A New Approach To Chronic Fatigue Syndrome
If we ask Nature a question and ask it well, Nature answers. Most of the time the answer refutes our hypothesis. This time, Nature didn’t refute the hypothesis. Robert Phair
Ron Davis mentioned one of chronic fatigue syndrome’s aces in the hole several times – the chance to make a huge difference in a major disease. You could work for decades on cancer and make a difference there – an important difference but probably a small one, or you can take on something like ME/CFS and potentially make a huge difference. One of the reasons we have so many creative researchers in this small field may be because the ME/CFS challenge draws inquisitive minds.

Phair got involved in Ron Davis’s ME/CFS work after reading about it in the Stanford Alumni magazine.
Phair certainly has a inquisitive mind. He was one of the most active participants in the three-day Working Group session put together by Ron Davis and the Chronic Fatigue Syndrome Research Center at Stanford University. On the fourth day, the Working Group was featured in the Community Symposium on the Molecular Basis of ME/CFS at Stanford University. Both were sponsored by the Open Medicine Foundation with a special thank you to OMF Board Member Dr. Deborah Rose.
Phair’s a rare breed – a biological systems engineer. When he started out he caught grief from both sides; his father couldn’t understand why he would top off a good degree (electrical engineering) with a PhD in a bad one (physiology), and the biologists in academia didn’t get him either. He ended up forming his own company, Integrative Bioinformatics, Inc., which produces “models for biomedical discovery”.
That dual focus, however, fits right into Ron Davis’s mold. Davis, geneticist and inventor, after all, is his own unusual blend. He doesn’t run the Stanford Genome Center, he runs the Stanford Genome Technology Center – a blend that adds a pragmatic bent to his efforts. Davis is not an ivory tower theorist: his forte is creating tools that impact human health. Robert Phair – engineer and physiologist – must have seemed like a kindred spirit. Davis said he’d been looking for a systems engineer for a while.
(It bears reminding that the originator of the Metabolic Trap Hypothesis – Robert Phair – came to ME/CFS via an article in a Stanford magazine about Whitney Dafoe and ME/CFS. Sharing makes a difference. The more stories we get out, the more people who know about the disease, and the better chance we have of finding someone who will solve it.)
Beginner’s Mind
He started off saluting what he called the best book on ME/CFS – The Doctor’s Guide To Chronic Fatigue Syndrome: Understanding, Treating, And Living With CFIDS, published by Dr. David Bell in 1994!
That reference to the 1994 book says something about Phair’s approach. He’s apparently read everything he can on ME/CFS and he’s done so with a “beginner’s mind”. Take the approximately 70 outbreaks that AAMES lists have happened between 1934 and 1990. Outbreaks were what ME/CFS was known for, for decades, but the last one listed happened over 25 years ago, and they are rarely discussed anymore.
With his beginner’s mind, though, those 70 or so outbreaks caught Phair’s eye. How many diseases, after all, have such a list of virulent outbreaks?
The fact that so many people became so ill so quickly stood out. A pathogen sweeping through a community was obviously one component, but the wide swath of people suddenly becoming ill suggested that the pathogen had taken advantage of a widespread vulnerability.
Phair’s Factors
Most researchers have looked for rare genetic tweaks but nothing rare could account for the masses of people that typically became ill. Phair reasoned that the susceptibility to the disease had to be common: if a genetic tweak (missense mutation) set the stage for chronic fatigue syndrome, it had to be one that was common – that many people shared.

Phair had a checklist of factors the metabolic trap hypothesis required to be present for it to be possibly valid.
He wasn’t looking for just any common polymorphism, he was looking for genes tied to systems that featured “bi-stability” – the system could be stable in two different states: a healthy state and an illness state. (The kind of thing only an engineer would look for…)
Plus, once one got into the illness state, the system had to feature a kind of “stuckness” which made it difficult to return to health.
Plus, the system had to be vulnerable to a sudden stressor and it had to feature the ability to suddenly “flip”.
Plus, the gene mutation had to have the ability to affect multiple systems – to generate the kind of widespread illness that ME/CFS is.
A common genetic tweak affecting a bi-stable system which exhibits remarkable “stuckness”, and affects multiple systems, and is vulnerable to a stressor and can suddenly “flip”: that’s a rather daunting set of preconditions to fulfill…
The Hunt
Common Gene Mutations
Nevertheless, the hunt was on. Phair had the complete genome data of 20 severely ill patients (and some more genome data from people who volunteered their data after the last blog on the metabolic trap). The first test of the hypothesis was to find a mutation which was ubiquitous in the severely ill group but which was commonly but not ubiquitously found in the general population.
Up popped a mutation that breaks or damages the ability of the ID02 gene to metabolize tryptophan. A single mutation (or another mutation in that gene, sometimes as many as 3) was found in all his ME/CFS samples but is only found in about 40% of the general population. (55% of ME/CFS patients had the most common mutation and everyone had one or more other mutations. Out of two possible broken copies of IDO2 (two chromosomes for each gene) the severely ill patients averaged 1.7 broken copies.) The first hurdle was passed.
Bi-Stable System
Next the gene had to be part of a bi-stable system in which the polymorphism could, under certain circumstances, conceivably cause the tryptophan-metabolizing system to take a strange turn.
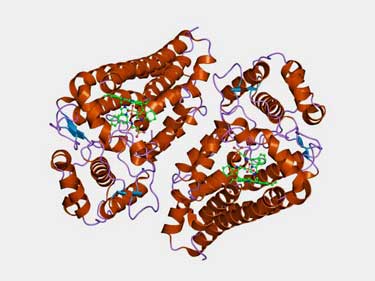
Every ME/CFS patient tested thus far has one or more mutations in the gene producing the IDO1 enzyme.
It turned out that the IDO2 enzyme was part of a system of tryptophan-metabolizing enzymes that also included an enzyme called IDO1.
IDO1 and IDO2 are two of three enzymes that catalyze the oxidation of L-tryptophan to N-formylkynurenine. (The last is tryptophan 2,3-dioxygenase (TDO). IDO1 is the first enzyme out of the gate. It breaks down tryptophan correctly most of the time, but two circumstances can make it fail: if it lacks the substrate it needs to do its job, or if tryptophan levels are very high.
The second circumstance leading to failure is probably the key one. Tryptophan is both required for an optimal immune response and is used by some pathogens to grow. Some pathogens can inhibit the IDO enzyme (in order to make more tryptophan available to them). Because IDO enzymes also regulate T-cell and NK cell functioning during infections and play a role in immune tolerance or preventing autoimmunity, inhibiting them can hurt immune functioning.
Thankfully, the other IDO enzyme – IDO2 – thrives in high tryptophan environments. But what if a mutation has blunted IDO2’s effectiveness? Then tryptophan levels rise, inhibiting IDO1 even more, resulting in less tryptophan oxidation, causing tryptophan levels to rise even more, and so on. You have a vicious circle resulting in higher and higher cellular tryptophan levels.
You’re stuck in what Phair calls the “IDO metabolic trap”.
Exhibiting “Stuckness”
Phair next had to demonstrate that the system can get stuck and be difficult to reverse. His mathematical models were devastating. Even if you completely stopped all inputs of tryptophan into the cell, ten days of stressor-induced, mutation-enhanced tryptophan concentrations would take 30 weeks to recover. If that situation dragged on for just 8 weeks, the system would essentially never recover – even if all tryptophan inputs were halted. This is a system which demonstrates remarkable “stuckness.”
Affects Multiple Systems
The serotonin system is in the midbrain. Phair pointed out six nuclei or neurons in the midbrain which control serotonergic pathways in the brain. Dysfunction in these nuclei could produce a long list of symptoms found in ME/CFS including impaired cognitive function, movement, and smell, dysautonomia, motor neuron problems, air hunger, dopamine production, POTS, and pain.
Plus kynurenine, which the model predicts will be low, affects hundreds of reactions in the body (including the production of ATP and regulation of immunity).
Testing the Hypothesis
“We can propose theories based on anything – from previous data to pure imagination, but the key (the thing that makes it science) is to use those hypotheses to make predictions and design a well-controlled experiment to test those predictions.” Robert Phair
Ron Davis got up and warned against cherry-picking data to fit one’s hypothesis. The real way to test a hypothesis is to use it as a prediction tool: if the hypothesis is correct, then when tested, “X” should show up. If it doesn’t, the hypothesis is wrong. Phair’s hypothesis is in the prediction phase – it predicted correctly that certain findings regarding tryptophan would be found in ME/CFS patients’ cells. Calling science a “constant disappointment”, Davis said most of the time hypotheses are wrong.
Passing the Cellular Test
But so far, not this one. Phair’s and Davis’s enthusiasm has grown as the hurdles from genetics to mathematical and other tests of the trap have been passed. The biggest hurdles, though, were the cellular tests. The group wasn’t sure if the trap even existed in the immune cells they were testing. It might have been only present in other cells.
The tests involved determining if altered tryptophan and kynurenine concentrations (high tryptophan, reduced kynurenine levels, increased tryptophan/kynurenine ratios) were actually found in ME/CFS patients’ immune cells. The Open Medicine Foundation stepped in to provide the funding.

The sample size is small but every ME/CFS sample tested thus far has displayed unusual levels of tryptophan and/or kynurenine.
The first six samples from ME/CFS patients fit the predicted profile while the healthy controls’ cells were absolutely normal. At that point, the trap appeared to have jumped from being a theoretical possibility to something potentially very real, and the level of excitement jumped substantially. Davis called the cellular test results “very strong support” for the hypothesis.
He grasped onto the cellular results like a mountain climber offered a rope. It was only after positive cellular test results that Phair and Davis felt comfortable explaining the trap in detail. The results are limited – just six severely ill patients – but they provide some scintillating new data to chew on. Whether Phair’s metabolic trap hypothesis is correct or not, Davis now has some new, very unusual, real world data about ME/CFS to think about.
