


Prusty (center) won a Ramsay Award in 2019.
Bhupesh Prusty’s goal seems to be to upend what we know about the herpesviruses, in particular, human herpesvirus-6 (HHV-6), and their relationship to chronic fatigue syndrome (ME/CFS).
His Twitter handle calls him “a passionate molecular virologist who believes in patient-oriented scientific research… Science is for society.” Thus far, Prusty has been the recipient of a Ramsay Award from the Solve ME/CFS Initiative and a Young Investigators award from the HHV-6 Foundation, and appears to have put both of them to good use.
HHV-6 has been like Epstein-Barr virus’s poor sister in the chronic fatigue syndrome (ME/CFS) world. It’s always been in the mix, but EBV has received the lion’s share of attention. HHV-6 has had something of a renaissance in ME/CFS lately, with several recent studies suggesting that it may play a big role in at least some patients.
A Latvian/Israeli team found that the more severely ill a person is, the more apt they were to have high HHV-6 loads and evidence of an active infection. Similarly, a 2021 study found HHV-6B/HHV-7 viral loads were correlated with symptom severity, cognitive issues, and autonomic nervous system problems in a subset of people with ME/CFS. Earlier this year, a study documented the existence of unusual HHV-6 enzymes called dUTPases in ME/CFS.
A 2022 meta-analysis of 17 ME/CFS HHV-6 studies concluded – without stating that HHV-6 was causing ME/CFS – that an “association between the HHV-6 infection and CFS” was substantiated.
HHV-6, like EBV and other herpesviruses, establishes a latent infection in the body – remaining present thereafter. The big question is whether it’s doing more than hanging around in ME/CFS and other diseases. Now that there is strong evidence that EBV is the causal factor in multiple sclerosis, it’s perhaps time to wonder what it’s sister virus – HHV-6 – is doing. Like EBV, HHV-6 prefers to target neurons in the central nervous system and has been associated with several central nervous system diseases.
Mitochondrial Fragmentor
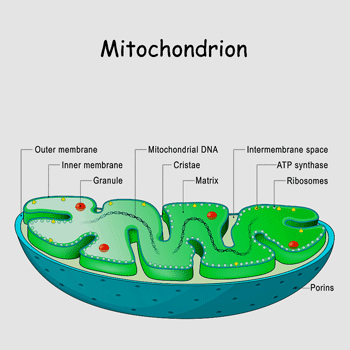
Prusty and Naviaux found that HHV-6 infection and serum from people with ME/CFS fragmented the mitochondria.
In one of the great leaps in research that I can remember, Prusty and Bob Naviaux pulled the HHV-6 and ME/CFS world in a new direction in 2020 when they showed that not only did HHV-6 appear to fragment the mitochondria but that it reduced the availability of the raw materials needed for the mitochondria to work properly.
That was just the warmup. Next, they showed that simply adding serum from people with ME/CFS into a cell culture produced the same strange state of mitochondrial discombobulation. They concluded that:
“HHV-6 reactivation in ME/CFS patients activates a multisystem, proinflammatory, cell danger response that protects against certain RNA and DNA virus infections but comes at the cost of mitochondrial fragmentation and severely compromised energy metabolism.“
Mitochondrial Fragmentation Explained
Prusty and his German colleagues were back in May of this year with a Nature publication, no less, “Selective inhibition of miRNA processing by a herpesvirus-encoded miRNA“, that cast a new light on how HHV-6 maintains itself in the body and does damage. HHV-6, it appears, produces microRNAs (miRNA) that trigger a “profound disruption of mitochondrial architecture.”
That, in turn, interferes with the production of the powerful antiviral interferon cytokines, thus allowing the virus to spread in the body and/or become reactivated.
They were also able to identify the miRNA (miR-aU14) that triggered the virus to waken up from latency and become reactivated. Even more encouragingly, they called the miRNA responsible for waking up the virus “readily druggable”, indicating that drugs could be developed to stop it. They concluded:
“our findings reveal a miRNA-mediated mechanism that a prevalent human herpesvirus has hijacked to interfere with intrinsic immunity, govern the lytic–latent switch and augment productive infection. However, viral miR-aU14 should be readily druggable using antisense approaches (antagomiRs)40, thereby providing an interesting therapeutic option for preventing herpesvirus reactivation.”
