It’s not pretty! The print is abysmally small, the margins are tiny, long stretches of text are common (it abhors paragraphs), the images are jammed together, and the formatting is designed to make you work and is decades out of date, but the fact is that when you’re one of the top research journals in the world, you can make your own rules.
PNAS or The Proceedings of the National Academy of Sciences of the United States of America – the official publication of the National Academy of Sciences – has been described as “prestigious”, “renowned”, “high impact” and even “sedate” (which in the research world is not necessarily a pejorative term).
PNAS, which touts itself as the second most cited scientific journal in the world, publishes a wide variety of scientific content. One statistical analysis concluded that, “Three journals have by far and away the most overall influence on science: Nature, PNAS, and Science, closely followed by the Journal of Biological Chemistry. ” Nobody else was even close.
PNAS may not be pretty – it’s probably intentionally unpretty – but getting published in it gets the word out. Since Rahim Esfandyarpour (the lead author) and Ron Davis’s nanoneedle paper was published in the most recent edition of PNAS, numerous media outlets have picked up their findings. I got to talk to both of them.
The paper immediately makes clear two things: the lack of a biomarker makes getting diagnosed with chronic fatigue syndrome (ME/CFS) a “lengthy and costly” affair (which, of course, precludes patients from getting treatments when they may help the most – while the disease is still new). From a research standpoint, the paper noted that lack of a biomarker may have tragic consequences if “heterogenous samples of patients” with “only marginally similar conditions” are being included in studies.
Obviously, getting a biomarker would fulfill a huge need, and other possible biomarkers may yet pan out. None, however, potentially tick off all the boxes (potentially very cheap, easy to use and very accurate) as well as Ron Davis and Rahim Esfandyarpour’s nanoneedle.
The first nanoneedle papers in 2013 indicated that the needle was originally designed as an “ultra-sensitive, real time” biosensor designed to cheaply detect biomarkers such as proteins inside (or outside) cells at a far more sensitive level than had ever been done before. One very promising feature of the nanoneedle was its ability to quickly identify biomolecules without needing to label them first (and then use a tool to detect them).
By 2016, a redesigned and improved nanoneedle was presented as a rapid, inexpensive, high throughput, real-time, label-free, highly sensitive alternative to the expensive or less sensitive devices available.
Then came chronic fatigue syndrome (ME/CFS). Focusing on the unusual problem of energy depletion following exertion (post-exertional malaise (PEM)) in ME/CFS, Davis and Esfandyarpour added a salt stressor to ME/CFS samples and used the nanoneedle to measure electrical impedance (a measure of electrical resistance). (The salt solution was used because it requires cells to generate a lot of energy (ATP) to maintain the appropriate salt concentrations inside and out.)
The increasing electrical impedance or resistance seen in the ME/CFS but not in the healthy control samples suggested Davis indeed induced a state of post-exertional malaise in his ME/CFS samples.
That was not surprising; what was surprisingly was how definitively he has done it. In one of the most evocative images yet to come out of the ME/CFS scientific literature, the graph below shows a dramatic separation between patients and controls. With the needle packing tens of thousands of biosensors, adding up to millions of data points, Davis has every reason to believe that the finding is robust, indeed.
Causes
A major question involves determining what biological process is producing the rise in impedance in the ME/CFS patients’ samples. Esfandyarpour pointed out that while answering this question is obviously biologically important, it’s not critical for diagnostics or drug screening to know what’s going on. Many drugs work, after all, without our knowing exactly why. Similarly, all a good diagnostic test really requires is that it be specific to a disease.

Ron Davis – senior author – on the left, Alex Kashi – second author, and Rahim Esfandyarpour – lead author on the right
(Esfandyarpour said “many” different drugs, inhibitors, enzymes, and substrates involved in cellular energy production have been tested to see if they can return ME/CFS samples to normal functioning in the nanoneedle. Two thus far (he didn’t say which ones) have returned the ME/CFS samples to normal functioning.)
Still, learning what the nanoneedle is showing should give us an idea of what has gone wrong in ME/CFS. There are several ways to do this – some of which do not require the nanoneedle. Different kits could be used to measure how the cytokines, etc. in the sample change following the introduction of a salt stressor. Adding an inhibitor to the sample that affects certain parts of the cells and assessing how the sample responds could provide clues as well.
One might think the Seahorse – a commonly used tool in ME/CFS that assesses cellular energy production – would play a key role, but it doesn’t. Because Seahorse cannot incorporate the potentially ever so important ME/CFS plasma into its testing regimen, if something in the plasma of ME/CFS patients is causing problems with cellular energy production, it will not be able to pick it up.
Esfandyarpour believes something in the plasma may play a critical role in ME/CFS. Davis’s team has been filtering out substances in an effort to learn what factor that might be. Nothing definitive enough to announce has been found yet, but exosomes (small extracellular vesicles involved in communication between cells) are a possibility.
Davis said he was encouraged by the idea that a substance in the blood is causing problems. Given that, so far as we know, this is rare in diseases, if such a substance is present, it may be quite specific to ME/CFS.
They’ll feel confident about such a substance if: a) when they remove it, the response returns to normal; and b) adding that substance to healthy cells causes their impedance levels to rise. If the substance turns out to be exosomes, the next step is to determine what in the exosomes is causing problems.
If something in the plasma is found to be playing a crucial role in the abnormal response of ME/CFS cells, a next step will be to determine which cells it’s affecting.
A Diagnostic Test for ME/CFS?
The nanoneedle passed a big hurdle when it was able to so definitively distinguish ME/CFS patients from healthy controls. The forty people in the study may not sound like a lot, but it’s not the quantity that tells the tale in this case – it’s the consistency of the result.
Probabilities tell the tale in research. The highest probability that is considered significant or meaningful in medical research is a less than one in twenty probability (p<0.05) that a result occurred by chance.
Studies are designed around the need to hit that probability. Miss it even by a small amount, and your results are worthless. Statistical analyses are done to determine how many participants are needed to hit that probability target.
There was no need for that in the nanoneedle study. The statistics indicated that the probability that the study results were due to chance ranged in the billions. That kind of probability is more commonly seen when trolling for a mega lotto win than in biology. Put another way, the nanoneedle study was actually far, far larger statistically than it needed to be to demonstrate that the ME/CFS samples could be differentiated from the healthy control samples.
Davis will continue testing samples, but for him and his long, long acquaintance with statistics – the question as to whether the nanoneedle can differentiate ME/CFS patients from healthy controls has surely been answered.
Differentiating ME/CFS from other diseases is the next step, but the big takeaway is that Davis appears to have produced a test that irrevocably demonstrates that people with ME/CFS have a biological illness. Developing a biological test for ME/CFS, Davis said, was his biggest goal.
Davis’s next task is to differentiate people with other fatiguing illnesses, such as multiple sclerosis (MS), from ME/CFS – a trickier project. Davis noted that some of the first symptoms to show up in MS are similar to those seen in ME/CFS, and that some people diagnosed with MS have been shown to have ME/CFS and vice versa.
It’s also possible that some people with MS will develop ME/CFS at some point. This type of disease convergence is not uncommon with fibromyalgia. Numerous studies indicate that a significant subset of patients with rheumatoid arthritis or other chronic pain disorders also have fibromyalgia. It’s possible that the same is true with ME/CFS and also some very fatiguing autoimmune diseases.
Davis, as Jarred Younger has, brought up the possibility that people with MS and ME/CFS – two illnesses often jumpstarted by an infection (and perhaps the same herpesvirus infection) – may start out on the same pathways but then diverge at some point.