Davis has talked about getting to the core of the disease but he’s never talked about possibly being at the core of the disease. He clearly feels the trap and the data gathered so far offer that possibility. Much testing remains to be done, more hurdles remain to be passed, but the trap hypothesis is clearly on much more solid ground now.
Davis once again noted that hypotheses often turn out to be are wrong. It’s possible that the trap could become another in a long stream of, as Robert Phair said, paraphrasing the great British biologist Thomas Huxley, “beautiful theories destroyed by ugly facts.” The only way to determine if the metabolic trap is causing ME/CFS is to forge ahead.
High Serotonin Levels / Low Kynurenine
Tryptophan metabolism can go one of two ways – to make serotonin or to go down the kynurenine pathway. It bears noting that a little serotonin goes a long way. The vast majority of the time (@95%), tryptophan is metabolized to kynurenine; just five percent of the time is it metabolized to serotonin. The implications of the trap – that high serotonin and low kynurenine levels are present – is highly significant.
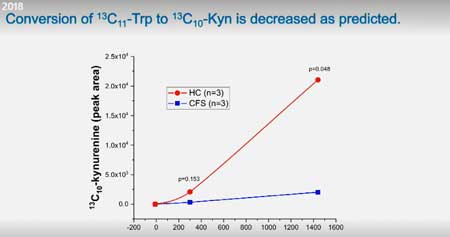
Thus far, tests indicate ME/CFS patient’s cells are metabolizing much less tryptophan to kynurenine compared to controls.
Phair’s slide of the serotonergic system in the midbrain – the same one that Cortene used to explain its hypothesis of an over-activated serotonin system – showed the serotonergic system’s immense spread.
Gerard Peirara of Cortene has asserted that the many more receptors for serotonin (14) relative to other neurotransmitters (dopamine (5), norepinephrine (5)) reflects how uniquely pervasive and influential that system is.
Davis focused more, however, on the low kynurenine levels found. Calling kynurenine a “very important molecule”, he noted it’s used to produce a substance called NAD, which plays a role in about 400 reactions in the body, including the production of ATP. Plus, because kynurenine is an immune system regulator, low kynurenine levels could also increase the risk for autoimmunity.
Phair, on the other hand, thinks that there is plenty of nicotinate (niacin) in the diet, so he’s not convinced that reduced kynurenine production will significantly reduce NAD. But, Phair emphasized, the biological activities of kynurenine pathway metabolites is a burgeoning field and has been for a decade or so.
Don’t Do This at Home
“This is a dangerous pathway to experiment on…by self-experimenting with these critical pathways, you could make yourself much, much worse.” Ron Davis
Self-experimentation is almost second nature to people with ME/CFS, but Davis strongly warned against it. Davis and Phair didn’t release the details of the trap until now, precisely because of the pathway’s dangers.
You can actually induce autoimmunity – an autoimmunity you cannot recover from – by taking tryptophan. Note that if the trap is correct, the brains of people with ME/CFS have probably become adapted to very high serotonin levels. What happens when a brain that’s been wired to high serotonin levels suddenly gets deprived of it? Nobody knows, but everyone can guess what might happen if the process is not done correctly. The results could be awful – and possibly permanent.
If increased tryptophan levels are the culprit in ME/CFS, the treatment has to be worked out using computer and animal models first. Davis requested that the ME/CFS community give them some time to figure out what’s going on.
Convergence on One Part of the Brain
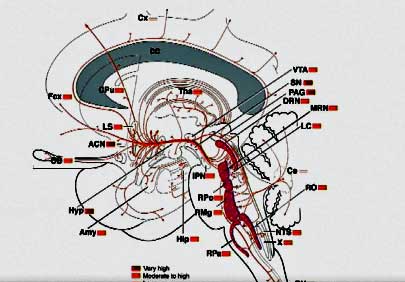
Phair and Cortene used the same slide to portray the reach of the serotonin system in the brain. (Modified from Phair’s slide presentation.)
Serotonin has hardly been mentioned in ME/CFS but 2018 has brought us two hypotheses from two entirely separate groups which are both converging on the serotonin pathways in the brain.
Cortene believes that stressor-induced dysregulation of the CRH receptors in the hypothalamus results in a serotonin explosion in the brain.
Phair believes that given the right circumstance, the mutations could produce what is essentially an evolutionary mistake at the cellular level (allowing serotonin to build and build), resulting in the same thing in the brain and elsewhere. Phair also noted, though, that depending on the cell type, serotonin levels could be high or low.
The fact that Cortene successfully exposed their brain model to hypothetical testing – which suggested serotonin dysregulation could account for sudden onset, gender differences, movement issues, metabolic problems, etc. – only buttresses Phair’s conclusions.
Working Group That Works
When they work, Working Groups provide new insights and new opportunities for collaboration. That certainly happened at the second meeting of Ron Davis’s ME/CFS Working Group hosted by the Stanford Genome Technology Center. A year of further data gathering – particularly from Davis’s group at Stanford – and an expansion of the group brought more energized discussions and more opportunities for collaboration. The fires that smoldered during the meeting of the first Working Group a year ago burst into flames in this one.
The Trap certainly highlighted how effective Working Groups can be. Different members offered to produce models of the trap. Among other things, those models will be able to provide ways to demonstrate that the trap is present in brain serotonergic neurons and to test potential therapies.
Next Steps
I wake up every morning thinking about what might be wrong with this theory and about experiments to test that worry. We’re working hard. We won’t give up. Robert Phair
A lot of work at Stanford has gone into gathering more and more data and that work is continuing, but with regards to the metabolic trap hypothesis, Davis and Phair are in a new realm. Now that they have a testable hypothesis, they’re going to continue to test the heck out of it.
The next steps include:
- First, more tests on ME/CFS and healthy control cells to provide convincing evidence that ME/CFS patients’ immune cells are handling tryptophan VERY differently from healthy controls’ immune cells.
- The trap needs to be shown to be present in immune cells in cell culture and it needs to be determined if they can get the cells out of the trap in cell culture.
- Concurrently, consultations with experts in metabolism and nutrition will commence, and computer simulations and experiments will be done to figure out how to get out of the trap.
- If all this works out, they will do a pilot clinical trial on a few patients, moving on to a larger clinical trial if that is successful.
Funding Request
Davis made an unusually explicit request for funding, stating that if there’s a big donor out there, now is the time to give. The rate-limiting steps in all of this are funding and resources. Everyone is spread thin. Julie, their lab ace, is working on seven different projects. Their mass spectrometer guy can devote only 1/7th of his time to this project. They simply need to be able to hire more people to quickly figure out if the metabolic trap hypothesis is right or wrong.
- Support the Metabolic Trap work via the Open Medicine Foundation. (You can direct your donations specifically to the Metabolic Trap work in the comments section.)
- An earlier Health Rising blog on the Metabolic Trap.






Wow, sounds super. Have you any idea of their expected timeline all going well Cort?. We all talk about 5 years or 10 years. Would within 5 years be plausible for treatment? Some of us won’t even last that long.
Jeez, I wouldn’t know but it would seem that if the Trap keeps passing hurdles more and more resources will come its way and treatment possibilities are certainly being thought about.
I a man bedridden, I have air hunger, I am so sick I can’t function mentally or physically. I got sick in 1986 after taking l-tryptophan. When I took I would gets burst of light Indy brain that felt like a lightbulb exploding. I would then fall asleep. That is what I took it for sleep. I started to notice my pupils were huge…ias only 23 at the time. Numerous symptoms came on……I was completely bed ridden within a year. I have never gotten better. I am currently 55. My life has been pure hell, torturous. I want to be used in a study. I am available if a participant is needed. Threasa Melton
dear theresa,
I am so sorry of the depht of your illnes! And I am sorry to that nobody else replied on your cry for help. Are we not a community who help eachother, especially the verry severe ill ones?
I am a severely ill one to. So I can relate to the depht of your illness and feelings.
I wish I could do more then say; hang in there. there will come a solution for us. We “just” need to survive until then. I hope you still have loved ones around you who take care of you and pull you through all the misery. I am since 2010 alone and it is so hard. being locked up in your bodu, in your bed, in yoour home. Not able to stand anyone arouns as if there would be many people after 30 years of illness. And the help here in Belgium is awfull. I hope you can pull yourself through each day after day until there comes a solution. Much strenght.
Absolutely care. Love and positive energy going your way to both of you and all severely ill.
Threasa I read somewhere that enlarged pupils are a symptom of high serotonin. Need to verify this. But it would corroborate your suspicion that that l-tryotophan set you up in a high serotonin state.
Miy sudden onset at 27 years of age came afterTo months of taking high doses of slo niacin. About 4 gr per day slo niacin is nicotinic acid. Not Niacinamide. The night a got sick I took 6 grams! In my ignorance I thought that I would inctease dose by 50 % that day to compensate having eaten a fatty pizza that day. I was given slo niacin to lower moderate high cholesterol. I am 54 now. This day I believe that set the slide into a metabolic trap for me.
Cort,
I am so sorry to here you have been suffering so long with this and I wanted to say to you I think your truly amazing in what you are doing and your style of writing to help us better to understand what the dand what the researcher are doing and thier findings! !ecause of you I have gain so much insight and understanding ?Hang in there and hold on as I see this rollercoast finally going up..blessings to u..?
So very sorry for all your suffering Threasa and Konijn. I hope that things improve for you. Please take comfort in knowing that people do care. I hope treatments and cures will come soon.
thank you Birdie for your compasion and wishes for us!
I’m so sorry … its so sad. you lived this hell, heaven may waiting for you.
I’m really so sorry
– Is the potential treatment of the metabolic trap theory already been tested on a patient? if yes, what is the result ?
– How many metabolic traps have been identified ?
– When we know that we have the defective genes, how to perform the PBMC (peripheral blood mononuclear cell) test to find out whether or not we are currently in the metabolic trap ?
To my knowledge no potential treatments have been tested on a patient. Ron remarked at how fundamental a pathway the trap involves. If the trap is determined to be present and causing ME/CFS then more modeling will be done and animal models will apparently be used to assess the effects of possible treatments.
