

Study Presents Alternative Findings

Neuroinflammation seemed like a slam dunk for ME/CFS – until this study showed up.
Neuroinflammation has almost seemed like a slam dunk in chronic fatigue syndrome (ME/CFS). Nakatomi’s small 2014 study (n=19) set the stage when it reported finding widespread neuroinflammation.
A widespread, low-level neuroinflammatory problem seemed like a match for a disease characterized by a wide variety of symptoms (fatigue, pain, cognition, sleep). Tony Komaroff called the Nakatomi finding potentially the most important in decades.
Since then, several studies have suggested that neuroinflammation is indeed present in ME/CFS, as well as fibromyalgia (FM) and Gulf War Illness (GWI). Shungu’s consistent findings of increased lactate levels in the brain’s ventricules, William’s dUPTase studies, Miller’s findings of basal ganglia deactivation, Hornig’s findings of altered cerebral spinal fluid cytokines, and Baraniuk’s CSF miRNA study all suggested that neuroinflammation was present.
Add to those the many hypothesis papers that have cited neuroinflammation as a potential cause of the symptoms in ME/CFS and fibromyalgia and you’ve got what seems to be a foregone conclusion: neuroinflammation MUST be present in ME/CFS/FM and similar diseases.
Fibromyalgia, Chronic Fatigue Syndrome, Gulf War Illness – the Widespread Neuroinflammation Diseases
Nancy Klimas’s models indicated that the first drug in her two-drug combo for GWI and ME/CFS should be focused on quieting down neuroinflammation. Health Rising has devoted over 25 blogs to the subject of neuroinflammation and has several pages devoted to potential neuroinflammatory fixes.
That speculation regarding neuroinflammation, though, has rested upon a pretty thin evidentiary reed. Until this Dutch study appeared, though, only Jarred Younger’s 30-person thermography and metabolite study more or less attempted to directly assess neuroinflammation, Younger, using a new technique, found elevated temperatures in widespread areas of the brain and then validated his findings by using MRS spectroscopy.
While only a few studies have directly attempted to assess neuroinflammation the evidence to date suggests it is probably present. Until now, though, no one has attempted to replicate Nakatomi’s small study results.
The Dutch Study
It seems remarkable that for a subject that has aroused so much interest and so many hopes, it’s taken 7 years for someone to attempt to replicate the 2014 study. In the 2021 “No Signs of Neuroinflammation in Women With Chronic Fatigue Syndrome or Q Fever Fatigue Syndrome Using the TSPO Ligand [11C]-PK11195” paper, Dutch researchers attempted to replicate and improve on the first PET scan study.
The Dutch researchers proposed a familiar scenario: an inflammatory hit from the original infectious event had reset the immune cells of the brain (the microglia). Now primed to act at the slightest disturbance, they were constantly producing the classic signs of “sickness behavior” (fatigue, pain, cognitive and sleep problems) – so often seen during the infection and so common in ME/CFS, FM, and GWI.
Like the Japanese study before it, the Dutch study was very small (n=16). Although the authors noted that better tracers are now available, the study used the same radioactive tracer that Nakatomi did. The study differed in that it added a post-infectious arm of Q fever patients, only assessed women, used a better-matched the authors thought, healthy control group, and an older patient set. (While the study required that symptoms reached a certain severity, it did use the Fukuda criteria). Finally, the researchers used a different, and they felt more accurate, reference point in the brain to assess their findings.
On the whole, the study seemed better designed than the 2014 study – plus it had the advantage of assessing a known post-infectious cohort. Because the study was so small, though, the authors noted that any of the differences between it and the Nakatomi study could have had an effect.
No Neuroinflammation!
The study found no evidence of neuroinflammation at all in either the ME/CFS group or the Q fever group. Some symptom scores were correlated with increased activity in some parts of the brain in the Q-fever patients but very little correlation was found between symptom scores, and microglial activation in the ME/CFS patients. In other words, the study was pretty much of a bust regarding neuroinflammation and ME/CFS.
