

Baraniuk believes chronic fatigue syndrome (ME/CFS), fibromyalgia and Gulf War Illness are related. In fact, he calls the post-exertional malaise found in chronic fatigue syndrome (ME/CFS) a “unique characteristic of CFS that is shared by Gulf War Illness (GWI) subjects”.
Both diseases often involve a triggering event that’s caused long-term disability. The impact the Gulf War had on many of veterans of that war is astonishing. Baraniuk reports that 25 years after the first Gulf War that from @25-30% of those who participated experience PEM (exertional exhaustion), pain, migraines, gastrointestinal distress and other medical problems. Symptomatically Gulf War vets appear much like people with chronic fatigue syndrome (ME/CFS) but some important caveats exist.
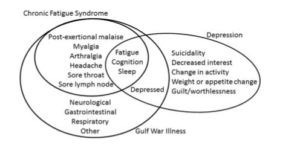
A similar symptom presentation occurs in both ME/CFS and GWI. Depression on the other hand is more distinct. From Baraniuk
The vast majority of ill Gulf War vets are men who were subjected to a variety of toxic exposures and vaccinations not usually experienced by people with ME/CFS. In his paper “Exercise – induced changes in
cerebrospinal fluid miRNAs in
Gulf War Illness, Chronic Fatigue
Syndrome and sedentary control
subjects“, Baraniuk suggests that that one logical hypothesis regarding GWI involves lesions produced by acute acetylcholine neurotoxicity caused by people with genetically reduced levels of acetylcholinesterase activity being exposed to cholinesterase inhibitors . No such hypothesis to my knowledge has ever been proposed for chronic fatigue syndrome (ME/CFS).
Despite their symptomatic similarities, the two diseases have very different triggers and likely have different pathophysiology’s. It gets more complex though. Baraniuk, a very creative researcher, used exercise followed by a standing test to identify two subsets of GWI patients. One called the START group (Stress Test Activated Reversible Tachycardia) responds to exercise by temporarily meeting the criteria for POTS, and altered brain functioning including reduced brain stem volumes. The other group called STOP (Stress Test Originated Phantom Perception) developed a pattern of basal ganglia and insula activation similar to that seen in phantom pain.
Baraniuk sampled the cerebrospinal fluid of the three illness groups (START, STOP and ME/CFS) and the sedentary healthy controls before and after exercise. He looked at the miRNA patterns in an attempt to determine changes in central nervous system functioning that have occurred over time. miRNA or epigenetic studies analyze the changes in small amounts of RNA which we accumulate over time in response to events. Because these small RNA’s are capable of turning on or off genes they’re a prime candidate in any disease which gets triggered by some event.
This is first study that I know of that’s examined cerebrospinal fluid factors before and after exercise in ME/CFS.
Results
“We clearly see three different patterns in the brain’s production of these molecules in the CFS group and the two GWI phenotypes. This news will be well received by patients who suffer from these disorders who are misdiagnosed and instead may be treated for depression or other mental disorders.” Baraniuk.
Baraniuk’s decision to introduce an exercise stressor paid off when, as we’ve seen at times in ME/CFS, all the groups showed no difference at all in their miRNA profiles at rest. After exercise, things changed, and they changed most dramatically for the ME/CFS group.
Reduced levels of three miRNA’s (miR-let-7i-5p, miR-93-3p, miR-200a-5) after exercise in the ME/CFS and GWS groups but not the healthy controls suggested some degree of commonality was present in the illness groups. Decreased expression of MiR-let-7i in a rat model of depression suggested that decreased levels of this miRNA might contribute to the mood alterations sometimes seen post-exercise in ME/CFS and GWS.
Chronic Fatigue Syndrome (ME/CFS) Group Stands Out
After exercise the 12 reduced miRNA’s in ME/CFS patients clearly differentiated them from people with GWI. ME/CFS was the only group to display no elevation in miRNA levels after exercise. (Five elevated miRNAs occurred in the sedentary controls, 3 in the START group and 1 in the STOPP group.) One miRNA found reduced in ME/CFS might be associated with pre-mature aging, another with Alzheimer’s and amyloid deposition, another with the leakage of inflammatory factors into the brain, and another with increased cell suicide in the brain.
