The authors showed how gut, immune and metabolic issues might come together to produce post-exertional malaise in ME/CFS.
We know the gut is in play in some way in ME/CFS. Gut symptoms are common and studies have evidence of dysregulated gut flora and leaky gut (gut bacteria in the blood).
The big question, though, is what it all means. We know that gut issues have the potential to affect the immune system, but do they? What effect does exercise-induced leaky gut have on the immune system? Do the metabolic problems that have been found during exercise have any effect on the gut? How might the gut, the immune system and the metabolism be intertwined in a way to produce post-exertional malaise?
Any answer to ME/CFS, fibromyalgia, long COVID, etc. is going to have find ways to link these different systems together.
This study, “Suppressed immune and metabolic responses to intestinal damage-associated microbial translocation in myalgic encephalomyelitis/chronic fatigue syndrome”, attempted to do that, but it was unusual in another way.
Led by an unusually determined researcher, Armin Alaedini PhD, it re-analyzed samples that had been gathered during a large 2014 XMRV study and a smaller 2015 study, and kept at -80°C in the Solve M.E. Biobank. The smaller study had assessed the degree of leaky gut that occurred during and after an exercise study in people with ME/CFS and healthy controls. Not only did it find a higher incidence of leaky gut in ME/CFS but it found that the gut bacteria had persisted in the blood much longer in the ME/CFS patients.
The study examined a wide range of immune factors (plasma levels of IgG, IgA, and IgM anti-endotoxin (LPS)-core antibodies, lipopolysaccharide (LPS)-binding protein (LBP), soluble CD14 (sCD14), intestinal fatty acidbinding protein (FABP2) (R&D Systems), IgG, IgA, and IgM antibodies to the gliadin protein fraction of dietary wheat IgM antibodies to casein, IgG, IgA, and IgM antibodies to bacterial flagelli, IL-10, IFN-γ, and TNF-α cytokines, LPS concentrations) as well as metabolomics.
Findings
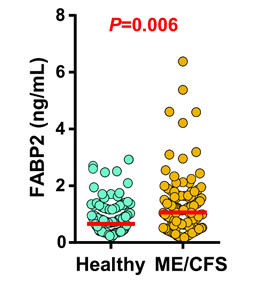
Note that not everyone with ME/CFS had elevated FABP2 – a sign of damage to the gut lining – but enough did to produce a statistically very significant difference (p<.006).
A dramatic increase in antibody responses to both microbial and dietary antigens in the ME/CFS patients suggested that their immune systems were responding to higher levels of bacteria and gut particles in the blood. The higher expression of FABP2 – fatty acid binding protein expressed in the epithelial cells linking the small intestines suggested that the epithelial cells lining the gut were damaged, allowing gut contents to spill into the blood in ME/CFS.
That made sense given what we know about ME/CFS, but the next finding did not. Given the evidence of increased leaky gut, they expected to find elevations in two early immune response factors (lipopolysaccharide (LPS)-binding protein (LBP), sCD14) normally associated with it but did not.

Similarly dramatically increased IgA and IgM antibodies to bacterial toxins in the ME/CFS cohort indicated that one part of their immune system was highly aroused.
Lipopolysaccharides (LPS) are proteins found on bacteria that present a red flag to the immune system, indicating that foreign bacteria are present in the bloodstream. LBP and sCD14 are immune factors that help to quickly rally the immune system to quickly remove the bacteria, yet both were low in ME/CFS.
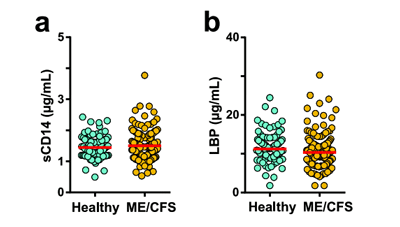
Given the evidence that more bacteria had leaked into the bloodstream of the ME/CFS patients, their early or innate immune response should have been going gangbusters. Their LBP and sCD14 levels indicated it was not.
The authors noted that another study had, to the authors’ surprise, failed to find increased levels of another early immune response protein called c-reactive protein. Inflammation seemed to be present in ME/CFS, but important early markers associated with the inflammatory response were not. What was going on?
THE GIST
- Talk about convergence. We’re used to hearing about how the gut can affect the brain through the gut-brain axis, but how about a study which shows how problems in the immune system, the gut, and the metabolism may be “conspiring” together to produce post-exertional malaise in ME/CFS?