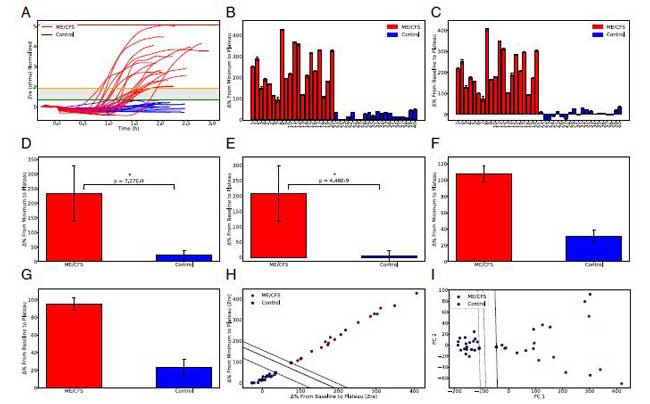
Notice the stark separation at all levels between the patients (in red) and the healthy controls (in blue).
Having some people with MS test similarly to people with ME/CFS on the nanoneedle is not a deal-breaker, though. Far from it. It’s not uncommon for diseases to share diagnostic markers. Doctors get around this by using a series of tests to diagnose patients. If some people with MS look like ME/CFS patients using the nanoneedle, then a brain scan which shows neuron damage in the MS patients will be able to differentiate the two diseases. People with MS and ME/CFS will be positive on both tests, while people with ME/CFS without MS will test positive only with the nanoneedle.
An even better testing regimen would use another anomaly more commonly found in ME/CFS, such as the red blood cell (RBC) deformity work the Open Medicine Foundation is funding. Since problems with RBC deformity probably do not occur in MS, a positive nanoneedle and RBC deformability result should be enough to differentiate someone with ME/CFS from someone with MS or many other diseases.
The point is that a diagnosis often does not rely on a single test: a single definitive test isn’t needed to diagnose a disease.
A handful of tests could enable ME/CFS to be clearly differentiated from other diseases in the fairly near future. It won’t take thousands of patients to show that either. So long as you have well-defined patients in studies produced by good researchers which produce good results (i.e. good probability stats), neither the number of studies or the number of patients need to be particularly large – the results simply need to be trustworthy.
Metabolic Trap
Davis has begun to separate out dendritic cells and test them to see if the metabolic trap is present. He brought up an interesting possibility – emphasizing that it was only a possibility – that if the metabolic trap is present, it could lay the groundwork for an autoimmune process to manifest itself. This is because dendritic cells are responsible for shutting down autoimmune producing B-cells.
If the metabolic trap is present in dendritic cells, it’s probably mucking up their ability to knock out bad B-cells. Furthermore, because the IDO1 pathway plays a role in immune system regulation, it wouldn’t be odd at all for pathogens to take a swing at knocking it out in order to evade capture. That – plus a possible genetic predisposition to problems with this pathway – could tell the tale in ME/CFS. That said – at this point it’s just a possibility (but it’s nice to have possibilities) – these are tangible ways of potentially explaining this disease.
The NIH Attempts to Take Credit
Director Francis Collins immediately jumped on the nanoneedle finding to trumpet the NIH’s role in funding the breakthrough. That was, in part, true – the NIH did provide funding for the early development of the nanoneedle – but then there is a second part to the story. The NIH later dropped its funding for the needle and it did so just about the time it was starting to be used on ME/CFS.
In 2016, citing a renewed emphasis on ROI grants, the NIH jerked Davis’s longstanding technology grant, undercutting his nanoneedle and other research. Ironically, for all its emphasis on accelerating ME/CFS research, the NIH, inadvertently it must be said, pulled the plug on funding for one of our most important ME/CFS efforts. In a recent conversation, Davis looked back:
“I used to have experts in almost all areas. We were on a roll developing all sorts of things. I don’t have that anymore because the NIH terminated my big grant on technology development.” Ron Davis
It turned out that the NIH no longer shared Davis’s emphasis on developing cost-effective tools that help people and reduce the cost of health care.
So when Francis Collins himself tweeted the good news that the NIH had funded Davis’s effort to develop a new test for ME/CFS…
“In @stanfordmed study, researchers developed new blood-based test that positively identified participants w/#MECFS. If findings can be validated in larger study, it may provide a diagnostic tool for clinicians & a target for new ME/CFS Txs. #NIH-funded https://stan.md/2IU7nYN“ Francis Collin
He crossed a line for Janet Dafoe and patients who went on something of a twitter rampage at the director’s gall. One wonders how much further we would be if the NIH had continued their support. Janet pointed out that the NIH hadn’t funded any of Davis’s ME/CFS nanoneedle work – which was, of course, what the paper was on; everything was funded through patient donations to the Open Medicine Foundation.
“That NIH grant was TERMINATED about 2 years ago because NIH said they didn’t want to fund that technology anymore. That grant had funded the initial development of the nanoneedle on cancer cells & bacteria. All the ME/CFS work started AFTER that & was FUNDED BY PATIENTS@OpenMedF. You don’t get credit for this good news for ME/CFS” Janet Dafoe
Then she pointed out that the NIH had muffed another opportunity to fund ME/CFS nanoneedle work when NIH reviewers savaged Davis’s NIH application, in part because they objected to the new technology he was using.
“In fact, Ron Davis applied to NIH for the nanoneedle work and was turned down! Reviewers were very critical and said it wouldn’t work, in spite of good preliminary data. STOP saying you don’t fund ME/CFS because you don’t get good grant applications! Patients funded this!” @OMF Janet Dafoe
Attacking the competence of Ron Davis – who’s been called a “frequent provider of disruptive core technologies“, and played a key role in the Genome project, etc. etc. was probably not the brainiest move by the NIH reviewers, who are now presumably busy wiping the egg off their faces.
The NIH is soon going to get another opportunity to fund nanoneedle work in ME/CFS. With the PNAS paper under their belts, Ron Davis and Rahim Esfandyarpour will be applying for another NIH-funded nanoneedle grant this June. Perhaps the third time will be the charm for the nanoneedle and the NIH.
Resources, Resources, Resources
With its potential ability to serve as a diagnostic tool, a drug screening tool, a tool to help determine if something in ME/CFS patients’ plasma is knocking their immune cells for a loop, a tool that could greatly enhance research studies (by ensuring that only ME/CFS patients are in them), the nanoneedle obviously presents immense possibilities.
The work seemed to be going slowly, though, and I was curious to find out why. Asking if the funding or resources were holding the work back elicited a big laugh from Rahim. Funding, not technology is the limiting factor, he said.

In its current iteration, the nanoneedle is a tortoise that, Aesop’s fables notwithstanding, really needs to be a hare.
It turns out that as sophisticated as the nanoneedle is, in some ways it’s rather primitive. In its current iteration, the absolute fastest it can work is to test two samples over a couple of hours. That’s not great in an instrument that needs to be involved in testing other diseases, in drug testing, in assessing plasma factors, in assessing the pathophysiology behind the reaction (mitochondria, membrane problems, immune issues, etc.). The drug testing alone is incredibly complex: Davis needs to test different drugs, different doses of drugs, drugs in combination, etc. in different patient samples. With its thousands of sensors, the nanoneedle may be a technological work of art, but it’s a sludge when it comes to processing samples.
Fortunately, a solution is present. Unfortunately, it will take something the ME/CFS field hasn’t had a lot of – resources. Developing a high throughput system that’s able to test dozens of samples at once is, Davis said, a top priority.
There’s apparently no mystery to developing that system. It’s simply a matter of resources. When asked, Esfandyarpour said that designing a high throughput system presented no technical challenges – the only challenge is funding – and not necessarily that much funding. When I pressed him, Esfandyarpour would not say how much money was needed, but when I asked if it was $1 million, he said no, it’s not nearly that.
Developing a high throughput system is the first and necessary step to maximizing the potential of the nanoneedle, but it’s only the first step. The logical conclusion to all of this is the transformation of the lab instrument into a portable one that can be used in a doctor’s office or medical lab. Davis said that doesn’t present any technical challenges either (!). If everything works out, a portable testing unit could be ready within two to three years.