Aging – Past studies suggest that premature aging may be taking place in ME/CFS and /or fibromyalgia. The CDC reported that telomere length – believed to reflect aging – was significantly shorter in ME/CFS patients. Telomere length also trended lower in fibromyalgia patients than healthy controls, and was associated with increased pain in that disease. FM patients scored about 20 years older than expected in cognitive testing. Reduced heart rate variability – found in both ME/CFS and FM – occurs as we get older as well.
(On a side note a study suggesting that age does make a difference in ME/CFS found that older people with ME/CFS experience more fatigue and depression and greater autonomic dysfunction (reduced parasympathetic/increased sympathetic functioning – i.e. reduced heart rate variability) than younger people.)
Dementia – No studies that I know of have examined the risk of dementia in ME/CFS but a recent fibromyalgia study suggested that an increased risk for dementia/Alzheimer’s may exist in fibromyalgia. (Despite the increased risk, the odds of getting dementia were still quite low). While Baraniuk noted that the miRNA patterns found in ME/CFS were different from those found in Alzheimer’s disease, one reduced miRNA in ME/CFS called miR-19b-3p was lower in both the cerebral spinal fluid and serum of Alzheimer’s patients as well.
Baraniuk didn’t mention it but his small 2005 cerebral spinal proteome study suggested increased amyloid levels could be present in ME/CFS as well. In an interview Baraniuk proposed that the aggregation of amyloid proteins in the blood vessels of ME/CFS patients could be causing small amounts of bleeding in their brains and questioned whether the location of those small blood vessel ruptures might determine whether one has ME/CFS or FM or GWI. Baraniuk speculated that these proteins might interfering with the brains ability to control the autonomic nervous system.
Blood-Brain Barrier – Nath has suggested that immune cell migration into the brain could be causing neuroinflammation in ME/CFS, and, in fact, the integrity of the blood-cerebrospinal fluid barrier ended up being a major topic in this paper. A finding of increased cell suicide in the brain after exercise might not be such a surprise if exercise is inducing inflammation. Cook has shown that cognition and brain functioning are altered one day after exercise in ME/CFS.
Caveats – Baraniuk mentioned some caveats to miRNA analysis of cerebral spinal fluid (CSF) including the “remarkable lack of consensus” regarding the levels of miRNA expected found in healthy controls. (Baraniuk noted that he used more CSF fluid than some other studies. ) Note that information on the functions of many of the reduced miRNA’s in ME/CFS is lacking, and the information on those that were found can be pretty sketchy; i.e. based on one or two, sometimes non-human studies. This issue of data acquisition out lapping our understanding of what it means is pretty common in “omics” studies.
Baraniuk has been exploring how exercise effects the brains of ME/CFS/GWS patients for years. His small 2013 study linking increased lactate levels in a subset of GWI patients to reduced exercise capacity was intriguing giving the high lactate levels Shungu has found in ME/CFS. As noted above, another exercise study uncovered two subsets of GWI patients: one developed increased pain after exercise and another temporarily developed POTS and exhibited signs of brainstem and basal ganglia problems.
The Baraniuk lab at Georgetown University in Washington D.C. is currently recruiting for ME/CFS patients for an NIH funded two-day exercise/MRI study.
Conclusion

Despite the similarity in symptoms, Baraniuk’s study of cerebral spinal fluid miRNA’s suggests that the central nervous systems of ME/CFS and GWI patients may respond very differently to exercise. The results need to be validated and interpreting them can be difficult given our limited knowledge of the effects that individual miRNA’s have, but they suggest, that too much exercise might harm the integrity of the blood-brain barrier, or alter cognitive functioning (studies indicate that’s true), interfere with cell suicide, or exacerbate an early aging process in ME/CFS.






A friend of mine explained some connections with choline and GWS and CFS. This was us discussing this on a POTS site back in 2012.
https://www.dinet.org/forums/topic/22052-need-help-finding-articlegulf-war-illness/?page=2
Appears some of us have connected our symptoms as similar. We have tried just about everything to tweak symptoms to see if it helps.
