

A Critical Look at the NIH’s Accelerating Research Conference
This chronic fatigue syndrome (ME/CFS) conference was different. Besides the normal goal of bringing researchers together to share their latest results, it had an ulterior motive – getting NIH researchers involved and highlighting the work of the new NIH-funded ME/CFS research centers.
I wasn’t able to get a full picture of the conference. After getting sidelined by a lingering cold halfway across the U.S., I watched the conference from a hotel room in Kansas City. But what I could see suggested the conference didn’t achieve either of its two goals.
While the upper part of the room was fuller, the lower part of the room near the stage never came close to filling up. Many of the questions put to the presenters came from patients, advocates and people with an agenda – not from NIH researchers newly interested in the field.
Plus, perhaps not surprisingly, given how long everything takes with ME/CFS at the NIH, we got precious little from NIH-funded research centers or from the intramural study, and the most interesting talks came from outside researchers.
As a showcase of what the new NIH approach is doing for ME/CFS, then, the conference appeared to be six months or so too early. The NIH, it should be noted, plans these things far in advance, and must have thought things would be further along by now.
This is not to slam the conference organizers. It was a worthy effort and an NIH ME/CFS conference is better than no NIH ME/CFS conference but the failure of the conference to pack the hall with NIH researchers was a pretty stark reminder that, once again, the half measures the NIH is taking on ME/CFS are not going to do it for this field. We have, after all, had NIH conferences, and some small NIH-funded ME/CFS research centers before. If the NIH going to fulfill on NIH Director Francis Collins’s promise to get serious about this disease, it must do more.
A Good Start
The conference presenters did a very smart thing – they started off with Cindy Bateman! Her sober, clear and impactful presentation was just what the doctor ordered for any skeptics in the audience.
The highlight of Dr. Bateman’s talk for me was her case report of a formerly active and now severely ill person with ME/CFS who is: a) getting excellent medical help; b) has the full support of her husband; c) has the ability to try everything; and d) is still severely ill. The patient – who is getting the best of the best – dramatically represented the limits of our current knowledge of ME/CFS.
It was a perfect way to present how dire the need is.
Metabolomics Session Disappoints
It was not surprising to see metabolomics – the hottest field in ME/CFS research – take pride of place during the first session of the conference. The surprise was how flat the session fell.
Bob Naviaux’s attempt to cram perhaps a decade of thought into a 30 minute presentation contained some of his most intriguing insights (Dauer, the mitochondria-illness connection), but it included almost nothing on ME/CFS and did little to advance the conference goal – accelerating research on ME/CFS.
Similarly, Oliver Fiehn – a metabolomics pioneer – was an impressive presence but spent almost all his time trying apparently to convince the audience that the metabolomics field was: a) legitimate; and b) ironing out its apparently still significant problems. His last second summation of his recent metabolomics results in ME/CFS – which he seemed almost apologetic for – did not seem likely to embolden NIH researchers to enter the field. It’s unfortunate we didn’t get a good overview of this important field at this conference.
Jonas Bergquist – on the other hand – whom the Open Medicine Foundation recently awarded funding for a Swedish ME/CFS Research Center gave a well organized and clear presentation. His talk titled, “The Biochemistry to Support the Evidence of Neuroinflammatory Involvement in ME/CFS”, wasn’t on metabolomics, but it was on ME/CFS.
The presentation focused first on his past proteomic work which he updated with some new proteomic work data. It was good to see Bergquist find alterations in proteins that fit with what we would expect – neuroinflammation, lots of cell damage and an upregulation in proteins designed to repair that damage. Getting results from different compartments of the body which highlight the same thing – in this case inflammation – is a good thing.
Bergquist’s inclusion of multiple sclerosis in his latest study paid off with ME/CFS falling somewhere between healthy controls and MS patients. The fact that he found many more protein differences in the cerebral spinal fluid than in the plasma suggests that the neuroinflammation – as Jarred Younger and Nancy Klimas believe – may indeed be the key.
Bergquist’s results – which are preliminary – will hopefully inform the Ian Lipkin / Simmaron Research Foundation new cerebral spinal fluid study – which now includes metabolomics. Those results of the Lipkin study are not available yet as the journal reviewers passed the study back to Lipkin asking him to get more healthy controls. Given the latest neuroinflammation findings, the first metabolomics study of the CSF in ME/CFS should be fascinating.
Bergquist has also found evidence of thyroid dysregulation in ME/CFS and has validated Carmen Scheibenbogen’s autoantibody results in ME/CFS. Given his results, it was easy to see why the OMF granted him that new ME/CFS research center. Let’s hope those studies will be published soon.
Immunology Session
Jose Montoya’s “Different Reality”

Working with Mark Davis and others, he’s been digging away at the impressive database of samples he’s gathered (200 patients/ 400 controls) with Ian Lipkin, Mark Davis, Ron Davis and others at Stanford.
Montoya’s big cytokine study suggested that cytokine levels do matter in ME/CFS but only (with two important exceptions) if they are correlated with severity.
TGF-B, which has now been found upregulated in five of eight studies is one notable exception, and Montoya asserted that we should now just put a pin in that finding and recognize that it plays an important role in ME/CFS. Besides other things, TGF-B appears to be upregulated in mold exposure and mast cells, and plays a role in Ritchie Shoemaker’s conception of mold-induced illnesses.
Montoya also reported that 10 of the 17 cytokines highlighted in the cytokine study play a role in mast cell activation. His “simplified model of mast cell activation” made one wonder, though, if that finding is a benefit or a curse.
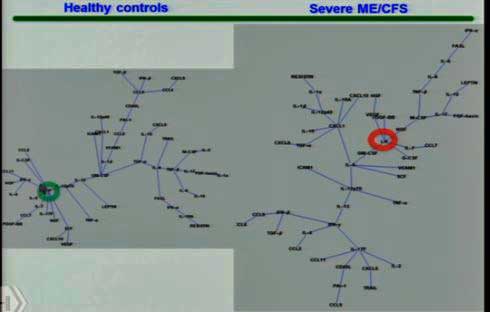
The hubs of the cytokine networks have shifted from one side to the other in healthy controls (left) compared to the people with ME/CFS (right).
Montoya’s finding that cytokine levels are highly correlated with severity appears to have caused him to dig more deeply and he’s now assessing cytokine networks – something Dr. Klimas has long been a proponent of. His overview of cytokine network findings underscored how dramatically different the networks of healthy people and people with ME/CFS are. Each network circled around different central hubs – suggesting that the immune systems of people with ME/CFS had dramatically changed. Gone were the neuroprotective factors found in the healthy controls – replaced by a pro-inflammatory network found in people with severe ME/CFS.
In another move that seemed reminiscent of Dr. Klimas, Montoya also discussed doing an “in silica” modeling of cytokine networks with the idea of trying to determine how to shift a network back to normality. He has also found evidence of, if I have it right, a genetic alteration in a HLA gene in ME/CFS which has been associated with a series of autoimmune diseases, and is working with Ron Davis on that. (Davis recently created a better and more efficient way to assess these highly variable and complex genes involved in our immune response to pathogens.)
