Small fiber neuropathy (SFN) describes damage to the small, unmyelinated, nerve fibers found in the skin and/or eyes. It is associated with symptoms like numbness, tingling, burning sensations, etc.
If all that people with chronic fatigue syndrome (ME/CFS), fibromyalgia (FM), postural orthostatic tachycardia syndrome (POTS), long COVID, and related diseases had to worry about was small fiber damage in their skin and/or their eyes, SFN might not be such a big concern. Because the small nerve fibers are found throughout the body, though, damaged nerve fibers could be causing a lot of mischief. For instance, some researchers believe that SFN may be causing microcirculatory problems in ME/CFS.
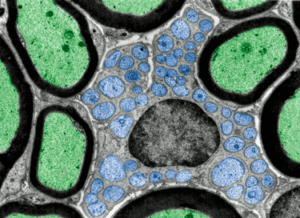
Myelinated (green) and unmyelinated or small nerve fibers (blue).
Numerous studies indicate that a distinct form of small fiber neuropathy called small fiber polyneuropathy, which includes damaged (small diameter) and missing small nerve fibers, are found in these diseases.
Peter Novak, MD, Ph.D. is right in the middle of all that. The Chief of the Division of Autonomic Neurology and Director of the Autonomic Laboratory at Brigham and Women’s Hospital, he’s a board-certified neurologist and autonomic specialist, and the author of a book on autonomic testing.
Novak discovered a form of orthostatic intolerance called orthostatic cerebral hypoperfusion syndrome (OCHOS) in people who became dizzy, fatigued, etc. upon standing; i.e. had orthostatic intolerance (OI), but didn’t experience a high heart rate (POTS) or low blood pressure (orthostatic hypotension).
Check out Novak’s papers and you’ll see some familiar faces (David Systrom, Donna Felsenstein). With David Systrom, Anne Oaklander, Ron Tompkins, Donna Felsenstein, and Michael Van Elzakker in the Boston area, Novak is working in the middle of a particularly fertile ME/CFS/FM research environment.
THE GIST
- Small fiber neuropathy concerns damage to the small, unmyelinated nerve fibers found in the skin and eyes of people with fibromyalgia, chronic fatigue syndrome, POTS, long COVID, and other diseases.
- Because small nerve fibers are also found in other areas, damaged fibers could conceivably, cause many more problems.
- A neurologist in Boston, Peter Novak, has been exploring the role small nerve fiber problems, blood flows to the brain, and dysautonomia play in a number of different diseases. Over the past couple of years, Novak has uncovered two different kinds of orthostatic intolerance.
- Novak and others have found a similar signature: high levels of small nerve fiber damage, reduced blood flows to the brain, and dysautonomia in long COVID, POTS, ME/CFS, and post-treatment Lyme disease (PTLD). When it was assessed, low CO2 levels (hypocapnia) were found.
- In the PTLD disease paper, Novak suggested that the vascular problems in PTLD may be similar to those found in diabetes.
- Larger studies are needed to confirm these findings in long COVID. If they do, they could provide the impetus for: a) long COVID researchers to demonstrate how important they are, and b) get at the cause and explore new treatment options that could apply to all these diseases
- Several COVID-19/long COVID studies have proposed a raft of treatment options that are available but have never been considered in ME/CFS, FM, etc.
Long COVID
Novak’s first long-COVID paper described a long-COVID patient who developed chronic fatigue, OCHOS, and small fiber neuropathy (SFN). First misdiagnosed with Lyme disease, the patient later recovered with IVIG.
Novak and company just put a small group of long-COVID and POTS patients through an intense battery of autonomic and immune tests (autonomic (Valsalva maneuver, deep breathing, sudomotor and tilt tests), cerebrovascular (cerebral blood flow velocity (CBFv) monitoring in the middle cerebral artery), respiratory (capnography monitoring) and neuropathic testing (skin biopsies for assessment of small fiber neuropathy) and inflammatory/autoimmune markers.)
The study findings mostly mirrored what other studies have found. Every patient in the study displayed dysautonomia. Narrowed blood vessels leading to the brain were clamping down on blood flows in both groups. Small fiber neuropathy was even more pervasive in the long-COVID patients (89%) than the POTS patients (63%). The POTS patients hyperventilated during the tilt table test but the long-COVID patients, interestingly, did not. Both, however, displayed low CO2 levels. Increased inflammatory markers were common in both groups.