The fact that what is holding us back is a matter of resources makes the NIH’s decision to end Davis’s ongoing technology grant sting all that more. In the end, though, the fact that its resources – not some technological breakthrough that’s needed – is actually very good news.
The resources are out there. I know of very wealthy people with family members with ME/CFS who could easily fund this or other research efforts and aren’t. I don’t know why. It doesn’t make sense to me, but the fact is that the more we spread the word, the more we get our story out, the more people we contact, the more people with resources who do want to contribute will show up.
The PNAS article will surely help with that, and it will help with the NIH. Davis now has a publication in an excellent journal to back him up. He didn’t have that before. “Our goal,” he said, “is to keep making it harder and harder for the NIH to ignore us.” The PNAS study just made it that much harder.
Resources are a problem, but what’s worse is a lack of possibilities. Thankfully, we’re not lacking in possibilities right now. The nanoneedle is just one of an array of possibilities the Open Medicine Foundation is funding under Ron Davis’s direction. These include the muscle biopsy work at the new Collaborative ME/CFS Research Center at Harvard, the spinal fluid work at the new Uppsala Research Center, the red blood cell work at Stanford and San Jose State University, the work on the metabolic hypothesis, and genetics and the severely ill patient study work going on at Stanford.
All are important. (If you want your donation dollars to specifically go to the nanoneedle project, though, – which to me at least seems to hold a special promise – note that with your donation.)
Rahim ended our talk by thanking the patients. He said, “I think we have very promising results and do not lose hope”. Then he said something interesting. Many diseases have had to fight for recognition over time, and in many of them, something happened to flip the research community from apathy to action. When it did, the resources of the research community were brought to bear.
He believes that will happen with ME/CFS. I couldn’t help thinking that the nanoneedle could – if everything works out – provide one of those tipping points for this disease.







Why on Earth has no one looked at
Dr. Ritchie Shoemakers biotoxin
pathway labs. Many of which I believe
deal with the “Innate Immune system.
I got sick from mold. I have CFS.
The hub of his pathway is a hormone
called M.S.H. This hormone affects
most of the symptoms that affect
CFS/ME, as well as fibromyalgia.
There are labs, one can follow progress or remission with his labs.
Why try to rewrite the alphabet???
Pride? Take a look. Call him.He lives
in Pokomoke Maryland.
There are many top docs in this field
using his labs and protocols.
Sincerely, Pam Lewis
If you want my experience with this
topic and doc. Happy to try to help.
I was a critical care RN for 32 yes.
Very promising. As highlighted a key next step is to see whether this response is unique to CFS or might be a ‘by product’ of a number of inflammatory disorders.
Another big question is whether the result is fundamental to the illness (correct it and you help or cure the illness) or just a downstream signal.
At the very least I am sure this will advance our understanding of the illness in some form.
I am not sure why the next step has to be to differentiate this from other diseases. Couldn’t that wait? Lots of diseases use the same diagnostic tools and the same treatments.
To me, the next step needs to be a clinical trial of plasmapheresis or transfusions, or IVIG — and see if people GET WELL or improve.
They are pretty sure there is a problem with the plasma. There are existing treatments which treat plasma, from “cleaning” it to removing and replacing it. Treat the plasma with the existing treatments, in a small trial, and see what happens! Please, let’s get on with it before we are all dead.
The “other diseases” differentiation has some sort of benefit, I’m sure; but cleaning, treating, or replacing the plasma might tell us if there is a treatment that will HELP US, right here and right now. That’s what we need, and that could be done very easily.
I’ll be the first to volunteer. I’ll even pay my way, and waive all liability.
Why one thing catches on in the research or medical world and another doesn’t I have no idea. I do know that history shows that introducing new things to medicine often takes time. Please contact me about a blog 🙂
The ME/CFS researcher community has the same problem with toxic mold as the
CDC/NIH has with all the evidence that has been “overlooked”
If they admit to Dr Shoemaker’s work now, it is automatic admission they have
been “overlooking” how toxic mold actually started the CFS syndrome.
So they go on acting as if this doesn’t exist.
Yes how would we access it.??? Something I was thinking about. Hopefully the doctors who have ignored cfs patients for decades would be so stunned that they could be influenced to at least give the test
So glad to see the comments on the liver. Why no comments
On toxic chemicals??
.
The Shoemaker was asked years ago by Scientists from the NIH to prove Mold was any Cause of CFS he never provided proof, as far as I know now there is No Cause of CFS yet found.
I have worked with Dr. Cheney who worked In the same town hoursShoemaker. When I was working with Cheney he discussed trading patients with Shoemaker. They found that the patients had similar physical and psychological problems but Cheney’s patients Differed because they had a sedimentation rate below 2. ESR< 2
Dear Pamela, I had severe dangerous mold illness and got over that part. It didn’t make me heal entirely though. Now, after 7 years of illness and treatments, I don’t think the horrible Mold-element is the initial main trigger. There are other factors present which make us as a consequence react to mold. Shoemaker was an important step but I think more info has come about now. My experience is that Epstein Barr and Herpes are star actors in these complex interactions. Whatever has been found, anything important in knowledge is going to, one day, come together and the whole puzzle will close in. In that light it is fascinating how core scientists findings are closing in with the findings of the Functional Medicine Field. We are moving ahead!
Very well written article for the most part however, before celebrating these findings, we have to look at the specifics of the study and its limitations. The first is thst is we only know that it’s sensitive, we have no idea if it’s specific. A test that has zero specificity is no good to anyone.
It is a very small study that only distinguished healthy controls from those with a clinical diagnosis of ME/CFS.
Now, they need to look at many other disease states with a “gold standard” to distinguish ME/CFS from other diseases. I also need to emphasize we’re not looking at cause-and-effect at this point, we’re only looking at cross-sectional information.
I suspect the nanoneedle test will have a very high false positive result as the potential for this test to be positive for any disease is very high.
We also have to keep in mind that they looked at moderate to severe ME/CFS patients. At later stages of this condition such patients have a number of significant comorbidities and a number of polypharmacy issues. We don’t know at this point if the test is positive for specifically ME/CFS or any of the many other co-morbidities or their medications. And as I said above, this test could be positive for a number of other diseases/conditions, hence not specific.
There will have to be much, much, larger studies to explore and validate all these questions through receiver operator curve stats, etc.
Hence, they are a long way from a diagnostic test that is specific (vs sensitive). Not trying to rain on hope, but, it is important not to create false hope for temporary glory. And, it is important as a to make sure to explain the full context for understanding the results of a study, particularly for those who are not familiar with how diagnostic tests are developed. The probability exclamation was very good, but it doesn’t tell the whole story. What is also missing in the probability explanation, is the likelihood of finding this marker in a general population vs not. Again, very involved very lengthy statistics and testing go into developing tests as stated above. Consider this and the current day controversies over mammograms …
Thanks Neilly,
This is a bit above my pay grade!
My interpretation is that this test primarily about showing a distinct biological abnormality in ME/CFS vs healthy controls. I think the main takeaway is that its a very strong test showing that something – we don’t know what – very biologically different different in the ME/CFS patients. I actually don’t know the breakdown in severe vs moderate patients. If its in the paper maybe someone can provide that.
And specificity – doing the test in other diseases – is next and we don’t know at all how that will turn out. We have no data yet! Given the potential overlap between ME/CFS and other diseases that’s clearly not an easy question to answer.
Ron did design this test specifically for ME/CFS and the PEM that appears to be at its core. While we don’t know what the test is showing yet, if it is showing that ME/CFS patients immune cells are unable to produce enough energy to handle the salt stress; i.e. are put into a state of PEM – then I think the test has a good chance of being specific as well as no other disease appears to produce PEM like ME/CFS does. Fingers crossed!