- Using samples stored in the deep freeze from two studies done in 2014/2015 (thanks to Solve M.E. Biobank), one of which examined the effects of exercise on leaky gut, they took a deep dive into the immune system, the gut and the metabolism.
- The results made it clear that exercise was indeed causing ME/CFS patients’ guts to leak – spilling gut bacteria and food contents into the blood. One side of the immune system – the antibodies – leapt into action. Another side – the early inflammatory immune response that’s designed to quickly neutralize the invaders – did not, however.
- Plus, an anti-inflammatory factor called IL-10 that both knocks down the early immune response and interferes with glucose metabolism in the gut was elevated in ME/CFS.
- That set the stage for the last finding involving impaired energy production. We usually think of energy production in terms of exercise, but when the immune cells find an invader, they need to ramp up their energy levels dramatically in order to combat it.
- This study found, though, that while exercise did indeed significantly increase the levels of two key energy factors (citrate, glucose) that power the early immune response in the healthy controls, it did not do so in ME/CFS. Instead, the levels of those factors remained flat. Once again, we see a strange non-response to exercise in this disease.
- That suggested that the early immune response against the bacterial invaders simply didn’t have the energy to get going in ME/CFS. That, in turn, allowed the toxic bacteria to persist in the bloodstream.
- The findings suggested that a metabolic breakdown that hobbled the early immune response allowed high levels of bacteria to persist in the bloodstream of people with ME/CFS after exercise. That produced a condition called “endotoxemia” whose symptoms (fatigue, cognitive changes, headache, nausea, increases in heart rate, and decreases in blood pressure) bear some similarity to ME/CFS.
The pattern was clear. Gut bacteria were invading the bloodstream of ME/CFS patients in large amounts. The antibody portion of the immune system was jumping into the action, but the early innate immune response that initiates a rapid inflammatory response (that was designed to neutralize the bacteria) was not. Given that, it was no wonder that the exercise study found that bacteria were hanging around longer in the ME/CFS patients’ bloodstream than in the healthy controls.
An Endotoxemia Disease?
Asserting that, “These data could be highly relevant to the clinical history and presentation of ME/CFS“, the authors suggested – for the first time that I can remember – that endotoxemia – which occurs when large levels of bacterial toxins are present in the bloodstream – could be producing the symptoms of post-exertional malaise (PEM). The symptoms of endotoxemia – “fatigue, cognitive changes, headache, nausea, increases in heart rate, and decreases in blood pressure” – are indeed, quite similar, to those seen in PEM.
The authors noted that if these bacterial toxins make their way to the blood vessels of the brain, they can turn on the microglia, producing neuroinflammation and problems with cognition. That was intriguing given that Cook – one of the co-authors of this study – found in 2017 that exercise “exacerbated symptoms, impaired cognitive performance and affected brain function in ME/CFS.” This new study suggests that an inability to clear bacterial toxins from the bloodstream could be contributing to the brain fog found in ME/CFS.
The increased levels of the IL-10 cytokine in the ME/CFS patients added an intriguing element. An anti-inflammatory, one would think, that elevated IL-10 levels would be welcome, but this and other studies suggests that the high IL-10 levels could be hobbling early inflammatory response to invaders.
The metabolomic results added another twist. We’ve almost always associated metabolic problems in ME/CFS with low energy and an inability to exercise. This study, though, suggested that the metabolic problems found (reduced utilization of glucose and fatty acids and increased utilization of amino acids to produce energy) could be having an effect on the immune system as well.
Immune cells need to quickly fire up their engines to fight off an invader or toxin, and the metabolomic results from the healthy controls indicated that during exercise they were able to access a significant increase in plasma glucose – the cleanest and most efficient form of energy available to our cells. Plasma glucose in the ME/CFS patients, on the other hand, remained flat. IL-10 – which was high in ME/CFS – is able to block glucose utilization in immune cells that protect the gut barrier.
The same pattern occurred with citrate – a crucial player in aerobic energy production – and the activation of the early immune response. Exercise triggered a rise in citrate in healthy controls but not in the ME/CFS patients.
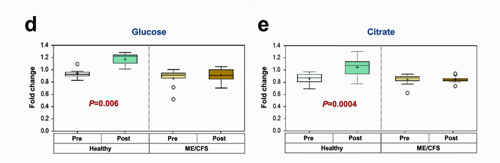
Note how exercise increased the citrate and glucose levels in the healthy controls but not in the ME/CFS patients.
High IL-10 levels and reduced levels of two crucial players in energy production may, then, have impaired the ability of the immune system to: a) protect the gut lining; and b) quickly clean up the bacteria that escaped into the bloodstream during exercise. The endotoxemia that resulted could help explain the post-exertional malaise (PEM) that people with ME/CFS experience after exercise.