Odd Man Out?
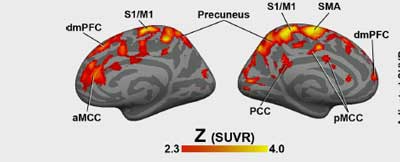
Loggia’s pet scan study found ample evidence of neuroinflammation in fibromyalgia.
It’s difficult to know what to make of this study. While the study appears to have improved methodologically on Nakatomi’s 2014 study, its results – particularly when ME/CFS’s sister diseases are taken into account, leave it an odd man out. Nakatomi’s and Younger’s studies in ME/CFS as well as PET scan studies of fibromyalgia, Gulf War Illness, and migraine all report finding a rather unusual signature – widespread areas of inflammation in the brain (and in many of the same brain regions).
Loggia is a key figure. Having investigated neuroinflammation in many other diseases (chronic pain, depression, anxiety, autism, amyotrophic lateral sclerosis (ALS), multiple sclerosis (MS), Huntington’s disease), Loggia has a long track record in neuroinflammation and has found similar neuroinflammatory signatures FM, GWI and migraine neuroinflammation.
While the basis for neuroinflammation in ME/CFS is still pretty slim – resting on just a few studies – thus far the evidence, notwithstanding this study’s results, still supports the idea that similar patterns of neuroinflammation are happening in ME/CFS, fibromyalgia, GWI, and other diseases.
While neuroinflammation makes a lot of sense, it should be noted that neuroinflammation is not needed to explain ME/CFS, FM, or other diseases. In the last of their ever-expanding series of hypothesis papers, Klaus Wirth and Carmen Scheibenbogen have found a way to explain the neurological symptoms of ME/CFS without ever calling neuroinflammation into play at all.
Time will tell if they are correct, but a great hypothesis should be able to explain all aspects of a disease and, so far, that’s what Wirth and Scheibenbogen, over three publications, have been able to do in their impressive effort. (A blog on their latest hypothesis paper is coming up).
The Future

The future – as it so often seems – should tell us much. Very large long COVID studies underway should be particularly illuminating.
The small Dutch study, notwithstanding, most evidence suggests that neuroinflammation is occurring in ME/CFS. The good news is that the future is, indeed, coming and it should tell us much. At least two major NIH-funded studies are underway.
Dikomo Shungu’s “Oxidative Stress and Neuroinflammation: Co-conspirators in ME/CFS Pathophysiology” study using PET scans and MRS spectroscopy, began in 2017, and should have ended, if all went well, in August of this year.
Jarred Younger’s “Measuring neuroinflammation in chronic fatigue syndrome with whole-brain magnetic resonance spectroscopy” study began in 2019 and is slated to end in 2024. Younger’s three-part study will search the entire brain using MRS for signs of neuroinflammation, will employ a good-day/bad-day approach to see if worsened symptoms are associated with neuroinflammation, and will do a PET scan study for neuroinflammation.
A first-of-its-kind Stanford study, “Imaging inflammation in the whole body and brain of ME/CFS patients“, is going whole hog – scanning and assessing microglial activation in the body and the brains of people with ME/CFS. The study encouragingly states that “our encouraging preliminary data shows increased [11C]DPA-713-PET signal in multiple brain regions of severe ME/CFS patients compared to healthy controls.” This smaller, exploratory R21 study began in December of last year and should be ending up now.
A 200-person long COVID study, “Understanding the long-term impact of COVID-19 on the brain through advanced MR imaging and spectroscopy“, brings us to a whole new level. This is surely just the first of many VERY large long-COVID studies that will be assessing neuroinflammation, neurodegeneration, hypoxia/ischemia, and other potential brain issues.
Other studies are surely underway, of which, Nakatomi’s larger and long-delayed ME/CFS follow-up validation study looms the largest.