The Brain Autopsy Study
For his next trick, Prusty and colleagues from the HHV-6 Foundation, the Ohio State University herpesvirus research team (Maria Ariza) and others, took the HHV-6 conversation in ME/CFS to an entirely different level.
In “Tissue specific signature of HHV-6 infection in ME/CFS“, they looked for evidence of the HHV-6 miRNA (miR-aU14) and HHV-6 proteins in brain samples from autopsies of people with ME/CFS (n=3), people with other diagnoses (n=3; anorexia, non-Hodgkin’s lymphoma, and breast cancer) as well as 21 other controls without ME/CFS.
They also assessed the prevalence of the following antibodies: anti-HHV-6B U94, anti-HHV-6B OHV3, anti-HHV-6 gB, anti-HHV-6 p41 and anti-EBV dUTPase. EBV dUTPase is an enzyme produced by smoldering infections of Epstein-Barr virus and has been a focus of study in ME/CFS. Finding antibodies in the brain would indicate that an immune attack had taken place – either against the virus or a part of the brain the immune system had mistaken for the virus.
Results
HHV-6 proteins (indicating the virus was present), miR-aU14 (the mitochondrial fragmentor was present), and/or antigens were found in 2 of the three ME/CFS patients across the brain in the choriod plexus, hippocampus, amygdala, dorsal root ganglia, and/or cervical, lumbar and sacral nerve roots. EBV dUTPase (smoldering EBV infection) was found in the brains of all three ME/CFS patients but not in any healthy controls.
The fact that an HHV-6 protein was found in the cerebellum of some of the healthy controls suggested that HHV-6 can exist in its latent state in the brain without causing problems.
The many more signs of HHV-6 and EBV found spread across the brains of the 3 ME/CFS patients suggested that something of a different nature was going on. Dramatic mages posted to Twitter by Prusty show how abundant some of the signs were.
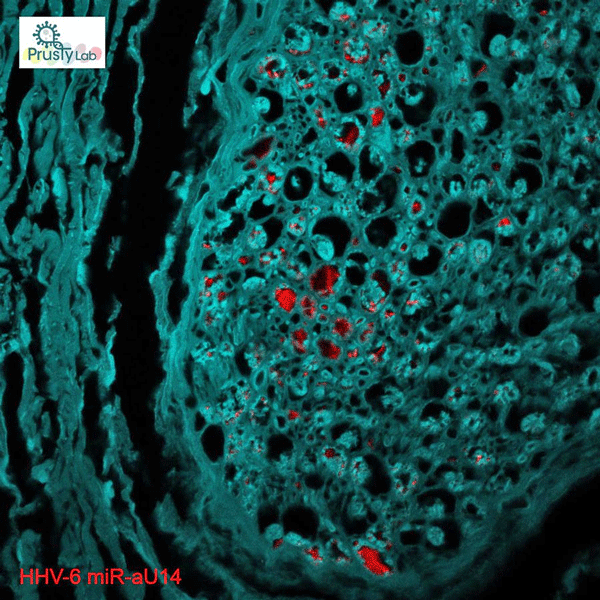
The red spots are signs of HHV-6 infection.
A Z-stack video showing presence of HHV-6 miRNA (miR-aU14) within Lumbosacral nerve root of one of the ME/CFS patient. pic.twitter.com/UMuaORBd3t
— Bhupesh K Prusty (@BhupeshPrusty) December 15, 2022
They were also found in quite interesting places. The choroid plexus secretes cerebral spinal fluid (CSF), and intracranial hypertension (elevated CSF pressures) appears to be a real thing in ME/CFS. The hippocampus and amygdala are part of the limbic system and play a role in memory and fear/hypervigilance. The dorsal root ganglia process the sensory signals entering the spinal cord and have been the subject of great interest in fibromyalgia. All of them appear to make sense with what we know of ME/CFS.
While we don’t know these patients died of HHV-6, it’s clear from other diseases that active HHV-6 in the brain is not anything that anyone would ever aspire to. HHV-6 has been found in the brains of people with a variety of neurological diseases, and the authors pointed out that active HHV-6 in the brain – and the inflammation it causes – has been linked to several of them, including mesial temporal lobe epilepsy (MTLE), multiple sclerosis (MS), and Alzheimer’s disease (AD).
Given HHV-6’s predilection for settling in nerves, and its ability to foment inflammation when active, an active central nervous system infection would fit in nicely with hypotheses proposing that neuroinflammation plays a major role in ME/CFS.
