

This was a surprising study – not because of what was done – but because it hasn’t been done before. Since the two branches of the ANS (the sympathetic/parasympathetic) control the heart rate, heart rate variability (HRV) is a good way to assess at a basic level, the health of the ANS.
EKGs are used to measure HRV – which are low in ME/CFS, FM, and other diseases.
Low HRV – the kind typically found in fibromyalgia (FM) and chronic fatigue syndrome (ME/CFS) – suggests that the sympathetic nervous system (SNS) or fight/flight system has achieved dominance – leaving the parasympathetic nervous system (PNS) or rest/digest system in the dust.
The HRV test simply tracks a known pattern: our heart rate speeds up when we inhale and slows down when we exhale. The inhalation triggers SNS activation while the exhalation triggers the parasympathetic nervous system.
Since we both inhale and exhale more when we exercise, an exercise test provides a nice glimpse into how good our autonomic nervous system is at handling stress. A healthy, flexible cardiovascular system should be characterized by a heart that’s able to speed up and slow down rapidly during the long inhalations/exhalations done during exercise. While that test has been done in fibromyalgia, it hadn’t yet been done in ME/CFS.
The test’s simplicity can be deceiving. Low heart rate variability (HRV) – or overdominance of the sympathetic nervous system – has been associated with a host of negative outcomes, including an increased risk of early mortality and many types of cardiovascular disorders. The fact that low HRV has been found in so many disorders – from ME/CFS to FM to IBS to cardiovascular, respiratory, and gastrointestinal diseases and autoimmune conditions as well as anxiety and depression – suggests not being able to shift into the rest and digest stage is common in many chronic illnesses.
Quite a few different statistical assessments of heart rate variability (e.g., time domain, frequency domain, and non-linear analysis) can be made, and more are coming. The low-frequency LF/HF ratio is the most commonly used and is usually taken to reflect sympathetic/parasympathetic activity. (This isn’t completely accurate as the LF measure is affected by both sympathetic and parasympathetic activity – but appears to be good enough.)
Given the difficulty that people with ME/CFS have with exercise – and the role the autonomic nervous system (ANS) plays in delivering blood and oxygen to the muscles during exercise – one would think that an assessment of ANS functioning using HRV would have been one of the first studies run – but no, the 2021 Van Oosterwijck paper, “Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome“, from the Nijs group in Belgium is the first to focus on this issue.
The Study
This 40-person (20 ME/CFS; 20 sedentary healthy controls) study put the participants through a submaximal exercise test that had the participants reach 75% of age-predicted maximum heart rate – a heart rate that is probably pushing ME/CFS patients’ hearts to the limit. The test, however, was quickly finished with the average time on the bike about 4 minutes. Heart rate, blood pressure, and various heart rate variability parameters were measured.
Results
The ME/CFS patients “demonstrate(d) a reduced functional capacity for exercise”. the authors
During Rest
As studies have often found, things were pretty normal at rest. Heart rate, blood pressure, and even HRV were all normal. One interesting exception occurred though. While LF/HF ratio (mostly sympathetic/parasympathetic) was normal – indicating that sympathetic dominance was not present – the values for each factor were lower than normal in the ME/CFS patients.
That suggested that the autonomic nervous system in the ME/CFS group might be underactive. While it might be responding in a balanced fashion, it may not have been responding enough. That, the authors asserted, potentially implicated something called the baroreflex.
The Baroreflex
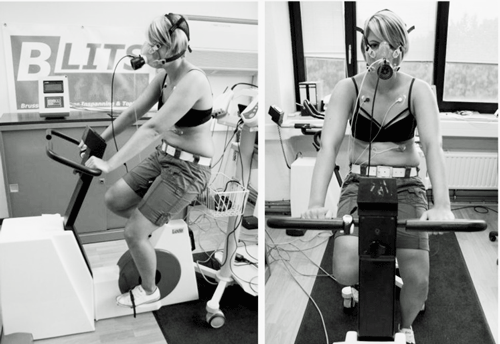
This short exercise study suggested that the autonomic nervous system problems in ME/CFS really kick in during the post-exercise recovery period when the parasympathetic nervous system fails to kick in.
