

“The story is always the same. When a pandemic happens, people always focus on the primary organ, but they forget about the brain.” Avindra Nath, MD
Most researchers, when they focus on post-infectious illnesses, go straight to the body to figure out what’s happening, but Avindra Nath is different – he goes straight to the brain.
“A Crisis in Neurology”
“Undiagnosed neuroinflammatory diseases carry a huge burden with devastating consequences.” Avindra Nath

Nath is strongly advocating for more neurological work in long COVID and other post-infectious illnesses.
Nath, the leader of the intramural study on ME/CFS, is a neuroscientist with an interest in neuroinflammation and post-infectious illnesses. Nath’s NIH page suggests he believes that neuroinflammatory diseases may be more prevalent and cause more problems than we know.
Nath was perhaps the first to raise the alarm that something was happening in the brains of COVID-19 patients. In his May “Special Editorial in Neurology, “Neurologic complications of coronavirus infections“, while he asserted that “Fear strikes the minds of the unprepared”, Nath emphasized how just unprepared the medical community is for a virus that affects the central nervous system.
He questioned whether the brainstem was involved, noted that several acute neurologic syndromes have been associated with coronaviruses, and decried the lack of drug development for infection-triggered neurological disorders.
“Although we all recognize the hundreds of viruses that can cause encephalitis and result in devastation to large populations, we have no treatment for any of these organisms except for herpes encephalitis.”
Calling the COVID-19 situation “a crisis in neurology”, he said: “The time to take action is now.”
Parkinson’s (ME/CFS?) Connection?
Next, Nath questioned whether COVID-19 might be a “perfect storm for Parkinson’s disease“. The parts of the brain affected in Parkinson’s (PD) – the midbrain and the basal ganglia – govern movement and reward and can produce severe fatigue.
These parts of the brain appear to be particularly vulnerable to an infectious trigger which induces a kind of chronic immune system sensitization, and likely, neuroinflammation. Nath suggested that the systemic and/or neuroinflammation that COVID-19 causes “might be ‘a perfect storm’ for the development of PD” over time. That’s an interesting idea as studies have implicated the basal ganglia in ME/CFS and fibromyalgia as well. Might a similar process be producing them?
Brain Autopsy Results
It wasn’t surprising to see Nath quickly check out what had happened to the brains of COVID-19 patients who had died. His recent letter, “Microvascular Injury in the Brains of Patients with Covid-19“, described the results of an examination of the olfactory nerve and brainstems of 16 people who had died of COVID-19.
Medical News Today reported that Nath was “completely surprised” at what he found. Instead of damage caused by low oxygen levels, he found “multifocal areas of damage (that are) usually associated with strokes and neuroinflammatory diseases.”
Leaky Brains?
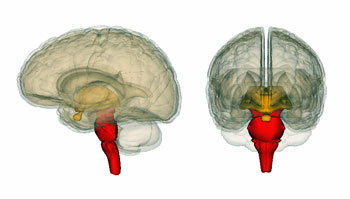
Nath found small blood vessel damage in the brainstem and olfactory lobes – but no virus.
The MRI showed punctate hyperintensities in about half of the autopsies. Punctate hyperintensities refer to lesions that are believed to signal small blood vessel problems in the brain. The walls of these blood vessels were also thinner than normal. A recent review of COVID-19 brain studies (done on live patients) indicated that MRI found white matter (WM) hyperintensities in about the same percentage of patients.
Nath found that these congested blood vessels were leaking fibrinogen, which he interpreted as being microhemorrhages. Activated microglia – a potential sign of neuroinflammation – (as well as macrophage infiltrates, and hypertrophic astrocytes) were found in most patients. Plus one report indicated that T-cells – which should not be found in the brain – were found surrounding these damaged areas. That suggested that immune cells from the body had infiltrated the brain and were likely causing damage.
The Gist
- In a May editorial, Avindra Nath referred to a “crisis in Neurology” when referring to long COVID’s impact on the central nervous system and the lack of drugs to combat viruses which impact the brain.
- Nath also suggested that the coronavirus’s ability to impact the basal ganglia and midbrain (via the immune system) could set long COVID patients up for Parkinson’s disease down the road. That’s an intriguing idea given that the basal ganglia have also been implicated in ME/CFS and FM.
- In a small autopsy study, Nath found evidence that the COVID patients had suffered from small brain bleeds or strokes across their brains. Nath speculated that the leaky blood vessels he found could have set off a strong immune response and noted that he also commonly found evidence of microglial activation.
- One of the earlier ME/CFS studies done found the same type of lesions (punctate hyperintensities) Nath found increased in ME/CFS. The finding was largely discarded because the lesions appeared to be more or less randomly distributed across the brain. That was the same pattern Nath found. He speculated that lesions in different areas were producing different symptoms in different patients.