The authors also referred to a string of papers by Matsui et al. which proposed that people with ME/CFS had a novel syndrome they called “cervical neuro-muscular syndrome” that Health Rising will dig into later.
EBV dUTPase also showed up big time in this study. EBV dUTPase is an enzyme produced by EBV when it’s present in a strange kind of broken, smoldering state. EBV dUTPase studies over the past decade in ME/CFS have shown that the enzyme is able to provoke an inflammatory response that could produce neuroinflammation. This is the first time, though, that the enzyme has been found in the brains of ME/CFS patients.
All in all, the herpesvirus studies continue to suggest that active herpesvirus infections could play a major role in at least some people with ME/CFS. Prusty’s study highlights the strong need for more autopsy samples and biobanks able to receive them.
Other Autopsy Reports
On that point, Prusty’s aren’t the only autopsy results that have shown up in ME/CFS, though. A series of case reports have looked for evidence of damage in the brain in mostly young women who died of ME/CFS.
Besides damage to the frontal cortex and basal ganglia, one young woman showed evidence of microvascular damage, and strange tangles of proteins, including amyloid plaques, were found.
“Substantial” amounts of C. burnetii DNA were found in Alison’s Hunter’s heart, lungs, spleen and liver. The authors concluded that the likeliest explanation for her decline and eventual death was a severe Q fever attack that infected her organs, causing brain and heart dysfunction; i.e. she had a post-infective fatigue syndrome.
The Gist
- HHV-6 has kind of been like a poor sister to ME/CFS, but recent studies have suggested it could play a significant role. It’s been Bhupesh Prusty’s work that’s really shined a spotlight on that pathogen.
- His and Bob Naviaux’s study found that HHV-6 was able to fragment the mitochondria and inhibit it from using the resources it needs to produce abundant energy. As they were doing that, they showed that serum from ME/CFS patients produced the same result (!).
- Next, in a Nature publication, Prusty and colleagues identified the microRNA responsible for fragmenting the mitochondria, and in a finding that could pave the way for the next wave of anti-herpesvirus drugs, it’s the miRNA that awakens HHV-6 from latency.
- In his recent autopsy study, Prusty found evidence of widespread HHV-6 activation across the brains of 2/3 former ME/CFS patients, and EBV activation in all three patients. Very little HHV-6 was found in the healthy controls.
- The finding possibly placed ME/CFS in the same general basket – at least with regard to HHV-6 – as other diseases such as multiple sclerosis, where HHV-6 activation in the brain has been found.
- A review of past autopsy findings found a pattern of damage to the dorsal root ganglia which are responsible for transmitting sensory and autonomic signals to the spinal cord.
- ME/CFS autopsies appear to be few and far between. Given the findings thus far, it would appear that a biobank that could hold them – if it’s not present yet – should be created.
- Prusty’s startling herpesvirus findings add more mass to the recent herpesvirus findings in ME/CFS and long COVID. We will certainly be hearing more about the herpesviruses in these diseases in the future.
A post-mortem examination found low-grade inflammation in 21-year-old Merryn Croft’s nerve roots and dorsal root ganglia. “Marked” dorsal root ganglionitis was found in one woman, and two other woman showed signs of dorsal root ganglia degeneration (nageotte nodules). The ME Association proposed that these issues probably disrupted sensory and autonomic nervous system functioning, causing hyperalgesia (pain sensitivity) and allodynia (skin painful to the touch), as well as contributing to problems with orthostatic intolerance (hypotension), light sensitivity, etc.
A Focus on the Dorsal Root Ganglia
With dorsal root ganglia problems showing up in five out of seven earlier autopsy reports, and two of the three ME/CFS patients in the Prusty paper, the dorsal good ganglion appears to be a hot spot for people with ME/CFS.
Dorsal root ganglionitis (DRG) refers to a disease of the dorsal ganglia. These are nodules found just outside the spinal cord and contain the cell bodies of sensory neurons. Because they rely on sensory signals to the brain and play a key role in pain perception, problems with the DRG could conceivably produce many of the sensory and pain problems in ME/CFS and fibromyalgia.
Bigger Studies Needed
Despite the different techniques that were used in the various autopsy studies, a clear pattern of dorsal root ganglionitis was found. These studies obviously present an opportunity to learn much, but we lack larger-scale, organized autopsy studies that would enable us to draw firmer conclusions.