The research community seems to have quickly glommed onto the role the autonomic nervous system plays in long COVID with at least 14 papers over the last six months focusing on it.
In June, the American Autonomic Society released a paper calling for more research into “long-COVID POTS”. Besides several case reports, including a large one which specifically noted that POTS often occurs after infections such as Lyme disease, Epstein-Barr virus, and influenza, several studies suggest orthostatic intolerance and POTS often follow a COVID-19 infection. POTS interventions such as ivabradine have been reported to be helpful in long COVID.
(Note that studies assessing the incidence of POTS or orthostatic hypotension, probably significantly underestimate the extent of orthostatic intolerance present as Novak and Van Campen et al. have shown that reduced blood flows to the brain can occur even in the absence of increased heart rates/drops in blood pressure. The Van Campen/Visser/Rowe studies, for instance, suggest low blood flows to the brain are found in everyone with ME/CFS).
The “Preparing for the long-haul: Autonomic complications of COVID-19″ paper warned about prescribing exercise, and specifically mentioned ME/CFS:
“Future research will also need to prioritize mechanisms involved in chronic fatigue of those with PACS, how it relates to the diagnosis of post-COVID ME/CFS”
The orthostatic intolerance/POTS/long-COVID connection seems strong but we need larger studies to firmly establish it. Those studies are surely coming.
Post-Treatment Lyme Disease (PTLD)
Both ME/CFS and POTS are often triggered by an infection and both have been associated with long COVID – but what about other fatiguing, exertion intolerant post-infectious diseases such as PTLD? Given that PTLD looks very much like POTS and ME/CFS, it wasn’t surprising to see Novak – who seems dedicated to exploring every subset in this group of diseases – turn to it.
Novak’s small 2019 study, “Association of small fiber neuropathy and post-treatment Lyme disease syndrome“, suggested that all these diseases may be of a piece; i.e. the PTLD group displayed the same general characteristics as ME/CFS, POTS, and long COVID: reduced blood flows to the brain, small fiber neuropathy, and mild to moderate autonomic nervous system failure (either sympathetic or parasympathetic). The authors proposed reduced blood flows to the brain were causing the cognitive problems and fatigue found.
That was an important study as just two older – and apparently long dismissed papers – have warned about autonomic nervous system functioning in Lyme disease. While cerebrovascular complications such as stroke are well known in acute Lyme disease, it doesn’t appear that anyone prior to Novak thought to check whether reduced blood flows to the brain are occurring in PTLD.
Back in 2009, though, a study, “Levels of sVCAM-1 and sICAM-1 in patients with Lyme disease“, found high levels of two vasoactive or blood vessel affecting markers (vascular cell adhesion molecule-1 (sVCAM-1), soluble intercellular adhesion molecule-1 (sICAM-1)) in both the serum and spinal cord fluid. It specifically warned that increased activation of the endothelial cells lining the blood vessels (i.e. blood vessel constriction) might be happening in Lyme disease.
A Diabetes Connection?
The sVCAM-1/siCAM-1 connection brought diabetes into the discussion. The same two vascular markers found increased in PTLD (sVCAM-1, sICAM-1) have been associated with vasoconstriction of the blood vessels leading to the brain in diabetes.
Diabetes is a metabolic disease often characterized by small nerve fiber problems. ME/CFS/FM may be a metabolic disease in which SFN often occurs as well. Could the two diseases be more closely connected than we know? Years ago, Marco wrote a blog for Health Rising suggesting that they might be, and recently metformin – a drug mostly used in diabetes – has been proposed for fibromyalgia.
Somehow, neither of these vasoconstricting factors have been assessed in ME/CFS, POTS, or FM but guess what? These factors have already been assessed several times in COVID-19 – and have been found elevated.
A Spanish group found those factors as well as a remarkable array of blood vessel stressors (endothelial stress products, complement activation, fibrinolytic dysregulation} in COVID-19. They noted, interestingly, that while endothelial problems occur in both COVID-19 and sepsis they differ. Sepsis is an intriguing potential model for ME/CFS because, if memory serves, blood vessel damage more than inflammation (cytokine elevation), plays the critical role.