The baroreflex or baroreceptor reflex refers to a remarkable process whereby tiny receptors embedded in the major blood vessels of our hearts respond in fractions of a second to the artery wall as it changes shape (stretches) in response to very small changes in blood pressure. The baroreflex receptors respond by increasing the heart rate when blood pressure falls and decreasing it when it rises. This all happens in a fraction of a second.
Having these baroreceptors be a fraction off might not seem like a big deal, but in a rather dramatic demonstration of how interconnected everything is, it turns out that small problems with the baroreflex could cause a lot of mischief.
This is because the baroreflex is heavily tied into the stress response. Baroreceptor activation (which usually occurs when the blood pressure rises) inhibits the activity of the fight or flight, or sympathetic nervous system (SNS). It also appears to tamp down brain activity, and reduce pain levels and emotional volatility.
Studies suggest that having more sensitive cardiac baroreflex responses may result in increased pain inhibition, thus reducing pain. A less sensitive baroreflex – the kind that may have shown up in this study – could leave the stress response turned on – resulting in increased pain, central sensitization, etc. A 2017 exercise study by this same group, in fact, linked elevated blood pressure during the rest period to increased pain levels in ME/CFS. The authors concluded – as they did with this study – that parasympathetic “withdrawal” and baroreflex problems were present.
One fibromyalgia study found all three branches of the baroreflex response were inhibited – both at rest and when stressed. The results jived with a 2017 study which found that a reduced baroreflex response was associated with reduced quality of life in FM. Reduced baroreflex responses have also been found in adolescents and adults with ME/CFS and/or postural orthostatic tachycardia syndrome (POTS).
The fact that the signals the baroreceptors emit go through the brainstem (possible factor in ME/CFS/FM), activate motor neurons associated with the vagal nerve (another possible factor), and inhibit sympathetic nervous system neurons in the spinal cord (another possible factor) makes the whole process of baroreceptor activation all that more interesting. Could the baroreceptors have gotten whacked by something? Or is the signal not getting through?
During Exercise
A host of problems have been found during exercise in ME/CFS, but this study found few. Heart rate and blood pressure were normal and sympathetic nervous system domination – a typical finding since the SNS exists in part to enhance our ability to exercise – was found in both groups.
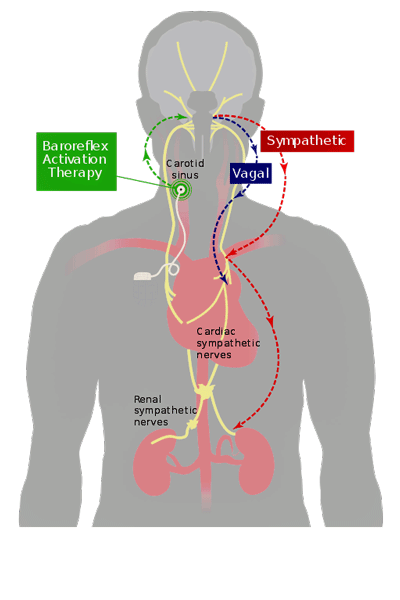
The baroreflex – which uses stretch receptors in the big blood vessels leading to the heart to adjust the heart rate – has been implicated in increased pain and other symptoms. Baroreflex problems have shown up in ME/CFS and in spades in fibromyalgia. (Image by Darrel Francis from Wikimedia Commons).
This wasn’t entirely unexpected. One-day exercise studies don’t pick up as many abnormalities as two-day exercise studies and the exercise period in this study was short and it didn’t measure the factors (VO2 max, anaerobic threshold, etc.) that are often abnormal in comprehensive exercise studies.