Montoya now also has funding for and is getting samples from the severely ill in his studies. All in all it was a good presentation with lots of food for thought for the NIH.
Mark Davis’s Search
Ron Davis got a rare major grant for ME/CFS to fund Mark Davis’s work on T-cell clonal expansion – a project which we learned originated in a hallway conversation between Montoya and one of Mark Davis’s students. That study was (and is) being funded by the OMF, and is the subject of the big NIH grant.
Talking about his T-cell work, Mark Davis started off by saying that ME/CFS more and more looks like an autoimmune disease. It was a complicated presentation full of twists and turns which showed that the deeper you look into the immune system, the more surprises it holds, and the less you can trust traditional thinking. Davis, for instance, found massive clonal expansion of T-cells in a multiple sclerosis model. That indicated that the cytotoxic T-cells had encountered something and were pumping out clones which then went to work obliterating the myelin on MS patients’ nerve cells.
When Davis tried to make the situation worse by adding more cytotoxic T-cells to the mix, the disease got better instead of worse (!). That made it more important than ever to find out what those cytotoxic T-cells were going after. Davis’s student developed an algorithm to do that, which – in a reflection of how difficult (and important) a feat that was – wound up getting published in the Nature journal.
Davis next found that T-cells in celiac disease were responding to the disease – but not the kinds of T-cells he expected. Apparently, the T-cells are capable of mounting a range of responses in autoimmunity – some of which were a surprise. He found the same in other autoimmune diseases. In another huge surprise, he found that the cytotoxic T-cells he was sure were attacking the gut membranes were not attacking them at all. The upshot for me, if I got all this right, is that the T-cell repertoire is much more complex than we suspected – perhaps good news for ME/CFS – which has this irritating tendency not to fit traditional boundaries and consistently fall through the cracks.
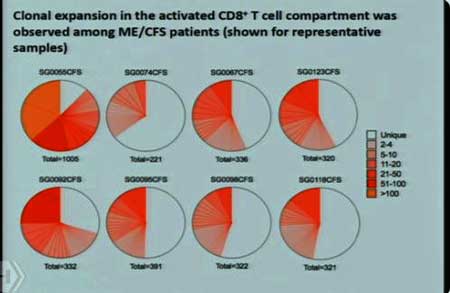
Which it proceeded to do once again as Davis was able to find evidence of T-cell clonal expansion in every ME/CFS subject tested thus far, but has failed to find a good reason why. Pathogens don’t seem right now to be the answer. To make matters worse, it appears the significance of the T-cell clonal expansion found in ME/CFS may be in doubt as Ron Davis the next day reported that a similar level of T-cell expansion was also found in healthy controls.
Davis, is, however, studying a new cohort and is going to broaden his search significantly. Something is causing the T-cell clonal expansion in ME/CFS and if Mark Davis can find out what that is – and it’s something clearly pathogenic – that could be very helpful.
Derya Unutmaz and the Unconventional T-cells in ME/CFS
Next came the first report from an NIH-funded ME/CFS research center. The fact that Unutmaz was able to gather his data so quickly was a bit of a surprise, but he did have an ace in the hole – the Bateman Horne Center – which needed to get so many ME/CFS samples (200) that it ended up creating a new ME/CFS outreach program in Salt Lake City.
Suzanne Vernon, the BHC’s research director, has warned about a welcome new need – the need to get enough ME/CFS patients to fulfill our field’s need as it ramps up. The good news is that the BHC showed that it can be done and has provided a template on how to do that. The very, very nice, secondary outcome of the ability to do that is that a lot more doctors in Salt Lake City now know about ME/CFS (:))
Unutmaz, another T-cell researcher said he was shocked, just shocked, to find significant differences in a first, rather coarse look at T-cells in ME/CFS. Upon probing more deeply, he found some of the differences disappeared when age was taken into account but others did not.
All in all, the finding suggested that the T-cells in ME/CFS may be aging more rapidly than normal – through exhaustion – which perhaps will surprise no one. (A similar finding has shown up in HIV, plus the Lipkin/Hornig cytokine study suggested the same. Plus, the CDC’s telomere findings in ME/CFS suggest the same, as do some fibromyalgia findings.)
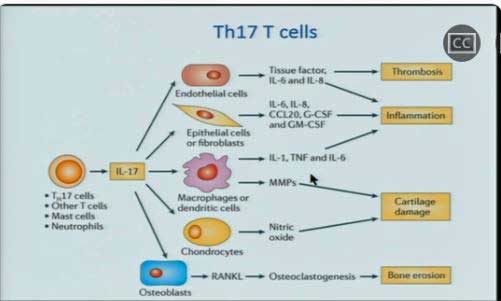
Unutmaz was keen to check out Th17 T-cells – key players in autoimmunity and inflammation that are highly regulated by the gut – in ME/CFS – but when he did, he got a surprise. (No!!!!) He found high levels of Th17 cells but low levels of the cytokine (IL-17) they produce.
That suggested either exhaustion or a malfunction. Something in the gut, probably, is triggering a massive Th17 cell expansion but the cells (perhaps quite fortunately) are having trouble getting activated. Some of the data from the EMERGE conference and from the next presentation may explain why. The immune cells, like so much else in ME/CFS itself, do okay at baseline but stress them out a bit and they punk out.
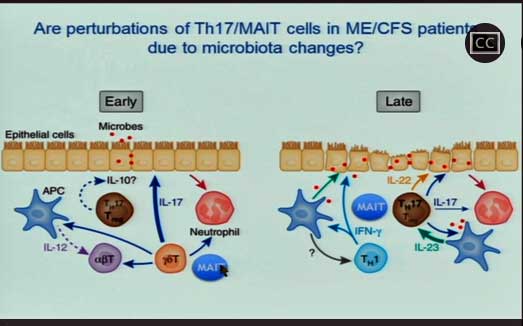
For now, MAIT cells apparently lurk in the gut lining as a kind of last line of defense against the bad bacteria getting into our blood stream. Once activated, they turn monocytes into macrophages, which then gobble up the bacterially infected cells.
Unutmaz’s findings suggest that the MAIT T-cells have been repeatedly activated in ME/CFS and evidence the same activated/burned out pattern just described. He and Oh are trying – just as Mark Davis is – to determine what bad actor has turned them on. If they can find out what that bacteria is – and remove it – they may be able to tamp down the immune activation that just about everybody believes is present in ME/CFS…
Maureen Hanson … er … Alexandra Mandarano – Immune Cell Exhaustion Explained?
Maureen Hanson then turned the microphone over to her student, Alexandra Mandarano, who appeared to explain just how those T-cells may be getting exhausted.
This project – done in collaboration with the Simmaron Research Foundation – found that ME/CFS patients’ cytotoxic (CD 8) T-cells were able to generate normal amounts of energy at rest but pooped out when activated. Since they’re activated in order to knock out infected cells, having lethargic cytotoxic T-cells could help explain why the colds that often trigger ME/CFS tend to be so severe.
We’ve long known that T-cells’ cousins, NK cells, are horrible at killing infected cells in ME/CFS, and a Griffith study, if I remember correctly, suggested that cytotoxic T-cells might have the same problem. Now we appear to know one reason why – they just can’t generate enough energy to get off their duffs and wipe out the invaders.