The authors hastened to say that because ME/CFS is a heterogenous disorder this process may be happening in a subset of patients. Bigger studies are needed to validate and expand on these findings, but the intersection between exercise, bacterial invasion of the bloodstream, and a suppressed early immune response – driven by metabolic problems – is really intriguing. I don’t remember many studies that have been able to bring so many different elements together in one package.
This study also highlighted the need to explore how the energy production problems in ME/CFS affect immune functioning.
The authors stated they hope to explore further why leaky gut is such a problem, how the immune system and metabolism play a role, and uncover biomarkers that can be used to assess treatments, and ultimately, to employ treatment strategies.
Check out an interview with the senior author of the study – Dr. Armin Alaedini





Wow. This could be why patient led studies such as @remissionbione using probiotics and several antibiotics provide some relief to people experiencing PEM.
This is good Cort. I also seemed to experience a reprieve in the intensity of symptoms after using a months worth of antibiotics in addition to pre + probiotics, change in diet, physical (started with strength training done from bed/home) and occupational therapy, LDN and B-12 (methylated).
Yes, fixing the leaky gut should stop the bacteria from getting into the bloodstream and then amping up the immune response and possibly contributing to neuroinflammation and god knows what else. They are very keen to understand better why the leaky gut occurs and how the immune system and the metabolism are involved.
I thought the IL-10 issue was really interesting as high IL-10 levels have been found fairly consistently in ME/CFS but those high IL-10 levels could be causing problems with glucose uptake in the gut….Who knew?
5 years ago my Functional medicine doctor slowly ramped me up on OrthoMolecular SBI Protect Powder and Metagenics Glutagenics to repair leaky gut. It’s hard to say if they enabled me to exercise more and feel better or if I was simply in a better place since I was younger. Im inspired to try them again. Ty for the article!
This is fascinating. I recently did some additional testing that pointed to high levels of inflammation and immune dysregulation in the gut. My symptoms began as solely GI specific before progressing, eventually leading to PEM and chronic fatigue. In all honesty, for people with similar presentation and history as I do with gut/IBS/SIBO symptoms (present in 60%+ of me/cfs candidates, this feels like it could be the missing link.
Really hoping for some more research in this area.
Maybe so! If you look at the graphs they show that a significant number of people fit this profile and others don’t.
I noticed in the paper that most ME/CFS patients aren’t really that different from healthy controls…but then the ME group has many more “outliers.” So while this won’t be an explanation for all…it sure fits me like a glove. I think, anyway. Anything that “enhances” the microbiome in me makes me so ill, anything that kills and ruins the microbiome almost always induces a mini-remission.
I’m truly excited about this research and hope to see more explaining WHY it happens and WHAT we can do to mitigate it.
Can you expand on the things you tried that are supposed to ‘enhance the microbiome, but made you sick? I tried eating a tiny amount of a fermented food – kimchi – the other day and it made me sick for two days.
Hey Ilan, that would have made me ill at this point in my illness too. Some of us can’t process the histamine in fermented foods; for others it can create too much lactic acid that permeates the gut wall; for others either the bacteria or the cabbage/carrots feeds SIBO; and for still others it’s healthy and helps them.
“Microbiome enhancements” that my body has rejected include: probiotics (including low histamine and low d-lactic-acid producers), prebiotics (including inulin), butyrate, FODMAPs, colostrums, s boullardi (a “beneficial” yeast). “Gut supplements” my gut hated included l-glutamine, medicinal cinnamon, oregano oil, h2 blockers, and on and on.
Kimchi has some strong antimicrobial properties in that one of the organisms is pediococcus which can kill off a number of bad pathogens. Best suspicion is that the kimchi killed off some bad guys thus making you sick.
Kira – Wow, you’ve been through the wringer digestion-wise. What are you able to eat? My main issue now is that I’m not getting enough protein. I started eating vegan a couple of years ago, but my body hasn’t like lentils and beans, so I end up eating too much brown rice and other carbs. I tried lentils and beans again recently, but I’m not sure yet if it’ll take. I’m considering trying small amounts of high-quality egg/chicken/fish again.
That’s what I figured as well. It was prophetic that I randomly found this article the day after I tried eating fermented food for the first time in a few years.
Absolutely FABULOUS article, Cort! THANK YOU!