One finding did suggest that the sympathetic nervous system might not have kicked in in the ME/CFS patients as much as it did in the healthy controls. That could make sense. We know that when the heart is put under load during longer exercise tests, in a process called chronotropic incompetence, it appears to fade. The system then seems hyperactive and underpowered at the same time. It’s “on” much of the time, but when put under stress it quickly bottoms out.
After Exercise
It was in the 10-minute period after exercise that the autonomic nervous system abnormalities really appeared. Once the exercise has stopped, the parasympathetic nervous system should quickly put the brakes on the sympathetic nervous system and return the heart rate to normal.
Note what happened during the entire test period in ME/CFS. The heart rates of the ME/CFS patients were essentially the same as the healthy controls at rest, were a bit low (but not significantly low) during the exercise period, but were higher at the end of the recovery period.
In fact, by the end of the 10-minute recovery period, the heart rates of the healthy controls had returned to baseline but the heart rates of the ME/CFS patients remained significantly increased (p< .020) compared to baseline: they had still not recovered. That might seem an obvious result to anyone with ME/CFS who had attempted to exercise, or indeed anyone who has exerted too much in other ways. Long periods of apparently increased – or at least very noticeable – heartbeats can easily ensue.
A similar finding was also found in a recent fibromyalgia study which also found reduced HR recovery; i.e. abnormally increased heart rates at 30, 120, 180, 300, and 600 seconds after exercise. The increased heart rates after exercise provided an interesting contrast to the reduced heart rates the researchers found during exercise. Once again, we see a system that seems to be turned on but then bottoms out when stressed.
The study only assessed heart rate after the completion of exercise for ten minutes, but it turns out that a quick return to normal after exercise may be quite important. A 1999 study that followed almost 2,500 people found that a delayed decrease in the heart rate during the first minute after exercise was, surprise, surprise “a powerful predictor of overall mortality”.
The relative risk of dying in the participants (average age 57) doubled over the next six years in those with delayed heart rate recoveries. This didn’t mean they were likely to die, but their risk of dying had certainly increased. Since then, several studies have shown that quick heart rate recovery after exercise is a function of a healthy and responsive parasympathetic nervous system.
The authors of this study suggested that future ME/CFS studies examine the extent of the heart rate drop during the first minute after exercise in particular.
Heart rate variability (i.e. the sympathetic/parasympathetic ratio (fight-flight/rest-digest)) returned to normal in the ME/CFS patients during the ten-minute recovery period, but abnormalities suggested that all was not well.
The strange inhibited autonomic nervous system response occurred. While the HRV returned to normal, the reduced magnitude of both the SNS and PNS response suggested that a blunting of the autonomic nervous system had occurred. Plus, it took longer for the ME/CFS patients’ HRV to return to normal (@ 8 minutes), and the sympathetic nervous system was more activated. All in all, the authors stated the results “demonstrate(d) a reduced functional capacity for exercise”.
Noting that it’s unclear how to use exercise to return the autonomic nervous system to normal in people with a “dysfunctional stress system”, they suggested that an HRV-informed training regimen be tried in ME/CFS. Decreased morning parasympathetic activity would indicate that more recovery time is needed. Increased parasympathetic activity, on the other hand, would suggest it’s time to get a bit more exercise.

The healthy controls were able to recover quickly. The people with ME/CFS did not.
Conclusion
This Belgium study filled in a hole in the exercise saga in ME/CFS when it assessed what’s happening with the autonomic nervous system (heart rate, heart rate variability, and blood pressure) during and after exercise. The low heart rate variability found in MECFS, FM, and other diseases has been associated with the dominance of the fight/flight or sympathetic nervous system, and a withdrawal of the rest/digest or parasympathetic nervous system.
The Gist
- This study, for the first time, assessed autonomic nervous system functioning measures such as heart rate variability (HRV) in ME/CFS before, during, and after a short submaximal exercise stressor.
- Nothing abnormal was found during rest -before the short exercise period. During exercise, the autonomic nervous system measures assessed were mostly normal. However, the magnitude of the ME/CFS patients’ autonomic nervous system response appeared to be limited – something the authors proposed could be due to an impaired baroreflex.