An overview of a fascinating presentation from the EMERGE conference will provide more information on this subject but, for now, it’s good to see energy production problems showing up in the immune cells as well.
Provocation Studies
As time has gone on, creative researchers have used different provocations to stress the systems of ME/CFS patients in new and different ways. Whether it’s Ron Davis and his nanoneedle, Maureen Hanson and the Seahorse, the Japanese and their cognitive tests, Peter Rowe and his tilt table testing, the Griffith group and their NK cells or even, come to think of it, Lenny Jason and his group of infectious mononucleosis students, researchers are provoking ME/CFS patients’ systems in different ways and coming to the same conclusion – that when you stress an ME/CFS patient’s system, it tends to fail.
The exercise tests that Workwell, Betsy Keller, David Systrom and others use provide perhaps the greatest stressor of all – and not surprisingly, bring a welcome clarity to the field. Staci Stevens of the Workwell Foundation – the creator of the two-day exercise protocol used in ME/CFS – has maintained for years that the exercise studies do for ME/CFS what virtually no other kind of study can – elucidate in clear and stark terms the central problem in ME/CFS – the inability to tolerate exertion.
Dr. Klimas on Computing a Pathway Out of ME/CFS
Dr. Klimas’s computational work in ME/CFS is well known. Collecting millions of data points during exercise, she’s literally observing post-exertional malaise come to life. First, exercise kicks off a burst of inflammation, which in turn triggers oxidative stress and causes the autonomic nervous and hormonal systems and others to go bonkers. She, Gordon Broderick and Travis Craddock have used a computational model of what has gone wrong in ME/CFS to identify drugs they hope will shove our systems back into health.
Dr. Klimas’s fascinating work will be covered in a separate blog.
David Systrom’s Trail Into ME/CFS
“Pathophysiology and treatment of exertional intolerance in ME/CFS: insights from cardiopulmonary exercise testing”
I think of David Systrom as a hopeful harbinger of the future – not just because of his exciting results – but because of how he came to this field. Systrom was not enticed into it. Instead, he came to it through his study of exercise intolerant individuals – many of whom, he later found out, had ME/CFS.
A pulmonologist working out of Harvard University’s main hospital, he brings two things we vitally need: credibility and contacts. He’s working, for instance, with Anne Oaklander – another Harvard researcher – who uncovered the small fiber neuropathy present in fibromyalgia.
Systrom’s invasive cardiopulmonary exercise test (iCEPT) is like the CPET tests we know, love and occasionally hate – on steroids. Using a catheter, these fantastically expensive tests provide much more information.
Systrom is finding relatively normal VO2 max in ME/CFS – not a surprise for a first day exercise test – but his invasive tests are revealing problems during that first exercise test that ordinary CPET tests cannot. Two major abnormalities are showing up: both the filling pressure of the heart and the systemic oxygen uptake are uniformly low during upright exercise in ME/CFS.
They suggest the heart is not getting the blood it needs, and the mitochondria are not getting the oxygen they need to produce the energy we need during exercise.
Systrom cited the story of a former competitive cyclist who had undergone a $1,000,000 workup – not that difficult a thing to do, he said, in the hospital he works in – to no avail. His testing revealed the problem – his heart wasn’t pumping the blood it needed to his muscles and even when the blood got there, the oxygen in it wasn’t being transformed into energy.
Systrom also brings the gift of a big data set – something this field has missed for years. Of the 200-plus patients he worked up in one data set, 72% met the criteria for ME/CFS. The man has basically been studying ME/CFS for years. POTS was next on the list (27%), followed by fibromyalgia and mast cell activation syndrome (MCAS) (20%).
This breakdown seems to fits perfectly with the degree of exercise intolerance found in these diseases – present in POTS and FM – but not nearly to the degree found in ME/CFS.
Systrom has identified five different subsets which appear to differ depending on their issues with oxygen extraction and blood vessel problems. The high blood flow group, for instance, has pretty good blood flows but is not using up their oxygen.
Dr. Klimas’s drug of choice – Etanercept – knocks down the same immune pathway Systrom’s preliminary results suggest exercise is upregulating in ME/CFS.
Most of this group had ME/CFS and forty percent of them also have small fiber neuropathy (SFN) – a remarkably similar percentage to the amount of SFN found in both POTS and fibromyalgia.
Unfortunately, the amount of small fiber neuropathy present did not correlate with the exercise findings. Systrom, however, believes that small nerve fiber functioning rather than nerve fiber density may be a better measure.
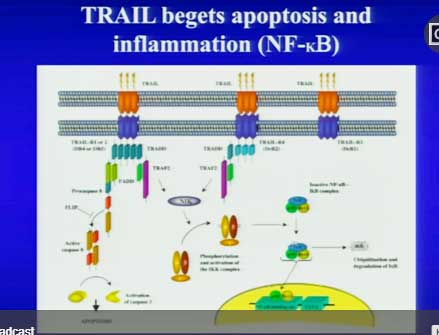
Systrom next surely opened some eyes when he proposed – citing a conversation with another researcher – a new potential immune pathway for ME/CFS called TRAIL. TRAIL involves an uber pro-inflammatory cytokine, TNF-a, that’s often provoked in response to a viral infection. Systrom’s early testing suggests that exercise may be temporarily upregulating that pathway in ME/CFS patients.
A temporary upregulation fits with Nancy Klimas’s model of a massive upregulation of inflammation occurring early in exercise in ME/CFS, which then dysregulates other systems. In fact, Etanercept, the drug Klimas has chosen to tamp down the inflammation in GWI and ME/CFS, is a TNF-a inhibitor (:))
Systrom ended a fascinating presentation with the news that we can expect a treatment study out – something rare in ME/CFS – which shows that pyridostigmine bromide (Mestinon) – at rather increased doses, as I remember, was effective in increasing both VO2 max and, most interestingly, oxygen extraction in the muscles (!).
Mestinon, an old drug, increases the availability of the parasympathetic nervous system neurotransmitter, acteylcholine. Dr. Jay Goldstein recommended it in ME/CFS decades ago but it’s hardly been used in ME/CFS until Systrom came upon the scene.
Check out the story, though, of a person with a longtime case of ME/CFS who is now able to exercise after getting Mestinon (from Systrom).
Systrom is also starting to do muscle biopsies and is incorporating the Seahorse and mitochondrial studies into his work. He has found positive acetylcholine autoantibody results (using the Mayo tests – he has not used the new German tests yet). Also, it’s important to note that all Systrom’s results disappear when the patient is tested while supine. He also noted that the results he’s finding in ME/CFS are not found in deconditioning.
Systrom’s first presentation at an ME/CFS conference was for me the highlight of the conference – not only because he brings such exciting results but because he’s clearly now immersed in this field. His first pure ME/CFS project is being funded through a grant from the Solve ME/CFS Intiative (SMCI).
Systrom has taken exercise testing to a new and exciting level in ME/CFS, but as Dr. Keller, the next presenter, will show, the non-invasive CPET studies are still providing plenty of juice.