This is not a diagnostic test but hopefully it’s the start of one – and a very good start.
Thanks for putting hope back in this conversation. I appreciate any ray of hope in this journey.
“Ron did design this test specifically for ME/CFS and the PEM”. The article only says that the impedance increased after the salt stressor is added. Did the impedance abnormality lasted several days? If not, I wonder how it tested specifically for PEM.
In the paper it says 5 of the ME pts were severe and the rest were moderate. They mention differences in results in the severe vs moderate with respect to the impedance change
Thanks for all your work. It’s really fantastic!
Thanks!
I’m not too concerned at this stage.
If you look at Jonas Bergquist’s presentation at the NIH Conference last month you’ll see that he presents evidence that ME is similar to MS. From memory the inflammatory markers in the cerebrospinal fluid samples showed lower levels of inflammation in ME compared to MS, but higher levels than healthy controls.
So possibly some people with MS will give a positive signal on the nano-needle. I.e. they will have mitocondrial fragmentation caused by something in exosomes in blood (Prusty – NIH Conference last month) resulting in a positive signal on the nano-needle. I think this will also apply to Lyme. I’m not sure that there are other disease which are currently misdiagnosed as ME and which would give a positive signal on the nano-needle. If false positives turn out to be a problem with the nano-needle then there are other tests which may separate these out – intracellular phenylalanine (see my comments below), mitocondrial fragmentation —. Also, as time goes on I’m confident that more biomarkers will be found.
Potentially the nano-needle/other tests will enable doctors to assess SS-31, and copaxone, as treatments i.e. before you get the drug they could check if your a likely responder.
As stated in the paper/by Cort the nano-needle etc. will enable researchers to identify homogeneous groups to study.
As highlighted in the article lack of funding is the main concern.
ME Action are lobbying for the validation/development of a diagnostic test for ME https://www.meaction.net/…/announcing-millionsmissing-2019…/
Thanks Francis – check this blog out for more on the spinal fluid differences between MS and ME/cF
http://simmaronresearch.com/2015/04/spinal-fluid-study-finds-dramatic-differences-chronic-fatigue-syndrome/
One conclusion was: “The fact that the alterations in the immune factors in the ME/CFS were almost as extreme as in multiple sclerosis – a disorder characterized by severe central nervous system dysfunction – suggests a major pathology is present in the central nervous systems of ME/CFS patients.”
Thanks Cort.
There’s a bit here about exosomes crossing the blood brain barrier; “extracellular vesicles can readily cross the blood brain barrier” [https://www.frontiersin.org/articles/10.3389/fnins.2017.00026/full]. So the inflammation of the central nervous system, in ME, could be the result of the exosomes [Prusty/Davis] crossing the blood brain barrier.
So ME might just have got a whole lot simpler i.e. now that we can see that the signalling (affecting cellular energy) is caused by something in exosomes (micro-RNA).
Possibly Ron Davis’s work will redefine MS, Lyme and ME [role of exosomes]. That may have benefits for ME i.e. make the delivery of treatments more attractive to drug companies and help to secure research funding from Governments. You did a great article announcing NIH funding for the study of microRNAs in ME in 2015 –4 years ago — what might have been! [https://www.healthrising.org/blog/2015/08/23/lipkin-nih-grants-chronic-fatigue-syndrome-2015/].
Just what I was going to say, only better. For fairness though, Cort mentioned combining it with other tests to specifically diagnose CFS: CFS if and only if A & B & C. Nano-needle, if proven in larger study against healthy control, could become another tool in the shed we could use.
I would add depression and atrophy as conditions to be tested with nano-needle. These are two conditions that some people still think are the causes of CFS and it’ll be important for the diagnostic tests to be able to separate CFS from them, especially if the diagnosis is to be used for the disability purpose. And disability is just about the only practical use of the diagnosis since there is no treatment available for CFS.
Which means I obviously disagree with Ron Davis that the criteria that are currently in use can result in “heterogeneous” samples and tragic consequences for CFS studies. In fact, relying only on diagnostic test to define CFS could lead to false-positive CFS samples if you preclude qualitative criteria like brain fog or PEM which are the hallmark for CFS. Unless you can also come up with objective test for them, of course.
Cort, I always love to read your explanations of these studies. You really clarify it for me. I watch the presentations on You Tube and try to understand the gist of it. Then your article helps me increase my understanding. Thank you for this work!
Really, really good article and very hopeful. You explained in detail very well. Davis’ group had already thought of all the ‘what ifs and objections.’ They are brilliant people. I appreciated learning the background of the nanoneedle, as well as the funding and lack thereof, interesting if infuriating. thank you
I was fascinated by the background of the needle as well 🙂
One key technology that may be important is Aethlon Medical’s Hemopurifier. It is a filter (substrate) that can filter out all exosomes from plasma. It’s a dialysis filter (and likely available in a lab-bench form) so it could filter CFS patients blood directly (possibly temporarily eliminating symptoms). It could also filter exosomes exclusively on sample plasma to test in the Nanoneedle.
thttps://www.aethlonmedical.com/the-hemopurifier/the-hemopurifier-in-cancer
There are also several “Exosome Release Inhibitors” compounds that can be tested on the Nanoneedle. They inhibit exosome release by disrupting sphingolipid metabolism which mediates exosome release. (Is this a connection to Naviaux’s results?!) This paper describes an exosome release inhibitor that dampens sepsis inflammation. Sepsis (and CFS) may be a exosome driven pathology.
https://doi.org/10.1016/j.bbadis.2015.08.010
Whoa! Thanks for passing that on. Ron said that Alain Moreau is looking into exosomes and Maureen Hanson is as well. If exosomes are the issue Ron didn’t think finding out what was in them would be that hard. From your comment it sounds like the medical field has already figured out ways to get at them.
The German Oncologist working on CFS used a treatment on CFS patients a very expensive procedure to do with exosomes, some responded but
relapsed not long after & I have heard something like 30,000.+ Euros for treatments that ended up failing
Thank you!!!! I can’t wait to read your links. 🙂
Excellent article, Cort! I think I can answer the question as to why more people of means don’t donate more abundantly to CFS research. If people with CFS were able to have the testing done on themselves that the researchers are doing in their studies, and their results were consistent with what they are finding in their research, I believe there would be a large increase in donations. If I was able to be tested with the nanoneedle and I tested positive for CFS, and I believed the test was legit, I would write a substantial check. I’m sure many others feel the same way.
I think you could be right, people who hand over a lot of money generally want something in return. That may just be a result in which case they will come on board a bit further down the line. Also I think there are many people who only think about a cure and I wonder if this is because of comparisons made to Aids. There is no cure for MS or Parkinsons but they have tests for proof and look at how much better they are treated by the medical profession and social services.
We need a biomarker just to get money we are entitled to now and it is still the foundation to everything else.
Thank you Susan – great point! As Ron noted in our talk – even our families often don’t believe us and who could blame them when doctors treat this illness the way they do. We’re behind the 8-ball from the beginning. I believe the stigma and lack of belief in this illness must stop family members from participating in advocacy and funding research. Clearing the stigma away with an accepted biological test will reap dividends!
As Rahim said, illnesses often have turning points and a biological biomarker would certainly be one. Let’s hope Davis can ramp up the nanoneedle testing and the excellent results continue and the needle will end up being able to differentiate ME/CFS from other fatiguing diseases. If it can do that we will have made a huge step forward – a step that should inspire people to donate to the research and participate in the research.