Some of us on Healthrising Forum are also talking about choline and it seeming to help. It does appear that many of us middle aged are noticing more severe brain fog/cognitive issues. Whether that’s due to a lack of blood, oxygen or what?????? It’s a problem.
It gets complex and complicated……..
Issie
“Some researchers believe ME/CFS occurs when genetics we are born with collides with some environmental factor.”
——————————————————
2012 CFSAC testimony by Jeri McClure Kurre
http://www.youtube.com/watch?v=EbcR9f7hwow&feature=player_embedded
Let me make something perfectly clear. The illness entity known as CFS, based on an outbreak of ME Plus, has never been fully investigated by
anyone. Not one researcher has examined all of the evidence that caused the creation of this syndrome. NOT A SINGLE ONE. After nearly 30
years. This is a disgrace of epic proportions. A disease that has brought brutal,relentless suffering to now millions of people and yet we have
evidence that has been around since the inception of the syndrome that has never been examined. The reason that I am able to be here today is
because I have been fortunate enough to be able to utilize therapeutic strategies based on evidence that has been ignored for over a quarter
century.
Incline Village survivor Erik Johnson, who served as a prototype for the syndrome, noticed a peculiar phenomenon in regards to biotoxins. He
devised a strategy in response to it which enabled him to achieve such an astounding recovery that he is able to climb mountains. Thanks to his
work and persistence there are now quite a few CFS patients who have used his techniques to achieve recovery. We are not cured. However,
many of us who were severely disabled are now able to live near normal lives. I think that it’s about time that researchers start looking into this. It
seems to me that discarding evidence for decades on a horrific, life-destroying disease is downright negligent. I want to know why NO doctors and
NO researchers are telling patients about this.
If it had not been for Erik Johnson, I would either be dead or at home still rotting in my bed. So, to him I would like to say this: with all of my heart
and soul, thank you for helping me. …..And to all of you CFS researchers who claim to be unaware of this particular evidence, I want to say this:
You are now.
What are you going to do about it?
-Jeri McClure Kurre
This is in response to Eriks post. Having found CIRS with myself, my sister and her son….I think some of us genetically are predisposed with inability to throw off mold and biotoxins. This can be found every where. None of us are immune. It’s what our body response (immune systems) do with this exposure as to whether we can throw it off or not. Genetically, I have the worst combinations and inability to detox. We know mold is an issue with me, it was found in a thyroid tumor biopsy. And yes, this can get in organs and look like cancer – when it’s not. But this type mold, that was found with me, can cause vascular tumors – I was told by my doc. Detoxing for several years now and getting treated for Lyme is now making me, finally, see light at the end of a tunnel. I feel a glimmer of hope now.
If our immune systems were working properly – we wouldn’t have the issues. If we can detox and get them to recognize what shouldn’t be there – we start to “climb out”. So much can not yet be detected with medical testing. There are a lot of organisms yet to be discovered. So just because one test may say negative doesn’t mean there isn’t something there. Just means the testing isn’t there to detect it yet. Science advances…..but us guinea pigs and pioneers are getting tired.
Whatever “triggered” the epigenetic changes (mold, biotoxin, virus, vaccine, bacteria, protozoa, stress)- if we could switch off this activation and get our bodies to function in a more positive direction and detect instead of ignore – what it shouldn’t – moving our bodies to a more positive response – we would probably be as good as imperfect bodies can be.
Issie
Having suffered from ME?CFS and now fibromyalgia for over 30 years,all I can say about researches is that they continue to look under the wrong rock: whether by ignorance, on direction by big pharma, or out of malice because suffers seem to be more informed than the medical fraternity is. Regardless, have found great results from addressing biotoxin containmination, mold exposure and most importantly, addressing L-glutamated neuro-toxicity in my body.. Going Paleo ( eating ketogenic/paleo diet), detoxing with bentonite clay to remove the negative gram bacteria in my gut, and avoiding all fermented foods and formaldehyde and excito-toxins has yielded wonderful relief from multiple system I have suffered from. While I never served in the gulf war, I did serve in the RAN prior to 1990 and was innoculated with the same dirty vaccines( except anthrax) that deployed vets were given. I was also exposed to pesticide agents ( anticholinergeric agents) which is the same for GW vets. What I find extremely galling about the whole medical establishments focus on ME/GWI is they flatly refuse to come out and say the ME/CFS/GWI/Fibromyalgia is caused by poisoning period. And that the real solution lies with detoxing, minimising further exposure to anticholinergic agents, and educating the poisoned suffers on how to self heal and manage any damage done and lobby politicians to force chemical manufacturers to stop polluting us and our environment any more.
