomic network has been damaged in ME/CFS
- The damage appears to occur in the signaling pathways that connect the network
- The low stressors employed in the study (sitting up) suggest Barnden has identified core issues in ME/CFS.
- The NIH Intramural study should be able to greatly expand on Barnden’s findings
Barnden put 50 ME/CFS patients and healthy controls into an MRI and had them wear a monitor of blood pressure and heart rate for two days. Then he determined if the blood pressure and heart rate abnormalities he found were associated with changes in functioning of the brainstem area.
All the ANS measurements were done while the patients were lying flat or sitting up – as close to resting as possible. Plus none of the ME/CFS patients met the criteria for postural orthostatic tachycardia syndrome (POTS) – so they didn’t exhibit the major autonomic nervous system problems that those patients have.
The lack of stressors made this study all the more interesting. Many studies use physical stressors to push patients’ systems into an altered state where it’s easier to find abnormalities, but these studies purposefully removed these stressors and individuals (POTS patients) who were likely to exhibit autonomic nervous system issues.
No differences in the total brain volume of the grey or white matter in ME/CFS were found but greater drops in grey matter over time in the ME/CFS group suggested they might be aging more rapidly. (We all lose brain matter as we age).
Changes in total brain volume were not, however, associated with the severity of fatigue or other symptoms. A regression analysis, did find a significant reduction in white matter volume in the midbrain that was associated with fatigue duration. The longer a chronic fatigue syndrome (ME/CFS) patient had been fatigued, the worse their midbrain white matter loss was.
Low activity levels could cause this, but if so one would expect to see fatigue severity affecting the findings because increasing fatigue severity should result in reduced activity and therefore, according to the inactivity hypothesis, reduce brain volume. Fatigue severity did not, however, affect brain white matter volume.
The midbrain, not surprisingly given its location in the middle of the brain, sits in the midst of a vast communications network that is constantly zinging signals from one part of the brain to another. We know now that even simple actions require different parts of the brain working together. Midbrain damage, for instance, can affect the functioning of the “central autonomic network” made up of the hypothalamus, brainstem and other areas.
The study suggested that altered blood pressure-pulse readings were, in fact, associated with a cluster in a part of the mid-brain that regulates the reticular activation system (RAS). The RAS regulates cerebral cortex activity and is a conduit through which autonomic nervous system signals from the brain to the body pass. That suggests that the white matter loss Barnden found could be contributing to autonomic nervous system problems and/or – because the RAS regulates cerebral cortex excitation – could impair motor and cognitive activities: i.e. movement and thinking.
The white matter of the brain is made of blood vessels, fluid, and glial cells and nerve axons – everything but the actual nerve bodies. Further analyses suggested reduced numbers of glial cells were the most likely cause of the white matter loss found.Since glial cells when functioning properly are protective, a reduction could impair the brain’s ability to fight off pathogens.
2015 Study
Autonomic correlations with MRI are abnormal in the brainstem vasomotor centre in Chronic Fatigue Syndrome Leighton R. Barndena,b,⁎, Richard Kwiatekc, Benjamin Croucha, Richard Burnet d, Peter Del Fantee. NeuroImage: Clinical 11 (2016) 530–537
Barnden expanded on the brain’s connection to the autonomic nervous system problems he found in ME/CFS in his 2012 study. In this study he again compared the MRIs of the two groups and then did regression analyses using the MRI findings and the autonomic nervous system readings.
Apparently because Barnden assessed parts of the brain that regulate “steady state” blood pressure, he did not include stressors such as standing, walking or exercising which other studies have used to pop the brain or the ANS into a dysregulated state
Even at rest, heart rates were increased and pulse pressure was reduced in the ME/CFS patients. Anxiety and depression were negatively correlated with pulse pressure when patients were standing; otherwise no correlations were found.