- The dead COVID-19 patients appeared to have mild forms of the illness before they apparently suddenly collapsed and died.
- Studies suggest that the microcirculation is impaired in ME/CFS, FM and POTS as well.
- A 1992 ME/CFS study and the 2020 COVID-19 study suggested the same thing was happening in both diseases – an inflammatory attack on the central nervous system.
- A large study involving researchers from 30 countries will be assessing the central nervous system in connection with COVID-19 over time.
No Virus
Nath only autopsied two parts of the brain – the olfactory lobe and the brainstem – but he may not have needed to go further to determine if the virus itself had made it to the brain. (Both these regions are of prime interest given the loss of taste and smell, and breathing problems found in COVID-19. )
No evidence of the virus was found in either part of the brain. Nath noted that it was possible that virus had been present at some point, or that they had been unable to detect it, but proposed that the immune system was probably largely to blame for the brain damage found. He believes the inflammatory process is the key:
“The inflammatory response is key to the neuropathogenesis of this syndrome, since we were unable to find virus in the brain.”
He believes that the virus triggers an inflammatory response in the nervous system, which damages the blood vessels causing them to leak factors into the brain – which then triggers another immune response. Jennifer A. Frontera, MD, a neuro-critical care doctor also agreed that a secondary (or immune) response involving “inflammation and hypoxia,” was probably key.
Strange Group
The patients Nath had autopsied didn’t spend weeks on a respirator in intensive care before they died. In fact, they only had mild COVID-19 symptoms before they suddenly apparently collapsed and died.
Obviously, nobody reading this will fit that particular pattern, but many of us do, in a way: many of us had “mild” symptoms before becoming coming down with ME/CFS and/or FM. We may have felt wretched, but we didn’t end up in the hospital.
Tony Komaroff showed this back in 1996 when his large study found that people with ME/CFS had significantly worse functioning than people with heart attack, heart failure or multiple sclerosis.
This finding demonstrates that it’s possible to go from mild to debilitated, or even dead, very quickly. It’s another finding that doesn’t really “fit”; it doesn’t seem to make sense to quickly go from a mild COVID infection to death but that weirdness, in a way, makes it perfect for the perennial odd man out – ME/CFS.
ME/CFS, FM and POTS and the Long Haulers
The Long Haulers
“Now we have leads. We know what to look for.” Nath
Nath has begun a long COVID neurological study in which people with ME/CFS will reportedly be used as a control group. One focus will be the microvasculature. Nath has said something similar (“We know what to look for”) regarding his ME/CFS studies.
“In the future, we plan to study how COVID-19 harms the brain’s blood vessels and whether that produces some of the short- and long-term symptoms we see in patients.”
For her part, Frontera is writing up a long-term follow-up of almost 400 COVID-19 patients.
Chronic Fatigue Syndrome (ME/CFS)
In the last blog, we saw quite an overlap in symptoms developing in people with long COVID and people with chronic fatigue syndrome (ME/CFS). Now we see some intriguing similarities developing in the brain and the blood vessels.
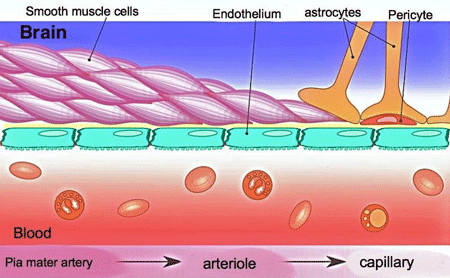
The leaky brain blood vessels Nath found in long COVID could be producing a variety of symptoms.
Punctate hyperintensities were one of the first findings in ME/CFS. A large percentage (78%) of patients had them compared to healthy controls (21%) but they were discarded, as I remember, because they didn’t fit a pattern: they were spread willy-nilly across the brain.
A similar pattern was found in the COVID-19 autopsies. Nath reported the lesions weren’t evenly distributed: “you would find a small blood vessel here and a small blood vessel there.” He said it was almost as if a series of small strokes had occurred across the brain.
Almost 30 years later, perhaps that 1992 finding may make more sense. The widespread nature of the punctate hyperintensities could reflect the widespread nature of the symptoms in ME/CFS/FM. The symptoms one experiences may reflect the part of the brain with leaky blood vessels. Nath stated that punctate hyperintensities found in the brainstem could produce problems with the heart rate, breathing and blood pressure.
Nath appeared to have hinted at this when he said:
“We hope these results will help doctors understand the full spectrum of problems patients may suffer so that we can come up with better treatments.”
Because they can also be found in healthy people, there’s been some question as to what these hyperintensities mean. They appear to be considered now, though, as “markers of brain frailty“. Increasing levels of punctate hyperintensities have been associated with cognitive problems, including executive functioning and memory, mobility problems, stroke and possibly dementia.