I hate to hear that we are at risk of dementia or Alzheimers. Are we getting any closer to figuring this illness out…what do you think Cort?
Yes that is hard to hear and I want to note that the risk was still quite low. It does make me want to implement this plan – https://www.healthrising.org/forums/threads/reversing-alzheimers-what-could-it-mean-for-chronic-fatigue-syndrome-me-cfs-and-fibromyalgia.4715/ – which is basically an anti-inflammatory plan.
I think we are getting closer but we still have quite a ways to go…..You never know what will happen though.
With CIRS, my brain is swollen and compressed. The compression part is due to the swelling of other parts – according to my doc. And it should get better when the inflammation comes down. The parts compressed do affect cognative function. The “brain on fire” – Dr. Ackerly speaks of happens with mold and biotoxins. Getting down inflammation is very necessary. But if it’s an internal cause – you have to focus on the WHY.
Issie
Cort, you made this statement above: “Studies indicate that anything which substantially stresses the body (infection, birthing procedures, adverse childhood events) can set up people for disease in later life.” Have you seen this abstract?
Rethinking childhood adversity in chronic fatigue syndrome
http://www.tandfonline.com/doi/abs/10.1080/21641846.2018.1384095?journalCode=rftg20
No I hadn’t seen that. That statement didn’t refer to ME/CFS but was based on broad findings. If the goal is not to get ME/cFS being interpreted as a psychological disorder. though, I’m not sure if how much that study helps. Here’s the conclusion from it
Now check out the conclusion from a recent study finding regarding adverse childhood events and autoimmune diseases – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3318917/
Many studies have shown that ACE’s increase the risk of inflammatory diseases later in life. I think I would rather be in that group actually.
“In an interview Baraniuk proposed that the aggregation of amyloid proteins in the blood vessels of ME/CFS patients could be causing small amounts of bleeding in their brains and questioned whether the location of those small blood vessel ruptures might determine whether one has ME/CFS or FM or GWI.”
-> As theorized earlier these potential “…small amounts of bleeding in their brains and… …those small blood vessel ruptures…” could play a factor in keeping blood volume low in ME(and FM?) patients. Higher blood volume (and associated blood flow) could increase brain bleeding and damage to levels that would cause acute danger and chronic decline. Too low volume and flow would deprive the brain from almost anything essential.
This would force the body to maintain a very small region between too much and too few (in order to operate on a very poor “optimum”). With it we would get both serious symptoms of too few blood volume like brain fog, POTS and increased exercise intolerance and on the other hand serious symptoms of brain inflammation and increasing amounts of permanent brain damage caused by ruptured small blood vessels and brain bleeding.
Short term pushing the body to increase blood volume could make one feel a lot better by removing the symptoms associated with poor blood flow. Experiments with medication or saline however very often showed the body keeps restoring the “bad” low blood volume and flow with a vigor. If Baraniuks theory could be validated it could help giving a better view on many of these “stuck” mechanisms. Other such stuck mechanisms could be too much/few inflammation, too low/high body temperature, too strong(auto immunity)/weak(body full of opportunistic pathogens) immune functioning… This could explain why restoring wrong values to normal without adjusting the “other side of the equation” often gives so poor results or fires back later.
I therefor do believe that giving the body options to move values in a wider range holds more promise than trying to fix “wrong” values. I do believe that avoiding extremes (compared to the average sick self, not to a healthy person) both low and high helps as well in this regard.
What about POTS after exercise in ME patiënts? It is not only in GWS, Baraniuk is wrong at this point!