Thanks for making clear that this is when NIH pulled out on Davis et al. What timing NIH has. Davis is one amazing scientist, and he is 77 years old now, still in peak form. Reading the Wikipedia article about him, and then reading some of the references on line, I learned that he has an amazing past, as to his scope of science, his patented devices for biotechnology like this nanoneedle, and his breakthrough work in genomics. He comes from Illinois, my state, that was news to me too. I hope his team keeps on working, and the move of the lead author to UC Irvine does not impede their fast method of moving right along with a rational proposal for their continuing studies. I hope another lab can reproduce what they have done, but who else has a nanoneedle?
Given Ron’s background it’s no surprise that something like the nanoneedle would come out of his lab. What’s really inspiring to me is his focus on relieving suffering by producing inexpensive tools medicine can use. I was not happy to see Rahim move to UCI either but in my talk with him it was clear that the nanoneedle work is moving forward and it was good to hear that he and Ron are working on an NIH grant application.
Given what I read about the invention it doesn’t appear that anyone has created a nanoneedle like the one Davis and Rahim have. It seems to solve several issues in the field at once.
Now I get what’s the idea behind the saline stress test. Moving salt across cell borders indeed cost a lot of ATP. Makes sense. Thanks for explaining it.
Their is still a lot of work to be done, but for the very first time I am hopeful we will have a working and affordable test to at the very least show that we are really biologically ill, even if this test were not able to discriminate between ME/CFS/?FM? and other serious diseases like cancer, Parkinson…
As to “Director Francis Collins immediately jumped on the nanoneedle finding to trumpet the NIH’s role in funding the breakthrough.”
It shows they feel how important this breakthrough is and that they can’t be seen as having completely missed that boat. Having them publicly turn their communication 180 degrees on a “controversial topic” in a single day is not common at all.
I wonder what Francis Collins role is in it here. Many may cry wolf at him, but he must have known this could create a storm of critic against his person and the NIH.
*IF* he truly wanted to invigorate ME research for years and couldn’t surmount the opposing forces within the NIH this may be a rather brave move.
It would be nothing short of saying to all inside the NIH who oppose a real big step forward in ME research by trying to dismiss ME as a disease in the mind
“You made a fool of me, NIH director, once. You know this can’t happen a second time, given the breakthrough and missed opportunities making a denial position totally untenable. Raise hands who on the next round is willing to take blame of making a total fool out of me when we denied to fund another big breakthrough. Sign your conditional firing papers and leave them on my desk already.”
Thank you for presenting the other side DeJurgen – that while Francis’s tweet got a lot of negative reaction – it also drew attention to the paper. He has publicly supported ME/CFS before – let’s hope that he does a Director’s Blog on this finding 🙂
Another potential diagnostic test is intracellular phenylalanine i.e. measured by RAMAN spectroscopy; you can see the paper here [https://sci-hub.se/10.1039/C8AN01437J ].
Again, from memory this is reliable; there’s a larger study underway (may even be completed).
Other options include mitochondrial shape (Prusty – NIH Conference last month).
So separating ME from other diseases may be possible using one of those tests.
Cort I was slightly surprised by your reference to MRI i.e. to separate ME from MS. MS is associated with demylenation of nerve cells (detected using MRI); however, recent MRI studies have shown abnormalities in ME. I wonder if MRI can detect the difference between ME and MS?
Regarding which cells are affected by the exosomes (or whatever), Karl Morten demonstrated the effect on muscle cells [https://www.youtube.com/watch?v=Yb3wbQt1BCc]; this was invitro work (muscle cells in culture) so not definitive. An expert of exosomes may be able to advise how they target cells (I’ve seen something about surface receptors); i.e. whether they are specific/general.
Regarding public funding for the development/validation of a diagnostic test – don’t let the European Union off the hook. The EUROMENE biomarker group are due to make recommendations next April (2020) on potential diagnostic tests [Dr Eliana Lacerda, Vice Chair of EUROMENE, speaking at 2019 Emerge Conference].
ME Action are lobbying, in Europe, for the validation/development of a diagnostic test for ME https://www.meaction.net/…/announcing-millionsmissing-2019…/
Great article.
ugh…Francis Collins, what a nerve….!
I’ll be he didn’t know what he was stepping into. I imagine that he’s met with few communities so aware of what the NIH has or hasn’t done as is the ME/CFS community. On the bright side it gave us another chance to remind him that the NIH is still neglecting this disease and that we’re very aware of what’s going on.
I think Ron Davis should actually take over the NIH
Two things start to get clear now:
* From the nanoneedle: the disease is clearly present in every single cell.
* From the Naviaux research and Ron Davis interpretations: there is something in the blood. ME cells do behave as healthy in clean control blood and control cells do behave as ME cells in ME blood.
I add a third one: our livers seem to be in a 24/7 overdrive compared to controls. The total work our livers do 24/7 to recycle the large amounts of pyruvate and lactate due to large scale anaerobic functionning plus fighting the (very?) large amounts of oxidative stress our cells produce plus flitering the likely large amounts of other toxins our body produces out of the body must place a very high constant load on our livers.
=> So what enables our individual liver cells to go 24/7 into kind of a crazy overdrive? What kind of chemicals does the liver filter out that makes blood behave like ME blood? Could this filtering be that profound that our liver cells “don’t even know they have ME”?
=> That creates shortcuts to gaining new insights. Like testing with the nanoneedle if liver cells indeed are ME cells or if they are shielded from this disease, testing if freshly liver-filtered blood does decrease how fast other cells poop out compared to feeding them with bulk blood or compare the metabolics results of liver cells versus other cells to spot candidate chemicals that “turn ME on” in healthy cells.
Amazing DeJurgen. Ron mentioned that the liver throws off a lot of the particles found in the blood. I just didn’t get it into the blog.
Interesting to know that Ron mentioned that the liver throws off a lot of the particles found in the blood.
It seems that much of the memory (of the disease) may be in the liquid. That’s both in the blood as well as in the cells. These influence each other. And memory in technical sense is the basis for a feedback loop or vicious circle.
So part of what keeps us ill may be not only in the blood, but in the bodies liquid at large being the blood, the intracellular liquid, maybe the extracellular liquid and interstitial fluid too.
And when the thing that helps locking this disease would be in the liquid then it also should be in the circulation and micro-circulation of the liquid. Combining the Naviaux and Ron Davis research and it seems like the “amount” of disease in a cell can be changed for example by putting healthy cells in ME blood or vice versa. Circulation and micro-circulation play a key role in the distribution of these chemicals and their concentration around the body.
So how well things like blood flow, micro blood flow, lymphatic fluid flood and interstitial flow function may in part determine disease expression and severity.
I am not convinced that it would be as “simple” as to purify our bodies liquids in order to get back to full health. Part of the particles in the liquid “keeping ME alive” may be messengers our body produces to keep hibernation on to stave of a real danger to our bodies.
Interestingly, when my mother, who also had a diagnosis of ME/CFS, underwent open heart surgery including being on a heart/lung machine she came out of it mysteriously recovered from ME/CFS and her Sjogrens has also abated. Could it be that her blood was cleansed? Or did she undergo a system reset?
@Hezza: thanks for reporting this!
“underwent open heart surgery including being on a heart/lung machine she came out of it mysteriously recovered from ME/CFS and her Sjogrens has also abated.”
That is in line with some recent thoughts of mine: “supportive” functions like blood (micro-)circulation and breathing are starved of energy as much as the rest of the body and function no longer enough to “initiate” spontanuous self healing.
It would be very interesting if you could post some more details if you had them. If any well medical schooled people would be present here I’d be very interested to have some more information about “standard procedures” regarding such surgery.
The idea I have is that during this surgery the following happened:
* blood flow was taken over and forced to “optimal” values by a machine/pump
* she was clearly on a hart/lung machine; I believe low blood volumes in ME patients have a detremental influence on blood flow in the lungs compared to healthy people and hamper breathing far more then currently believed; the hart/lung machine temporarely (tries to) optimalizes this too
* likely she got IV saline to optimize amount of blood volume (likely pushing open the most restricted capillaries)
* optional: blood viscosity was checked and if necesarry medicated to bring to optimal
I believe this (the near complete /synchronuous failure / energy starvation of “self-healing functions”) may be an important part of what keeps the vicious circle on in ME and to some extent in potentially many auto-immune disseases.