What we do know is that every autopsy study has found evidence of damage and/or pathogen persistence either in the brain or elsewhere in the body. Hopefully, Prusty’s new findings will help support the emergence of biobanks that can support these kinds of studies.
In general, the COVID-19 autopsy study results seem to line up well with these results. They suggest that a widespread inflammatory condition in the brain results in blood vessel leakage, punctuate hyperintensities, and inflammation. Those punctuate hyperintensities have been found in ME/CFS as well. We’ll certainly see more COVID autopsy studies in the future.
With interest in the herpesviruses picking up – and findings suggesting that HHV-6 may be knocking off the mitochondria in ME/CFS – it’ll be interesting to see what the future will bring.
BIG (Little) Donation Drive Update
Thanks to the over 260 people who have supported Health Rising thus far in its end-of-the-year donation drive.

We try to make sense of all of it. If that appeals to you, please support us.
Bhupesh Prusty is one of the people Health Rising is committed to following, which is why we covered not just his most recent finding but his other ME/CFS findings in this blog. Health Rising was also the first – and is still perhaps the only website – to bring the ME/CFS autopsy reports together to look for a pattern. In this blog, we added Prusty’s new findings to the list – and, lo and behold, a pattern showed up. If you appreciate that commitment to completeness, please support us.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



Thank you for this interesting and informative post. It is a good reminder of the critical importance of brain donation. Being able to study brains is one of the best ways to advance understanding of brain health and brain diseases. I have registered to be a brain donor (when I am done with it) and I encourage everyone to join me. http://Www.braindonorproject.org is an excellent organization that will direct you to the best brain bio bank for you. Check it out.
Thank you! I just pre-registered there, too!
Did you receive any information after registration? I registered a year or so ago and never received any information. It’s strange. I was expecting a letter or contact information…anything to give to my family so they know what to do when I die. I attempted registration numerous times. I found it frustrating.
Try contacting Tish at the Brain Donor Project. +1 (513) 393-7878
She was my contact there and I found her extremely helpful. I’m sorry to hear you’ve had this experience; I don’t know if it is at the level of the Donor Project or the lab but Tish can help you either way. I did have to contact my lab several times to get the paperwork completed but in the end I got all the instructions for the family and a card to carry in my wallet. I think these labs run with limited personnel. Keep trying.
Do you know if there is something similar in the UK?
I’m sorry I do not. The Brain Donor Project was started by a family as a way to honor their deceased husband and father. You could try by contacting them via braindonorproject.org to see if they know of one. Also, I would expect that there are Brain Biobanks in the UK; perhaps you could try contacting one of them directly. It is one way we can all take part in advancing the science of understanding brain diseases.
English organ donation law has changed. All adults in England are now considered to have agreed to be an organ donor when they die unless they have recorded a decision not to donate or are in one of the excluded groups.
NHS website 2020. I always assumed this meant all organs
Thank you for your reply, Jane.
Thank you for the link, Ann.
Donating organs is different to donating your body or parts of it for scientific purposes. You have to be quite specific when you donate your body for science research, otherwise it can be used for medical student training, for instance, which would be a waste for ME/CFS. This is why organisations like the Brain donor project exist, to ensure the right part goes to the right organisation.
This sentence gives me hope again:
“However, viral miR-aU14 should be readily druggable using antisense approaches (antagomiRs)40, thereby providing an interesting therapeutic option for preventing herpesvirus reactivation”
Indeed! @Cort, are there any other treatment implications involved in Prustry’s findings thus far?
I imagine antiviral drugs of which there is reportedly much more interest now would fit the bill if either HHV-6 or EBV or both were found to contribute significantly to ME/CFS.
A LOT of immunotherapy work is being done in MS as well. It could point to something for ME/CFS if the EBV connection is validated in ME/CFS as well.
MS research with its now vigorous focus on EBV-induced aberrant immune responses may indeed be something we should follow in the ME/CFS field.
Here, an especially stunning piece (to me) seems to be the fact that one can obviously halt the progression of early MS through vaccination with BCG:
https://www.sciencenews.org/article/bcg-tb-vaccine-diseases-diabetes-multiple-sclerosis
It was also shown that BCG may be able to rein in viral reactivation (human herpesviruses included):
https://pubmed.ncbi.nlm.nih.gov/31055165/
Taken these findings/suggestions together I would not hesitate to study BCG as an option against ME/CFS.