Plus, studies in other diseases are underway. A fibromyalgia study that is using PET scans to determine if cannabidiol (CBD) reduces microglial activation should tell us both about neuroinflammation in FM and whether CBD can help. My experience with cannabis products suggests it just might.
Conclusion
A new and improved Dutch version of Nakatomi’s 2014 neuroinflammation study found no neuroinflammation at all in either ME/CFS or post-Q fever syndrome. While the study was quite small, the result is a reminder that whatever the popularity of the neuroinflammation hypothesis, it rests on a rather small direct evidentiary basis.
Besides Jarred Younger’s duo thermography/MRS study, the best evidence for neuroinflammation in ME/CFS probably lies in Loggia’s neuroinflammation findings in fibromyalgia, Gulf War Illness, and migraine. An accomplished neuroscientist with many neuroinflammation studies under his belt, Loggia’s findings of a rather unusual brain signature (widespread neuroinflammation) in FM, GWI, and migraine jives well with Nakatomi’s and Younger’s findings of the same.
While these studies provide indirect evidence of neuroinflammation in ME/CFS, more and larger studies that directly assess neuroinflammation are clearly needed to determine. Thankfully, several large NIH-funded studies are underway, including a 5-year study that was slated to wrap up a couple of months ago. Very large neuroinflammation studies are also expected in long COVID, one of which – a 200-person study – has started.
Health Rising’s BIG (little) Donation Drive is Roaring!

If you appreciate deep dives into the literature, please support us.
Thanks to the 80+ people who have gotten the 2021 fundraising drive off to a roaring start.
This neuroinflammation blog pretty much sums up what makes Health Rising different. We could have just presented this (rather disappointing) finding, but that’s not what we do. We take a finding, try putting it in historical context – showing how compares to past findings in ME/CFS and similar diseases, do our best to assess how rigorous the study is, and then see what the future may bring.
If you enjoy that level of effort, please support us!


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



these study sample sizes are far, far too small to extrapolate
n=100 min, and n=1,000+ far better
To be a bit more specific, I would say the study size of n~10 is smallish for a null result. For other results like the iCPET work where there was a very strong effect — clearly separating the cases and controls for one variable — n~10 can be and is (imho) meaningful.
Small sample sizes are common in this kind of research. While bigger studies are more powerful – and we will be getting them – the authors did assess the sample size needed for an effect:
“The sample size needed was estimated using the effect size (Cohen d) as calculated from a previous [11C]-PK11195 study by Nakatomi et al. in 9 patients with CFS vs 10 HSs5 and the [11C]-PK11195 binding potential (BPND) in HSs from a previous study performed by our group”.
Not being much up on statistics I really don’t know that means (lol)
Finally! Regardless of the result, I’m glad someone actually attempted to replicate the 2014 study. We need all the facts on the table before going too deep into hypotheses.
It possible that 2014 study didn’t match the age; older brains are naturally more inflamed and that could be what Nakatomi’s team saw. Or, it is possible that the Dutch team’s cohorts were mild patients who weren’t constantly in PEM and therefore their brain weren’t on fire during the test. It now calls for a second replication study. 45-140% more inflammation to none is a huge difference after all and someone should get to the bottom of it.
Oddly enough, it was the opposite – the Dutch study used an older cohort and questioned whether the longer duration meant there was less neuroinflammation – and that the neuroinflammation was higher earlier in the illness.
I meant that the age matched control, not the cohort, was older than Nakatomi study.
I wonder if there will be a response from Nakatomi team. If anybody could tell why the difference, it should be them, I would think. Any event, It’s good to know that there are more studies lined up for neuroinflammation. And I’ll look forward to your report on their results!
Right. I keep hearing that the Nakatomi study results are coming out and then they don’t. I hope there isn’t a problem.
I originally saw real promise in the neuroinflammation theory but have become increasingly skeptical over time.