- The baroreflex refers to a reflex triggered by stretch receptors in the arterial walls of the blood vessels leading to the heart. Increased or decreased blood pressure that causes the arterial walls to stretch triggers the baroreflex to adjust the heart rate.
- Because the baroreflex also inhibits the activity of the fight or flight, or sympathetic nervous system (SNS), regulates some brain activities and affects pain levels and emotional volatility, a balky baroreflex response could be quite problematic. Problems with the baroreflex have been found in spades in fibromyalgia and in postural orthostatic tachycardia syndrome (POTS) as well.
- The major finding was that while the heart rates of the healthy controls returned to baseline during the 10-minute recovery period, they remained elevated in the ME/CFS patients. It also took longer for the sympathetic nervous system to calm down in the ME/CFS patients, and their autonomic nervous systems displayed the same kind of inhibition that was seen during the exercise period.
- Some similar findings that have been found in fibromyalgia suggest that dysautonomia is common in that disease as well.
- The authors reported that their results “demonstrate(d) a reduced functional capacity for exercise”.
- This and other study’s results suggest that parasympathetic withdrawal is allowing the sympathetic nervous system or fight/flight system to remain active longer than normal in ME/CFS and fibromyalgia.
The baroreflex refers to a reflex triggered by stretch receptors in the arterial walls of the blood vessels leading to the heart. Increased or decreased blood pressure that causes the arterial walls to stretch triggers the baroreflex to adjust the heart rate.
Because the baroreflex also inhibits the activity of the fight or flight, or sympathetic nervous system (SNS), regulates some brain activities, and affects pain levels and emotional volatility, a balky baroreflex response could be quite problematic, and problems with the baroreflex have been found in spades in fibromyalgia.
The major finding in this study showed up in the 10-minute rest period. While the heart rates of the healthy controls returned to baseline, they remained elevated in the ME/CFS patients. It also took longer for the sympathetic nervous system dominance that accompanies exercise to calm down in the ME/CFS patients, and their autonomic nervous systems displayed the same kind of inhibition seen during exercise. Very short-term delays (within 1-minute) in heart rate recovery have been associated with an increased risk of mortality, and need to be assessed in ME/CFS and FM.
With the authors reporting that their results “demonstrate(d) a reduced functional capacity for exercise” in ME/CFS, we can add heart rate variability and autonomic nervous system tests to the invasive and non-invasive cardiopulmonary tests that have found the same in ME/CFS. Meanwhile, similar findings that have shown up in FM suggest that similar problems with autonomic nervous system functioning during exercise are present as well.
Once again, we see a withdrawal of the “rest and digest” or parasympathetic nervous system that allows the fight/flight or sympathetic nervous system to remain activated. Future blogs will search for ways to boost the rest and digest system.
While this study did not measure them it should be noted that very short-term (<1 min) reductions in heart rate recovery have been associated with increased mortality overall. Those studies, though, remain to be done in these diseases.






Oh my goodness. I have measured my HRV for quite a while. I would be considered mild ME, and just positive for FM. My HRV is relatively normal. The unusual stat is total power, and especially LF. Total power can be as low as 500, and LF often <100. Generally the ratio is around 0.1! I have contacted Elite HRV and asked about this and in other ME groups. I have discovered that it is a baroreflex problem. I am on the verge of pots, with HR increasing ~25-30 beats on lean test.
What could cause a baroreflex underactivity? And is there anyway to stimulate it? I’m so glad you are writing about this!
Baroreflex – my God, that is an interesting thing. I don’t know if those receptors have been damaged – that can happen if the nerves are damaged – or if the signal is getting disrupted on the way to the brainstem or after the brainstem. Probably other things can happen as well. Will definitely be checking the baroreflex out more.
I’m hoping that the trauma treatment that I participate in will relieve the fibromyalgia. This article is very helpful.
Do you have a sense of how this work relates to one of the studies the CDC shared at a webinar last year?