One endothelial-focused COVID-19 paper proposed that a variety of diabetes-associated drugs – none of which are currently being used in ME/CFS/FM/POTS – may be helpful in long COVID. (Blog coming up.) Another recent review of the endothelial connection in COVID-19 – which bluntly stated that COVID-19 is a disease of the blood vessels – similarly suggested a wide array of mostly immune-oriented drugs may be helpful. None are currently being used in ME/CFS/FM/POTS.
Once again, we’re seeing COVID-19 highlighting understudied aspects of ME/CFS, FM, POTS, Lyme, and related diseases and presenting new treatment possibilities. The vast majority of these treatments, it should be noted, are already available and could quickly provide relief if studies show they are helpful.
A Mast Cell Activation Syndrome (MCAS) Connection?
Novak keeps digging – and finding new connections. Over the past couple of years, he’s uncovered two new forms of orthostatic intolerance and unearthed evidence of endothelial damage and orthostatic intolerance in PTLD and long COVID. In 2019 he turned to another understudied disease – mast cell activation syndrome (MCAS).
In “Mast cell disorders are associated with decreased cerebral blood flow and small fiber neuropathy“, Novak put people with MCAS through a similar barrage of tests (autonomic testing encompassing Valsalva maneuver, deep breathing, sudomotor and tilt tests with cerebral blood flow velocity (CBFv) determination, and skin biopsies for small fiber neuropathy (SFN).)
The same suite of results found in long COVID and POTs popped up: reduced blood flows to the brain, small fiber neuropathy (found in 80%), and mild to moderate dysautonomia.
Next Up – Ehlers Danlos Syndrome (EDS)?
Several studies suggest that POTS and orthostatic intolerance are common in hypermobile EDS. I couldn’t find any studies on endothelial dysfunction, small fiber neuropathy, or blood flows to the brain in EDS.
Conclusion

Finding core connections in these diseases would be a godsend.
Novak and others have been unearthing a similar suite of biological abnormalities in COVID-19/long COVID, ME/CFS, POTS, and PTLD. Most of the studies have been quite small but if the findings are validated by larger studies, they could point to core biological problems present in all these often-post-infectious diseases. They include endothelial cell issues which reduce blood flows to the brain, small fiber neuropathy, evidence of dysautonomia, and, when it was assessed – low CO2 levels (hypocapnia).
Exercise studies finding reduced energy output, early entry into anaerobic energy production, reduced heart rate compliance, etc. have already shown up in long COVID and ME/CFS. Time will tell if similar issues show up in POTS, PTLD, MCAS, and EDS but thus far, a nice core appears to be emerging in these diseases. Finding core issues in these diseases is what we’ve been hoping for, as a breakthrough in one could lead to breakthroughs in the others.
The monster long-COVID studies being produced in the NIH Recover Initiative should tell us if this suite of seemingly connected abnormalities is present. If it is, the real fun starts when these massive research efforts begin to dig into them and try to figure out what’s causing them and how to fix them. That would be sweet, indeed, for a bunch of often disabling diseases that the research world has paid too little attention to for far too long.
BIG (little) Donation Drive Update

Connections, connections, connections… I’m a lumper to the core – I seem to be genetically predetermined to look for connections. That’s why Health Rising looks not just at ME/CFS, long COVID, and FM literature but at diabetes, POTS, EDS, PTLDS, multiple sclerosis, and many others. If finding potential connections with other diseases causes the blood flows to your brain to increase, please support us.






My friend has heds…exact same symptoms. Improved on abilify. Has pots etc.
I really hope the connection is made as I truly believe it’s the cause of autoimmunity and dysautonomia. Viruses etc, just take advantage of the weakness.
I really hope regenerative medicine gets a handle on it
Such good reporting Cort. I wish someone with budget authority followed your blog and chased these things through. It’s frustrating when there’s so many leads, but no Batman to track the Riddle down.
Time will tell but it seems, to me. at least. like there are some real possibilities here. I remember Ron Davis saying whoever figures out ME/CFS will figure out a lot. Let’s hope he’s right.