I only know of the one metabolic trap but references to more potential metabolic traps have been made.
Others would know better but my guess, given the limited amount of samples that have been run, is that the tests to determine tryptophan breakdown and kynurenine production are probably available only to researchers.
No treatment has been tested on a patient yet. See “next steps” in Cort’s article.
They have identified quite a large number of potential traps but more investigation is needed to determine if they fit all the requirements to be traps.
The way to test if someone is in the trap is to check the tryptophan/kynurinine ratio in white blood cells, not plasma. Regular medical labs are not equipped to do this. Scientists at the genome center are working on a simpler and cheaper method for doing this. Right now it requires a $100,000 – $1,000,000 mass spectrometer!
Dear Janet. I find this hypothesis both deeply interesting and extreemly scary. I have severe ME my self and my daughter has the same. We were told by our medical advicer to start to take TRP. We have done so for app. 6 weeks. Stomachpain has become worse and she strives breathing. We will stop the treatment imideately. But I am afraid I have worsened her condition by this experiment. And this breakes my heart. Is there anything I now can do to reduce damage -given the hypothesis fits?
Are there any advice considering diet/other thing even there is no treatment and no ways to prove if levels are to high.
From desperate mom
The United Kingdom Government laboratories [FERA (England) AFBI (Northern Ireland)] carry out Mass Spectrometry monitoring programs on food etc. Prices start at approximately 200 pounds sterling ($250 ish) i.e. for a mass spectrometry test. This price is for the $100,000 mass spectrometer; if you need the $1,000,000 mass spectrometer (I suspect you might) then the cost will be much higher. However, my point is don’t rule out mass spectrometer as a diagnostic test; your Government is already using it to test whether your chicken etc (food) has antibiotics etc (chemical residues).
Here’s another potential intracellular diagnostic test [https://pubs.rsc.org/en/content/articlelanding/2018/an/c8an01437j#!divAbstract]. It measures intracellular phenylalanine, which is also elevated in ME/CFS.
Once we know more then further (cheaper/better) test will I assume be found. But we need to see a relatively affordable test now.
Good points! My guess is that mass spects are going to play more and more of a role in ME/CFS.
I’m assuming if they caution against Tryptophan, they probably caution against 5-HTP as well. So many alternative doctors try to give CFS patients 5-HTP and Tryptophan based on low levels that apparently show up on nuerotransmitter tests. As always….so CONFUSING!
I agree! I don’t understand this either. 5-HTP certainly does work for some people. SSRI’s and SNRI’s do as well. All I know is that it’s a very complicated system….
Methylation pathways pop out ~ I wonder how much they have delved into that.
Ron says they haven’t yet explored the other side of the pathway leading to seratonin. It’s just as complicated as the pathway leading to kynurenine.
What is the best way to test for tryptophan levels? 5-HIAA in blood or urine? How to differentiate endogenous tryptophan levels vs that being produced by gut pathogens?
This research is all based on tests of white blood cells. Not plasma or urine. A regular blood test will not be informative. In fact, sometimes the results are opposite. This reaction occurs IN CELLS.
Aside from minimizing any physical exertion, the only thing I know to feel a bit better is by fasting. Does that fact fit into this hypothesis?
And while we wait for further experiments but with the thought that this could be a credible theory, is there anything an individual might do that could offer a little relief in the meantime?
Forever indebted to those making the effort to help solve this perpetual misery so many are stuck in.
Feeling better from fasting is possibly relevant, but not understood so far.
Janet, it warms our hearts that you and Ron are active on these research article discussions. We’re all hanging in there, cheering you guys on! Thank you for your tireless activism on the behalf of those of us not able to do so in person. Thank you Ron for pushing ahead with your solution oriented research!
Fasting would make one feel better as we are making sure foods which deliver tryptophan are halted. I would imagine that good which give tryptophan would make us feel worse??
I find intermittent fast to be very helpful. From research I’ve done – there can be several reasons for this helping. It ups BDNF (better brain function) in the brain, can help glutamate to go down pathway to turn it into GABA (calming) instead of being excitatory. Assist in brain detox. So many positive reasons why it may help. It also has greatly helped balance my blood sugar. I don’t have any issues with a 16 hour fast. If seratonin is too high – fasting helps increase dopamine which would balance things more. Fasting has been shown to help not only diabetes, but also other neurlogical conditions.
Hi Greg,
not commenting here on the validity of the trap hypothesis, but on https://en.wikipedia.org/wiki/L-tryptophan I did found:
“A common assertion in the US is that heavy consumption of turkey meat results in drowsiness, due to high levels of tryptophan contained in turkey.[18][20] However, the amount of tryptophan in turkey is comparable to that contained in other meats.[19][21] Drowsiness after eating may be caused by other foods eaten with the turkey, particularly carbohydrates.[52] Ingestion of a meal rich in carbohydrates triggers the release of insulin.[53][54][55][56] Insulin in turn stimulates the uptake of large neutral branched-chain amino acids (BCAA), but not tryptophan, into muscle, increasing the ratio of tryptophan to BCAA in the blood stream. The resulting increased tryptophan ratio reduces competition at the large neutral amino acid transporter (which transports both BCAA and aromatic amino acids), resulting in more uptake of tryptophan across the blood–brain barrier into the cerebrospinal fluid (CSF).[57][58][56] Once in the CSF, tryptophan is converted into serotonin in the raphe nuclei by the normal enzymatic pathway.[59][54] The resultant serotonin is further metabolised into melatonin by the pineal gland.[7] Hence, these data suggest that “feast-induced drowsiness”—or postprandial somnolence—may be the result of a heavy meal rich in carbohydrates, which indirectly increases the production of melatonin in the brain, and thereby promotes sleep.”
Basically: eating varied protein plus *too* large amounts of carbs results in high insulin. High insulin pushes amino acids that can be converted in glucose into the cells IMO, increasing the ratio of “glucose cleavable” amino acids to “non-glucose cleavable” amino acids, with tryptophan IMO the latter.
Note: I am not against “normal” carb ratios in diet IF it is in a diet with restricted caloric intake and is such that it prevents high combined (fructose + glucose) blood sugar peaks. Then it could provide even anti inflammatory benefits IMO.
The above mechanism could also be involved in PEM.
Exercise and even more so anaerobic exercise uses alternative sources of glucose too. This means that after exertion the ratio of “glucose cleavable” amino acids to “non-glucose cleavable” (including tryptophan) ones should be reduced and remain so till after the next meal.
IIRC ME patients rely more on proteins for providing glucose than healthy persons. If so, that should compound potential problems.
Having poor availability to “glucose generating proteins” for many hours may reduce our metabolism for a prolonged time, potentially being an important phase in PEM.
Personally, for all of my life including the many pre ME years I remember that doing activities after a meal impacted me a lot more then most others of my age.
An earlier article here pointed me to http://www.drcourtneycraig.com/blog/intermittentfasting , which describes how fasting may help.
Basically you start with a low-carb diet and after a while you add intermittent fasting once or twice a week. Among other effects, you’ll have ketosis during the fasting, which reduces neuroinflammation (I’ve read an article saying that neuroinflammation in lab mice disappeared when their ketones increased, but sadly I didn’t bookmark it).
Now, Google says kynurenine is a ketone, so if ketones are important for keeping neuroinflammation at bay then it makes sense that being low on kynurenine causes neuroinflammation.
I hope to try this eventually.
Thank you so much Cort for this update on the Metabolic Trap! Fantastic content as always.
I have to say I find it interesting about the perceived overlap with Cortene’s hypothesis regarding the over-activated serotonin system…
Me too. Never thought about serotonin pathways until this year! I remember Ron saying to keep our minds open – we really don’t know where the answer will lie. These two groups have demonstrated that viable answers may lie in systems we have hardly thought about. That’s kind of exciting…
Cort,
Thank you so much for sharing this!
I hope Dr. Phair’s metabolic trap hypothesis “opens up a can of worms.” I say this literally. I believe the gene mutation that Dr, Phair discovered may be caused by an active pathogen(s) in ME patients. Not necessarily a worm ?, however; isn’t Dr. Davis exploring a hypothesis about a trypanosome that causes African Sleeping Sickness and a potential mimicry ME pathogen that may yet be discovered? I’m curious as to what the role is that L-Tryptophan metabolism plays in Trypanosomiasis and other pathogenic infections? Also, what is the role that these IDO enzyme gene mutations play in ME cluster outbreak patients?
Perhaps these two researchers can collaborate and expand their hypotheses and clinical research by studying some of the survivors of the most recent ME cluster outbreaks ?? Compare and contrast the suspected etiologies and if they coexist with gene mutations? Sort of reexamine the pathogen “hit and run” hypothesis. ?
The pathogenic “hit and run” hypothesis needs to be challenged. Why have so many scientists/doctors/researchers concluded that the pathogens that may have caused ME/CFS be gone? Could they still be in us? Could the gene mutation Dr Phair discovered be reversed if the pathogen(s) that caused/or is causing his discovered gene mutation be destroyed or suppressed? How many patients studied by Dr Phair had previous exposure to herpesviruses versus enteroviruses versus bacterial/fungal/parasitical pathogens versus no exposure to a pathogen whatsoever during onset of illness? Please compare their IDO gene murations. Food for thought when it comes to the metabolic trap ???high serotonin in the brain hypothesis. Is tryptophan a “food” for active pathogens and are ME patients neurons keeping serotonin ? up in the ? as a sort of defense mechanism? What happens to people who take SNRIs? Reuptake inhibition right? Are SNRIs good or bad for us? I guarantee you that a lot of doctors are prescribing SNRIs for ME/CFS patients as we speak. If that isn’t the ultimate experiment in serotonin regulation then I don’t know what else is. I’d be more worried about doctors experimenting on patients more than patients self experimenting on themselves. ?. Thank you again for posting this article Cort and to the researchers like Dr Davis and Dr Phair and Dr Prusty who are all getting closer and closer to finding potential answers for us ALL!
So much to think about. One thing it makes me wonder about is the role that epigenetic modifications might play in some patients (???) I don’t know if that’s a possibility or not.