Betsy Keller

She added data on two more twists to exercise issues showing up in ME/CFS.
Chronotropic incompetence (CI) refers to the inability to increase one’s heart rate appropriately during exercise. That issue was first identified in ME/CFS by Workwell researchers in 2016, and updated by them in a new study “Chronotropic Intolerance: An Overlooked Determinant of Symptoms and Activity Limitation in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome?” this year. Dr. Keller showed that the CI in ME/CFS gets worse on Day 2 of an exercise test.
Keller also showed that using age-predicted heart rates in ME/CFS studies ends up in ME/CFS patients working much harder than their “age-predicted heart rates” would predict. This means that sub-maximal exercise tests which rely on these heart rates aren’t sub-maximal at all. If Keller is right, (and I have this right) people with ME/CFS in these studies are exerting near maximal exertion over extended periods of time. This is a big issue in the exercise physiologist subfield in ME/CFS.
Keller also added ventilation – the ability to push oxygen in and out of the lungs during exercise – to the list of exercise abnormalities found in ME/CFS.
She presented the case of an ME/CFS patient who was able to dredge up only 22% of her expected ventilation during exercise. Not only was she not delivering oxygen to her muscles, she was not removing CO2 from them either. Keller also found that this person’s blood pressure actually dropped (instead of increasing) during the second exercise test (!). It was no wonder she couldn’t exercise!

Then she dropped a stunner. Referencing a 1989 Montague study, she showed that the exercise problems in ME/CFS have been hiding in plain sight for thirty years.
The team of exercise physiologists at Workwell have repeated ad nauseum that the NIH has never needed sophisticated technology to uncover the key issue in ME/CFS and this was proof. It’s possible that the technology needed to solve ME/CFS simply wasn’t present in1989 but the technology needed to demonstrate that ME/CFS was a serious disease affecting energy production and exercise was there.
Thirty long years later, it feels like the pioneering work of Staci Stevens, Christopher Snell, Mark VanNess at Workwell, Betsy Keller at Ithaca University and others, is finally starting to come to fruition. David Systrom’s invasive exercise tests and Nancy Klimas’s immense data gathering efforts during exercise are providing important insights. The NIH’s decision to make an exercise stressor a key component of its intramural study means the NIH has finally got it about exercise and ME/CFS.
It’s been a long time coming, though. Over a generation of patients have had their lives immeasurably blunted as the NIH has sat, and is still, for the most part, sitting on its hands – pretending as if this disease was an afterthought. It has hardly begun to make up for that neglect.
Watch the two-day conference here:
Day 1 – https://videocast.nih.gov/summary.asp?live=31636&bhcp=1
Day 2 – https://videocast.nih.gov/summary.asp?Live=31640&bhcp=1
- Check out a superb Medscape article by Miriam Tucker on the exercise issues and orthostatic intolerance findings presented at the conference.
Check out the second day of the conference
Your Support Keeps Health Rising on the Web



 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!





I hope so much that all this and especcially the treatment studies come to belgium. I am so severelly ill, can no more, am totally alone and here they still talk about the bio psycho social model and GET and CBT; I even could not go to a docter 98% severe bedridden so even a consult if there was a good specialist would be to hard and a hospitalization also. I need them to come to my bed. so desperate…gets worse and worse an in belgium no one is interested, no docters, no specialists, no goverment.
Hi Konjin, I wish the members of the Trans-NIH Group and the Institutes they represent would think of someone like you when they make their decisions not to provide even a small amount of funding for ME/CFS. My most hopeful moment during the entire conference came, though, when Carole Head stood up and said I am sure that the NIH is going to come around. It’s not going to happen right now but it will happen. I trust her judgement. May that day come sooner rather than later.
thank you for your kind words cort! just reading I couls crie so ill and touched by your words!
hi cort, sorry for another comment from me. what does the trans-NIH group and institutes do? are they working international? could I somehow tell their/mail their my story?
do you think if treatments found, they will also become available in Belgium or is that still a 10 years wait?
thank you for everything!
The Working Group is where the funding for ME/cFS comes from. My hope is that the new NINDS group will make recommendations this fall that move ME/cFS into NINDS and out of the WG.
Keep an eye on Dr. Klimas. It is moving slowly but she is testing available drugs on ME/CFS. If it works out I would hope it would be possible to get them.
Cort Johnson, was there any mention anywhere on (HATS) Hereditary Alpha Tryptasemia, Eagle Syndrome or the Alpha-Gal Meat Allergy I just got my DNA mouth swab kit from the Houston,Texas lab I now have one possible
genetic Doctor here who may sign the form to send it back to them, I also did the Panoramic x-ray at my Dentist Office it shows two bones
one on each side of my scan it looks like Eagle Syndrome I will not know more until I get the proper CT Brain scan done for (ES) to measure
the length of each bone & if they are calcified at the bone or the veins some say best to have either contrast others say refuse the
contrast & ask if they can do the CT Scan images in 3D. I am going to try sending my Panoramic x-ray to the Team with Dr. Ron Davis I am also
still trying to get the Alpha-Gal Meat Allergy test done hopefully next month. Eagle Syndrome can cause also inflammation in the Brain & also
there are numerous medicines used some respond well others need Surgery but the medicines carry them forward until the operation is done. Also,
I was told the Cure can lie in removing the entire bones not partial as some ENT Docs do
Hi Aidan,
Not that I heard. Good luck with the testing. It seems there are so many possibilities with this disease.
So many possibilities because it affects so many systems and functions in the body. But, yet, very disappointed in the progress.
What we have is a lot of possibilities. What we really need is the funding to narrow them down and then be produce a concerted effort on assessing the viability of a few of them. We don’t have anything close to that yet. We need the NIH to step up…
I think a patient asked a question in day 2 that referenced alpha gal, but my brain can’t come up with more than that. No presenter I heard mentioned it in the talks, but I missed some & haven’t caught up online yet.
Hi Aidan, when running “Hereditary Alpha Tryptasemia protein misfolding” trough my browser I did found an interesting link:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3725216/
Disorders of Protein Misfolding: Alpha-1-antitrypsin Deficiency as Prototype
It apears that Alpha-1-antitrypsin is stuff that, like its name suggests, counteracts trypsin. Now trypsin is not tryptase but there seems to be similarities https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4583378/ with title “Trypsin, Tryptase, and Thrombin Polarize Macrophages towards a Pro-Fibrotic M2a Phenotype”
So if too few Alpha-1-antitrypsin creates too much trypsin and therefore activates increased protein misfolding, then too much (alfa-)tryptase may do something similar. It’s still a bit far fetched, I did not find research supporting this yet. But that may just be something that has to be done. It kinda feels similar.
I surely don’t say ME equals an immune response against misfolded proteins, but a chronic strong immune response against them (or against a potential overabundance of alpha-tryptase) may well create a “fertile” ground for chronic high ROS and inflammation. And that is likely to lower the threshold for getting ME a lot.
Hypothethic, but maybe of value?