Baraniuk’s 2005 cerebrospinal proteome study suggested that localized bleeding (and the deposition of amyloid-like fragments) was present in ME/CFS patients’ brains as well.
Tracking
Thus far, Nath’s findings are tracking with some prominent themes in ME/CFS and fibromyalgia. Several studies suggest the basal ganglia may be involved in both diseases. A recent study suggested that the basal ganglia is the center of a fatigue nucleus in multiple sclerosis.
Other studies have highlighted possible microcirculation problems in both diseases as well as postural orthostatic tachycardia syndrome (POTS). Blood vessel problems in the abdomen and lower legs are believed common in POTS. Systrom’s results indicate that a significant subset of people with ME/CFS have microcirculatory problems which are allowing blood to escape into the interstitial spaces, and reducing blood flows to the heart.
- Narrowed Small Blood Vessels Linked to Fatigue in ME/CFS
- Low Energy Production and Pain in Fibromyalgia – Is Your Microcirculation To Blame?
- Could Poor Microcirculation Be Causing Chronic Fatigue Syndrome (ME/CFS)?
Plus, the brainstem is an area of focus in both ME/CFS and fibromyalgia.
Back to the Future?
Compare the conclusion from that 1992 study, “A chronic illness characterized by fatigue, neurologic and immunologic disorders, and active human herpesvirus type 6 infection” (co-authored by Cheney, Peterson, Ablashi, Buchwald, Paul Gallo and Komaroff)
“the patients may have been experiencing a chronic, immunologically mediated inflammatory process of the central nervous system.”
with Nath’s recent statement about the long haulers:
“The inflammatory response is key to the neuropathogenesis of this syndrome.”
While it’s still early yet, and we haven’t gotten the big studies needed to truly validate a pathophysiological link between ME/CFS and long COVID, thus far it seems like we may be coming full circle. Plus, there’s some good news on the brain front in long COVID. It has seemed that in the search for immune issues that the brain was being left out in COVID-19, but the brain research community is stepping up.
The Alzheimer’s Association reported that representatives from more than 30 countries have “formed an international, multidisciplinary consortium to collect and evaluate the short- and long-term consequences of the viral infection on the central nervous system (CNS)”. The study will be evaluating COVID-19 patients for up to 18 months.
Check out a large study which found similar symptoms to ME/CFS in people with long COVID.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



Thank goodness! I really enjoyed your first thought from Dr. Avindra Nath, “The story is always the same. When a pandemic happens, people always focus on the primary organ, but they forget about the brain.” I believe this goes back to the failings of dualism, in which the body and the mind, or body and brain, are mutually exclusive realms. Obviously the brain is involved with the body–in fact, monitoring and directing it but medical science largely handed it off to the psychiatrists who did a very poor job, in my opinion, even handling the part that was theirs. Anyway, three cheers for the neurologists and all their future research!
“Nath reported the lesions weren’t evenly distributed: “you would find a small blood vessel here and a small blood vessel there.” He said it was almost as if a series of small strokes had occurred across the brain.”
When Issie discribed the effects of her 2 TIA (Transient Ischemic Attacks) due to stroke, the symptoms to me were indistinguishable from what happened with me when I had a very bad crash:
Massive loss of abilities, like the ability to (know how to) open a door and passing it without getting the order wrong and bumping into it and many long term memories lost.
The possible biggest difference *might* be that my loss of abilities was more “random”, not something that could be pointing to a single zone in the brain like for example the language or math zone.
For me, that points to the same mechanism: hypoxia and reperfusion injury. While with stroke a large zone is blocked from decent blood flow, with very poor blood flow and volumes plenty of small dot sized zones could be blocked from decent blood flow.
When doctors speak of stroke, they think of a big blood clot blocking blood flow to things like the heart or part of the brain. Those are easiest to find and diagnose too. But it seems like the other side of the coin, plenty and plenty debris blocking the smallest capillaries all over the body, is barely considered to be even a problem.
But hey, largely blocking double digit percentages of your smallest capillaries blood flow will deprive plenty of zones from bare minimum blood flow. That should create plenty of hypoxia and need to use extra anaerobic energy, as seen in ME brains, and plenty of recurring reperfusion injury dotted all over the brain. It is often said, for survivors that is, that the reperfusion injury (the injury made by the immune system to clean up after hypoxia) after stroke is more damaging then the hypoxia itself.
So, with plenty of local clots and plugs in capillaries the body sort of needs to either try and “flush” them out towards the bigger veins or destroy the local cloths.
The first option? That requires things like showering the zone with histamine. And that can be best done by having fairly strong mast cell activation. The second option? That’s basically letting much of the “destructive parts” of the immune system “go crazy” on that clot.