Barnden assessed anxiety and depression because of concerns that they could affect blood pressure and heart rate. (In the original study he found evidence of mild anxiety and depression.) Adjusting for anxiety and depression did affect which clusters in the brain showed up in the study but did not affect the number of clusters that showed up.
One of the potentially malfunctioning clusters in the brainstem that popped out in the ME/CFS patients is designed to inhibit heart rate. Other clusters in the midbrain and hypothalamus that increase heart rate could be contributing to the increased heart rates found during sleep. Another cluster associated with the default mode network may also be affecting sleep. Several others found in the prefrontal areas regulate blood pressure and heart rate responses to stress.
The vasomotor center of the brainstem was particularly affected. Sitting in the center of autonomic nervous system activity, the vasomotor center communicates with sensors that regulate steady state blood pressure and heart rate. The hypothalamus and areas in the midbrain, in turn, regulate the vasomotor center. The fact that each of these areas of the brain have popped up in Barnden’s studies suggests that much of the central autonomic nervous system network in the brain may be affected in chronic fatigue syndrome (ME/CFS).
When the Barnden group simply compared the MRIs of the patients and the healthy controls without assessing autonomic nervous system functioning, however, they found no significant differences at all. This suggested that signaling between these autonomic nervous system nuclei is the problem, not the autonomic nervous system nuclei themselves. Something appears to be interfering with the proper flow of autonomic signals between these different parts of the brain.
This finding is not out of sorts with other findings in the past. While alterations in brain volume have been found in ME/CFS, signaling and connectivity issues may be more of a problem.
Barnden’s 2011 and 2016 study results were thankfully consistent with each other. Both implicate the midbrain and/or the brainstem in the autonomic nervous system problems in ME/CFS.
Barnden’s studies have been notable for their lack of stressors used. Exercise or other stressors are often used to enhance findings in ME/CFS. The fact that none were needed to flush out Barnden’s findings was significant. Barnden’s ability to connect brain issues with autonomic nervous system problems that emerged when ME/CFS patients were doing nothing more than sitting up suggested the study identified core problems in ME/CFS.
All this came using limited autonomic nervous system readings (pulse pressure and heart rate). One wonders what would have emerged if Barnden had been able to do more extensive autonomic nervous testing.
Hopefully an expanded study is underway in Australia. At the very least it appears to be underway in the U.S. in the NIH’s larger (80 person) intramural study which Nath reported will be employing top autonomic nervous system experts and doing multiple brain imaging tests. Since Barnden was able to get results with his autonomic nervous system testing regimen, hopefully the NIH with its more refined testing procedures and larger study size will be able to find much more.
The NIH Intramural study, like everything else it seems at the NIH, is going slowly. Nath reported in the Feb. 1 telebriefing that six healthy controls have gone through the study thus far – two of which they had to kick out after brain imaging abnormalities showed up. ME/CFS patients, thankfully, are apparently not in short supply. Nath reported that they’ve screened many of them and they’ll start, one at a time, going through the week-long study in March. It’s going to take more time than we wish but it’s a powerful study and could tell us much.


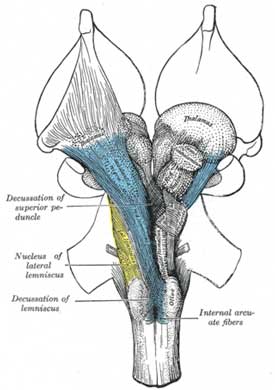
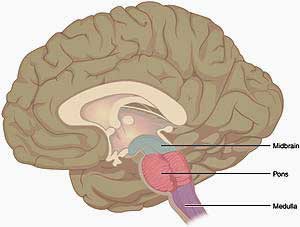




If we are losing gray matter and white matter more rapidly than normal how can full recovery be possible as Dr. Naviaux suggests? It is my understanding that once brain tissue is gone- it’s gone forever.