“Epigenetic modifications.” You summed up in two words what I was trying to suggest in 1000! This is another reason why I like you so much Cort. You are concise in your responses yet you still capture and highlight the detail perfectly! Your articles and feedback are exceptionally written and greatly appreciated, ?? equal to that of the research itself that is being conducted. ?
I’m afraid, to be honest, that it took just a couple of minutes in the Workshop listening to the likes of Robert Phair, Bob Naviaux, Neil McGregor and others for my eyes to start to glaze over. It is truly a humbling experience. My goal is simply to capture the main points! Thanks, though, for your nice words 🙂
Ron says: We are exploring trypanosomes and all known parasites because we are looking at any possible cause for this disease.
The IDO2 mutations we are looking at are not caused by pathogens. They are inherited.
Read in Cort’s article about why the outbreaks imply that the mutations have to be relatively common.
We have the hit and run model because the organisms that are hypothesized to cause CFS have not been found to be present. See Ron’s talk and Wenzhong’s talk at the symposium. More studies are being done looking for RNA viruses or other pathogens.
Have you determined a connection between inherited IDO mutations and HLA-DR mutations? I saw a few articles on it being found together. If that’s true – possibly mold (CIRS) and other fungus could be at play. With me, I have the worst genetics and inability to throw off mold and chemicals. Extremely sensitive and aware when in wrong environment.
I also saw articles indicating IDO mutations can be connected to tumors. Dr. Fry Lab found a type mold/fungus in a thyroid biopsy of mine. This fungus is known to cause tumors. Having positive markers for CIRS. Would be interesting to connect a IDO mutation here and confirming this as a contributor to tumors and possibly genetic. However, if this is true – there appears to be a fine line as to activating the immune system and this gene. Could backfire. (Cancer connections.)
Since this also had connections to kynurenine pathway – would explain my own hypothesis in connection to this pathway and faulty conversion with glutamate and it going down proper pathways. Also causing too high seratonin and too low dopamine.
Even though I wasn’t found to have antibodies in calcium channels – I do have mutations in TRMP3. Treating my MCAS and POTS with mild calcium channel blocker meds – helpful.
Another possible pathogen is Lyme and it’s co-infections.
Addressing CIRS, MARCONS and Lyme has been my best results to date.
Hi Issie,
I’ll try to give it an initial shot:
* Let me start with: for IDO, I consider 40% rate of mutation not to be defects; if they were only disadvantageous there number would be reduced by selective survival chances. (Nice idea of Robert Phair to think that way BTW!)
* This mutation plays a role in gut motility and has a link to fructose malabsorption (more on that in another coming comment).
* With my likely fructose malabsorbtion goes hand in hand (intermittent) diarrhea and frequent blood in the stool.
* I had a clear improvement when I treated potential “undigested protein” in the bloodstream with eating papaya with each protein rich meal in order to try and reduce amount of undigested protein in the blood. Results: reduction from *very* frequent aphthuous ulcers (immune response to protein) to normal rates and remarkably better health.
* The idea here seems to be: when eating fructose rich food I get a lot more frequent blood in stool; somehow it causes “holes” in my gut (or stomach, heliobacter pylori?) that allows undigested protein and other garbage into the bloodstream.
* Last piece from https://en.wikipedia.org/wiki/HLA-DR: “The primary function of HLA-DR is to present peptide antigens, potentially foreign in origin”
It’s still weak, but IDO mutations may go hand in hand with increased chances for leaky gut symptoms, leading to undigested proteins in the blood. Those are seen as a threat by the immune system. HLA-DR mutations may allow to better deal with that threat. Both might go hand in hand because at 40% some IDO mutations might be useful but at a cost that must be compensated (potentially by having simultaneously a HLA-DR mutation modifying reaction against undigested protein).
Long shot but you might be able to use it if it were to hold any value.
Dr Dafoe/Dr. Davis,
Thank you for taking the time to respond and clarify. I haven’t found Dr. Wnnzhong Xiao’s presentation on the Open Symposium link yet, however; I did watch your husband’s presentation in great detail. ?. Without going into too much depth, we are lucky to have both of you and the remarkable minds and perspectives that go with it.
I do have an important follow up to your response. I was prescribed two different SNRIs (Cymbalta/Savella). Both SNRIs triggered a rare side effect to occur. As a result of this side effect, a pathogen was identified, streptococcus. The pathogen could only be detected via biomarker when I was actively taking the SNRIs. As soon as I stopped taking the medication, the side effect would disappear and the strep could no longer be detected. Something bizarre occurred along my brain-gut-immune system axis when taking the SNRIs. I believe that this took place as a result of serotonin intake inhibition along neural/gut/GI pathways. In my layman opinion, when the release of seratonin from my cells traveled along these pathways, a biosynthesis of serotonin may have caused a pathogenic awakening. How do I know this? I don’t. This is pure intuition as a result of self-observation of a personal experience and a hypothetical conclusion based on three positve results for strep everytime I exhibited the side effect from the SNRIs. I hate “cherry picking” data in the way Dr Davis warns researchers not to do. I’m not attempting to self-diagnose. I am not attempting to acquire special consult. I am attempting to share a personal and very private observation in an effort to promote further discussion and awareness. Maybe someone out there has experienced the same thing I have. Maybe not. All I know is that we are lucky to have people like you both in our corner. And every single time I find myself feeling sorry or not having enough patience to wait on the experts to figure things out, I think of Whitney Dafoe and Jamison Hill and put things into a much more mature perspective. I am reminded. I am humbled. I am guided. I am inspired!
And prion (proteinaceous infectious particle)?
I don’t claim to understand but there does seem to be some link between IDO2 variants and human African trypanosomiasis- I presume this means having variants would make you more susceptible to the sleeping sickness reaction to the parasite? An epigenetic type of thing?
http://pathcards.genecards.org/Card/african_trypanosomiasis?queryString=IDO2%20
Though it might mean that trypanosomiasis impacts IDO2 function? there’s some relationship between the kynurenine pathway and trypanosomiasis ( https://www.ncbi.nlm.nih.gov/pubmed/28013248 ) so these ideas perhaps aren’t entirely separate?
Just speculation.
Actually numerous organisms have been found to cause a CFS like syndrome, including C. pneumoniae, Coxiella burnetii, and Giardia lamblia. There has never been a credible hypothesis for a living organism as the cause of Ramsay/Dowsett ME.
ME is hypothesized to be caused by a virus, not an organism. And it’s always been hypothesized to be caused by the RNA virus enterovirus, which you haven’t even looked for yet. So I’m not sure how you can state something that you haven’t even looked for hasn’t been found.
Enterovirus is the most replicated pathogen finding in ME research, so if you’re speaking in general about them not being found, you are dead wrong.
https://me-pedia.org/wiki/List_of_enterovirus_infection_studies
I started taking tryotopan 4 weeks ago. I have felt more pain in my stomach, but nothing else.
I will stop imediately, but now I am deeply afraid that I have made my condition very much worse? And maybe for ever? I am very afraid now.
My guess is that the main issue for all of us is whether the trap is present or not. If it’s present then I suppose – speaking as a laymen – it’s possible that you could have juiced the trap a bit but to me the presence of the trap is the main thing. If you’re in the trap you’re stuck and the goal then is to find a get out of the trap. Phair’s models suggest that if you’re in the trap even for a little while it’s very hard to get out of.
Personally I wouldn’t think that giving more fuel, if that’s what’s happening, to the trap really makes much of a difference. The trap is the issue – not the substances going into the trap. If Phair’s models are correct the trap is so stuck that if they can find a way to get anyone out of the trap then I imagine it will work on everyone…
In short I wouldn’t worry. I think most of us have tinkered around with this system using supplements. Ron’s group is already actively checking out the treatment end.
Thank you Cort!
Besides being the most updated, pedagogic person you also function as a psychologist. You’re the best ?❤???
Oh jeez… I’ve been taking tryptophan and 5-HTP every night to help with sleep. Maybe that’s why I’m going downhill faster than my friends with the illness.
Is melatonin safe to take?
Also I’m taking the highest dose of an SSRI. I’m gonna start today to lower it and hope all goes well.
This is so worrying. The very things I’m doing to try to survive might be making me worse.
These questions bring up a big problem! We have a limited number of samples and don’t know if the trap is present or not. As Phair stated more tests need to be run to determine if it is present.
Whether or not to change one’s treatment regimen based on a promising hypothesis which nevertheless still needs quite a bit more testing is a good question to ask. Particularly with regard to drugs like SSRI’s which may be helpful and which can produce withdrawal problems. If you’re going to drop the dose down be sure to check with your doctor about the best way to do that.
When is the right time to prudently act on this information? I don’t know but Ron emphasized this is a very tricky area to play around with.
Ron thinks Cort is giving good responses to these questions.
Some of us have depression as well as ME/CFS, right? So they must have to be very careful about changing doses of antidepressants.
But I remember Dr. Shoemaker saying that for people who have what he calls chronic inflammatory response syndrome, taking SSRIs would be like pouring gasoline on a fire.
“If you’re going to drop the dose down be sure to check with your doctor about the best way to do that.”
I totally agree with Cort on this one. I can cut off one SNRI at will without withdrawal symptoms, but when withdrawing from another medicine I thought for the first half hour of the day I could fly.
I had the believe that “floating” down the stairs would be easier then walking down the stairs. Luckily, I had still a faint idea that somehow it were wrong. Even better, I was too tired to get out of bed in half an hour after waking ;-). Trying out that floating thing might have proven destructive.
The hallucinations disappeared when I realized it was related to my sudden stop of taking this drug so I went back to a normal dose and build down gradually.
Emily do not take melatoning, make you body produce it natually. You do this by getting AM sunlight, then blocking out all artificial blue and gree light at night. See Dr Jack Kruse work.