Thank you for this article. Once again you give me hope when I feel like I have none. I am donating and looking into monthly donations
Thanks, Gina! Prusty seems to be breaking new ground with every paper. Let’s hope he and others get the funding they need to really explore what’s going on with the herpesviruses. I imagine that the MS finding has produced an explosion of interest in EBV and what to do about it….
I also watch MS research Cort, because they have been breaking ground on EBV. One company I have been watching is an American company called Atara Biotherapeutics. They are doing some interesting anti EBV research. 🤞 Check out their press releases.
“All in all, the herpesvirus studies continue to suggest that active herpesvirus infections could play a major role in at least some people with ME/CFS. ”
Funny thing, I remember my father handing me an article he’d cut out of the New York Times in the mid-80s. I was a teenager who had constant sore throats and fevers, DSPS, and no energy, and the article suggested that herpesviruses could be the culprit.
I guess the difference that ‘suggesting’ it could be the culprit and proving otherwise are differing matters. That said, so many theories and suppositions were aired and then everyone moved on to the next thing. As research ability improves and testing becomes more ‘advanced’ i guess we can go back and trawl past theories for examination under todays microscopes.
The first guy who suggested oranges/fruit as a way for sailors to avoid scurvy on long voyages was ignored til 100 years later when someone else suggested the same thing!!!
The world just does not change does it!!!
Me. Too. Since teenagerhood. Awful. Plus many other additional illnesses. My father once broke down and cried when he saw my energy failing and my unwellness
I thought it was temporary. I couldn’t imagine that it would last a lifetime
Many years ago, some of my favorite cousins unexpected died. Husband and Wife. Although separate deaths, we are certain that the Husband’s death was from heartbreaking after Wife died.
But to my point…
They were in the medical field and had donated money to an organization that was supposed to take their bodies for science. I don’t know the name of the company/ organization? However when the time came, the organization refused their bodies. Stated that they no longer needed donations. Now, all the years of donations were spent otherwise… no specific information was given. Ultimately the family cremated Wife and Husband and ashes wete scattered over their favorite places.
My point here is be careful and aware of what you’re donating to. Check place and organization Etc… very thoroughly.
This is nothing new since Dr. Martin Lerner knew this many, many years ago!
Yes, Lerner told us the disease was due to an active viral infection. Dr. Dantini in Florida has been treating ME/CFS patients with antiviral meds for years.
Hasn’t Dr John Chia been saying much of the same thing for years? Regarding ongoing active viral infection?
Cort, Great presentation of Prusty’s work. I’m anxious to see Dr. Pridgen and Dr. Duffy’s anti-viral drug trial results.
I was a patient of Dr. A. Martin Lerner and was on his Cidofivir protocol circa 2014 before he died. Reactivated HHV6 and EBV is what I was diagnosed with. People really should have listened to him.
Did the Cidofivir result in significant improvements?
Yes, for me Dr. Lerner’s protocol helped me tremendously
Yes, I consider myself one of DR Lerner’s successes. I was under his care from Jan 2014 until his death September 2015. I proactively worked with a local doctor, who agreed to continue dr Lerner’s protocol based on his research and testing. From bedridden to having a life again. If not for fibromyalgia and arthritis pain at 71 years old, my activity be amazing!
i hope i’me getting back to the right person as i don’t know how to work this sight correctly.
DR. LERNER? yes, he wanted to intravenously feed antivirals in me but i was too ill. please let me know how he helped you and who you saw/see now for this illness. i would love to hear about your life with this illness. thanx!
Far from convinced sorry.
Neither am I. They didn’t test for other viruses, which we’re all surrounded with. And this sentence was telling:
“The fact that an HHV-6 protein was found in the cerebellum of some of the healthy controls suggested that HHV-6 can exist in its latent state in the brain without causing problems.”
That to me suggests, it’s not caused by a virus.
Back in the early 2000s, there was HHV-6 brain research happening at Stanford and (I think) Cornell. Back then, HHV-6 was one of the viruses that I tested positive for (along with EBV, CMV, and some other usual suspects), and I always wondered about it. It’s good to see it back again in the research; hopefully they’ll get some answers this time.