Taken a group of these functions over during surgery may have been what caused both ME and Sjorgens to abate. I think this also point to the potential value of IV-saline over longer periods (there has been a research reporting good results!) and the “failure” of the Rituximab study (where both treated and controls improved A LOT, but where IV saline treatment was part of both groups)
See other comment I was already planning to write beneath this one.
Hi Cort,
Great article! As you say, the fact that resources, rather then tech, are the problem is good news. But it’s also frustrating…imagine if we could get just one billionaire philanthropist to take up our cause!
By the way, have you ever considered presenting big news updates such as this in a video? Maybe even a healthrising youtube channel? I think it would be helpful for sharing with friends and family. It may also be helpful for those of us whose PEM inhibits reading more than a few paragraphs at a time.
Thanks
Nice idea. Maybe some short video’s that go over the key points? If I didn’t embarrass myself too much that might work. 🙂
Gentle, minor comment here: the “Donate Now” button in this particular article could easily be taken as “Donate for the Nanoneedle” by people dealing with brain fog [raises hand]. Maybe you could add a link for donations just above, then add a dark line and a header of “Donate to Health Rising”.
LOVE the article. Thank you!
Yes, that just happened. I’ve fixed it.
Sorry cort, did you say the ME/CFS cells started to struggle when they were exposed to saline or mistaken illness beliefs? This may truly signify the end of an era.
LOL Debsw i really needed that. Thank you!
What surprises me is that Davis can distinguish 100 percent ME patients from controls. That’s weird in the medical world. But also worrying. It seems too good to be true. This indicates a very selected group of ME patients. Selection bias wouldn’t surprise me. although the ccc criteria are used, his findings using the Fukuda or Oxford criteria will not hold.
100 percent seems to be very optimistic indeed. We should be aware that there is still a long way to go. An important question is how well it can discriminate between other diseases.
But if such a test was affordable and would say “has some very nasty physical illness” then I’d take it tomorrow and have the medical report in my pockets for all who feel they can be abusive towards me because I can’t prove my disease is real.
Also this is but a very important first step. The research indicates that the cell itself may hold some memory of two things:
1) the patient has ME or at least a severe disease.
2) how much this cell has been exerting too intense recently.
Now number 2 may be a major thing: would it point to a cellular basis for PEM? That could improve another two things:
1) provide a means to better diagnose ME patients with the nano-needle: if stressing the cell “creates a memory of how much the cell over-exerted” then removing the stress should show how fast the cell recovers from this over-exertion. In ME patients this should take a long time too. If so, diagnosis could be based on both the impedance going up within x time when stressing the cell and going only slowly down after y time when removing the stress. As PEM is a key characteristic of ME it would be a very strong diagnostic tool if the nano-needle test could be modified to show both IMO.
2) provide both “proof” of the existence of PEM (hopefully allowing to recognize that ME patients’ disease can’t be dismissed if a patient can do some errands during 5 minutes and allowing to recognize the patients need for pacing and resting) and allow to better examine and understand the cellular basis of PEM.
Good point. I imagine that if it was all up to the CCC criteria or other criteria the number would drop quite a bit. Because we have no biological marker Ron had to find the best patients he could to assess the nanoneedle. I was surprised that even after doing that he got such a good result. I imagined that different pathways would be operating in different patients…
The next steps will be very interesting. Is the nanoneedle picking up some general abnormality that’s present in other disease states – like a marker for inflammation – or is it picking up something more specific to ME/CFS? We have no data at all on how other diseases will show up on this brand new instrument. It’s truly unknown.
I can’t wait for the answer!
Great blog, thanks Cort.
So the logjam is the current nanoneedle setup, interesting. Do you know if the NIH grant application in June is for developing the new high-throughput platform? If so, any idea of timescale for the decision from the NIH?
And if that’s not the purpose of the grant app, is there another plan to raise the funds for this? Like you, I think this has special promise and would appeal to donors. I would certainly be willing to donate myself.
Any answers appreciated.
Thanks for a very interesting post on a very important subject, Cort.
I agree with Simon that this is very interesting. You mention that Rahim Esfandyarpour says that funding is the limiting factor for developing the nanoneedle to be a high-throughput system and an amount of “not nearly” $1m would be needed. OMF has been very successful in raising (I think) $5m last year – do you know if they have enough funds available in the pot to fund that work and if so, are they planning to do that? It seems very high-priority to me, and time is of the essence for all of us. If that’s the roadblock, I’d like to see it cleared!
Great question. I have been wondering about that as well. I don’t know if the grant is for technology development or straight research or if the two can be combined. I will try and find out.
Thanks
I was told that the grant application is being written now and they’re not sure what will be in it. Getting the grant funded if it happens later this year, will help – how I don’t know but I assume getting money for the nanoneedle will allow them to move faster (i.e. hire people). Regardless they are moving forward with the needle – not as fast as they would like – as Rahim reported that at this point – resources are a big limiting factor – and they have not begun creating a new more high throughput machine.
Hi Cort – in your message below (which doesn’t have a ‘Reply’ button next to it on my screen), you say:
“I was told that the grant application is being written now and they’re not sure what will be in it. Getting the grant funded if it happens later this year, will help – how I don’t know but I assume getting money for the nanoneedle will allow them to move faster (i.e. hire people). Regardless they are moving forward with the needle – not as fast as they would like – as Rahim reported that at this point – resources are a big limiting factor – and they have not begun creating a new more high throughput machine.”
Were you able to put my question (below Simon’s) to OMF, about whether they already have enough funds available to fund that work themselves and if so, are they planning to do that? The nanoneedle work looks as though it might offer the holy grail of a diagnostic bio-test, which would be transformational – and so speed would seem to be of the essence.
Thanks!
I put that question in and am waiting for the answer.
Simon I know all grant applications submitted to NIH in June will be granted or rejected by the end of September as that’s the end of FY2019 at NIH.
Thanks Eimear. I was wondering about that as well.
Thanks
I disagree some with your statement “Studies are designed around the need to hit that probability (p<0.05). Miss it even by a small amount, and your results are worthless."
The p<0.05 criteria has been in long use, but the statistical and health professions are getting away from it. The American Statistical Association has recently come out against its use, and some journals are no longer allowing authors to discuss results around p<0.05. Clearly a very small sample leading to a p that is close to 0.5 does not provide a lot of information. But any result, no matter what the p value is, provides some information. Further, the .05 criteria is completely arbitrary. One could just as well use .06 or .04. Finally, if one has a very large sample size, you will almost surely get p<.05 for a treatment, even if it shows only a trivial effect.
Yes, it’s arbitrary – how could it not be? I didn’t mean worthless in the sense of not being meaningful, I meant worthless in the sense of not being considered “significant” and therefore the experiment being considered a failure. It’s become a kind of magic target that researchers shoot for whose importance is clearly out of bounds.
What would be cool is to take this to Workwell and be able to correlate a positive result with a certain level of disability as demonstrated by the 2 day test. If it could be used to prove disability, then it would be immediately useful to people who desperately need it, and who cares then whether it’s specific to a certain disease?
Maybe it doesn’t work that way. I’m still excited that Davis is so confident.
On the other hand, getting a true diagnosis isn’t going to impact me personally. My doctors just refuse to engage with this illness at all. Even if there was a cure available tomorrow, I’m not sure how I would access it.
When overdoing it by exhausting my mind two days ago I once more had the experience that exhaustion and pain “sunk to my legs” and spread to the rest of my body. Then I made a link to this blog and some of the comments I made on it. Much of it came down to the following question:
“How does the brain know that our cells are exhausted?”