Here in Australia we are still fighting poor quality exercise studies which when adverse physiological events are actually measured they are explained away by people on the panel advising our lead research organisation on ME/CFS the National Health and Medical and Research Council (NHMRC). The ties to the UK are strong with a couple of the panel members supportive of PACE and the MRC. A 14 person pilot study found resting heart rate and exercising heart rate deteriorated in all participants but because their hand grip improved and they didn’t report symptom increase during the 4 week trial this physiological deterioration has been ignored. The full 6 month trial is going ahead with promotional material saying NO ADVERSE effects…
oh kayla, even in australia! I wished it was better for you there and for many others! IN’94 I got the GET and CBT and totally went downhill. I wish so much for you that the situation would be different and that they would take you and other sufferers suriosly! much strenght!
Ouch! Well, I hope they keep their eye on the exercise study research.
I wanted to mention I know one Woman now she had the Surgery done needed only one side for Eagle Syndrome (EG), her daughter as well shows also positive on scans she has 2 of them one each side but very small she is
to be monitored now for growth every 5 years & scans will be performed. Research links also (EG) to a Genetic born condition I think it is possible maybe the (HATS) just might be the Gene involved as a
possibility. Maybe Jarred Younger could look for Eagle Syndrome it will likely not be seen on his MRI’s CT 3D is best. I was just told now I do not have EDS 3 or even any partial Marfans the Genetic Doctor said I
have (HSD) Hypermobility Spectrum Disorder so I guess this is because on the new criteria definition put out now…
Thanks for the good write-up Cort! Hope you get better quick.
Some progress, huge amounts of work to do. ME/CFS: the final frontier (Star Trek…)?
“Something is causing the T-cell clonal expansion in ME/CFS” (quote from Mark Davis) combines with “TNF-a, that’s often provoked in response to a viral infection.” (quote from David Systrom) into: Ahpthous ulcers (I had masses and still have very much of them).
https://en.wikipedia.org/wiki/Aphthous_stomatitis:
“The mucosal destruction is thought to be the result of a T cell (T lymphocyte) mediated immune response which involves the generation of interleukins and tumor necrosis factor alpha (TNF-α).[6] Mast cells and macrophages are also involved, secreting TNF-α along with the T cells. When early aphthous ulcers are biopsied, the histologic appearance shows a dense inflammatory infiltrate, 80% of which is made up of T cells.[5] Persons with aphthous stomatitis also have circulating lymphocytes which react with peptides 91–105 of heat shock protein 65–60,[2] and the ratio of CD4+ T cells to CD8+ T cells in the peripheral blood of individuals with aphthous stomatitis is decreased.”
This includes a bonus link to “Montoya also reported that 10 of the 17 cytokines highlighted in the cytokine study play a role in mast cell activation.” (quote from Jose Montoya)
Then there is a link with “Up-regulation of Th17 and related cytokines” (quote from Derya Unutmaz).
There is an auto immune disease where having plenty and plenty of Aphtous Ulcers is part of the deal https://en.wikipedia.org/wiki/Beh%C3%A7et%27s_disease:
Under the list of things it affects:
“Brain: CNS involvement most often occurs as a chronic meningoencephalitis.” -> Chronic ME?
“Research suggests that previous infections may provoke the autoimmune responses present in Behçet’s disease. Heat shock proteins (HSPs) are present in some bacteria and serve as a “danger signal” to the immune system. However, some HSPs share a similarity in bacteria and humans.”
Following research paper https://www.clinexprheumatol.org/article.asp?a=6609 has title:
“Up-regulation of Th17 and related cytokines in Behçet’s disease corresponding to disease activity”
Both Aphtous Ulcers and Behcet’s disease origins are still unclear, but both seem to react strongly to “some sort of proteins”, be it HSP, unfolded proteins or (more alternative explanation) poorly digested proteins leaking through the gut. Eating a slice of papaya with every meal gave me a near instant (2 weeks until full change) improvement of about +80% health relative from where I was before. Aphtous Ulcers dropped by over half ever since. Papaya contains a very powerful protein digesting enzyme called papain.
-> This leads me to believe that partly/undigested proteins leaking through my gut into the blood stream cause(d) a massive immune response in my body (I had aphtous ulcers every other day at worst, where 3 to 6 times a year is normal for the 20% of people having them).
-> Many patients not having aphtous ulcers does not disqualify this idea IMO. The links of above findings are too high IMO. The location of this massive immune response may differ from patient to patient. For me, it’s focal point seems to be the mouth mucosa. It’s probably high elsewhere in the body too, but my immune system seems to have chosen my mouth as the main battlefield (to fight the rogue proteins?) as too speak. For others, it may be any of the places found in Bechets disease (mouth, skin, mengities (brain), bowels, lungs, blood vessels…). For still others the bulk of the immune response may be near the myelin and lead to MS.
A massive body wide immune response against rogue proteins also is in line with “no specific immune marker against a known pathogen”
“That suggested either exhaustion or a malfunction. Something in the gut, probably, is triggering a massive Th17 cell expansion but the cells (perhaps quite fortunately) are having trouble getting activated.” (quote from Derya Unutmaz).
I increasingly tend to believe that in the gut, the immune system works different compared to the rest of the body. When a bacteria is discovered in the blood stream then it is simple: attack and kill it.
When a bacteria is discovered in the (large) bowel, then it is normal: the large bowel contains much more bacteria then the human body has cells. Thus attack and kill every bacteria found in the gut is no option.
But wha then? Only attack those bacteria that are “known offenders”? That is, only attack those bacteria that the immune system has memory of as being “bad”? Doing that in the digesting food mass seems to be a very difficult thing to achieve.
But if the gut contains about ten times more bacteria then the human body contains cells and if a significant portion of those were bad, fighting them off at the gut wall/interface would provoke an extremely massive immune response in this very thin gut wall on a daily basis. That seems to be utterly destructive. I can imagine that would destroy the gut in days.
So what could be done? I get my inspiration from how nature handles insect pests and how cops handle massive demonstrations with potential uprisings.
For nature, picking away one by one adult insects yields very poor results. Some eggs are washed away, some dry out to heat, some freeze to death, some infant insects freeze to death…
There seems to be many “mechanisms” for “population growth and numbers control”. Nature does not eradicate the insect pests but “seeks” to control their numbers so that they do not grow out of control in a balanced ecosystem. With far more then one hundred species of bacteria in a healthy human gut, the human gut is sort of an ecosystem too.
As to “crowd control” by the cops in massive demonstrations, going all in frontal to root out any problem is very hard to achieve too. Here again, an approach of “controlling and moderating” the problem is more realistic and more often used.
-> Maybe the Th17 cells in the gut are not activated to “crush and kill” (as that would create a gut-destructive battlefield), but are ordered to slow down the growth rate of bacteria. Remember bacteria themselves have plenty of mechanisms to decrease the growth rate of their competition (like producing lactic acid, acetic acid, antibiotics…).
Why wouldn’t some human immune cells be able to do the same trick? It would be far and far less destructive then going into a straight outright attack against the bad bacteria. And given that bacteria growth is an exponential thing, reducing their growth rate just a bit is more then enough to for example reduce their numbers tenfold (within a certain time frame like the time the food passes through the gut). That decreases the problem at hand very much.