“Exercise Testing in the MCAM Study” by Dr. Dane Cook,
https://youtu.be/4utGRlsrRCo
That was a very interesting study. I’ve only heard parts of the webinar but will certainly cover it when the study is published.
Taking metoprolol or propranolol, commonly prescribed for hyper POTS or Benadryl, prescribed for MCAS, significantly lower my HRV.
What does this artificially lowered HRV say for health?
Interesting. Generally we want higher HRV but if it’s helping you that might indicate that you need more sympathetic nervous activation – not less. You wouldn’t be alone in that. We are heterogeneous group. I would imagine that in your case it’s helping your health.
I have hEDS (possibly TNXB mutation), I find that beta blockers help a lot. It could be that instead of helping directly perhaps what they do is reduce the triggering of the bodies reaction to what would otherwise be an overactive sympathetic nervous system. Perhaps one such reaction is the release of GPCR-aabs.
I wear an oura ring who alsomeassures HRV during sleep. since booster HRV had not been normal anymoer. declined also a lot. hr during day and night are totally crazy since booster. wish i never had it! before 2 vaccines also declined a lot! what does this mean? i do not seem/feel to recover anymore….still strugling for eating and drinking
I am experiencing same issues since Vaccin astion#1, illness, #2 passed out while driving!, Booster, eating and sleeping issues.
When in my 30’s, (30 years ago)experienced elevated heart rates, over 250bpm, 20 minutes after aerobics still walking it off, trying to get rates down. As a child, hypersensitive to the hammer on the knee, hand and elbow test. Jumped out of my skin! Very fast sprinter and highjumper, but long recovery after events. Now vaso vagel issues. Cleveland Clinic says Heart supposedly good, can’t figure my case out.
Where can I go from here?
Just within the past couple of months since having a relatively mild case of COVID( never got vax due to worries) I have had a terrible problems with my heart beating so hard, waking me up in the middle of the night. Passed out twice last weekend. Seems to have made whatever heart stuff I had with post Lyme CFS worse.
Thanks Cort, very interesting. Did the study say anything about the severity of the patients? I’m guessing they might have been all mild, given the rest of us wouldn’t sign up for an exercise study?
Wondering if this HR effect could be exaggerated in more severely ill people. I’m moderate (99% housebound), and after a 10 minute gentle walk last week my resting HR remained elevated by more than 20bpm for several *hours* afterwards, and didn’t go back to my usual resting HR for about 36 hrs.
A somewhat similar study worth mentioning:
https://www.frontiersin.org/articles/10.3389/fphys.2021.771899/full
They also did 2-day CPET and looked at heart rate recovery and variability. Somewhat similar results, but probably not specific enough to ME/CFS to be used as biomarker
My group here in Cambridge UK carried out a pilot study measuring HRV response to sub maximal exercise challenge for 18 PwME and 16 non-ME. We found no difference at all. It also left us skeptical of past literature on HRV and vagus nerve response as reported differences were close to the noise floor of the measurements. I’ll have to take a closer look at their instrumentation
Did they look at Total power and LF/HF as well?
It seems that the same article had already bern published in 2015:
https://www.physiotherapyjournal.com/article/S0031-9406(15)02014-3/fulltext#relatedArticles
I saw that. I have no idea what’s going on.
Have the authors not read any of the new information coming out in the last years? Like from Systrom?
I’ve been dabbling in HRV for a while. Tracking it with my Apple Watch. I’m about to get this device- https://getlief.com/ Have you heard of anyone with CFS using it? Over all, I do think if we can balance our ANS and elevate our HRV, exercise might not be so bad….just my thoughts.
Check out this – Your Crash in a Graph? How Heart Rate Variability Testing Could Help You Improve Your Health – https://www.healthrising.org/forums/resources/your-crash-in-a-graph-how-heart-rate-variability-testing-could-help-you-improve-your-health.353/
It’s worth looking into the ‘unique’ way that Apple tracks HRV (it averages measurements through the day rather than taking a morning reading) – avoiding the Health app altogether is recommended.