While that may sound a trivial or philosophical question, it remains largely unanswered. So let us look at some possible answers:
* The brain keeps track of what commands it sends to organs and estimates how much that should fatigue/exhaust cells and organs. The problem with this option is that it is very hard (near impossible) to make a good guess of the accumulative effect of exhaustion on the fatigue of the cell themselves. It would have to know very well in what condition all parts and subparts of the body are and how exercise affects them. I can’t see that option to be viable at all.
* The brain senses, trough the nerves, how exhausted our cells are. Problem with that idea is “what does it sense”? Normal touch sense doesn’t register fatigue. So would it register the presence of certain chemicals indicating fatigue and/or exhaustion? If so then the nerve system still could only get a very rough view of fatigue at the scale of (part of) organs. The nerves themselves (nerve + “wiring”) are consisting of many cells so it couldn’t sense in every single individual cell.
* Each cell keeps track, with chemicals stored inside the cells themselves, of recent fatigue and exhaustion inducing activities. This could be done by producing chemicals inside the cells for every activity that consumes energy. Activities that are more intense produce these chemicals at a significantly (more then linear) higher rate. In that way, when the cell has been asked to produce a lot of energy recently the levels of these chemicals inside the cell rises. Each cell thus keeps track of it’s own amounts of exhaustion.
Now the last option with some tweaks is quite an interesting one. It shares many components with a very often used mechanism called an “accumulator”. This control mechanism is used extensively in both engineering system design as well as throughout nature itself. It is a very simple and robust design.
As tweak let us assume these fatigue/exhaustion tracking/accumulating chemicals do not use an active mechanism to traverse the cell borders, e.g. they do not use ATP to be moved inside or outside of the cells. There exist plenty and plenty of chemicals like this. The key point of this tweak would be that spreading (through the cell walls) the chemicals would be a process of diffusion.
Now these chemicals potentially spreading through diffusion would again make for a simple and robust system. The system would require few energy to signal fatigue/exhaustion. That is a clear advantage as if cells are exhausted they could easily lack the energy to transport these signals across cell boundaries. Another key advantage would be that accumulating and high levels of fatigue/exhaustion signals would (near inevitable) leak throughout the rest of the body from the fatigued/exhausted cells via the bodies liquids. That is from the liquid inside the cell into the blood, lymph, interstitial liquid…
These liquids would “message” that somewhere into the body levels of fatigue/exhaustion are accumulating. The levels of these chemicals would be the closest to the source of fatigue/exhaustion but would spread trough the entire body.
As “very old” (like last month) information about exhaustion is way less valuable then recent information (like an hour ago, a few days ago…) these chemicals should constantly be reduced in amount over time. Like say by the liver clearing out constantly part of the signalling components.
Having such chemicals spreading throughout the body would spread the message to all other cells (both brain, muscle and other) into the body: “some system(s) are in need of a good rest”. If levels rise then this message to all cells would be closer to “some system(s) are in need of a very urgent big rest”. So these chemicals would/should have the direct ability to modulate the amount of energy individual cells are allowed/able to produce and consume. Cells closer to the source of fatigue would see higher levels of these chemicals indicating higher fatigue/exhaustion and automatically inhibit these cells more.
If the levels of these chemicals would become excessive in many parts of the body it would inhibit both glycolysis and mitochondrial output of massive amounts of affected cells throughout the body.
This mechanism resembles a lot what seems to be already at large scale working in ME. If much of our symptoms align with having such a (protective an essentially necessary) system, it may be at work already.
(note: comments not nicely placed where they should nested under others, browser problems)
In the last comment I often used fatigue/exhaustion signalling chemicals. I believe the same chemicals that are at work in healthy individuals may be at work in ME too (to a large degree). Many patients may disagree as ME exhaustion is way and way beyond what any normal fatigue ever is. But I’ll make counter arguments:
* In most of human history people with ME wouldn’t have lived long enough to survive and get kids, so there was no evolutionary force for developing special “ME procedures and chemicals”
* Investing in creating a whole different way to deal with ME compared to fatigue when being utterly exhausted seems “wasteful” and hard to do when resource depleted.
* I *believe* I recognize many feelings of deep fatigue (when I was still healthy) to be more then vaguely familiar with how ME exhaustion feels. Being able to remember and compare how I feel now to back then (even if over a decade ago) is a feat that helped me a lot in my battle against ME. Note that I absolutely do not say both feel the same, they are in totally different orders of magnitude but still appear to have similarities and be on a linear scale. A bit like sensing a feather with touch senses and being hit by a hammer on the finger both use the same mechanism but are on a totally different scale.
So what would make the difference between “normal” fatigue and ME exhaustion in this model? Amount, time and extend of the signalling.
When being healthy and getting a nasty bout of flue, one starts body-wide with low levels of these fatigue signalling chemicals. The main “organ” under heavy fatigue for about a week would be the immune system. So only a small part of the body would produce these fatigue signals and it would not accumulate on top of already high levels. Still, as a healthy person experiences during the flu it can deplete energy levels of the body a lot.
Likewise running a marathon without proper training largely will deeply fatigue the muscles and motor cortex. But again one would start from low levels of fatigue signalling chemicals. The main “organs” under heavy fatigue for only a few hours (I discuss *accumulating* of fatigue, thus the period of effort only) are the muscles and motor cortex. Yet running a single marathon can badly fatigue an untrained person.
Something likewise holds for studying twenty hours apiece before an examination. It can deplete a person for days.
Now see ME as having all of these organs (plus some others) produce these amounts of fatigue signalling chemicals 24/7. That would trump any “normal” definition of fatigue by a large margin and would end us into ME exhaustion territory.
Part of these chemicals could be “made on purpose”, part of them could be “plain waste”.
Your brain sure don’t have problem firing, are sure you have CFS? 🙂
The cytokines are commonly understood as the “fatigue” signal, I think. Your body constantly produces it in order to recruit immune cells to repair the damage. Muscles produce myokines whenever you move. They take humoral path to brain and produce general feeling of fatigue. Or take neural path via receptors to generate specific regional fatigue sensation.
Both physical and mental fatigue could be taking the same “last mile” in the circuitry. You use your brain, does damage and immune system gets to work to make necessary repair resulting in fatigue sensation. The physical inflammation could be getting translated to the same brain mechanism to cause the fatigue sensation.
Lactate could be another signaling molecule and trigger more immediate fatigue sensation, in both in mental and physical exertion. There could be more than a few signaling molecules, who knows.
Under this model that chemicals signalling fatigue/exhaustion are produced for every exercise of every cell in both healthy people and ME patients, diffusion of these chemicals are a key component.
As I have recently written about “micro-circulation of blood” the blood in the most constricted capillaries should be “more polluted” then blood in the less constricted arteries. But as diffusion works to transfer chemicals from higher to lower levels, the amount of these signalling chemicals would be even higher in the cells located near these most constricted blood vessels. These cells would see the “full onslaught of ME and inhibition”.
The reverse of that would happen in the liver. In the cells of the liver the lowest amounts of these chemicals throughout the body would be seen by far. The liquid in these cells would be a lot cleaner then that in the blood leaving the liver (due to the nature of the diffusion process).
As our livers are “fired on” massively by a good located liver supplied of ample blood flow and plenty of adrenaline (much higher then in healthy controls) stimulating the liver, many ME patients may have “clean” liquid inside liver cells competing with or beating that of healthy controls. So our liver cells would not “know” they have ME or maybe even find fatigue/exhaustion chemical levels inside them to be below normal. That hypothesis would help solve why our livers seem to work in full turbo 24/7 while (most of) the rest of our bodies are in near full hibernation.
Now what could this model/hypothesis learn us if it were at work in ME?
Let us look at a clear potential for how it could create a vicious circle.