My money is on Carmen cracking the CFS puzzle
I think, as long as they throw not the amount of money as for long covid, for ME/cfs, we stay in mostly small pilot studys, not replicated, and mostly only hypothesis.
and i may have overlooked but mhy did the dutsh study use the fukuda criteria and wich criteria use the other study’s. And with all the diferent definitions. and even long covid, we know, that with ME/cfs patients dit not all got it from an infection. to ill, to old. how do they say:lost generation…
and still, why can’t they work more together so that the pilot studys are at least not so small. it is as if they all want to have found it themself. and this for such complex deseases.
i can for long time because to many promisses, foundings, promis on treatments, broken promisses, reading for decades, not believe anymore… untill they really throw big money on it…
Having everybody use the same criteria would certainly be helpful and using different criteria doesn’t help this field. The authors did require that the participants reach a certain symptom severity.
Big money would help a lot! At least we’re going to see that in long COVID and those NIH studies should be pretty big as well.
Yep lots of promise not delivered on in our field, I won’t mention names.
and with such a much higher rate on suicide, they could have for decades do autopsys to learn much more of what is going on and subsets, etc but no, no money again or what? no intrest? i do not understand…
Like you, I’ve also become skeptical over the past 25 years. All those theories. That’s how science works. Nevertheless, 1 good research can make all the difference. Autopsy could be one. But that is also difficult. One thing I do know how little scientists actually know about diseases.
The best example is covid. See the effectiveness of the vaccines. Very disappointing. After half a year it almost doesn’t work anymore. Is it really wise to vaccinate all people and and give them booster shots?
Booster shots haven’t even been tested in a double-blind study. Everyone is going to get covid with or without vaccination. And let’s hope we all get through it. All those vaccinations can never be good in my opinion.
Well, I would say that 90% of the people in the hospital struggling to breathe and dying in the US are unvaccinated and that ICU are overflowing in Michigan and other states once again – and almost all of that could have been avoided. About a 1000 people are still dying of COVID19 every day in the U.S. . COVID-19 is also accounting for about 10% of lung transplants being done in the US. (Imagine having to get a lung transplant.)
The vaccines are working very well to prevent death and hospitalization. They are also limiting the spread of the virus because vaccinated people are less infectious.
I’m sorry for those who have not done well with the vaccines. Fo me I just got my booster shot. Thankfully, I just had mild symptoms for one day and that was it. I still mask up but it makes a world of difference to me knowing that my risk being hospitalized or dying has dramatically dropped. It’s a big weight off.
80% of dying covid patients in Europe are overweight, have diabetes or other underlying conditions. And are over 70 years old. I know that overweight is a big problem in the VS. Death among young people is almost non-existent. Why should they be vaccinated? You are almost as contagious with your vaccination as you are without.
there is no RCT study for booster shots, only elevated antibodies. Which in turn does not work well in the elderly. In addition, antibodies are not everything. You also have T cells etc…
So 20% of deaths from COVID are in healthy people – like this specimen of health?That’s actually pretty scary. This guy was climbing mountains just weeks before he got COVID. He died within a week. – https://www.theguardian.com/world/2021/aug/04/fit-and-healthy-man-42-from-southport-who-rejected-vaccine-dies-of-covid
While most people who die are older or have health problems many young healthy people have also either died or now have long COVID. https://abcnews.go.com/US/wrecked-lives-families-young-adults-died-covid-19/story?id=74148061
I understand the reluctance that some people with diseases like ME/CFS have regarding getting vaccinated. Healthy people who choose not to get vaccinated are playing with fire.
Check out this “COVID-19 has killed thousands of young Americans. This is not just a tragedy for the elderly.”
https://www.businessinsider.com/young-americans-dying-historic-numbers-covid-19-2021-1
Chance of death after positive corona test before the vaccinations a year ago. Age: 10-29 0.00%, 30-39 0.01%, 40-49% 0.02%, 50-59 0.05%, 60-69 0.33% 70-79 2.08% 80 -89 7.77%, 90 > 12.04% overall 0.40% Data base Health organisation
There are just as many counterarguments to your arguments. Many ME patients and healthy people regret taking the vaccine. Many young men have heart infections and other health problems after vaccination.