Since ME/CFS basically presents the best example of the failure of the ability to exert oneself I would think he would be right. Fundamental things are going on.
Re: SFN & EDS. A correlation between these disorders seems likely. David Saperstein, MD, of Phoenix, AZ, a neurologist known for his expertise in treating EDS patients routinely tests EDS patients for SFN. He ordered a punch biopsy for me. I was surprised when it was negative.
The finding of possibly universal low cerebral blood flow in ME/CFS is intriguing. I probably also had tick-borne disease dating to the time of onset of my illness, meaning I have hEDS, ME/CFS, CCI, TBD (not exactly PTLD since it went without treatment for over 20 years), and MCAS. I hope to avoid adding long-COVID to the picture!
As I understand it, eds is very much a field in flux. It’s just not received a lot of attention and scientific enquiry. I wouldn’t be surprised if there are more types to be discovered.
Have you heard of the rcccx theory. I totally think that’s spot on.
Also the reduced brain flow is also a feature if autism and is often because of incomplete healing cycles in part.
Autistics have connective tissue disorders too.
I hope and am pretty sure all these connections of been made tho. What suprises me is that it seems obvious to me that stem cell and crispr seemed to be a root cause solution. I’d love to see money thrown at that
Here’s a blog on the RCCX hypothesis
https://www.healthrising.org/blog/2018/08/09/rccx-chronic-fatigue-fibromyalgia-eds-pots/
I went from probable Lyme, to ME/CFS (with SFN and OI/POTS) to MCAS. Totally makes sense to me that they are all related! This is looking hopeful that they will find a cure or at least management options soon.
Hi Cort, although not mentioning endothelial cells proper, this might help you in your next blog on EDS and dysautonomia;
https://onlinelibrary.wiley.com/doi/10.1002/ajmg.c.31951
It comes from a brand new batch of collected research articles on EDS.
Interesting and bang on I’d say.
I can’t wait to see my docs face when all this comes out.
Last week, she said to me, the reason your sick is that you didn’t take therapy seriously. I actually did.
Ugh, so sorry Oliver.
Thanks another Anne
look at that! Just what I was looking for – and hot of the presses. 🙂 Thanks
Es un gran problema el uso de “Central” pues hay una corriente aquí en España que diirectamente aplican eso para la derivación a psiquiatría. Pues consideran una desrregulación de neurotrasmisores = patología psico…….
I represent another orphan group under the CFS umbrella : Fluroquinolone Toxicity Syndrome. Many/most of us have CFS, along with POTS,MCAS, ANS dysfunction, neuropathies,CNS damage and immune disorders post Floxing. This research is connecting the dots and validates the connections. It’s encouraging that all this research is progressing with with the speed that Long COVID syndrome precipitated. A silver lining ?!
Thank you Cort for keeping us informed !
Thanks, Carolyn for representing the fluroquiolone toxicty syndrome patients here!
Wow Carolyn, I’m sorry. I didn’t know Fluroquinolone Toxicity Syndrome was actually a recognized syndrome. Just looked it up. I did know that tendon ruptures were listed as potential side effects and eventually the FDA put a black box additional label warning people with connective tissued disorders NOT to take any of the drugs in this category.
Some years ago I took one dose of Cipro and afterwards I could not control my eye movement. They were just rolling around everywhere. I was very scared! I can’t imagine what they did to you…
Here’s a blog on the RCCX hypothesis
https://www.healthrising.org/blog/2018/08/09/rccx-chronic-fatigue-fibromyalgia-eds-pots/
Great
Excellent article! I have a screenshot of Dr Rowe’s presentation at the Dysautonomia International presentation w/ Stanford back in September. His results include POTS, ME/CFS, AND hypermobility (I don’t believe it specifically states EDS). I remember because the average reduced cerebral blood flow for people with POTS, ME/CFS, & hyper mobility was 33%! I believe it was about a 25% reduction for ME/CFS on it’s own! Mind blowing stuff!
I need to search again to find the full study…
Thanks as always Cort! I can’t tell you how valuable THE GIST section of your blogs is for me. When I’m in an intense flare up of brain fog (which is the majority of the time these days, unfortunately) I can at least still follow your blogs.