Thanks very much for sharing this article Cort – very clear and helpful explanation of the research as always, still giving us hope. It’s going to mention the Atara company that Wayne mentioned above, they seem to be developing a cool drug that goes after EBV infected B cells if I recall – in the setting of MS, but could see how it might be applicable to multiple other areas like ME and fibro. Fingers crossed Prusty can keep up funding for his great work!
This is possibly the most exciting study of the year. Two comments:
Markers of viral reactivation were found in nerve roots and in the brain. In the brain, the reactivation affected
– the chorioid plexus (which is made up of ependymal cells, i.e., specialized neuroglial cells
– astrocytes
– Iba1 positive neuroglial cells – i.e., microglia
So it seems that the playing field of viral reactivation in the brain is the neuroglia – which could bring the reactivation hypothesis of ME/CFS together with the neuroglial hypothesis (https://www.frontiersin.org/articles/10.3389/fncel.2022.888232/full ).
Here it needs to be remembered that microglia is highly connected with the peripheral immune system (among many other things!) and that astrocytes are important in the regulation of local blood flow (again, among many other things!)
A second comment. Theoretically it is possible that ME/CFS may result from or reflect ongoing reactivation of a single, specific viral species (like HHV6 or EBV). However, given the copious findings of (co-?)reactivation of other microbial species in ME/CFS, it is also possible, that ME/CFS may be sustained by the reactivation of a whole „orchestra“ of microbial species. Indeed, as Prusty discusses in his Nature paper, activation of HHV6 and subsequent mitochondrial dysfunction is associated with a restricted interferon response – which could be no less than a door opener for reactivation of other microbial species any person may harbour.
So the question would be if the severity and clinical course of ME/CFS may reflect a) the severity of the reactivation in a given person (i.e., reactivated viral load) and/or b) the number and identity of the concurrently reactivated microbial species (i.e. the make-up of the reactivated „orchestra“).
Food for thought! And thank you for this article!
Herbert, this may be the most exciting news of the decade. Between Long Covid, and all of the data pointing to an EBV cause for MS, the folks with the $$$ for research are less inclined to ignore us.
As always, thank you Herbert for sharing your insights! I always learn something.
Pretty sure this is the cause of my CFS. Glad there is hope for treatment!
Could HHV-6 linger in female vaginal yeast infections?
Could you expand please
Jane, If a male had unprotected sex with a female who had a severe fungal and or bacteria vaginal infections, could there be HHV-6 in the vaginal infection and therefore passed the HHV-6 on to the male. I’ve read where vaginal infections carry herpes viruses not herpes — I don’t mean genital herpes but a soup of herpes verses including HHV-6 and EBV.
Maybe slightly off topic but the last years I’ve noticed sporadic skin lesions resembling pityriasis rosea lesions whenever I had a bad PEM. Apparently pityriasis rosea has been linked with HHV-6 reactivation.
Does this sound familiar to anyone?
Yes, it does, Kristien! I didn’t have a name for it, but I’ve had these scabby places since I became housebound about 18 months ago. I haven’t been able to see a dermatologist to determine what they were. Thanks for providing a name. 🙂
Kristien, I have these, too. One I’ve had for years and years. The doctor told me it was fungal, but no fungal medication works. I have them on my thigh, back and shoulder. They are pale and scaly, not red. They do not itch.
If viral infection or re-infection are causal, does this mean nebulized hydrogen peroxide would help?
Kristien, I just found this:
“Clinicians should be aware of the association between COVID-19 vaccination and the development of pityriasis rosea. Pityriasis rosea has been linked to reactivation of human herpesvirus 6 and human herpesvirus 7 and has been reported following administration of the influenza and human papillomavirus vaccines. Pityriasis rosea is a self-limited, cutaneous eruption that resolves within 6 to 8 weeks, and patients should be educated on the benign nature of this condition.”
https://www.mdedge.com/dermatology/article/249518/infectious-diseases/pityriasis-rosea-associated-covid-19-vaccination
So what does it mean if it doesn’t ever go away?
In 1986, two doctors (Salahuddin & Ablashi) in the Gallo AIDS lab discovered a new virus. It was so lytic (able to kill) that they had trouble keeping enough of it alive to study. When they finally did, they discovered that it was killing B cells so they named it Human B Lymphotropic Virus (HBLV). Further research revealed that it was also killing T cells, so the virus was renamed HHV6. Then oddly, it was subdivided into HHV6 A & B. HHV6 B is the cause of common roseola in children, but HHV6A is much more troublesome. Visit The HHV 6 Foundation website to learn more https://hhv-6foundation.org/.