It would be even better if these immune cells were able to selectively reduce the growth rate of badies compared to goodies. Then the goodies would further help to reduce the badies as all bacteria try to reduce the numbers of their competition.
And that may not be too far fetched. In a recent blog Cort wrote about some good gut bacteria that evolved to alter the immune activation and expression of human immune cells in the gut. So cells that evolved over millennia in the human gut (and hence must be good enough because otherwise the human carriers would have died of disease) are able to tone down the immune response, allowing for a selective response of the immune system against at least some bacteria.
I recently research the Salk and Sabin polio vaccines. I am old enough to have been part of the early/first groups to receive Salk. But I did not recall why the Sabin came into being. I also had that course. It was given on a sugar cube.
I discovered the Salk was not that good; some still developed polio and some died. Salk was an injection.
Sabin discovered it had to be taken internally and work in the gut to be effective. The rest is history.
The gut was able to use this oral vaccine.
I find that idea very intriguing and I have upped my probiotics and added SBO.
I feel it is too late for me. I already have severe me/cfs, and have been going through the worst crisis since 2012. I was an agrarian individual, living on a farm most of my life with many animals and a lot of dirt. Yet I still got this. But if something could be discovered early enough, and a similar oral vaccination .. . . . . . I pray it happens.
Such insights DeJurgen. One would think – given it’s ecosystem properties – that gut researchers could learn from ecologists. How to shift an ecosystem? Give it too many or too few nutrients. Burn it (don’t burn it), flood it (don’t flood it), add new species which exploit an unexploited niche. In some ecosystems timing is so important. Get a gnarly species in there too early it stunts the growth of the whole ecosystem. The California grasslands were completely altered by the addition on annual non-native grasses which germinate earlier, grow faster, and keep the non-native perennials out.
Related to this is the concept of NETosis. That’s an alternate immune reaction mainly aimed at capturing fractured DNA / protein residues.
It is specialized in leaving very little permanent damage compared to other immune reactions. For example the aphtous ulcers being found upon biopsy to be filled with 80% T cells reveals a truly massive immune response. Despite that most of them heal without a single scar. I have some scar tissue in my mouth due to them, but it is no way near what is supposed to be when looking at how many I had over the years.
This is similar to what happens in ME: despite a supposed very strong increase in (auto-)immunity, there is few very clear and “massive” evidence of permanent tissue damage/scars/lesions. It shows up more in small fiber neuropathy. That is not totally surprising as nerve cells show far lesser regeneration rates then for example mouth mucosa.
NETosis, as the name says, creates NETs a bit comparable to spider webs. When that happens in the blood vessels I can only imagine it decreases flow speed of larger particles like RBC a lot. Decreased blood flow is a prime finding in ME too.
As a general/innate type immune response it leaves very few markers as to what the immune response is against. That’s again a common element with ME.
For mor information on NETosis, see https://en.wikipedia.org/wiki/Neutrophil_extracellular_traps
Thank you Cort for this massive report, especially when you were ill, hopefully you are feeling better?
Feeling a little bit better. It’s a weird 6 week long lingering cold that’s thankfully not getting worse but taking its time to move out.
Thank you Cort as always I look forward in reading your overviews (thoughts) of/on the happenings, articles and as well as others comments..ect..
My fav. Comment Award goes to Dr.M.:
His assertion that “a different immune reality” exists in ME/CFS – which he called a “chronic incapacitating inflammatory disorder” – hopefully perked up some ears.
Dr.M.,” I tip my hat to you,Sir and thank you!”
Now let’s see if they will rename CFS to CIID after all it is what it is …in reality CIID ?
Montoya is very good at getting his points across!
In my lifetime I have gave this Award to one other..
His Comment to me at a very young impressiable age was:
You are your own Doctor, Drs. are Detectives and Healers…hence why I went into the field of medince..thank you for your gift of wisdom..
World Renown Heart Specialist, Dr. DeBakey ?
Hi Cort, Thankyou for all your hard work.. Do you know if any of these researches, have ever looked at the HEAVY METAL load of CFS patients. As I have MCS/CFS, chemical sensitivity triggers all my symptoms and I have a high heavy metal load. After having my gut wreaked from the drug FLAGYL, and lived in a high pesticide/herbicide using area, 24yrs ago.
Heavy metal poisoning has a very strong capacity to increase oxidative stress / ROS levels.
I see plenty of evidence that the body *must* decrease energy production a lot as a protective mechanism if oxidative stress reaches too high levels, no matter if that oxidative stress originates from a strong immune response, metal poisoning, misfolded protein response or something else…
The only one I know who has is Ron Davis – in the severely ill patient study. If I remember correctly I don’t think he’s found much but we have a story coming up from an ME/CFS patient who’s problem was heavy metal poisoning and who is doing much better.
Not surprised on Naviaux. His work has always seemed intellectually indulgent to me. Interesting, sure, but I’ve never been convinced. The autoimmune research sounds much more promising.
Naviaux is so interesting to watch. I don’t know if his grand scheme is right – and there really wasn’t time to present it well there – but I can tell you that he’s also got the nuts and bolts nailed down. He can go toe to toe on biochemistry et. al with anyone. His recall and command of the facts is astonishing.
Cort, I was surprised that Naviaux didn’t provide an update on the new group who will be making Suramin. I’d love to know where they’re at in that process and if/when they will be trialing this in more autistic children and ME patients.
Thanks Cort. Great job of summarising complex ideas into layman’s language.
Thanks
Cort you don’t seem as hopeful as you used to be. Have you no faith left in NIH?
I have such mixed feelings about the NIH. Recently I’ve been looking more at funding which gets me into a really bitter mood about the NIH – and I am very disappointed that they are not doing more for us – very disappointed – which is made worse by the fact that I don’t know why. That can just gnaw at me – which is not a good thing.
My upset, though, lies in the gap between what the NIH could be doing for us and what it is doing. The NIH does splendid work. It funds huge, complex studies. It is easily funding more ME/CFS research than any other organization – I just want it to do more! 🙂
I really like the people I’ve met at the NIH. Love Vickie Whittemore. Very impressed with and had a good time with Avindra Nath. They are throwing much more than I thought into the intramural study.
I have a lot of faith that the NIH, when it finally really supports this illness, will make major headway with it.
It’s basically a love/hate relationship at this point (lol)
Cort, thanks for presenting this in layman’s terms so that our brains aren’t overtaxed any more than needs be. I’m curious, do you also suffer from ME/CFS?
Yes – to a lesser extent than most – but far more than I would ever wish..
Hi Cort,
Are any of the presentations online yet for viewing? If not will they be? Would love to watch them. With hope.
Cheers, Tamara
Yes they are
https://videocast.nih.gov/summary.asp?live=31636&bhcp=1
https://videocast.nih.gov/summary.asp?Live=31640&bhcp=1
I get it. I hover between 35% – 65% in severity. Also getting over a “cold” (takes longer for us than others). Thanks for all you do.
Thank you as always for wading through all of this and summarizing it! So very helpful when my energy/brain is so limited 🙂 Hope this cold passes soon for you!