I once thought, like many, all symptoms started in the gut. But the truth is it started in the brain. Many studies show injuries to the brain such as trauma, PTSD, stroke, etc cause gut permeability. It was no wonder why I wasn’t responding to all the SIBO/LEAKY GUT treatments I was first started on shortly after onset years ago. I found the right doc who told me it starts in the brain with neuroinflammation. Calm the brain and immune system and everything else starts to calm and heal. He said NUMBER 1 I must remove exposure to endotoxins (food and environmental sources alike). Upon doing so I immediately felt better, then I began powders that help to remove endotoxins which help a tiny bit but still helpful, and those in combination have done better than any of the antibiotics, antimicrobials, antifungals, herbs, you name it that I so desperately tried thinking those were the answer.
I didn’t think of finding ways to remove the endotoxins once they are in the bloodstream – thanks for that idea. A key appears to finding out why our metabolism is not using glucose or fatty acids well – and finding ways to boost them. Of all the metabolic findings in ME/CFS I find that our preference for using amino acids – not a good energy substrate at all – the most intriguing.
I think the problem of using amino acids for fuel has to do with the itaconate shunt affecting the Krebs cycle. On another note taurine has been big in the longevity news lately. One of its many benefits is tightened intestinal cell wall junctions. Maybe not getting at the root but could be a help.
I don’t know what a ta is, but I sure will drink ta urine if it cures my ME/CFS. 😂
What’s of interest is my doc says they see a shift toward aerobic glycolysis and there is a great debate as to why. One hypothesis is that is simply our body’s response to neuroinflammatory disease states. As stated in some of his findings, “…aerobic glycolysis has been shown of be protective to neurons and initiates proliferative physiology in the injured state. The twin engines of neuroinflammation and aerobic glycolysis appear to be underlying the mechanisms of injury and repair observed…”
In other words, his findings regarding this hypometabolic shift that isn’t entirely characterized but includes aerobic glycolysis remains a mystery. The jury is still out but his theory on this shift toward burning sugar seen in his research resembles a “brain on ice” rather than a “brain on fire”. Essentially, aerobic glycolysis is a secondary pathway the organ systems use to rebuild themselves and is consequential, not causative. Others debate the opposite.
What are the specific powders that helped you to remove endotoxins?
From what I’m told, the only substances found to date that match the exact particle size and charge (gram negative) of endotoxins for binding is Cholestyramine (cholesterol drug), which is most potent and effective, followed by the less effective Welchol (cholesterol drug), and to an even lesser extent Okra powder for those who cannot tolerate the above prescription drugs.
Finally! Endotoxin gets some attention
I was reading the post on Prusty’s most recent findings. There was mention of the flu-like/immunity symptoms of PEM being signs of autoimmunity. I hope I’m paraphrasing that more or less right.
I was going to comment there, that if you don’t know about endotoxin, it could be under your nose and you wouldn’t see it. You might even have idiotic thoughts such as the body is attacking itself, when what it is reacting to is endotoxin.
Also, little is actually understood about the microbiome.
“Human microbiome myths and misconceptions”
https://www.nature.com/articles/s41564-023-01426-7
Read and think about this when you reach for a pre/probiotic…
Since stress (any source) increases gut permeability and endotoxin into the bloodstream, you start to understand why keeping to your energy envelope and even the mindset programs can be beneficial. Because they reduce stress, reduce endotoxin, reduce serotonin, reduce inflammation, etc.
This is probably true for ANYONE with a chronic disease, any and all of them, not just ME/CFS.
So people, stop pushing!
Go watch some of Toby’s free videos on youtube for figuring out your baseline / encouragement.
When I hear of people who get better just from mainly not overdoing things, I believe it. A recent post on a man that got better with a lot of deep rest, not just his body, his brain too – I believe it!
It was not my case. I did a bunch of things to get out of the severe bed/housebound states – none of it would have worked though if I had kept pushing, and not kept to my energy limits.
I first learned of endotoxin and its role in degeneration and disease from Ray Peat. I got ideas for what to do and try by reading his work and applying it to my situation.
Shea, what were the powders you took to remove endotoxins?
From what I’m told, the only substances found to date that match the exact particle size and charge (gram negative) of endotoxins for binding is Cholestyramine (cholesterol drug prescribed off label), which is most potent and effective, followed by the less effective Welchol (another cholesterol drug prescribed off label), and to an even lesser extent Okra powder for those who cannot tolerate the above prescription drugs and prefer the natural less effective route.
Of additional interest are how oxalates play into this gut dysfunction. Looking for reasons for the leaky gut, many of us are reading about the role of oxalates, acquired hyperoxaluria, and a created metabolic disarray are playing into this.