If I reember well, listening to the conference, or Dr Phair, or Dr Davis (allthough verry excited about the metabolic trap)said at the end that all the other researchers on the congres who spoke, could be right (in finding the cause). I am severely ill,I hope Dr Phair is right and we have “a quick fix” but I do not forget what they said at the end of the conference. All the researchers can be right…
So it is waiting again…after decades and not knowing how long to survive anymore.
Yes, it’s good to point that out. Phair openly said someone else may be right. The Trap – while exciting – remains a hypothesis – they are very clear about this. I think it’s important to point that out so as not to assign blame or be upset at anyone if it doesn’t work out.
On the other hand, Ron Davis and Phair are clearly excited about the progress the hypothesis has made. They feel it’s made enough progress to be released into the world – and to ask for more funding.
My guess is that a testable hypothesis has appeared more quickly than Davis might have expected and that’s good news.
we have to be carefull with our harts and our hopes. Ofcourse I hope to, verry much, but I do not want to be first totally excited and convinced and then hartbroken again. And the “stress” it brings with it.
Maybe indeed a testable hypothesis has apeared more quickly than Dr. Davis has expected, who knows? And it is good that their hypothesis has made enough progress to be released into the world and ask for more funding.
But the research from other scientists also asks for more funding.Alls researchers who spoke at the conference and others who are working worldwide to solve this verry verry complex illnes.
konijin and Thresea my heart goes out to you❤️️
thank you Peach!
Neuroinflammation II: the Kynurenine Pathway in Fibromalgia and ME/CFS – Health Rising
https://www.healthrising.org/blog/2015/06/28/neuroinflammation-ii-the-kynurenine-pathway-in-fibromalgia-and-mecfs/
I’ve been talking about thinking this was a part of the picture. Good to hear it’s being looked into.
If they would look into calcium channels too – may be enlightening.
Building on the work being done by Sonya Marshall Gradisnik and Don Staines at Australia’s Griffith University?
https://www.meaction.net/2017/03/16/calcium-channel-ion-defects-research-from-australias-griffith-uni/
Ron says: We do not find that those mutations that are reported from Griffiths come out as significant in our studies (see Wenzhong’s SIPs study slide on mutations from the symposium). Furthermore, those mutations appear to be common in the European population of healthy people.
A lot of info on this post on kynurenine pathway and it’s connection to a lot of possible connections/symptoms we may experience.
https://www.healthrising.org/blog/2015/06/28/neuroinflammation-ii-the-kynurenine-pathway-in-fibromalgia-and-mecfs/
Again here’s link
“but in the presence of inflammation, whether in the body or the brain, things change.”
Makes sense:
https://en.wikipedia.org/wiki/Dioxygenase#Heme-containing_dioxygenases:
The IDO mechanism needs Fe2+ as a key reagent. That very likely does make the reaction speed of the tryptophan to kynurenine reaction quite depended on oxidative stress conditions
https://en.wikipedia.org/wiki/L-tryptophan
“Like other amino acids, tryptophan is a zwitterion at physiological pH…”
Blood pH likely plays a role in how fast the chemical reaction occurs, and we seem to have changed blood pH. The impact of this one may however be far smaller then of oxidative stress due to the Fe2+ dependency.
Wondering that it means that my preference for ham (sodium needs for dysautonomia) shifted some years ago to turkey…?
Turkey does not have high levels of tryptophan. People get sleepy after Thanksgiving because they ate a big meal! LOL
Ham can contain high levels of nitrites and nitrates and these can mess with your hemoglobin (blood) and myoglobin (muscles).
From the pt perspective, the hypothesis rings a lot of bells. I know of many examples where SSRIs made this condition much worse instead of better. This includes my teen-aged son whose paed started him on a dose of SSRI (not for depression mind, but to settle his CNS down). Over the next six months, he went dramatically down hill, his personality changed and he was eventually hospitalized for suicidal ideation. It wasn’t a standard adverse reaction, classsic allergy, or seratonin syndrome. He didn’t have a mental illness and after he was weaned off it, felt mentally much better (but took 12 months to lose the shadow of that trauma.) His fatigue and cognitive fog never returned to pre-SSRI levels. This proposal that tryptophan can be high or low perhaps reflects the contrary finding that some find great relief from SSRIs. Above all, it’s a great reminder tread very carefully on this seratonin pathway. Shelley.
Thanks for bringing this to us, Cort, in such detail as usual. I believe I have always had high serotonin levels as I don’t tolerate antidepressants and I have always had runny bowels. Perhaps the tryptophan metabolising polymorphisms are the body’s way of trying to stop the serotonin level getting too high with the unfortunate side effect of a diminished kynurenine pathway. The question is what caused the high level of serotonin in the first place. I agree that it is linked to high cortisol. I myself took tryptophan almost 20 years ago. I only took it for a while as there was no improvement but I didn’t seem to get worse at the time. My husband didn’t like me taking it as he said it smelt very bad! Maybe it did have some effect because I continue to get gradually worse over the years.
Sorry, I meant to say the high serotonin level is linked with low levels of cortisol, not high.
Thanks again for the clear way you present information to the brain-fogged Cort! Just of interest-I quickly developed ME|CFS after taking a course of Prednisone for Inflammatory Bowel Disorder. Still sick after 17 years, but have never had Inflammatory Bowel Disorder in that time. Does Prednisone have the potential to affect or reset, tryptophan or the kyurenine pathway?
If this hypothesis is correct, are there implications for patients taking SSRIs and SNRIs? They are so commonly prescribed, especially for those with ME/CFS, that I imagine it will be a question on many people’s minds?
Around a year ago in New Zealand the government drug buying agency changed the brand of venlafaxine (a common SNRI) to save money and performed an interesting experiment on the 45,000 unsuspecting citizens taking this drug. I’ve been keeping an eye on the growing number of reports of adverse affects and found several of the symptoms overlap with ME (fatigue, dizziness, brain fog, joint and muscle pain etc) – which made me wonder – how many of my symptoms were ME and how many were SNRI related?
Fascinatingly when I binned the government funded drugs and bought the old brand out of pocket by POTS improved SIGNIFICANTLY to the point I think it might be gone! As we all know – very hard to tell if the drug change caused that or something else entirely. However it does make me wonder…?
Ideally I’d like to wean off SNRIs to see if my ME improves, however realistically I don’t think I have the spoons for that right now. In any case I look forward to greedily reading any further information that comes out on this topic!
Very compelling info, Cort! If the serotonin and/or tryptophan levels are out of whack, it might help explain Dr. David Bell’s “Tired but Wired” symptoms of ME/CFS where you’re totally exhausted but can’t sleep or shut of the mind games.
I have a hard time with anything artificially sweetened with Aspartame (which breaks down to tryptophan.) Does 23andMe or any of the other genetic sequencing companies test for these mutations? Sure would be interesting if this turns out to be true.
Do you know how long it will be before we have another update from Ron or Robert? It would be also really useful to have an idea of the alternative meds that could be implicated in this pathway. Thanks to Ron and Robert for their work – I’m UK and I watch and listen each day with eagerness and hope that one day I may be better.
I don’t know. It could depend, come to think of it, on how they do with funding. More funding = more testing the hypothesis = more quickly the work goes = the more quickly we know if the trap is doing what Phair thinks it might be doing.
You can watch Dr. Phair’s IDO Metabolic trap presentation at the Stanford ME/CFS symposium via the live stream replay: https://livestream.com/accounts/1973198/ME-CFS-2018/videos/180981460
6h42m mark.
I started taking tryptopan 3 weeks ago after having it recommended from.a doctor.
My stomachpain is so much worse and keeps me up at night and my burning pain is worse. I was told it was completely harmless. Can I have wosened my ME tremendously by these 3 weeks of “treatment”.
Severly worried now ??
If anyone knows if this can having caused my little function irreversible damage or if there is anything I can do (except throwing this shit away) I would be very pleased to know ❤?
I can see you answered this over! Thank you ?
If you got runny bowels lately then your body disposed of most of the extra tryptophan content by itself at your discomfort.
Extra tryptophan means likely increased serotonin means increased bowel movements means more tryptophan disposed down the toilet.
Just stopping taking it if it makes you sick makes sense IMO. Give it a month of moderate food intake (see comment to Greg: eating too heavy meals likely makes it worse) and it should disappear by itself IMO.
Eating no more (but no less either) then needed has plenty of good health effects BTW including being anti inflammatory.
Chris, excellent insight with your observations that parasites like trypanosomes (flagella) feed on tryptophan r their survival n reproduction.
It makes complete answer that these parasites infect peopl, turn on a genetic switch for excessive tryptophan production and simultaneously may downregualte IDO which has been shown in labs to dramatically increase the number and density of trypanosome parasites.
African species of trypanosomes cause ‘Sleeping sickness’ – which is exactly how ME/CFS appear, to be always sleepy and tired needing rest. There must be other species of trypanosomes that have not been discovered and are causing ME/CFS onset and potentially chronic stealth infection. Chagas is a trypanosome parasite that has now spread globally. If infects the guy in chronic conditions when untreated and can infect the brain. All of which mimics many symptoms ME/CFS sufferers have in the gut and brain.
The researchers need to find these stealth parasites that are causing excess tryptophan for their lifecycle. It makes complete sense. There may also be helminth worms that create the same dynamic of excess tryptophan production, many people who take ivermectin with ME/CFS / Fibromyalgia find a lot of relief and reduction in many symptoms.
Which points to the anecdotal evidence that stealth parasites of many kinds are involved in chronic ME/CFS and just the same way toxoplasma gondii or borreliosis hijack molecular pathways to produce excess amounts of what they need – whatever parasites require tryptophan will be implicated in ME/CFS patients.
This needs a lot more study!
I’ve written a blog post about this too https://tipsforme.wordpress.com/2018/10/13/understanding-the-ido-metabolic-trap-hypothesis/
When I was searching for info to write it a curious detail I came across is a link between African trypanosomiasis (sleeping sickness) and IDO2 and IDO1. Sleeping sickness is something Ron Davis has mentioned in a different context. He said the gene expression of pwme is very similar to gene expression when people have trypanosome parasites.