Cort, thank you so much for your continuously comprehensive and coherent work reporting on current research of M.E. and progress–or lack of–at the NIH. Your reporting is immensely valuable and appreciated!!!
I wish we could just call the disease M.E. The term chronic fatigue grossly misrepresents describing this disease. And I wish we could change pem to pei, post exertion incapacitation. Perhaps we could use both, with the latter to apply to those who are more seriously affected, like myself. I am always incapacitated by exertion.
Which comes first, taking the disease seriously and, as a result, changing some of the terminology to represent that? Or changing the terminology to represent the seriousness of the disease in hopes that those in charge would take it more seriously? Those doctors and researchers who do take it seriously use the stronger language, as you reported from the conference, but it still seems to fall on deaf ears, doesn’t it?
When I describe my symptoms to people I encounter, I use the stronger language. When I communicate with other M.E. patients in my support groups, I use the stronger language.
For more seriously ill M.E. patients like myself, I encourage us all to use the stronger language to help us advocate for more support, more awareness, and more intensive approach by the physicians we encounter and organizations like the NIH.
I agree. The term “chronic fatigue syndrome” just so sticks in the throat. I use it only for search engine purposes. I don’t know how much a difference changing the terminology will make to researchers – to patients and the public yes, but fibromyalgia has an excellent name and it gets much less funding per person than “chronic fatigue syndrome” does.
It’s the whole basket – the NIH neglects a whole series of diseases- good name or bad – that mainly affect women, are largely invisible, cause enormous amounts of pain and fatigue and are not acknowledged as killers. They include ME/CFS, fibromyagia, migraine, POTS and others.
Cort, I hope you continue to feel better every day. Thank you for your on-going and great reporting, always putting it into a context that is easily understood.
For those of us long, long time sufferers,
the road has been indeed so frustrating and at times sad when we keep reaching dead ends. All we ask for is just a bit of the research money pie. It could make the difference.
You must feel at times like just giving up, letting go of the reins, letting someone else do all that you do. Always remember how much this community is dependent on you to give us a glimpse into what is happening in the research world. Without you, we would be in the dark, thinking that no one is out there at all.
You sacrifice much, my friend.
You are so greatly appreciated, admired and loved.
Thanks so much for your support. Following the research of David Systrom, Ron Davis, Maureen Hanson, Staci Stevens, Derya Unutmaz and others is exciting and inspiring. I feel like we have lots of good leads – what we need now is the oomph (funding) to tease out the bad ones and focus full bore – with big studies – on the good ones. In some ways we seem so close….
Dear Cort
Thank you once again for all your hard work. Just a small correction: yes, Goldstein talked about Mestinon, but you may recall that Dr Teitelbaum and Dr Holtorf who owned the chain of Fatigue-Fibromyalgia enters in various cities of the usa in the 2000s also used Mestinon in their treatment plans.
Of course, the issue is that many patients could not tolerate the drug, and became quite sick from it. Dr Systrom should consult with these doctors, who have lots of experience with this drug. Sometimes I get discouraged, because stuff just gets repeated…as it it was not ever done before. And the patients wait and wait and suffer.
Anyhow, thank you for your hard work. Best wishes to you.
I agree with Perrier on two counts: a deep appreciation of your hard work in an effort to keep us all informed, Cort, and the sentiment that many of these drugs that are listed as “new, effective treatments” have already been tried unsuccessfully by some of us who have battled this illness for decades (I include LDN in this category as well).
For those of you for whom these medications help, that is great. However, I do wish we could move past this repetitive state and explore more productive and efficacious treatments.
Thanks Perrier, I didn’t know they used the drug but I’m not surprised come to think of it. Systrom will have a study out on the effects of the drug. My guess is that the news will be similar to other drugs: it really helps some, moderately helps others and doesn’t work in a substantial group of people but we shall see.
Yes Cort, thank you for all you do. We would be lost without you. I always look forward to your write ups as they bring glimmers of hope. I hope you will be reporting Breakthroughs in the not too distant future 😉
I hope that for 25 years now. We’re not even close to it. This generation of ME patiënts will never know what they have had in this life. I even think they will never find the real cause of this disease.
Sorry if i am to negative 🙂
Completely understandable Gijs. The hard truth is that, unless we get lucky – and we could – I am thinking of something like Cortene here – we need a lot more funding to really get at this disease.
Well it’s a choice I choose hope!
On the other hand, as you note Eimear, resignation will get us nowhere, it leads to despair and inaction. Action guided by hope, on the other hand, is the only way out.
We are certainly making progress. Look at how quickly this field has elucidated problems with the mitochondria and energy production. A couple years ago we had very little on that. That is a major and important theme that research is cohering around. A presentation from the EMERGE conference – coming up – was just fascinating.
Hi Cort, sorry you’re still fighting the cold [tried zinc?] Thank you for the excellent summary and the link to Miriam Tucker’s article summarizing Dr. Systrom’s presentation. I also found his presentation the most exciting of all (and a lot of them were pretty exciting). Elucidated a lot of things. I’m hoping he has published some of the work he presented so that I can find it and take it to my pulmonologist. I think it would help explain why they keep saying I’m giving “poor effort” on the breathing tests though I’m almost bending over with effort to give my all. Maybe someone in this comment stream has seen something… Thanks.
There is just something about exercise studies and explaining exercise intolerance that just floats my boat! You should be able to find links to Systrom’s publications on these blogs. Note that the studies do not talk ME/CFS – they talk about exercise intolerance but as Systrom made clear, most of the people in those studies met the criteria for ME/CFS
https://www.healthrising.org/blog/2017/08/16/invasive-exercise-tests-chronic-fatigue-fibromyalgia-autoimmunity/
https://www.healthrising.org/blog/2016/07/04/exercise-intolerance-fibromyalgia-chronic-fatigue-pots-explained/
thank you!
Pyridoxine is an antinerve gas agent provided to soldiers in the Gulf War in case of exposure to nerve gas. It is very interesting that it would be useful as a treatment in ME/CFS.
A big fail on rituximab.
https://medicalxpress.com/news/2019-04-benefit-rituximab-chronic-fatigue-syndrome.html
Given the failure, hard to conceive that CFS is autoimmune.
That’s what I thought but I learned that it’s not true. Rituximab effects B-cell mediated autoimmunities but some autoimmune diseases are driven by the T-cells.
Thanks for that insight, Cort. Hope you are feeling better.
Still I wouldn’t dismiss rituximab research totally.
As Fluge and Mella do generally treat cancer patients and use the drug to IIRC treat certain forms of blood cancer, their initial “test audience” was a very selective subgroup of ME patients.
If I remember well (need to check), some forms of blood cancer carry both the “cancer” and “auto-immune” label. So if the result of rituximab for that specific subgroup ended up removing the cancer and helping about a third of patients to get rid of ME/CFS and another third seeing significant improvements were for real, then it could give an excellent opportunity to study what *can* be part of the complex web called ME for some well defined subclass of patients.
Given that understanding of the disease looks like a black hole at the moment, I wouldn’t like to see that opportunity wasted. Understanding what happened better could reveal part of the disease.