The Small Fiber Neuropathy connection is fascinating. I’ve always thought all of these labels are different names for the same condition. Or at the very least connected by different post-infectious reactions. I still don’t know what possible treatments are though for SFN.
And while interesting, I don’t think the RCCX hypothesis is the one. Genetic mutations have lead to a lot of failed research, especially the more we learn about epi-genetics and how quickly genes turn on and off.
I have a strong hunch to the opposite. I guess time will tell. I hope you’re right. I think it may be easier to treat.
But autoimmunity, smouldering viruses, dysautonomia, IBS, anxiety etcall group together in tissue disorders.
This is such an interesting area for me. I’m becoming more aware of the possibility that some of my most persistent issues may be related to blood flow in my brain.
Every day, the one symptom I’m really vigilant in picking up quickly and fear the most, is a dizzy, lightheaded feeling. I have had the racing heart and nausea too but more often I just feel a bit, or sometimes very, faint. I’ll also have difficulty thinking clearly. I’ve experienced the feeling that my brain is clamped and sort of shrunk – not a good one either.
I wonder whether I have MCAS, as I have do many immune reactions in many areas of my body. I am very sensitive to most food and I think my brain flow issues may be related to reactions I have with certain food. I take antihistamines, which seem to help but it’s not easy finding enough food that I can eat, that don’t cause issues. My food sensitivities also seem to trigger my sympathetic nervous system.
It’s so good to see that Peter Novak MD PhD, is thoroughly focusing on this area.
Off topic. Cort, can you make a poll with experiences of Novavax and ME/CFS/POTS?
Maybe this vaccin could be an option.
So many possibilities, and so many here who are ‘dying’ to have their particular markers sleuthed out, but no researcher has enough money to do so much that is needed for soooo many.
Challenge to those few rare philanthropists out there who can invest in research—- could you work alongside researchers to hire lab techs and testing solutions/ needed items, and allow cfs me etc patients to pay a small fee to get answers and provide additional data points for researchers? That would be outside of the limited size and scope of their research budgets?
But benefit patients and enlarge info to help crack this?
Please, for people have been sent to the moon……please help people here who have not just their two feet on earth, but for most of the day their whole body flat on earth—sort of like the astronauts when they come back—- but we haven’t gone anywhere.
Please believe, and become invested wholeheartedly.
Believe me, people here would be over the moon to receive in time and targeted help.
sincerely, sunie
I’ve just seen a reminder from Amy Proal PhD about her webinar (Thurs Jan. 6th, 4-5pm ET) put on by the Global Interdependence Center and the Long Covid Initiative.
Amy Proal tweeted:
‘Hey! Join me at this webinar where I will attempt to connect #SARS-CoV-2 reservoir, herpesvirus reactivation, #microbiome dysbiosis, autoantibody production, microclots, vasculature issues, neuroinflammation, vagus nerve signaling + small fiber neuropathy in one #LongCovid talk!’ Register here:
/us02web.zoom.us/webinar/register/WN_-64g3ph2RS-IX5KgOg6rmQ?mc_cid=398b1c2cd1&mc_eid=cf0cc0ae81
That link didn’t work, so this one might and then you click to register…
https://solvecfs.org/solve-mecfs-initiative-webinars/
Scientists May Have Solved the Mystery of Long COVID – with ME/CFS mention:
https://www.zmescience.com/medicine/scientists-may-have-solved-the-mystery-of-long-covid/
Guardian article by the scientist postulating the microclots, Resia Pretorius:
https://www.theguardian.com/commentisfree/2022/jan/05/long-covid-research-microclots?CMP=Share_iOSApp_Other
I have OCHOS but before I was diagnosed I tried Metformin, because we thought I might have reactive hypoglycemia. Unfortunately it made me feel really sick (side effects) and I had to stop it after a short time.
I wonder whether Dr Novak ever tested any of his OCHOS patients for sVCAM-1 and sICAM-1. If so, he hasn’t published on it.
Dr Novak mentioned in a 2023 conference talk that his lab had assessed several hundred patients with EDS and found that their cerebral blood flow fell abnormally during tilt table testing.
Unfortunately the conference talk wasn’t published because the audio quality was too poor, but I presume he will publish the results in a journal soon.