It’s not something I claim to understand but has anyone else suggested a connection between IDO2, trypanosomiasis, Suramin and Cell Danger Response?
Oh boy! This is a little scary… about a year prior to and immediately before and during the period I was developing M.E I was in a relationship with a guy from South Africa who had only recently moved here…. makes me wonder now…
Relation IDO to IBS, fructose malabsorption, oxidative stress and cancer.
* At 40% of global population, these mutations are not rare at all. IMO this means that they do provide some advantages as well as disadvantages for not their occurrence would be reduced a lot by evolution. Potential side effects:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4266036/: “Indolamine dioxygenase (IDO) is the key enzyme involved in the conversion of tryptophan in the intestine. The enzyme catalyses oxidation of tryptophan to kynurenine in a reaction that produces peroxide and gives rise to highly reactive and potentially harmful oxygen and hydroxyl radicals. The IDO reaction may therefore contribute to what is generally called oxidative stress”
and https://en.wikipedia.org/wiki/Indoleamine_2,3-dioxygenase “A wide range of human cancers such as prostatic, colorectal, pancreatic, cervical, gastric, ovarian, head, lung, etc. overexpress human IDO (hIDO).”
-> So having mutations that reduce IDO effectivity may reduce cancer incidence and increase longevity.
-> In people with other “high oxidative stress genes” this additional IDO mutations may compensate as these genes reduce oxidative stress. As such, epigenetic effects of too high oxidative stress may increase the expression of these mutations. In other words: reducing oxidative stress may be double important in ME patients with these mutations.
* Fructose malabsorption has potentially a double protective effect against too high tryptophan levels:
https://link.springer.com/referenceworkentry/10.1007%2F978-3-540-29676-8_1787 “Abnormal tryptophan availability can be seen in subjects with fructose malabsorption… …subjects with fructose malabsorption have a reduced intestinal absorption of tryptophan due to chemical affinity between tryptophan and high intestinal fructose concentrations”
-> eating fructose removes tryptophan.
-> the other way around: eating tryptophan reduces fructose uptake as this chemical affinity works two way. That may be interesting as (see comment to Greg) simultaneous high uptake of tryptophan and sugar leads to high insulin worsening the problems. I do know that insulin reacts against too high levels of glucose, but both sugars are often found in the same food and both contribute to glycogen and fatty acid construction out of carbs so reducing fructose intake has a longer term effect on reducing insulin levels. There are plenty of scientific links between high fructose intake and insulin resistance.
* If there is plenty of fructose left in the gut (due to fructose malabsorption), then there is plenty “fast fuel” for gut bacteria. IMO it allows some strain of bacteria to grow at an explosive rate causing gut disbalance and poor diversity. Ideal conditions for IBS / SIBO IMO. The idea here is: fructose is just as much a “far quicker” fuel then for example (resistant) starch for bacteria as it is for us humans.
I wonder if it could help solve my prolonged oscillating between loose stool / constipation mystery after eating fruit. Likely having an unconfirmed fructose malabsorption the loose stool thing seems reasonable to me.
But having it so often swing from the one to the other as it has been for so much of my life is more difficult to understand.
So far I observed that eating too much (not even that much…) fruit at once and nothing beside it induces first a strong onset of diarrhea. About 3 days later I get few, hard and slow pellet like stool for some days. Often this comes with additional about 2 weeks of blood in the stool (or more if I keep up the for me bad diet). The swings between both stop after over a month only if I remedy my diet.
Could it be that:
* Eating fructose causes an explosion in bad gut bacteria as discribed above.
* That is flushed by strong bowel movements in the gut.
* Bowel movements are strongly increased by serotonin. Having serotonin modulated strong bowel movements should cost a lot of serotonin reducing the stock left in the bowel. Note that the bulk of serotonin is stored in the gut.
* IF the reaction would consume much of the serotonin stock in the gut during the 2 to 3 days of diarrhea AND IF rebuilding it would be slow, then the gut would have no “capacity” to induce diarrhea the coming few days or even have rather slow motility.
* As I have written at other places: poor blood flow in the capillaries very likely increases receptors for signal molecules a lot near those small capillaries. This should cause strongly overreacting responses to them with changing blood flow. Diarrhea would induce dehydration reducing our already reduced blood flow a lot.
* The 2 above could lead to a combination of reduced seratonin per ml/blood coupled with a strong reduction in blood flow in the gut capillaries (blood flow in capillaries is disproportional effected with reduced blood volumes). That combined would restrict bowel movements a lot.
* During this period of constipation, few water is excreted and more food (and tryptophan) becomes available in the gut again, potentially leading to the reverse of the above. If buildup is sufficient of both diarrhea could be quickly reached due to an overreaction of both continuing the cycle.
This large swinging is not that common with IBS, but it is consistent with “classical” system control theory in the sense that reduced blood flow leads to higher amplification constants leads to less system stability leads to more oscillation.
For those with diarrhea only IBS, I believe it is common to swing a lot between near normal and very plentiful bowel movements too. Duno if that is correct.
Since increased oxidative stress has always been found in ME/CFS that oxidative stress finding is an intriguing one!
If the IDO mutations were a mechanism in place to compensate for an increased amount of other pro oxidative stress genes then it would:
* Make people with those other pro oxidative stress genes more vulnerable to all kinds of biological stress going hand in hand with increased oxidative stress.
* Extra activation of this anti-oxidative mechanism due to ME would shift patients more towards faster bowel movements once ME starts. I think on average this does indeed happen.
So far it goes with what we observe.
But if increased oxidative stress would increase IDO gene expression then it also would reduce kynurenine in blood and with it oxidative stress.
The latter is a negative feedback mechanism and should help unlock the stuck state.
Combined with the finding that there are plenty of scientific links on the net showing a clear correlation between oxidative stress (and aging) and higher kyrunenine levels my gut feeling says:
If the metabolic trap is there for real, it may be a curing mechanism rather then a mechanism making us sick:
* It has a strong anti-oxidative stress effect and it may well kick in very viciously when oxidative stress is excessive.
* It seems to kick in such a vast way making itself stuck in its ?oxidative stress fighting action?
* Despite this stuck strong kyrunenine lowering / oxidative stress decreasing reaction it seems that oxidative stress in ME is still rampant up to a ridiculous level. Cell Defence Reaction found in ME by Naviaux and Davis does produces copious amounts of hydro peroxide.
* Low IDO should reduce bowel inflammation a lot IMO (see quoted source) yet many of us have rampant bowel inflammation so something stronger is “overpowering” the potential healing IDO mechanism.
=> As an engineer and an intuitive thinking person I tend to say: the trap is likely real and we see a strong correlation between the trap an ME rather then causation. It seems to be the tripped fuse/alarm activating a fierce likely healing emergency mechanism. I’d rather suspect an “oxidative stress” trap / runaway mechanism behind it.
Wow. This makes sense. Something causing massive oxidative stress and we go into a cell danger response. I have massively high 5hiaa and severe ribofavin deficiency. Also deficient in b3, b1 and b6 and glutathione. Sometimes I wonder if it’s a carnitine or riboflavin deficiency affecting FAD enzymes Many me/cfers have low iron too. And riboflavin needed for iron status. Do you think iron being a co factor for IDO could help? Do you know of any other IDO cofactors? Maybe I have this backwards…
I wonder whether Ron and Robert think the metabolic trap is operating in less severe cases of ME/CFS or whether this only happens in the severely ill subset. Might it apply to more ambiguous cases of ME/CFS or to patients floating between ME/CFS and fibromyalgia?
I also still wonder about the debates over viruses. I was watching a recent Montoya lecture and he still seems to think that HHV-6 plays a major role in a subset of cases (Cort also recently mentioned unpublished Klimas data indicating this). But I would think that Ron and others would have found evidence of this and I imagine he and Montoya bump into each other on Stanford’s campus now and then. Do they have a fundamental difference or this or is it a subset issue?
The question regarding less severe cases can be extended to: if it is a bi-state trap, what with the many of us that have been at different levels of disease at different instances of time?
I am of the progressive onset rather then sudden onset subgroup. I had many distinct levels of “stuck semi-stable health” until something happened good or bad that brought me to a better or worse “semi-stable level of health”. It’s only recently that I managed to break out of it and have a semi-continuous slow increase in health.
Something similar seems to happen with many sudden onset patients: they are within a relatively fixed range of health with PEM superimposed until something happens and this level shifts to another state for quite a long time. That shifting happens from time to time but it often has a “distinctive” character.
I’ve been wondering about the same thing. I can imagine three ways that the trap hypothesis could handle this: 1) more cells in the affected parts of the brain enter the trap; 2) cells in other parts of the brain enter the trap for the first time; 3) IDO2 isn’t the only trap. In any case, at least one trap must be really stubborn.
My understanding is that evidence for genetic origins the trap comes from both the very severely ill and from some less ill people. Determining its provenance in the less severely ill will obviously be quite important.
I am still totally confused about the viral angle as well. Ron Davis is still searching for viruses but as of yet has found nothing. Nor have some other groups yet some still believe that herpesviruses or others may play a major role in ME/cFS. For me, it’s still hard to give up on that idea – time will tell.
Don’t be confused. He started looking for the wrong viruses (DNA viruses) in the wrong place (blood). That’s why nothing has been found.
Cort– I don’t know what sort of technique Ron Davis is using to search for viruses. I wonder what he would make of Bhupesh Prusty’s recent Nature paper on RNA biomarkers for HHV-6 reactivation (with negative PCR) and whether his technique would pick up something like this.
Weyland– Can you point me to a paper or some evidence that’s convinced you the cause is an enterovirus?
JC,
The most solid evidence comes from 3 post mortem studies showing the presence of enterovirus in the brains of ME patients at time of death. There’s dozens of other studies providing support for this hypothesis. I’d recommend this MEpedia page if you want to dig into the evidence:
https://me-pedia.org/wiki/List_of_enterovirus_infection_studies
An Organic Acid Test measures neurotransmitter metabolites such as Tryptophan metabolites (5-Hydroxyindoleacetic) i.e. 5-HIAA from serotonin, Quinolinic and Kynurenic Acid and also Dopamine Metabolites. I guess if your levels are within range, you are safe ?