Having immune cells bombard the wide zone with plenty and plenty of strong and toxic chemicals eating bit by bit the clot away till it becomes small enough for a strong “histamine shower” have the local veins expand and the clot be flushed away into the larger veins (where the more destructive parts of the immune system will continu to eat it away but with less damage to nearby blood vessels as most micro debris will now float away from the blood vessel walls.
Anyhow, such plentifull and widely spread reperfusion injury and the immune system going all crazy on hard to remove local clots, that are stuck and surrounded 360 degrees by blood vessel walls, is a good recipe for blood capillary damage (and increased chances to auto immunity to blood vessels following in its trace).
So I’m sold on the idea. Knowing that Covid is known to be able to let clot (“coagulate”) blood of certain subgroups of patients and that some patients have extensive bruising as their main Covid symptoms, I can see the parallells.
Finishing this line of thoughts, Covid patients where things go badly wrong tend to have a very strong immune response. There are several possible ways to generate a very strong immune response. One of them is the body forming massive amounts of antibodies in an attempt to target and deal with the unknown pathogen. Very large amounts of antibodies are also know to… …let blood clot.
And in COVID, they are finding that there is massive histamine response. They are having to get on it very fast. (Dejurgen explained why this possibly happens. It can help clean up things.) But too much is toooooooo much and to fast. So in an accute situation it has to be tamed down. (This is different than how I handle my daily chronic mast cell issues. I am trying to reset my histamine receptors. Have been off anti histamine for months now. And doing better. Unless I have a need for this autoimmune response and then mast cells are more quick and unpredictable. I still only use a mast cell stabilizer and herbs when this happens.)
And with my TIAs, despite there not seeming to be a clot, my MRI showed flares and that was indicative of things going on in the brain. And sadly, it did affect my ability to do math as well as before, and that didn’t come back as well as I once had it. There were other neurological issues that stayed with me too. Dejurgen “episode” seemed so similar and he had neurological issues too. Much of his came back though.
I do believe that the small capillaries are affected and that can be body wide, not just in the brain.
There is even a COVID rash that appears to break veins and cause inflammatory responses. That part of COVID isn’t talked about much. The purple toes, called COVID toes are spoken of more. Lack of blood flow to the feet, or blood pooling and lack of blood flow. So there seems to be issues with massive inflammation and autoimmune response in a huge, BIG and fast way. If too much, too fast……the body can’t clean up fast enough. And that could have dire consequences. And if it does manage to survive, all the things Dejurgen spoke of with repurfusion damage and Supreme use of energy…..throwing one into a bad ME/CFS spiral. I can sure see how this can happen!
Note: “Dejurgen “episode” seemed so similar and he had neurological issues too. Much of his came back though.”
That didn’t happen by itself however. A DIY (as ME patients don’t get them even if they lose plenty of skills) rehabilitation theraphy was needed for that. I had to diligently try and retrain many skills from the ground up again. Some showed quick improvement, others are still an ongoing process over 5 years after after my worst crash.
I wanted to say Dejurgen isn’t all the way with his recovery from that “episode “, but didn’t know if he would want me to tell that. (I still notice some neurological issues with him, like speach impairment. He is OK with me writing this, as I asked if it was okay to tell.) But since we have been friends and researching together (going into 2 years now), I have seen some nice improvements in him. He still has more to go, but more forward, positive steps in the right direction…..is GOOD!
How are you dealing with histamine issues? How did you get off them? Which ones were you taking?
@sue, I’m resetting my histamine receptors to work better. There is a thread on the forum with a link to a very technical book from a doctor, from another country, that tells how he did this for his patients. I’m not using what he used. But I do tell more about what I’m using on this forum thread.
I was on anything and everything that could be thrown at MCAS. I had it really severe to point of being in intensive care with Kounis Syndrome, when it hit my heart. I was on antihistamines daily for over 8 years. It started affecting my cognitive function severely. When I read this book, it made sense to me and even though it would be risky for me to attempt this, I did. And have been off for months now. If I don’t get on top of it soon enough, I still have GastroCrom that I will still use occasionally. But the herbs/tea and watching histamine foods have helped. I also believe that a combo product of bee Propolis, bee pollen, royal jelly along with nettle tea has been my best help here.
Here is the link to the thread. There are other threads on MCAS that Dejurgen and I have commented on this further there. But this one is where this idea and my research and experiments started with getting off antihistamines. I’m really glad I did. My brain is back and I have even less issues with MCAS than I did while on the antihistamines. There was bad rebound when those wore off.
https://www.healthrising.org/forums/threads/mast-cell-histamine-immunotherapy-with-histamine.6233/page-2#post-36690
On the ischemia-hyperperfusion issue – I assumed that was happening as well and apparently do did Nath but to his surprise he didn’t find evidence of that. If I’m reading him right instead he found evidence of an inflammatory attack which was attacking the walls of the blood vessels causing them to leak
I must admit I cut some corners on both the froming of the idea part and the part of trying to explain it.