Let me explain my thoughts differently:
Many cases of ME/CFS are caused after a sudden onset of a (viral) infection. That somehow starts up a very quick acting and strong vicious circle that is very hard to break out off. Chances are best when fighting it off in the first year, but still are poor.
So far, it is believed that a strong immune reaction to the infection may play quite a big part in setting up that vicious circle.
Now what if the initial patients of Fluge and Mella had something similar but with a “blood type cancer and blood type auto-immunity onset”? Then maybe this different and probably less sudden type of onset started up a hard to break out vicious circle creating ME/CFS. The process being slower is no counter-indication against ME/CFS as there are many patients with a gradual onset type of ME/CFS.
Now if that would be the case, and if solving the combined blood cancer blood auto-immunity solved a large portion of those patients out of their ME/CFS then studying the process could shed more light on:
* what can trigger ME/CFS onset?
* is ME/CFS a disease where an chronically increased immune response is critical for ME/CFS to remain active after initial onset?
* how does the !first ever! drug that breaks that vicious circle for a well defined subgroup of ME/CFS patients at 30%+ success rate work to not only remove the initial onset but also the element pertaining ME/CFS?
* what unresolved elements do remain with those third of patients that only see a temporary improvement?
* what unresolved elements do remain with the other third of patients that sees no improvement?
As a bonus, it could help shed light on when the blood cancer / auto-immunity began, shedding further light on this types of cancers.
I see so much opportunity over here. If the initial results were for real in this group of patients, the 1/3, 1/3, 1/3 division offers incredible (near perfect!) potential to study multiple partial processes of this very elusive ME/CFS disease!
I know the “failure” of the rituximab study will make it a hard sell, but personally I would start two (at different research centers) large scale trials on rituximab treatment for ME/CFS patients that have (had?) this type of cancer.
As one does want to treat all these cancer patients as good as possible, setting up a double blind experiment may be a tat more difficult, but unless one tries to do so one does not know if it is possible.
If those initial test would be successful I’d pour millions of research dollars into digging deeper here.
OMG – you should be working in this field. We should get you well so you could contribute to it on a professional level. 🙂
@Cort: There is value to be found on finding out why promising research failed to duplicate, just as there is value to be found when practices that are a clear hit for some patients don’t work for others.
While it’s clearly a less comfortable path exploring those than walking straight successes, we have no shortages on failures to replicate partial successes in this disease. I see a true goldmine waiting to be explored. And the more failures, the easier it should get to find out what is needed to get it working. We are good here too.
dejurgen, are you a doctor? You are brilliant on your comments! I agree with Cort!!
No medical degree here. Just a patient getting creative due to necessity.
Thanks for the kind comments both ;-).
Dejurgen, The 1/3 for each type of response to rutuximab brings to mind Jarred Youngers still unpublished work on tracking fatigue. His results divided up into rough thirds, one third fatigue varied with CRP (infection), one third with fractalkine I think it was (immune) and one third still undetermined. https://youtu.be/QHIvcw9SNFo
Thanks Tiffany for the link!
Two things struck me about the 1/3, 1/3, 1/3 thing:
A) From a statistical point of view it’s near perfect to have 3 clear near equal sized subgroups.
B) If it were 3 consecutive processes that are set or not set this division could offer great potential for insight in how the disease forms and locks on.
Then there also was this vague feeling of recognition. I attribute great value to such feelings. They often indicate the subconscious mind has detected some kind of pattern. I don’t remember exactly but I feel like somehow there popped up a roughly 1/3th subgroup in several earlier “failed” viral researches.
So yes, I could find myself in a 1/3 viral or infection subgroup, a 1/3 immune subgroup and a 1/3 other subgroup.
My case has a very strong “gut” drive. If that would not turn out to belong to either the 1/3 viral/infection subgroup (pathogens leaking through gut) or to the 1/3 immune subgroup (immune reaction to pathogens and undigested proteins plus toxins) then this may be the 3th subgroup.
For now, I’d consider it a non auto-immune non-infection immune driven subgroup. It may also belong to a protein misfolding and NETosis driven subgroup.
All three subgroups would drive a major part of the immune system into chronic action. So it would not be really different diseases but more like one common disease with three different “front ends” to drive this disease if my “gut feeling” is wright.
Cort, I just tried to watch the 7+ hours of the conference presentations, and I just couldn’t do it. My head was spinning. I got through about 3 hours of presentations in two sittings (lyings, actually), and I couldn’t go on. It is immeasurably valuable to have your concise accounts of the presentations, and I’m so grateful to you for this distillation. Thank you!
Oh, and, I’m sorry you had that cold. I can’t imagine wading through all of this material with a cold on top of ongoing ME/CFS. I’m glad you stopped in Kansas City.
Thank you so much for your work cort!
I have recently found this study here and wondered if this could also be an approach for ME:
https://phys.org/news/2019-04-mice-mitochondria-alleviates-chronic-inflammatory.html?fbclid=IwAR1WuiEmcIG1tQdvdqoK76Kw29tpChR9rKHabmVgFhcxThOx7_2cIoJSSqE
Whoa! Now that is an interesting study given how things seem to be winding up. Thanks for pointing it out 🙂
No significant advances have happened in nearly 4 decades for a reason.
CFS patients are not sick in the way proposed by many.
They actually have highly accelerated immune systems and biological markers.
This is a gift of an advanced human being, not an illness.
So instead of trying to analyze the indifferences in the bio markers, in a negative light. They must describe what makes these differences in CFS patients so special and teach how to balance the energetic systems..Mind, body, Soul and Spirit.
This will allow CFS patients to thrive in the environment with their high frequency sensitivities.
Lovingly,
Michael
Hi Cort thanks so much for your report it’s very much appreciated. You make everything so clear and easy to understand. Wanted to know if you have heard of the supplement Moducare for modulating autoimmune conditions? I might try it because it seems quite promising. Thanks again.
Thank you, Cort, for this excellent summary. It is difficult for many patients to watch the presentations on video, so the summary is really valuable.
Thanks Sten 🙂
We should dispatch with the notion that CFS is mysterious. There are slew of fatigue disorders like post-viral fatigue, cancer fatigue, post-concussion fatigue, over-training syndrome, and most of all, aging. Before my mom passed away in her 80s, she looked just like a CFS patient. She would have a burst of energy one day and run around doing house-keeping. Then she would be bed-ridden for the rest of the week.
Calling it mysterious can cause people to search for novel pathology in immunology, gut microbiome, metabolism, etc when studying the fatigue and exercise intolerance that comes with aging or over-training may yield more productive results.
Great point TK. I think what you said is why Ron Davis thinks that solving this will open up new insights into so many other diseases. Plus studies in both ME/CFS and FM point to the possibility of premature aging (yuck!).
Over-training is such an obvious cohort…If this field was bigger I’m sure we would see that and when it is bigger I’m sure we will. The very interesting question to answer there is what is different between athletes who overtrain, stop training and recover, and people with ME/CFS who stop moving around – and DON’T recover…
Just wanted to take a minute out of my day to say how much I value your site and how much every single person I’ve ever met with CFS values your site. Sincerely, thank you. You’ve helped.
Thanks Bridget. I’m very glad the community gives me the support to be able to dig into this stuff 🙂