Urine levels would be reflective of plasma levels of these biomarkers. Dr. Phair’s study looked at intracelluar levels inside Peripheral blood mononuclear cells (PBMC).
It would be good to know of any association between cell and plasma levels.
My understanding is that Phair is, as stated, focused on the intracellular concentrations of these substances. Whether increased intracellular concentrations should show up in some way in the plasma seems a bit controversial.
Seeing that serotonin can be an issue I wanted to add my experience. About 10 years ago my Homeopath suggested 5-HTP for sleep. I took 50 mg. a night for about 6 months and when she tested my serotonin levels….yikes! They were 6 TIMES the highest range number. So, needless to say we tapered and stopped it and thankfully, I didn’t seem the worse for wear.
The article is fascinating and as always Cort, so well written and understandable, even for a lay person.
I just watched Forgotten Plague again last night on Amazon (for the umpteenth time) because I’m going to be giving my new doctor a copy of the DVD. Amazingly we have a new Osteopath in our small Texas town of Winnsboro, Dr. Janett Phelps D.O. She’s an alumni of Nova Southeastern where Dr. Nancy Klimas is doing her research. Dr. Phelps graduated in 2009, a few years before Dr. Klimas started her lab and got her crew of scientists together with their awesome 10,000 computers, but I’m hoping that I’ll finally have a decent doctor who will at least be empathetic and interested in the latest and greatest regarding ME/CFS.
Watching the DVD and knowing how much worse Jamison Hill’s heath is (I also follow his blog), made me wonder about Whitney and how he is doing? Maybe Janet could share an update? I know it’s such a race against time and I can imagine how very difficult it is for his family.
Dr. Janet Dafoe regularly updates about Whitney on this Phoenix Rising forum thread
https://forums.phoenixrising.me/index.php?threads/whitney-updates.47052/page-24#post-1006854
Thanks Tally. I signed up for the site. NOW I realize where all the ME/CFS patients are ‘hanging out’, other than Cort’s site. I see there are over 25 thousand members. It will take me a good long time to check out the forums.
Hi Stephanie, please let me or us know how Dr. Phelps turns out. It’s encouraging to hear about new practitioners. I want to find as many as possible and possibly interview them 🙂
I certainly will. My appointment with Dr. Phelps is Nov. 19th. I spent over a week….s-l-o-w-l-y….getting many of my previous tests copied, a cover letter with a brief bio and my ME/CFS history, list of meds, supplements,’typical day’ scenario, etc. and included the Forgotten Plague DVD and a few of your articles, especially the ones featuring Nancy Klimas and my dear huggy (oopss, that’s dear hubby, lol) mailed it to Dr. Phelps yesterday. I thought it would save an enormous amount of time if she was familiar with my case before my visit. What is so interesting to me is that in the last 16 years I’ve had ME/CFS (I’m 64), that in the Houston area where I previously lived, other that perhaps Dr. Patricia Salvato, it was a virtual wasteland of anyone that knew anything about ME/CFS, let alone had any empathy. I dreaded my yearly appointment with my Endocrinologist, Dr. Vivian Rodriguez. She was great about treating my Addison’s or hypothyroid issues (she would prescribe Armour or generic Porcine in special sustained release from the compounding pharmacy), tested all my thyroid #’s including RT3 and such, but I’d want to literally smack her because EVERY year she’d tell me to do things like ‘take a bus go to the YMCA and take a swim class’ and other idiotic suggestions. And Houston is supposed to be a mecca of medical facilities and advancement. I guess that’s so if you’ve got cancer or diabetes. For ME/CFS..not so much.
Good luck!
Thank you for posting as I have servere Osteoparosis and I am clueless as whom to see, where to go or what to do..Do you have osteoparosis..is she Osteoparosis dr. ?
Completely wrong and backwards. ME doesn’t cause increased serotonin it causes deceased serotonin probably due to constant immune activation driving IDO to push tryptophan towards kynurenine rather than serotonin production. One of the most replicated findings in ME is excessive increased serotonin receptor sensitivity. This won’t be caused by increased serotonin levels, but decreased levels. You’re not going to see this by isolating and looking at immune cells, you have to look in the brain. This is an infectious neurological disease, not an immune system disease.
If you actually review the literature you will see this, and you will note that several of the studies that showed excessive serotonin receptor sensitivity also showed that the patients that had this were chronically infected with enterovirus. You know, the only viral agent ever to be isolated from the brains and bodies of ME patients. Why are we wasting time and money looking for parasites when we already have the answer to what is causing this disease?
Oh and by the way, increased IDO activity is immunosuppressive and allows viral activity in the brain to increase. So in a chronic viral disease such as ME, decreased IDO activity would be beneficial and preferable for viral clearance.
That’s why we don’t need to assume that ones “symptoms/deficiencies/excesses” are illness – they may well be compensations. It could be the lesser of the two evils. But if found – then you can look for the WHY. Is it a compensation and if so why/what for. We don’t want to treat something until we know WHY. It may be the better thing – our body could be trying to save us. Immediately trying to change something as a knee jerk reaction may make you way worse. We need to go deeper than symptoms and get to “core”. (ie – Autoimmune and inflammation?????) If we need a bandaid to cover over symptoms and it gives more quality of life – I’m all for it. But unless it’s the core issue – it only “covers over/puts a bandaid” on symptoms.
Issie
I agree with weyland about they kynurenine pathway being more likely involved with ME than the serotonin pathway. TRP makes my symptoms worse. So do easily-digested carbs, 20 minutes later, which seems consistent with insulin, and the effect is blocked (or delayed and spread out) by taking BCAAs. Some of the nastier kynurenines, such as quinolinic acid, could explain many of the neurological symptoms. While TRP makes my symptoms worse, 5-HTP doesn’t, so the problem doesn’t seem to be the TDO pathway, but the KYN one.
IDO, which is mainly triggered by IFN-g, and in the brain is produced by microglial cells, depends on superoxide for activity, and on peroxynitrite for breaking it down again. That means that mitochondrial function is critical: dysfunction would affect IDO activity and kynurenine production. I seem to be very sensitive to peroxynitrite scavengers (cinnamon, turmeric) which make my symptoms much worse, and to events that increase IFN-g. It seems logical that a low peroxynitrite/superoxide ratio would result in longer half-life of IDO and thus increased kynurenine production.
I suggest testing CSF to look for abnormal levels of kynurenines in ME patients. Some experiments with superoxide and peroxynitrite levels might show something interesting too.
How would an ssri have caused my cfs? I’m not disagreeing. It’s obvious to me that a subset of cfs patients have serotonin dysregulation. And I am proof of this. I was an extremely fit professional ballerina and in three month on lexapro became bedridden. All my symptoms began on that Med, and 13 years after stopping I have not improved. There are many post ssri people that have developed cfs from ssri’s.
Hi Cort, thanks for the blog! Is there any mention or advice on whether or not to slowly reduce tryptophan/5htp dosage? Ive been taking it for 2 years now… i also wonder if ssris are what causes disease permanancy as the first thing a GP does is chuck you on an SSRI.
Basically the advice is to just sit tight and let them work, test the hypothesis further and get back to us with the results. I have no idea about SSRI’s but it’s an interesting question for sure given, as you point out, that that’s generally the first thing docs do….
I am really leary of SSRI’s as having a bad side effect of one as I read one of the comments about Strep. has any one look into A Strephoccus as a possible biomarker as that might be the keep of key…
Yes! I have wondered how many cfs patients have not connected their illness starting from an ssri. Seems the rise in cfs/me correlates to them coming in the market and being over prescribed. Obviously not everyone develops cfs from ssri, but certainly could be genes that influence the outcome. COMT for instance cause neurotransmitter to not recycle as fast and I’m homo for this.
Very interesting article. Having heard some anecdotal reports of people recovering with high doses of cyproheptadine (and similar) to reduce serotonin.
I find this helps a bit. I have severe me/cfs and high 5hiaa which is also a marker of hyperactive platelets. I also have very high vegf, scd40l and il 8. I get serotonin syndrome from a few drops of methylene blue.
How does this effect intraceullar calcium levels?
From the work done at Acumen, and by Dr Myhill and cheney it is apparent that every person with CFS has a problem with calcium efflux.
This means that once 5G come in NOBODY will be able to recover as nnemf causes calcium to flood into the cell.
Hmm. The lifespan of a WBC is only 20 days max.
If the met.trap theory is right, how does this condition propagate from generation to generation of cells?
To clarify. Although the bad ID2 gene gets passed on, The pathology requires a stress trigger to elevate tryptophan levels rapidly within the cell. But every 20 days or so, new white blood cells will be born, which have normal tryptophan levels in them. Therefore, the metabolic trap should only last for 20 days in a white blood cell line. Maybe longer in other cell lines, according to their life spans. Most cells have a lifespan of months or in some cases a few years. So why is the metabolic trap persisting across cell generations? CFS triggers don’t last forever. Most of them get resolved after a while. Are we assuming there is some unknown factor that forces new cells to take on too much tryptophan? Or perhaps the stressor is not actually the underlying pathology, but some broken signal to the new cells? Just trying to understand how this works.
Hi Cort, would you be able to explain the link between high tryptophan and high serotonin? From the article, tryptophan needs to be broken due in order to produce serotonin, so the fact tryptophan is not being broken down (due to the faulty enzymes), means that serotonin should be low? So why is it high? Or am I confusing something?! Hope you’ll be able to clarify. P.S Thank you got such an amazing article that has brought me a lot of hope!
It is confusing. I think the idea is that if tryptophan is not getting metabolized to kynurenine then it must be being metabolized to serotonin. Phair was able to show very little production of kynurenine in one of his tests. That’s my guess.