Let me fisrt ask: where did you find this exact information? Does it talk of Nath’s finding regarding Covid, ME/CFS or both? I can’t find easily find that info.
When it comes to people who died from Covid, finding no real reperfusion damage makes sense:
Most patients who die from Covid see sharp drops in oxygen saturation levels early on in their disease. If their condition got worse on their declining path to death, it is very fair to expect that their oxygen levels got worse and never have returned to anywhere near normal or in other words they’ll never have met the medical definition for having reperfusion injury: having oxygenation returning to normal after hypoxia.
So these patients may be victim to both the cloting, hypoxia and / or an attempt to bomb the clots with wat remains online of the imune system during this hypoxia as explained above. But the cycle hypoxia / reperfusion injury might not be a main suspect in deceased Covid patients.
ME/CFS patients may have more “options” for likewise damage.
Let us first look at diabetes patients. Untreated diabetes patients often have toes, skin nerves and part of limbs like feet dying from poor blood flow (and hence oxygenation) due to sticky thick blood slowing blood flow in the capillaries too much.
So let us say that lasting unresolved bad hypoxia causes tissue death. With that, cells first go into programmed cell death and are cleared whenever possible by the immune system. So lasting bad hypoxia is a very inflammatory process if it leads to massive programmed cell death.
Our low blood volumes, reduced heart pumping, narrow blood vessels, inflexible RBC, poor RBC oxygen release… all point to poor to very poor blood flow and hence also oxygenation near everywhere near the finest capillaries. Yet, our tissue sort of escapes massive dying and forming of necrotic tissue.
When being for longer time in partial hypoxia, the body can produce chemicals like HIF in order to let the body better adapt to low oxygen situations with less damage. But such could also happen in diabetes. There, it doesn’t work.
That points to the thick blood creating poor blood flow being more a constant thing in diabetes versus us having more a swinging between stronger hypoxia and less strong hypoxia where we have periods of enough oxygen to let things repair.
In my case, I had years that needing to breathe like a horse and still starving for oxygen was a recurring nightly thing. During those hours, oxygenation (more so near the smallest capillaries) probably was rather bad. With it, the immune system likely had very few “ammonition” to fight pathogens during those hours. As hypoxia, even with adaptation to low oxygen levels, also causes damage, the combo of poor “immune patroling” plus stronger hypoxia during the late night likely created damaged in need to be cleaned up every morning.
If it wouldn’t be cleaned up, much of my body likely would have gone the untreated diabetes way. So, when oxygenation returned “somewhat” in the morning, it is reasonable to assume that the immune system was pushed into double gear in order to clean the mess up. If not, damage would keep pilling up and making being at a very low but stable baseline without clear further decline impossible IMO.
So, (likely quite a bit) more damage created during the night and it needing to be cleaned up during the day before going to bed again, the immune system had to work harder to clear “hypoxic damage” in the fewer then 24 remaining hours of the day. I can see how that would create damage to the capillaries.
I can also see why this wouldn’t be a daily repeating “real” hypoxia / reperfusion cycle, as oxygenation in those capillaries likely never returned to normal. It probably oscilated between “damaging bad” and “barely good enough to clean up the mess”. As such, it would not meet real hypoxia reperfusion criteria and I can imagine that the exact immune processes would differ too.
In both Covid and ME/CFS I still see however a clear role for hypoxic damage due to poor blood flow triggering nearby damage to the blood vessels. In ME/CFS there may in addition be something *similar* (I omitted that word in the orgininal comment) to hypoxia reperfusion damage going on.
Probably you refer too https://www.ninds.nih.gov/News-Events/News-and-Press-Releases/Press-Releases/NIH-study-uncovers-blood-vessel-damage-in-COVID-19-patients ?
““We were completely surprised. Originally, we expected to see damage that is caused by a lack of oxygen. Instead, we saw multifocal areas of damage that is usually associated with strokes and neuroinflammatory diseases,” said Dr. Nath.”
I wonder, as most damage is done by the reperfusion part of ischemia reperfusion and many death Covid patients have a strong decline in oxygen levels without it really recovering, if there is a strong knowledge of and schooling in the difference between “damage due to lack of oxygen” and “damage due to reperfusion”. I don’t know.
hey cort,
great read!
On another point, we are still waiting on the fall of 2019 McGregor update, any news there
Hi Cort. Wasn’t the drug Nimodipine (aka Nimotop) that Dr David Mason Brown had a lot of success with, developed to help increase blood circulation in the brain for people who have had a stroke? I think you wrote about it a year or two ago. I wanted to try it after reading Dr Kristina Downing-Orr’s book Beating Chronic Fatigue but my doctor wouldn’t prescribe it for me. I seem to remember that Dr Downing-Orr said that she improved by about 80% using Dr Mason Brown’s protocol.