Actually it is possible to regain some brain volume and in rather simple ways. Exercise, for instance, has been shown to increase brain volume in the elderly (http://s3.amazonaws.com/academia.edu.documents/3480158/colcombearticle.pdf?AWSAccessKeyId=AKIAIWOWYYGZ2Y53UL3A&Expires=1486159319&Signature=09MyulCCrPUYSFR3rqwt3OU4snI%3D&response-content-disposition=inline%3B%20filename%3DAerobic_exercise_training_increases_brai.pdf)
Another article suggests that Omega 3 fatty acids may be able to.
Brain volume losses are also normal as we age; they don’t necessarily mean that we’ve lost a lot of capacity. It’s a bit different with these studies though; they suggest that specific regions of the brain have lost volume that may affect autonomic nervous system functioning.
The brain volume could be due to neuroinflammation which, if stopped, could, I would think allow the brain to recover (hopefully)
Lithium has shown to restore matter.
I was in one of the studies, I had my scans in 2010. My results said I had reduced cerebral blood flow.
I have been diagnosed with an inflamed pituitary. It means my adrenals are not getting signals to produce cortisol. I have secondary adrenal insufficiency. SAI
Primary Adrenal Insufficiency PAI is caused by an adrenal disease otherwise known as Addison’s.
Anyways, I have Hashimoto’s too.
I read at the HHV-6 Foundatiom that the Shingles virus, HHV-6 has been implicated in causing Hashimoto’s, so if HHV-6 can attack the thyroid gland I think it can also attack anything, and the HHV-6 Foundation is connecting that virus to other diseases like MS too. So I think that since I have been battling the Shingles virus for over 20 yrs that maybe it is causing the inflamation of my pituitary. Maybe it is also causing my ME/CFS too.
I had Epstein-Barr virus, sever problems on right side of body; felt like I was straining against my self, blown out thyroid. Later, thyroid insufficiency, adrenal problems, nerve problems, circulatory problems
occurred. Found I was extremely low on B-vitamins (B-12- used up too fast, or always depleted), magnesium deficiency constantly, low potassium, insufficiency in iodine. Low thyroid/adrenal nutrients, low on serotonin.
Increase all B vitamins, need extra multi-vitamins, needed D3 5,000 units for immune system, very low also on minerals. Need B-12 liquid shots 1-2 to start(not 5-hour), LESS SUGAR, MORE PROTEIN, 2 FISH OIL CAPSULES (brain needs)
also for heart/circulation and COQ10. Need B-12 immediately! Pills do not work fast enough, need liquid.
believe different viruses cause CF- some affect the brain, some the body more;
concerned about additives to vaccines; my Epstein-Barr virus affected my thyroid,
parathyroid, adrenals, and caused some problems with circulation in limbs. Did have a virus
that impaired breathing (for several months), since recovered. Will keep you all in my prayers!
I have the same concerns re: v addictive or preservatives.All the best !
I think mitochondria(in the brain have been impaired also due
to the viruses, also correlation between dementia and impaired
cellular energy. with people with heart problems, doctors don’t
mention that CoQ10 is needed for the heart muscle and heart metabolism
Jsuzor, I’ve had chronic shingles for seven years concomitant with ME/CFS (13 years). I see Dr. Chia and he has measured in me elevated titers of reactivated EBV (HHV-4) and HHV-6 (as well as enteroviruses and Chlamydophila pneumonia, a bacteria that acts more like a virus). I know that all of the herpes viruses can present with shingles-like rashes, but it’s HHV-3, herpes Zoster (VZV), which is shingles. Having HHV-6 yields similar symptoms and presentation, but it’s not Zoster/VZV/HHV-3/shingles.
This is fascinating, once again. Thank you, Cort, for your excellent explanation of these studies. It is so encouraging to see that research really is moving along now .
Thanks Jeanie 🙂
Try Lymphocytic hypophysitis autoimmune pituitary, NO GH or Test and TSH
How do I go about getting into this study? I have been in bed for the better part of 2+ years, with occasional bouts of “energy”. I have not exercised once in that time, and sitting up is an accomplishment. Any information would be very much appreciated. Thank You.