Some ME experts warn against vaccinations in ME patients. Associated New Zealand ME Society (ANZMES), found nearly 20 percent of the 359 survey participants had experienced a relapse, with worsened symptoms that had not returned to baseline, after taking the Pfizer vaccine.
The findings will be submitted to the Immunisation Advisory Centre (Imac) and the Ministry. Tate has also submitted data to Imac over his concerns.
https://www.rnz.co.nz/news/national/456598/expert-says-chronic-fatigue-sufferers-should-be-exempt-from-covid-19-vaccination
I think we agree that corona is a serious virus. But in my opinion, (this) vaccination is not the solution or the holy grail. Whether or not to vaccinate must therefore be a well-considered decision based on good information. There is now a promotional campaign in the media and often one-sided information. Because otherwise the willingness to vaccinate will decrease. I argue for more nuance And more transparency.
But lets go back on the topic neuroinflammation. Very interessting blog.
Beware that the Dutch MECFS landscape is highly politicized, and that a number of this study’s authors have a vested interest in not finding anything.
The Netherlands certainly has quite a history. It and the UK really drove the CBT/GET work for decades but GET has been officially discarded and hopefully Dutch researches – there are some really good ones (Van Campen/Visser spring to mind) will focus more on biology.
I just went through the papers each author has published and I’m seeing almost all biological work. The lead author has been studying Q-fever, sometimes in combination with ME/CFS, for quite some time. He’s done multi-omics, cytokine, genetic, mitochondrial, B-cell, interferon, CXCL-9, monocytes and now neuroinflammation studies. (Quite an effort there). Yes, I do see a meditation/CBT and a CBT paper but the bulk of his work has been tracking down the biological basis of Q fever syndrome and sometimes ME/CFS.
https://pubmed.ncbi.nlm.nih.gov/?term=Raijmakers+R&cauthor_id=34815320
Interestingly enough Mike vanElzakker just came out with preliminary findings of neuroinflammation in ME/CFS. And he was wise enough to do this with patients meeting ICC and not Fukuda:
https://twitter.com/MBVanElzakker/status/1465774712886214658
Thanks Herbert. That’s good to hear. It’s not the widespread neuroinflammation yet that some other studies have found but the anterior cingulate cortex makes a lot of sense.
How does it make sense to ME/CFS Cort? I don’t understand.
If the anterior cingulate cortex becomes damaged, however, the patient will struggle to associate certain actions with emotions such as fear or sadness. This can lead to anti-social behavior because the person does not realize that their actions can hurt others or cause them distress.
I wouldn’t say the ACC would be my brain region of choice to be disturbed in ME/CFS but the ACC actually does a lot more than that and it’s been implicated several times in both ME/CFS ( https://pubmed.ncbi.nlm.nih.gov/26602611/ https://pubmed.ncbi.nlm.nih.gov/30430664/) and FM and central sensitization ( https://pubmed.ncbi.nlm.nih.gov/24508406/) . It affects pain perception, cognition, attention, effort and others.
Upregulation of the ACC could conceivably result in a boatload of body sensations which I would suggest are almost as much of a highlight of ME/CFS as is PEM. I am often drowned in body sensations.
It also appears to regulate the autonomic nervous system (https://pubmed.ncbi.nlm.nih.gov/33041747/),
This is a link to a very interesting video by Michael VanElzakker and Amy Proal called ‘How environmental factors may impact vagus nerve/brainstem/neuroinflammation in ME/CFS and Long Covid.’
https://www.youtube.com/watch?v=UY2vwDbLlno
This study employed the Fukuda criteria, which does not require Post-Exertional Malaise as a symptom. It is the same flawed criteria that was used in the PACE Trial study. Study the wrong people, get a wrong answer.