Very recently I had an “event” and upping my blood flow too quickly fired back HARD on me, and set me back, for a long time. Dejurgen warned me to be very carefull and go low and slow but it still took me by surprise. I had a very strong inflammatory reaction, likely reperfusion injury, to regions that saw poor blood flow for decades. We are trying to work out the details and I’m now recovering from this. But it did set off more POTS, MCAS and ME got much worse.
about nifedipine:recently my physician told me I should take a medication to lower blood pressure.I suggested nifedipine because it also prevents fatal lung disease in altitude sickness
the alveoli of the lungs fill with fluid, oxygen levels fall very quickly below 70% and inflammation follows.That is exactly what happens with Covid infection (ref Klinghardt)This is a bonus I thought. But nifedipine was not available any more so she gave me amlodipine which is quite similar.
But when I augmented the dose from a quarter to halve a pill
it gave me horrendous muscle pain; continuous waves of pain
day and night; like giving birth without sedation -level 10 of pain
I suppose-(and yes I am that old to have experienced it before)
I did not sleep for 10 days but then happily my son read on the prescription; side effects; muscle cramps. After stopping the medication the cramps ceased. It was traumatizing.
Nimodipine and nifedipine both calcium channel blockers but many different actions. Many folks have reported impressive improvements with nimodipine. I tried it for a month and did not change.
Can we recover from this – I am coming up to my covid anniversary ( although it was a slow burning fuse and i didn’t get very ill till May ) I am a lot better perhaps Level40-50 from level10 on the Bells but still way off any one in normal health , let alone the level pre- covid.
Cort, Is this what they call UBO unidentified bright objects, found in many ME-patiënts?
I believe so.
I also think inflammation is causing major problems with micro circulation not only the brain but all the body. Was wondering if anyone has tried Pentoxyfilline for thinning of blood and improving blood flow, also helps red blood cells more deformability. I have not tried and would like to know if someone has.
I just answered a similar question as to my having too thick blood on another blog. This is what I use to thin my blood due to genetic issues with Factor 8 and Collagen Binding. Some with ME/CFS have also tested positive for APS. (They normally do this test 2 times and both times they need to be positive. I had it positive once.)
I use many different herbs, but daily Ginkgo is in a brain formulation I take. I find Ginger helps pain and to thin blood too. But that will burn my stomach, so has to be with food or put into tea or coffee. (Powder version) Enzymes, I rotate with Serrapeptase and Vascuzyme by Empircal Labs. The enzymes help dissolve fibrogen and break down biofilm in the blood vessels. It has been found to help get rid of pathogens that reside in the biofilms and blood. It also helps pain, if taken away from food.
Cort, thought this might interest you and wondering if the same cells have ever been found in MECFS patients.
https://www.medscape.com/viewarticle/945774#:~:text=The%20long-term%20neurologic%20symptoms,cells%2C%20a%20new%20report%20suggests.
Whenever I read articles like this – about ME/CFS and now about long-Covid – I always think about this line of research that shows that trichothecenes have the ability to damage the blood-brain barrier (as well as the gut lining).
It would be interesting to look at whether people who get long-Covid are more likely to be living in buildings with Stachybotrys problems, therefore.
http://www.paradigmchange.me/wp/karunasena/
Lisa Petrison, Ph.D.
Now that vaccines are available, does anyone have any insights into whether or not ME/CFS patients should consider them.
Hi 3D
Check out our polls on how ME/CFS patients are doing on them here
https://www.healthrising.org/blog/2021/01/28/coronavirus-vaccine-effects-chronic-fatigue-syndrome-fibromyalgia/
and ME/CFS experts on whether to take the vaccine
https://www.healthrising.org/blog/2021/01/03/chronic-fatigue-fibromyalgia-experts-coronavirus-vaccine/
I I have been very ill since my second moderan dose on 4/9. I have a list of 24 symptoms and no answers. All test results have come back normal so far-same as many others. Those of us suffering from vaccine injury have many (if not all) of the same symptoms as Covid long haul sufferers. It completely makes sense that our issues stem from a Neuro issues. I have no seen Neuro yet. I have not seen a lot of specialists because I am fighting everyday for health care. Dr’s don’t know whatnto do with me, so they have written me off as a psychiatric patient, or just passed me off to someone else. Or, they just refuse to even see me. We need help. I need help. I am slowly losing my life.
Hang in there Kelly! Hopefully long COVID answers will apply to you as well.
Hello Kelly and Cort, after reading this article I was wondering the same as Kelly. I have been dealing with chronic headache/photophobia for 16 months now since getting two Moderna vaccines. Could this article apply to us Cort? Please answer me back regarding my question. I agree Kelly, it’s so hard to get answers from the medical community. Health Rising articles have been very helpful. My PCP is supposed to interface with a doctor through the Clinical Immunization Safety Assessment Project team with CDC any day now. Need answers please!