While Quest labs can test for HHV6, they don’t separate A from B. But, testing for A can be done by hair and nail samples as well as uterine biopsies offered through a specialty lab.
That HHV-6 can have a deleterious effect on immune function is not debatable. HHV6 is a major cause of opportunistic infection in HIV-infected individuals. HHV-6 infection can upregulate HIV replication and accelerate progress towards AIDS.
The question is whether HHV6 can cause illness in people who are immunocompetent for other reasons: anti-rejection meds, chemotherapy, exposure to environment toxins, genetic immune incompetence.
In the early 1990’s, I suggested to an epidemiologist studying Gulf War Syndrome that it might be caused by HHV6 and exposure to a pesticide which was widely used in the Gulf environment, but has since been taken off the market.
On the HHV6 website, there are some immune stimulants mentioned: Immunopro ( a whey based supplement); Nexavir (an antiviral made from pig’s liver) and a mushroom complex.
Antivirals will never “cure” a herpes virus, but a strong immune system can contain outbreaks.
Dr Cheney asked me to serve as “a prototype for a new syndrome”
At first I refused and told him to get someone else.
But he said “You are the only member of the original 160 in Incline Village group investigated by Dr Gary Holmes who is EBV negative but positive for the new HBLV virus. You have to volunteer. It is your duty, and you would be performing a service for the entire patient community.”
Reluctantly, I agreed.
This is how I became the first prototype for Chronic Fatigue Syndrome.
Nothing to add except that I am so impressed with Corts’ writing skills. I only wish I could write half as well. Thx Cort! I will be donating next week.
Thanks Scott! Lots of practice helps! 🙂
There are many anti-viral herbs and they usually cost less with fewer side effects. Consider….
https://www.healthline.com/nutrition/antiviral-herbs#2.-Sage
Comment *herbs dont help
My EBV IgG levels have been super high ever since getting cfs. They’re usually over 700, but this only means I had I a prior infection like most adults. I’ve never once had positive igm antibodies. Does this mean I might benefit from an antiviral?
Can damage to the Dorsal Root Ganglia be reversed? And I gave up on most supplements but I have to say that monolaurin cuts down my regular herpes outbreaks, better than just lysine. I do think it helps the fatigue too.
Hopefully we will be hearing more about EBV and further confirmation of this finding: “EBV dUTPase (smoldering EBV infection) was found in the brains of all three ME/CFS patients but not in any healthy controls.”
It is small sample but finding something in all the ME/CFS patients and none of the controls doesn’t happen too often.
Far too small to have any value at all, beyond a VERY speculative possibility.
https://www.youtube.com/watch?v=YH1wn3D9HNg&t=40s
This was on Youtube last year. Prusty appears in the final third covering this issue. All translated
I think in MECFS the whole system is depleted by prolonged stress or other factors. We are not able to control the pathogens we harbour anymore and become sick.I’m convinced herpesvirusses play a huge role in maintaining this illness,because they react to mental and physical stress. It is such a complex disease,but there is progress,on many levels now. I’m grateful for that.
Interview with professor Scheibenbogen
(..) We found that many ME/CFS patients have a stronger antibody response to one piece of the EBV virus, the so-called poly-arginine region. By doing a modern bioinformatic analysis and looking into sequence homologies, we found that there are several structures in our own body’s proteins which are similar — including adrenaline receptors and mitochondrial proteins.
The other interesting thing is that this poly-arginine region is not specific to EBV, but is also found in other viruses — for example enteroviruses or human papillomaviruses (HPV), which are also known to trigger ME/CFS. (…)
https://phoenixrising.me/myalgic-encephalomyelitis-chronic-fatigue-syndrome/autoimmunity-me-cfs/
Professor Scheibenbogen and her team do great work.She is one of best researchers in Europe. She’s extremely dedicated and gets a lot of invitations lately,to appear on German TV,to talk about MECFS and Long Covid. Good sign!
I get red spots and hard burn like areas on my fingers and hands which correlates with the occasional times when I get considerably worse, I eventually identified these marks as being identical looking to ‘EBV rash’ or mono rash that you can see on Google. I did Joshua Liesk’s antiviral protocol last year and did initially get the expected headache response which required a certain thing to counteract, and then also a strong immune response initally, but it didn’t sustain / intensify and cure my disease, it did change my functioning while I was on the protocol though. I think his protocol was aimed at removing latent HHV-6 virus from the body, particularly the liver, which he theorized was causing ME/CFS. Now I think that guy was really onto something but maybe just not quite right.