To be honest I’m not convinced that using the Fukuda criteria is automatically going to lead to a null or negative result. While it would, of course, be better to use the Canadian or similar criteria, studies used the Fukuda criteria for decades before the others came into use and many of them did find abnormalities. It might be as simple as they used the wrong criteria but it’s not necessarily so IMHO 🙂
How the hell can a person have ME/CFS without PEM? Those criteria sound useless imo. Seriously, CFS without PEM?!?!? Would that not just be a different condition altogether?
I just read a most interesting article on the difficulties of researching ill defined illnesses, especially long-Covid but also ME/CFS.
https://www.wired.com/story/to-help-people-with-long-covid-scientists-need-to-define-it/
Apparently the one factor that most predicted long-Covid symptoms was the belief that a patient had a Covid infection–whether it could be substantiated by antibody tests or not. This led the author to speculate that symptoms might be from Covid or something else including a confluence of factors–not necessarily psychological in nature.
The long-Covid studies are having the same difficulties as much of the research you just discussed, Cort–how to define and what to test.
In my personal life, I am having noticeable success with using oxaloacetate, albeit at a much lower dose than recommended. I did find a cheaper (probably one time only or I would share) source for it.
I am also trying proteolytic and fibrolytic enzymes to help with some scarring I have and think that they might be helping my fatigue as well. Some of their functions are to promote digestion and circulation in addition to helping the body with tissue remodeling. They are also proported to help with inflammation although the research on that is thin, probably because they can’t easily be be patented. Given that the biome and ME/CFS seem to have some sort of connection, this makes sense to me.
I do hope researchers will still keep open minds instead of glomming on to neuro-inflammation as the main suspect.
@nancy B.
may i ask how much oxaloacetate you take? and is it with the vitamine c from jubilance or the pure form from benaene? how many mg oxaloacetate an how do you take it? in the morning, split up in doses, etc? I had contact with alan cash and he wrote that nobody in kaufmans study was so bedridden (98%). may i ask your level of functioning and what you mean by noticeable success with using it. And how fast you noticed something and if you know of interactions with meds? and does the effect build up over time like kaufman said? thanks!!! sorry for the many (personal) questions. ps and are you in menopause? i am. the jubilance is made for pms, that stands on the bottle.
@konijn
You ask a lot of questions! That’s just fine so apologies for a long answer.
I started out with Jubilance 150mg once per day and within about 2 weeks did notice a very slight increase in energy. I try to experiment with one thing at a time so I will know if it has any effect. About three weeks on, I bought another two bottles so I could take one capsule in the morning and one in the evening–total 300mg. per day. The positive effects were stronger. I decided to go for a higher dosage but even the ME/CFS package price offered by Jubilance made me cringe and was almost the same as buying single bottles on their monthly plan. I scoured the Internet until I found a more economical source–but it wasn’t tons cheaper.
And yes, I’m in menopause. Before the Jubilance, I was functioning at about 40% most of the time with an occasional ‘better’ day. To put it another way, I was couch bound, but not bed bound. I could barely do anything actively productive (like dishes, showering etc.) on my ‘off’ days.
Now with oxaloacetate it’s about evenly balanced with one day ‘off’ and one day ‘on’ with the ‘on’ days at about 60% of what a normal person might accomplish. I must admit that I have problems with pacing and can’t always tell where my energy boundaries are so some of the ‘off’ time might be because of PEM.
The vitamin ‘C’ in Jubilance is used to stabilize the product. My new batch doesn’t have ‘C’ in it and each capsule is 200mg. I will be starting at 2X per day soon. I’m exceedingly sensitive to substances and am also a very tiny person and so think I may not need as much as Kaufman’s study. So far I haven’t read about any significant interactions nor have I experienced any symptoms I could attribute to adding the oxaloacetate.