In order to get into the NIH study you’d have to be much healthier I’m afraid. You’d need to be able to withstand a weeks worth of testing.
I don’t know if another Australian study is underway but in that case you’d probably have to live near Griffiths University on the Gold Coast I believe.
Wow
Has there been any efforts to correct this signaling ever?
Thank you Cort for this information.
I don’t know but if neuroinflammation is involved it might be possible to correct by using treatments that reduce that.
Interestingly, Naviaux believes that signaling is the key issue in ME/CFS as well.
And there are age testrictions. I think over 60 and they won’t accept you
Thank you for sharing this. Are there any studies like this for Fibromyalgia?
Or are these findings applicable?
I would be surprised if they are NOT applicable. People with FM have the same heart rate variability test results as people with ME/CFS and a small fibromyalgia study found that a vagus nerve stimulator was able to rebalance the autonomic nervous system and reduce pain.
The brainstem has also been shown to be effected in fibromyalgia (https://www.ncbi.nlm.nih.gov/pubmed/24064248). I wouldn’t be surprised if the same thing was not happening.
Yes. .I have mild heartbeats variability. What can be done to rebalance this? I have read some of your posts on Vagas stimulation..but those devices are pretty expensive yes? Are there other options?
Heart rate variability lol
What do you mean by “mild” heart rate variabilty? You might be lackingvsome infirmation.
Everybody has heartrate variability and it is actually a very good thing.
It is what shows our “fitness level” for lack of a better word.
I am using an app called “visible” – free to download and use.
Check it out! with live from Montreal.
If I could do a happy dance without paying for it later, I certainly would. For the years and years and years that I have been ‘accommodating’ this disease I have believed it definitely had something to do with a ‘misfiring’ in my brain; my signals are getting crossed somewhere along the line. I am so encouraged by this study.
I would very much like to be part of this study, is it in the United States?
Thank you do very much for your interest and efforts in finding a cause and cure for this life robbing disease.
There is the NIH study. You can find out about being in that here. They are looking for people. https://www.healthrising.org/forums/threads/the-nih-wants-you-be-in-the-me-cfs-intramural-study.5241/
Hi Cort,
Is this thinking along the lines of the Gupta retraining program?
Hmmm….Well anything that rebalances the autonomic nervous system could be helpful and mind/body practices like the Amygdala Retraining program, breathing exercises and meditation all aim to suppress sympathetic nervous system functioning and increase parasympathetic nervous system functioning. If they can do that they could be helpful.
The symptoms that lead me towards a Fibromyalgia diagnosis were alot of gut pain, migraines..muscle spasms and alot of audio issues..hyperacusis…tinnitus..Audio Processing Disorder…I imagine this is linked to dysregulatin of nervous system…Vagal issues etc. But what I really want to learn more about is the RAS and what things I can/should be doing to focus my efforts in regards to these “guiding symptoms” Any suggestions?
.
Is this the same as volume loss in the cerebellum ?
No. the cerebellum is in the back of the brain.
Congrats to our Aussie researchers once again. I can certainly relate to changing pulse and blood pressure. Pulse ranging 68 – 120, BP from 150/82 to 200/105. And that can be while sitting reading in a chair. How do I know? It feels like an internal earthquake and lasts too long. The possibility of it being due to intermittent signaling is heartening. It is not all relating to mind and emotion!
Wonder if part of the reason your BP is that high due to lead or other heavy metal toxicity.
Mine had been high but recently dropped substantially after lead I didn’t know was there unexpectedly dumped out of me.
Interesting. In my view, the autonomic nervous system is the core problem in at least a subset of CFS / POTS patients. However, there is a question that nobody can answer. Is this a primary or secundair problem. In other words, is this a compensatory response or not!
In my view the CNS symptoms are due to low enegy output purposely enforced by our bodies to dampen the immune system. Thus protecting us from a more damaging autoimmune response. The better the dampening, the better the patient is, but in stasis. Protective stasis.