I’m wondering if there has ever been a survey done using FM and CFS suffers experiences? There may be many blogs out there who have thousands of members wanting answers. If we could all list our eg.
Most helpful medications/supplements/Lifestyle/etc
Maybe we can gather some insight into what is going wrong …researchers would also benefit from the info.
Thanks Cort for your generous contribution to us all. Cheers Berit😟
Check out the website “Stuff that works” as they are doing precisely what you described. It’s like crowd sourcing for health research.
CFSFMExperimental Yahoo group
Re: Remark – Heart disease causing cfids?
Sun Feb 13, 2005 2:36 pm
Don’t prejudge Dr Cheney’s work by focusing on this aspect as if he
might be claiming that “CFS is nothing MORE than heart disease”.
The Johns Hopkins work points to a circulatory disorder that
consists of an irreconcilable conflict between the circulating
Catecholamines from the tissues telling the heart to deliver more
Oxygen – and the mechano-receptors in the heart which sense excess
pressure from irregularities induced by autonomic dysfunction and
cause the left ventricular pooling.
Like a combination between Lerners viral defect and Krebs cycle
metabolic dysfunction.
As I recall, Dr Cheney is a pretty sharp dude.
-Erik
—————
Back when the CFS Experimental group was criticizing Dr Cheney for talking about this.
I can feel so great while I am exercising, at least for the short time I can handle it. Or at least some things. I can’t run. I can’t walk on level ground, I do much better going uphill. But I can shovel snow, use a scythe, and feel good just while I am doing it.
And then feel so awful. It’s soooo maddening, because exercise is the only thing that makes me feel really good.
Another thing I wonder if anyone else has this problem, and it seems to fit in with this…I can be totally exhausted, but as soon as I lie down to go to bed,my body feels like it’s plugged into the electric outlet.
Exactly!
Overactivity of the ANS and high heart rate can be the result of compensation due to insufficient blood flow and oxygen uptake. It is a sign that you have gone beyond your limits. Or that you have a serious oxygen and energy shortage. But it goes both ways.
In my opinion we have an abnormal stress response with all its consequences. But this can also be a necessary reaction of the brain to provide more energy with adrenaline.
Anyway, I hope there will be more research on red blood cells, blood volume, vascular system and the brain stem 🙂 Because there is also a breathing problem or dysfunction. This gets far too little attention.
Thank you Cort for your good work and your optimism. I lost it somewhere after 25 years 🙁
Perhaps they should also look at small fiber nerves as factors in baroflex failure. SFN has been found in a large % of CFS and FM. They are found throughout the body and play a big part in autonomic regulation. Especially the cardiovascular system.
Very interesting to read this study. I have not been able to exercise as I would” flare” after exercise. I do walk my dog on a daily basis and I just manage that. Interesting thing is that I have been put on Propranolol about 6 months ago and I think the betablocker has improved my FM slightly as I don’t feel as tired when walking the dog and I am no longer breathless. Would be interesting to see a study into this as it looks like I am not the only one who has noticed this.
Can morning HRV be used to determine if it’s safe to exercise later that day?
Have not all the studies, papers and youtube recordings of their work also been instrumental in these studies?
Hi as someone that was pretty fit Swam 30 lengths.played badminton.gym for hours lifting anything.Cycle raced all types and did long rides 100 mile plus
Then got ill after pushing it one day then went down with Fibro type things and Raynauds i found at first HR would climb way too fast to quickly and knew my rates for place rode as always used Polar HR monitor and i had a resting pulse of 20’s/low 30’s.
Later and years on found/find never recover properly would get out riding noticed wouldn’t gain performance as should be getting fitter but struggled more.
Was doing a Gym programme at a disability gym at first ok enjoyed it but after a few weeks noticed i wasn’t gaining but loosing performance.
I’m Deemed Autistic/Asperger’s and that also has a bearing on this stuff as sleep and stomach issues come about with it plus Processing things with Asperger’s.