I have noticed that the improvements seem to be related to dose and I will continue to titrate upwards with morning and evening dosing. It hasn’t gotten me ‘cured’ but the effects are very encouraging. Hope this helps.
thank you so much for the energy you took to explain everything!!!! it gives me a bit of hope because Alan cash said that in kaufman’s study where not so bedbound ME/cfs people tested.
i can only hope it works for me to because i am desperate and decline and decline. thanks for “the spoons” you used for me!!! if you want and can, let me know, maybe in another post from healthrising, how it works without vitamine c, the doses, and how it is going. thanks so much!!!
@konijin,
Thank you too. I’m always happy to help!
I’ll keep in mind further reporting…
My brain appears to be highly sensitive in many different ways. I don’t have a formal diagnosis of anything or receive any medical help, so I work things out myself by trials and errors. I’m aware of Jarred Younger PhD and some of the other studies into neuro inflammation.
My main issue is with food sensitivities and that makes life fairly complicated. However, I’ve cobbled together my own version of an anti inflammatory diet and have focused on other changes to hopefully reduce inflammation, like sleep, circadian rhythm, lessening my sympathetic nervous system dominance & trying to enhance my parasympathetic nervous system response and so on. Whatever I’m doing, something is working.
I can still easily fire up my brain, so this is an ongoing project, I focus on daily. This aspect of my brain’s irregularities specifically started after I had a high temp 41°c, for probably weeks, in 2017. I wonder if I had swine flu? It took me months to make any improvement & I’d just had a bacterial infection & IV antibiitics + two more courses of antibiotics & then I had this very nasty flu. My brain reacted when I started to eat – particularly anything that was processed and high sugar. Also if I irritate my stomach (gastritis) or my gut (IBS), then I’m in big trouble with my brain. It feels like it’s smouldering and I can feel this having a wide ranging effect.
It’s been nearly four years, so the passing of time may have helped but I can still trigger it now. (I’d add that my persistent lack of energy did not seem to be necessarily linked directly to this. That’s another issue for me.)
After taking 100mg of oxaloacetate I get a headache. Does anyone else find this?
Why is time and money being wasted on studies using any definition other than CCC or ICC??? All other definitions will include patients who don’t actually have ME/CFS and will dilute the findings of patients with actual ME/CFS.
I don’t understand why researchers are still using other definitions. There is already a severe lack of funding for research into ME/CFS…why waste those precious dollars on studying a group of patients who may not have the actual disease being studied. This is so obvious that I can’t understand why any researcher would do that.
One of the ME/CFS organisations should contact the researchers in the field and advocate for the use of a strict definition that increases the likelihood that the subjects in the study group actually have ME/CFS!!!
I’d be interested to hear what results they found for their neuroinflammation autism study, i recently heard of (new) research finding roughly 80% of ME patients to be autistic (something I found out about myself just over a year ago – after 18 years of ME – I’m autistic, with ADHD and PDA additionally. My assessor told me that in the majority of the late stage assessments for autism she did, most of them have ME, so I think there is truth in this. Will be interested to see more research on both these topics.
Talulah, I am very curious about this. Can you provide some links to this research? Thank you!
I started ME/CFS at 24, diagnosed ASD at 68. Would be very interested in this idea.
I offered myself to participate in this dutch research. I explained my symptoms. I definitely fit in the ICC ME category, looking back that is. But the researcher could not include me because I did not fit the criteria. I said to have extreme fatigue with decline in bloodflow to brain when standing. Because of later I was not included in research. I think they only selected pure on Fatigue. They do not have a clue on what me/cfs is about.
The Dutch study uses a old method on a very small number of patients without taking into account the severity and stage of their disease. Therefore I think nothing can be concluded from it. Too bad for the energy and money lost and also for us…
I started ME/CFS at 24, diagnosed ASD at 68. Would be very interested in this idea.