I know how to turn the energy on in my body. My body turns on really well. But. Then inflammation churns up and there is hell to pay. My body also knows how to clamp
down harder to stop energy rises.
It is very clear to me I have an autoimmune process. But when I turn my energy on, my mind is sharp, mood excellent, feel very energetic. At first. Then it turns ugly
One way I can ramp up energy is by taking gnc amplified wheybolic extreme 60.
But If I take it for too long, or too much heart rate goes up and my system goes into a furious faster and strong heart beat, and strong flu like symptoms.
I learned not to mess with the homeostasis state my body is in. I think the key will be to get rid of the antigen(s). Then the protection will be unclamped. Energy increase will happen next. I beleive that is why rituximab works.
By the way, if u are thinking about testing the product above, I learned to start at very low dosages. Effect kicks in more manageable at 5 tsps for me. But just one per day for me.
I do not advise its use. I have to stop anyway. Two days and my body fights something so hard I can not sleep well and feels like it is a runaway train.
But it was nice to feel great for an hour or two. It showed me it can all be eventually reversed.
In the comment above there is a link for the NIH study. Those unable to undergo an MRI scan are not eligible. Does anyone have the problem taking it? I experienced extreme, unbearable heat at the end of the procedure. I am definitely not claustrophobic.
It must have been a 3T MRI or higher (3T 3= is the strength t = tesla is the unit of measure). I had a spinal and then brain 3T MRI. I certainly felt a very hot spine and my head felt cooked from the inside out. Very unusual and quite scary.
Do they want gadolinium contrast? It has a FDA black box warning and has been known to deposit in the brain producing neurological symptoms.
Is this a form of dementia?
Cognitive problems are present but no one has called it dementia that I know of.
Time they told truth
Sure it is demensia
There’s no reason for the research commmunity to lie about cognitive tests – particularly since the researchers doing the tests are ME/CFS researchers committed to helping people with ME/CFS. Its possible that some people with ME/CFS do have dementia but the test results do not indicate that dementia is present for most people.
Thank you Cort! It’s nice to know that there is someone in my ring. Keep us posted please on any further advancements. Dont give up!
To the post by jsuzor: How did you find out your pituitary was inflamed? I have “something” on my pituitary…a microadenoma. Cort Johnson, how do I get this message to jsuzor? Thank you.
Cort, can you please comment on this news story which gives details of a story showing that the was no difference between CFS and fibromyalgia in terms of impairment of motor function.
Do you have a link to that? Or did I miss it?
Oops, I forgot. Here’s the link
https://www.dovepress.com/gross-and-fine-motor-function-in-fibromyalgia-and-chronic-fatigue-synd-peer-reviewed-article-JPR
What is a cluster?
Very Interesting Cort! I’ve been suffering from a CFS/ME ever since I cold turkey’d off an SSRI after being on it for 15 years. It’s now been 18 months and I’m still suffering. I have chronic fatigue, muscle pain and weakness, joint pain, blurry vision, breathing problems, heart palpitations, blurry vision and cognitive issues. I never had ANY of these symptoms prior or during my time on the SRRI. Since SRRI’s effect the brain and neurotransmitters, I’m almost 100% convinced it has messed up the signaling in my brain. I hope that I have not permanently damaged my brain. Do you believe that coming off an SRRI could cause an autonomic nervous system dysfunction?
Whoa! I don’t know but withdrawal can cause a lot of problems in some people. It’s not permanent but it can be tenacious. I know Klonopin is different but check out the Klonopin withdrawal issues that sometimes crop up.
There must be a way to ameliorate these symptoms, though.
Cort, so what were the blood pressure abnormalities in the CFS/ME patients? Low blood pressure/orthostatic hypotension?
Hello, do you know if there have been any studies done regarding a possible link between Central Sleep Apnea and ME/CFS or brain stem problems? Thank you for all the good information