I understand a lot more than many think inc quacks as also did coaching with having to study lots on muscles/bones and so much more but also i can work so many things out by being open minded with my Asperger’s and i was the one that worked out had Pituitary tumour when quack gave me the Default no no no.
I was always Taught Always listen to your body.
I started with CFS in 1985, have added Chronic Migraines, Fibromyalgia, Mult Chem Sensitivities, and Sjogren’s Syndrome. I had to retire my career as a Dermatology physician, and my level of function and mentation continue to drop. I feel like I’m becoming an Alzheimer’s patient, Extensive Psych and medical testing did not point to any specific diagnosis. My life is going down he drain, and I understand progressively less about what is causing it.
What would you suggest for further work up, UCSD, or Scripp’s Clinic, or who else? I am currently a Kaiser patient, for years I would appreciate any suggestions.
Hi Sandy, I have been wondering how you are, and trying to find you. (At least, I think you must be the Sandy Stedinger I used to know.). I knew you had become ill from, I think it must be, at least 15 years ago, and I’m so very sorry you still do not feel well. I got married in 2002, no kids. I can’t drive at the moment, and my dear wife doesn’t like me dating or spending any time socializing, but I would love it if we could meet on Zoom and get caught up. Have you learned Spanish? I go to the Peninsula Spanish Language Meetup group on Saturday mornings sometimes. Lo siento que sientes tan mucho dolor, y esparo hablar contigo.
21 DAYS after the moderna shot 4-21 I had problems after a very modified activity in the yard. Rests every one minute and every 40 mins rest for 10-15 mins. It was the 2 ND DAY IN A ROW. 2.5 hours of pathetic work. My respiratoy rate did not come down from 30 beats per min(looks like your running and out of breath). After aspirin and ativan to calm the overexcitaion of the CHF/nervous system. I was not anxious. I was in bed for an hour still with a RR of 30. Full easy breathing with full expelling of residual air. Episode was 1.5 hours. The next day I went to the ER. My CK was “critical” 1027 thats muscle damage. (go figure) I screamed, “Thank God after 17 years finally objective data. Yes It shows I have a significant muscle weakness problem” The ER dismissed me as having asthma and extreme exercise can cause a high CK. Both DX were so wrong and did not fit my story. OK for them to make crap diagnosis up, and not listen to me saying what is actually going on. I even demonstrated and told them the person having an asthma attack does not need to be told they were not getting enough air. They know it. I was getting plenty of air. My body was driving the high Resp rate. And you can see the muscle damage-CK. I had a pulmonary exercise test a month ago. Pulmonary function is fine. I begged and cried to the MD doing the test not to dismiss anything unusual. I told him the story. He wrote in the report my bp did not go up during my peak exercise. Thats it. My Blood pressure was taken 2/3 times while I was killing myself walking. He did not write all the bp numbers in the data. (dismissed). My heart rate was over 100 for 20 minutes after the test stopped while I was in a sitting position. “slow to recover” Thats a 50% higher chance of dying in the next five years. I wrote a postal letter to the MD who ordered the test and called the office 4 times. I was told my pulmonary function was fine. I am completly burnt out. I gave the MD overseeing the test the slow to recover article.Then begged to not dismiss anything. I have so little fight left in me. But a level one pulmonary exercise test can show some problems. If your looking for objective data. I know you are. I need a therapist but cannot go to one who does not get it. Anyone have a pyschologist that gets the CFS nightmare? MA.
CFS/SEID I finally tried melatonin. For years I did not try it because I have slept like the dead since being sick. Nothing wakes me. So I figured I did not need it. WRONG. I started taking it to re-adjust my bedtime. After a few weeks I started waking up easier. HUGE deal. Less of a fight to wake up. less of a heavy sleep. A more normal, wake if you hear something, which was my old self normal. So if you sleep like a rock and have huge trouble waking up maybe it will be good for you too. I started w 3mg but take 10mg sometimes. Peace.
Congratulations D! Thanks for sharing your breakthrough 🙂