Your description sounds a bit like what happens to me.
You might want to check for pellagra. I find the old medical literature much more helpful. Its presentation is so varied, it doesn’t fit into the cookie-cutter 4Ds they make it out to be nowdays.
I find that both vtamin D and B3 help get rid of the rash. It reoccurs if I expose my hands to sun or supplemental B1 – which, wheat flour is usually enriched with it, so having a baked good will do it.
I’d appreciate if someone can help with this question. I’m under the impression almost all people get HHV6 as babies/children. Wouldn’t everyone then show positive in HHV6 testing? (Or, do MECFS patients show IGM positive as well as IGG and regular people show IGG positive only?). Also I wonder the percent of false negatives vs testing blood vs tissue.
Hi Nancy,
The problem is that there are two versions of HHV6… A & B. B is very common in children and is a cause of childhood roseola, but we have no statistics on the incidence of HHV6 A because testing is not widely available. HHV6 A seems to be associated with more serious outcomes in adults.
Thank you ☺️
With these miRNAs identified, can a special radioactive marker (may need to be created) be tagged to them and then be identifiable on MRI or PET scan?
Thanks so much, Cort, for this elaborate summary on the herpes viruses in ME/CFS question. This is such wonderful developments. I was so excited to read your blog.
I was looking for Herpes in ME/CFS research from the beginning, because before I was diagnosed and from the first inflammatory episode I was convinced that I had herpes reactivations. Even when the main symptoms differed from a HV simplex 1 outbreak it was clear to me that the syndrome was generally the same.
I had a very slow deterioration of symptoms and it was only when it got worse that I realised the now also the brain was inflamed. When I had the first severe episode of more than two months I begged my throat-nose-ears doctor to prescribe me aciclovir on speck. Because I had read about Mollaret meningitis. And it was wonderful, because one hour after taking the first tablet of 800mg the inflammation’s heat, pressure, and movement in the area of the frontal lobe subseded, just as did my susceptibility to PEM. And after a two week’s cure I was cured. It was about four months later only that the next episode was triggered by many weeks of stress.
For several months now I tried to find a specialist in herpes meningitis or encephalitis. But just encountered gaslighting by a neurologist and an infectiologist claiming that my ideas about what was behind my illness were a mere fabrication and the effect of aciclovir had certainly been placebo and thus a mere fantasy of mine.
I was extremely desperate but succeded to bring the second severe episode to an end by my own good care. It was then only three weeks after that I watched my first documentary on ME/CFS and then realised that I might have this syndrome and then have my GP refer me the the right specialist in the end.
Over the last year I wasn’t always able to understand the research on herpes viruses because I was overstrained with the process of acceptance of a severe illness and learning to pace myself. But now the first thing that I did over the last weeks was to try to update myself on the herpes research in ME/CFS. But it was difficult to interpret only by myself. So when I found your blog I was so excited.
Since I already knew all the studies superficially I was not blown away by the news, thanks god. But still I was so happy to have found it because I could understand and interpret them more easily with your help.
Personally, I find the information of inflammation in the area of the brain that regulates anxiety very important. Because I struggled with severe anxiety during episodes. I was managing it well because I am a long time yoga and meditation practioner. But still, it made it all the more difficult to relax and pace and get well again. I thought it was psychological and the stress of being ill just made it worse. Now it’s so good to have a hint that it might be directly caused by the brain inflammation.
And so good, to have all this evidence now of herpes actication, brain inflammation, even the damage in the mitochondria… I am more than convinced that it’s going to take 5-10 years max. and the situation for ME/CFS patients will have completely changed.
I would like to suggest to call ME/CFS Lerner-Encephalomyelitis and I hope that Prusty and his colleagues are going to win the Nobel prize in medicine in 2034!
I am late to the game on this one because I only recently joined. I have long suspected there was a connection based upon other research. My 26 year old son suffering with ME/CFS, mostly PEM, has very high HHV6 and EBV antibodies (as well as mycoplasma pneumonia). They were high enough to have been considered for this trial …..way back in 2013. Are doctors willing to actually administer antivirals or is there too much risk? – this study used valganciclovir. https://pubmed.ncbi.nlm.nih.gov/23959519/