Some pwme are getting worried that this means they need to check all their supplements for whether they can affect the kynurenine pathway. For example I myself am getting significant benefits from curcumin, and I was also planning to start NAC. But I’ve just found out that these can both reduce kynurenine… But am I right in thinking that the warning from Ron was really just to not intentionally try to mess around with this pathway (E.g. by taking tryptophan)? Not an issue regarding taking supplements which also effect this pathway a bit but are being taken for the purpose of other benefits?
On another note, I am very interested in this theory, because my ME started around the time that I started taking an SSRI…. However, I did also have a really bad flu shortly after starting the SSRI, so it may be unfair of me to blame the SSRI.
I’ve just done a little bit of reading about kynurenine pathway and apparently there is a relationship between kynurenine and pro-inflammatory cytokines. Wasn’t there a study that found increased levels of these cytokines in pwme in the first 3 years of illness, then reduced levels after that. So would be interesting to see whether Phair’s kynurenine finding is different for pwme in first 3 years of illness vs longer-term pwme.
Studies have found neuroinflammation in pwme so I wonder does inflammation cause reduced kynurenine or vice versa?
PS my first time commenting on HealthRising – feel like I have reached some kind of pwme milestone lol!
“my ME started around the time that I started taking an SSRI”
For many patients stress/anxiety/psychological symptoms did pop up early on their disease.
For me for example, at about age 26 I changed from a person having very few trouble with stress to a person nearly consumed by stress. From that age on, my health started a slow but continuous decline until I ended up with full blown ME over a decade later.
However now I strongly believe it weren’t the far too high levels of stress that caused the decline of my health, but the early decline of my health and hidden start of my pre-ME illness that caused huge hormonal changes that caused the stress.
Starting to take SSRI medication could hence just as well be to deal with a symptom of pre-ME manifesting itself. While sudden onset with ME is very common, many patients who get better to some extend see that the disease was present for a far longer period of time in their life than they originally thought.
As to supplements: listen to your body for stuff you are already taking. If you feel it clearly helps you, then you have found more then many of us ;-).
Same happened to me, what brought me down completely in the end was NAC though.
Are you german speaking? Your name sounds like it. I am from Austria.
Interesting – I haven’t come across many others who’s CFS has been caused by an SSRI – mine undoubtedly was. I was given the SSRI for anxiety – had a very strong reaction to it – significant anxiety for a number of months – then started to feel fatigued, headaches, foggy brain – and into full blown CFS less than 6 months later. Was a highly energetic marathon running high paced professional prior to taking the SSRI. Biggest mistake of my life taking those little pills.
But the serotonin link of SSRI to the IDO trap seems to make some sense. It’s interesting they’ve released this finding on the IDO trap – they must know that CFS people are desperate and are undoubtedly going to try to solve it themselves. I’m already googling how to lower tryptophan and serotonin levels. Slightly concerned by the talk of needing to investigate animal and human models – sounds like years of research before a treatment.
I could guarantee your cfs was triggered by an ssri as mine was. Lexapro specifically. Lexapro is the biggest offender as it is considered a clean ssri. It targets only one or two 5ht receptors so strongly. Although I’ve seen damage from all of them. I was completely healthy and three months on lexapro my life was forever changed.
I have classic cfs with high viral titers and low nk function. Autoimmune issues. All began as soon as I took lexapro.
And one more thing. Through the years docs have tried me on various other ssri’s for my cfs. It’s obvious my serotonin system was altered dramatically because I can take a minuscule amount and immediately have awful symptoms.
But when I started lexapro for the first time back in the beginning, it took weeks to feel the effects. So it permanently altered something.
Liz – very interesting. I was on SSRIs as a teenager and in my twenties, and then stopped taking them. A year ago under my doctor’s supervision I tried Wellbutrin (first) and had awful headaches, jitteriness and some nausea. We discontinued that. Later we tried Cymbalta and I was sick as a dog for a week. I’m surprised there hasn’t been more research on serotonin syndrome in pwme.
I just spent valuable energy writing a comment but it hasn’t appeared after I submitted it 🙁
Me also
In 2007 a study found that eating dark chocolate with a high cocoa content benefited ME/CFS patients. This has always been my experience too. My love of all things chocolate perhaps suggests I have low serotonin but I don’t often suffer from depression and after an absolutely horrible experience with SRIs now wonder if I have high serotonin. After reading about this exciting new theory, it sounds like many patients are having negative reactions to tryptophan supplements. However, chocolate is high in tryptophan. Does anyone have any idea why tryptophan supplements are dangerous but chocolate isn’t? So confusing!
Anything concentrated and not in a whole form is a very potent medicine. Foods and herbs in whole forms have many more properties than just one substance.
Issie
Prof Gilles Guillemin at Macquarie University (Sydney, Australia) leads a team that studies “metabolites deriving from tryptophan metabolism [including the kynurenine pathway] and their involvement in several neurodegenerative diseases” such as MS, Alzheimer’s disease, ALS, and MND.
(see https://www.mq.edu.au/about/about-the-university/faculties-and-departments/faculty-of-medicine-and-health-sciences/staff-profiles/gilles-guillemin)
Cort, I am amazed at your ability to write these complex reports. I literally cannot wrap my head around this anymore. Ha! But I have to share – my husband had mono about 25 years ago. The idiot doctor gave him an SSRI for depression. He took one pill and hallucinated all night. I also was given an SSRI for my me/cfs, and was up all night going crazy with anxiety. In both cases it seemed we had a serotonin overdose. So again, this brings me back to speculation that me/cfs is infectious.
Wow…
Cort,
As I haven’t figured out how to navg.on here yet..not techie Steph A was another gift my mom gave me and I inturn gave it to my daughter is why I said biomarker from what I read so far and by reading others comments on here
I actually had a lightbulb moment..I hope they look into this..
This is really confusing because I respond VERY well to serotonin boosting drugs. I was taking Venlafaxine (Effexor) and I noticed after discontinuing the drug, within 3 months I declined rapidly. Practically bed bound unable to tolerate noise and light (GABA/NMDA dysfunction possibly).
It’s only until recently I had added 1+1 and it clicked in my mind that looking back to when I discontinued the Venlafaxine, that’s when things went terrible for me ESPECIALLY with how my brain was reacting to daily life.
Currently I’m on a quest to find the right natural supplements to balance out my GABA levels and serotonin. As for serotonin I have had success in microdosing with Psilocybin (active ingredient in magic mushrooms) and in the past I’ve felt good results with 5htp.
However, 5htp isn’t good for long term use (apparently), and using psilocybin causes an increase in glutamate (I think) as I get horrible headaches after consumption, which is a shame!
Female, Ill from 16 after tick bite & contact with tick bloods or EBV wo appropriate resting(both in same year).
Never any therapies offered.
SSRI when burnt out after 3 months of full time work: did not improve – was bedbound for ±1 year.
Now 30 years of age…
Got prescribed L-tryptophane: went well for 2 days!
And then … the uppermost strange and scary feeling I ever experienced, kind of being nausea in my head – left me going to my bed to feel sick and whine but laying did not help!! It was like I was living unter a shell it felt very weird and unpleasant while I experienced all kinds of neurological symptoms (for these 15 years among them extreme vertigo and dizziness to the point where i can’t get my body up straight)…
I stopped the pills immediate.
Then started again some days later with a half one.
Feeling did come back…
Did not touch the L-tryptophane again.
Maybe even more interesting: as is known, SSRI may elicit suicide in certain people, of whom my father has been one.
Suppose these people were not depressed, but having CFS symptoms?
The L-trytophane feeling was not suicide ideation or any kind of that. But I felt so strange in my head, no matter what I did or how nice it would be – it followed me everywhere and could not be ignored anyhow, if it would stay, it would certainly not make me happy but make me consider it because my life quality all the sudden was like gone.
I can’t describe the feeling it was like your head feeling very heavy, nauseas, extremely sunisitis, malaise, feverish, and if you are in a waze – living under a dome, as if so hit you on the head or you have a brain concussion. It was horror …
It was even nothing like brainfog (!!!!!) which I experience almost 3/4th or half of my awake time or cognitive problems or anything and stood completely apart from that…
Excess serotonin has been well documented to cause excessive perspiration. I remember the night sweats that came earlier during the course of my ‘sickness’, a stack of clean dry t-shirts next to the bed. I send as often as I can to OMF, so grateful for the hope
Typo in the caption under the enzyme illustration – says “IDO1” has mutations, but it’s *IDO2* in Phair’s hypothesis.
I’m just catching up on all this; exciting stuff to be talking directly about modelling a plausible central mechanism like this. I’m very keen to know of updates on this! 🙂
In his emerge talk, in March, he mentions that more accurate testing of the intracellular metabolites should be complete in about 3 months (now-ish!): https://youtu.be/Quh-77gvw4Q
Do you think we’ll get an update on that (and whatever else Ron alluded to) at, or before(?), the next annual symposium?
Yes..
Lol! Redundant question, eh? Thanks for digging this out the the spam bin. 🙂
I checked my Dante Labs results and I’m heterogeneous for IDO2 rs21608099A;T0 & rs10109853(C;T) & homozygous for IDO1 rs7820268(T;T)
Speaking of self treatment, I recently took ivermectin for horses since I couldn’t get a doc to prescribe ivermectin.
The ivermectin did exactly what the package states…”some horses experienced reactions with swelling and itching shortly after treatment.in most such cases the horses have been diagnosed with carrying heavy infections of Onchocerca microfilariae and it is assumed the reactions were from heavy die off In large numbers”
along with itching and swollen joints, i ended up with high fever and was in bed for 1.5 days. Thus has all subsided but now my immune system seems to be trying to regulate itself. A week after this happened, I received a parasite kit from a new gastroenterologist specialist so I am hoping they may find something with all this heavy die off.i said some research on this above parasite…its a parasite obtained from mosquitoes. I can tell you with uncertainty, that if mosquitoes are present, I will not only be the first to be bitten but will also have the most bites. Just thought y’all may find this interesting as I know long haul covid folks pull out with ivermectin
Can you tell me what evidence supports this statement : “You can actually induce autoimmunity – an autoimmunity you cannot recover from – by taking tryptophan.”?
Thank you.