Cort! I thank you for this article. It raised some thoughts in my mind. As you are aware of there is a hypothetis that EDS and ME somehow might be either misdiagnosed with one another (oh, language issues again!) or be connected. My thought when I read this is that we EDS’ers bleed easylier due to faulty collagen in the walls of the blood vessels, and therefore get the same sort of leak and thereby more susceptable to ME? Beside of brainstem and cervical instability? Or have I misinterpreted your text complitely?
My brain was definitely negatively affected, when I developed a very nasty virus in Feb 2017 (Swine Flu H1N1?) . I’ve never been so unwell (I don’t think I’ve had Covid-19 yet) for so long. I became very hot 41°/105f. Afterwards my brain seemed to get fired up, when I ate certain foods. I’d have a splitting headache and my brain couldn’t function. I went to my doctor and when I asked him if he could give me something for my inflamed brain, he smiled and just shook his head.
So since then – nearly 4 years – I’ve made a lot of progress. As Dejurgen says above – DIY – as there was no one to help me. That’s the tightrope I walk – what to eat, that is nutritious and gives me energy, that doesn’t set my brain off.
‘A Neurologic Crisis’ – Thanks Dr Nath that sums my situation up nicely ?
Hi Tracey Anne,
This recent research may provide clues: https://medicalxpress.com/news/2021-01-scientists-reveal-mechanism-bowel-syndrome.html
It seems that having a food that gives easy rise to allergies or intollerances at the same place and time of gut infection creates a sort of associative mistake: the body forms antibodies to both the pathogen and the food that is present there. That happens easier if it is food where one easily builds intollerance to.
So maybe there are two clues to be found here:
* Try and triple avoid eating food that is known to cause food intollerances when having a gut infection. Note: with many Covid patients the gut is infected.
* Eating only in-season food *might* help and wane off food intollerances as the affected regions might lose some or even much of there association / learned immunity / food antibody generating immune cells if you leave off foods for over half a year each year.
@Cort: this research on the plausible cause of IBS and the involvement of the immune system (and it’s potential relationship to Covid infection turning to long Covid by creating gut issues and ways to avoid this) might be worth a Blog all by itself.
Thanks Dejurgen, that’s so interesting. I’ve had noticeable IBS, since in my 20’s but it used to be confined to my gut. If I had a little graze through the cupboards and fridge – I knew I was in for trouble during the following days. The worst offenders were dairy products, oats, chocolate digestive/oat biscuits and so on…
My current situation is made a great deal worse because I think my microglia in my brain are now primed (Dr Datis Kharrazian and Jarred Younger PhD) and so I think an Irritable Bowel now instigates an Irritable Brain too.
Writing this now Sunday morning – I’m fine. However later in the day my brain energy will wane and I am able to eat some 60% dark chocolate (no dairy) which definitely helps. The supermarket ran out of it a few months ago and I had to turn to a 70% dark chocolate but it did set off, what I think is a histamine response – my brain would become sluggish and my balance would be affected. Anyway the 60% is back in and I’ve switched back to that. It seems to give my brain just what it needs… I just have to limit how much and how frequently I eat it, which I’m fairly hopeless at.
Also sometimes I need to Stop Rest Pace and not think eating chocolate, is the answer to everything! My brain does not work well if it is tired and my view of my self, others and the world are definitely not rosy if I’m wrecked. So this week I’ve tried to remember that and have a rest – which does actually work. So instead of getting annoyed and frustrated, I have a nap and then feel better. I find it quite challenging to live with myself at times ? I find it helps if I can see the funny side – which doesn’t always occur.
I have been taking 10mg of cetirizine and they help but I’ll stop taking them now, as I may have the Covid-19 vaccine soon and I don’t want them to lessen my immune response. I also do take notice of Issie and what she says about antihistamines.
So I definitely feel my brain is affected by what I eat (or don’t eat) and apparently our digestive tract is a major part of our immune system and so that all makes sense to me.
@Tracey Anne, just keep in mind with stopping antihistamines, the rebound. When your body kicks back into working again, histamine will be felt even worse than before. Likely the receptors won’t know when to stop and shut off. I find Nettle tea before a histamine response to give me enough histamine to tame down the response. It also gives a boost to serotonin. But don’t drink it when in a flare. I drink it about an hour before bed and sleep better. I also find the bee supplement to be VERY helpful. Both my sister and Dejurgen have taken up the experiment with me and both are doing better. Yayyyyyy!!!!
When I first started this journey with trying to stop antihistamines, I used a transdermal histamine cream. It was tricky to use. And it had to be a small amount. But I was trying to get my body to not release its own histamine, by giving it some externally. That started my H2 receptors to kick in better to stop and slow down histamine response. And without my own body releasing from H1 receptors, it also didn’t release as many inflammatory substances either. I have less pain now too.