Human bodies may seem frail compared to steel machinery, but they have one astonishing feat. They can do a variety of hard to anticipate tasks for decades with remarkable little wear and tear except for aging. Hip replacements are a new thing on the timescale of human history. Before humans kept working by their “self repairing and self healing” part to heal wounds, repair damaged and infected cells, regrow bone…
In ME it feels as if there is no healing and repairing left, but without it we wouldn’t last more then a few days if even so much. The biggest problem may be: our “supporting, healing and repairing” functions may be inhibited just as much as our “work producing cells” like muscle and brain cells.
Under “supporting, healing and repairing” I do count thing like regular breathing, blood flow, lymph and fascia functioning, sleep, cell DNA repair, immune functioning, digestion…
When our bodies would be “bathing” in these fatigue/exhaustion signalling chemicals all cells including those providing these restorative functions would be inhibited by the high levels of these chemicals. So (much of) our restorative functions would go into hibernation just as much as our “work producing cells”.
This is a clear point where ME can start and lock in. When a “completely” healthy person gets the flu, as said above, this person gets “only” (or at least mainly) the immune system producing these fatigue signalling chemicals. Levels would rise enough to entice the person to take some decent amount of rest but would not accumulate high enough throughout the body to inhibit “self repair and self healing functions” so much that the body could not pull itself out of it. If the onslaught is to strong, lasts too long or more likely a hidden problem is slumbering inside the body weakening it already at the start (even if one is not aware) then the following can happen:
* levels of these fatigue signalling chemicals were already higher before but high levels of stress hormone (can be physical stress!) did hide it from the person. An additional big source of these chemicals and not taking sufficient rest is problematic and kick-starts ME.
* some supportive functions were already weakened inside and cannot help to pull a person out of an acute flu infection. Think of having a bad life long gut issue. When this issue is for instance a food intolerance the person could have been largely unaware of this hidden weakness.
* some key functions were already weakened like having genetic problems with RBC, high uric acid levels, fat metabolism. This problem may be worked around mostly until adding an acute flu onto it makes pulling out of the combined assault is too much to do.
This model would also help explain why the “economic debt model” does not work in ME. With this “economic debt model” I mean:
* When having debt, things are going from bad to worse as long as expenses are higher then income.
* When income finally gets permanently above expenses by for example cutting in expenses, debts start to gradually reduce and debt levels get better and better from that moment on.
=> As anyone here will have experienced, saving energy by pacing for example or improving general health by taking supplements does very very rarely create a condition where energy expended becomes smaller then energy available and where that leads to ever improving health by “clearing up the health/energy/waste debt”.
One often at best gets a small and quickly stagnating improvement as long as one keeps doing the changes. Understanding why this is so is rather important in ME IMO.
This model of chemicals inside the cells would help explain this one. When finding a new thing that helps and keep doing it lifelong, all cells see lower levels of these inhibiting chemicals inside them. That means all cells are put a little bit less deep into hibernation. That also means that both “energy producing and consuming” and “restorative” cells alike get their metabolism turned up. Hence, this mechanism automatically “consumes” a large part of the improvement by increasing metabolism a little bit from it’s very low state !body wide! Even when still pacing as much and not “using/spending” the improvement, the cells are put into a higher state of metabolism and “readiness” to do things. So there is (near) no “extra, surplus” energy to invest in continuously improving health. Most likely you are more able to do an activity a bit longer if you are willing to do an activity. Having this readiness by itself requires higher metabolism and consumes part of the saved energy.
So not being able to “chose” which cells see the additional influx of energy is part of the inability to use it to create an ongoing positive spiral.
But that doesn’t need to be bad news. Let me finish with some good news:
* This potential core mechanism of ME does not *require* any permanent damage. Of coarse living the the dire conditions ME creates for years or decades can create plenty of permanent secondary and tertiary damage but IMO ME does not require any damage to be permanent. This also ignores the fact that people with pre-existing conditions that can create permanent damage may have been doing so for much of peoples lifespan. In my case my gut may be in a dire condition that can’t be fully repaired as I only recently know I am intolerant to about half of food I used to eat.
* This idea opens potential to better separate “functions” into “producing and consuming energy” and those being “restorative”. This opens options to better support “self healing potential” at a low “energetic” cost in order to improve health and if one is very lucky to break the vicious circle. The same idea holds for symptoms being “just bad and nasty” and “those being very unpleasant but being side effects of the restorative attempts of the body”. Think of having moderate fever as being helpful to fight of the flu, but then examples better suited for ME.
Can you direct me to articles for individuals like me who are alone isolated with no fame
Ily who have to deal with cfs entirely alone. I have been doing this alone for thirty years but now that I am 72 it gets harder and harder. I have to drive myself to the emergency room thanks. I know this does. Not fit with this article but I need to know. Thanks
Great article Cort. Sure brought out the heavy thinkers who have experience and substantial insights to contribute.
Excuse me for going off Topic a minute. Do you know if Ron Davis has tried the Dr Goldstein “Resurrection Cocktail” with his son Whitney? Or any of the other Goldstein treatments?
Thanks
I don’t know. I would be surprised if they haven’t tried it – they’ve tried so many things – but I don’t know. They have found that Ativan is the one thing that can rouse Whitney temporarily.
For those unfamiliar with the “Resurrection Cocktail” it’s something Dr. Goldstein used to get his ME/CFS patients going when they really needed to be up for something. You can find it here – https://www.healthrising.org/forums/resources/dr-goldsteins-resurrection-cocktail.342/
If this nano-needle technique can measure cytokines in real time and in high precision, I’d love to see how the measurements correlate to the symptom severity after exercise. It has been done with athletes by taking blood every hour for several days and they showed that inflammatory cytokines spike up in waves for several days. And the waves seem to coincide what I’ve observed in a mild/moderate patient that I am: crap out an hour after; quasi-paralysis after 5 hours; crash after 24 hours. It could be that just walking from bedroom to bathroom may be resulting in the same pattern, albeit in much lower intensity, and severely ill CFS patients react violently to it.
Brilliant! Brightest ray of sunshine in a long time. Thank you!!
The thinking behind this is amazing and resonates with my research and my personal experience with this disease for the past 30 years. It comports well with the multiple cause scenarios that we see, especially the virus connection and the immune system abnormalities.
If this device proves to be near as useful as we hope, it will get funded. The Brits’ fully socialized health system has reason to drag their feet: This test could bury them in an avalanche of disability claims. Take note Obamacare fans. Social medicine is not always your friend.
But here in the U.S., market forces still have more pull. If indeed there are 2+ million and counting, M.E. sufferers in the U.S., I’m guessing that at least half of them would pony up a good sum to get their diagnoses settled.
I’m not doubting the altruism of the men who are developing this, but someone — trust me — SOMEOME knows that a technology like this could not only be extremely beneficial, but could also generate billions in revenues worldwide. If it takes charitable donations or a GoFundMe campaign to get R done, fine. Sign me up.
We hope that it doesnt end up like that PET scan “breakthrough” that apparently went bust. In any case, this is very exciting and I , for one, will be closely following it. This could be huge.
I don’t care about separate entity recognition or politics amongst the ME/CFS groups. The only thing I care about is a diagnostic test. The FM test with its low false positives and negatives was a game changer in Diagnostics and made doctors listen. I want that for myself and others. I’m sick and tired of the medical community not acknowledging ME/CFS until another disease like covid-19 comes around with similar systems and people are called long haulers. I’ve had CFS since childhood and gone through a number of name changes for the disease Yuppie flu, CFIDS, CFS, ME, Epstein-Barr, post viral syndrome, etc etc. If the Nano Needle can give me a definitive diagnostic then I’m all for it!
This is really fascinating, and I’m eager to see further research. I’m VERY curious about whether there’s a correlation between impedance change and symptom severity.