I still have MCAS. But it is soooooo much better. And diet is a key part of it. Sadly chocolate will trigger it for me. I have to be doing really good to eat it and then limit it.
We need histamine. It helps with our immune system function and also our cognitive function. We just don’t need too much of it.
They are using histamine to help with narcolepsy, MS and Alzheimer disease. And some other things too.
For me, as severely dysfunctional as my histamine receptors are/were, I do have GastroCrom on hand in an emergency and liquid H1 blocker in a liquid, if I get my timing off and don’t get on top of things fast enough. I also have epipens. (But thankfully have not had to use those.) I have had to use the mast cell stabilizers some, but not often. (GastroCrom).
The book listed on the thread on the forum is very technical, but can give more of the science than I can. And Dejurgen has also written quite a bit about the science in regards to MCAS.
Thanks Issie, I’m not too bad as long as I avoid the foods that set me off – which is most food but I manage to eat a fairly healthy diet now. The chocolate is a tricky one – it will trigger a histamine response – I just have to try and keep it to a minimum – I just haven’t found anything else that works. If I run out of choc or decide to eliminate it, then after a day or so, my brain energy drains away and isn’t replenished. I used to do well on peanuts but they’re not tolerated anymore. I can now eat almonds, walnuts and brazils again, which is a relief.
Stress makes everything worse too, particularly really difficult, overwhelming issues. I’m continually trying to work at the stress levels from two angles – reducing the stressors (if possible) and attempting to feel more empowered in dealing with them, if I can’t get rid of them. I have a lot of projects on the go!
Some people crave chocolate because of a need of magnesium. Maybe that would help? Using a spray on, transdermal one seems to agree with me better than oral.
But sometimes its the comfort feel it gives. It would up dopamine then. Coffee can also up dopamine and Korean Red Ginseng.
I take magnesium, I don’t think it’s that. I’d agree with you in relation the dopamine and I think getting glucose to my brain. I’m working on it…
I’m continuing to go to two of my homecare clients. I won’t go to new people at the moment due to the increased risk to all of us. I do know that face to face contact is a health benefit (risky with Covid-19) and I have always been better off in a group or with others but feeling isolated (as against enjoying being on my own) is not good for me. I might try the Korean Red Ginseng. For whatever reason, I’ve never tried Ginseng.
I’m doing much better with my energy too. For me they’re all slightly different areas in which to focus on and I’m reasonably aware of what’s improving or getting worse, even if I don’t know exactly why it’s happening.
“Nath has begun a long COVID neurological study in which people with ME/CFS will reportedly be used as a control group.” Cort, where did you get the information that ME/CFS patients would form a control group? It is not on the clinicaltrials.gov posting that I see. Thanks!
That was either told to me or I read it somewhere which is why I put “reportedly” :).
I’m not sure the findings of microvascular injury in the brain can be generalised to longcovid or ME symptoms in general. It could be that all patients with such microvascular injury in the brain will unfortunately die.
Secondly, “inflammation” is too non-specific. Inflammation is normally useful as it leads to elimination of the infection. The problem is when it happens in the wrong places – specifically, places where inflammation doesn’t normally occur. This happens for a reason – perhaps the immune system is targeting something it shouldn’t or perhaps a sneaky viral protein is disrupting signalling leading to leukocytes sticking around when they normally wouldn’t or doing the wrong thing. I note that the SARS-CoV-2 spike proteins bind to quite a list of cell surface receptors (not just ACE2) and likely uses other receptors for cell entry in different cell types as well.
Thirdly, leaky blood vessels doesn’t have to be caused by inflammation. Or the inflammation might be an indirect consequence of other sneaky virus behaviour.
Viruses are known to disrupt cellular adhesions and often target cell junctions of endothelial cells.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7049484/
https://www.sciencedirect.com/science/article/pii/S1534580702002988
SARS-1 for example has demonstrated such sneaky behaviour:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2982091/
And likewise for SARS-2
https://onlinelibrary.wiley.com/doi/full/10.1002/pro.3936
Some of you may be thinking, well what about the herpes viruses? Well they have around five times as many genes as coronaviruses and are persistent buggers, so what do you think? 😉
Along this same line of thinking, I was sent this article today on antibodies and how they can attack self, even after the virus is gone. So if COVID can do this, and it is giving symptoms like ME/CFS, why not other virus, pathogens etc. (Think of even Post Lyme syndrome. The body does the same thing with that.)
https://www.eatthis.com/news-side-effect-covid-autoantibodies/
This article was taken from a New York Times Post, according to the writer of it. I don’t have an account with them, so wasn’t able to read that article. But here is the link.
https://www.nytimes.com/2021/01/28/health/coronavirus-antibodies-immunity.html