The probiotics never seem to be the answer, just an adjunct to creating better health overall. Butyrate is also entering the conversation and it’s relationship with oxalate. I’ve tried using the butyrate and have had significant, but not curative, results. A low-oxalate diet is also suggested which is making a difference in most.
Another excellent article Court. Thank you.
There has been alot of research into viruses and bacteria but not much into fungi. Do you know of any?
I personally think the problem is possibly metabolic ( change in the way we produce energy following a period of high demand ( i.e. stress, illness ) and the rest is a cascade of adaptive changes which lead to pathological outcomes. I am a member of several online groups where people share heath concern and blood test results and the number of people reporting metabolic and endocrin changes as well as insuline resistance post Covid is unpresedented.
Someone commented above that everything starts in the brain and I would tend to agree. My issues started with 6 weeks of headaches following a summer flu. Then i woke up one morning not being able to get out of bed. I didn’t experience GI problem originally. They appeared two years later.
Neli’s comment about believing the metabolic shift is a result of the initial stressor/trigger/event and that it all starts in the brain is precisely in line with what I’ve written above about my doctors findings. It’s interesting and extremely wise to be watching the patterns of what people are saying in those FB groups. That is good information. Logic would say everything starts in the gut but one could just as easily argue the brain is the root cause cascading down to the gut. Like my doc says, as a good example, so many patients think they need to supplement vitamin D after lab results come back low. Yet, the patients who treat their neuroinflammation find their vitamin D levels normalize without supplementation or intervention because the receptors are able to properly function again. All in finding and treating the root cause.
And vitamin D upregulates IL-10. As I read this article I wondered how high my 25-hydroxy Vit D levels were summer 2020. I was in sun the entire summer stuck at home so I didn’t bother checking it that year. Wondering if levels that are too high are harming us just as levels too low do?
Vit D experts suggest 40-60ng/ml optimal levels (Michael Holick).
Was my level too high and producing excess IL-10 when I got COVID? I have to wonder.
Hi Cort,
Great and stimulating article.
Some questions; the answers to which I shall present to my GP.
1) What is the best way to test for LEAKY GUT SYNDROME?
2) What is the best way to treat it? (N Harkness recounts using OrthoMolecular SBI Protect Powder and Metagenics Glutagenics.)
But is the best according to the most recent science?
3) What are the chances that reducing Leaky Gut will reduce PEM,
which affects me severely.
PS. I’m 80 years old and also have Hashimoto’s Thyroiditis.
I found this:
L – Theanine – L-theanine is a unique amino acid that is found in certain tea leaves.
Known primarily for its calming properties and as a means to balance out caffeine,
L-theanine also has a unique ability to reduce exercise-induced leaky gut. During times of stress when blood flow is reduced to the gut, more bacteria are able to pass through the intestinal lining.
L-theanine has been shown to be able to reduce the amount of endotoxin absorbed during exercise.
L-Glutamine is also supposed to help heal a leaky gut.
Thanks a lot Cort!
I was always sure, that the beginning of all my problems with me/cfs started in my gut and that therefore all my efforts and supports must be concentrated by healing this area.
After changing my nutrition into ketogen a lot of problems, like brainfog, leaky gut, diarrhea, shortness of breath, mess with SIBO and candida became much better! Bell Scale went from 30 to 40- sometimes 50.
It´s a hard task to change the system of glucose metabolism to a fatty one, but i think i was right to change it.
I am looking forward for more research and results in these theme.
Thank you for this excellent article. When I had the fibromyalgia blood test done by (I can’t remember the name now) but it showed my peripheral monocytes we’re hardly reacting at all in the presence of a stimulant. (84% less than healthy controls) This seems to correlate with the article.
I think I might try butyrate. I know that I used to have a leaky gut because I had undiagnosed celiac disease for a while and that causes damage to the epithelial lining of the intestines, so I got off gluten. I developed fibromyalgia after getting off gluten so even though it can* contribute to a leaky gut I wouldn’t say that it’s the cause. I drank bone broth for a couple of years and that helped also with healing my intestines so that I’m not lactose intolerant anymore. But The fibromyalgia still persists.
The part about the immune system affecting metabolism was very interesting. Even though the article talked mostly about IL-10, in my research I also came across that people with fibromyalgia have very low levels of lL-6, which people would think is good because it’s inflammatory, but having super low levels is also not good because it’s protective when it’s at normal levels. Too high and you get rheumatoid arthritis, too low and you get stuff like fibromyalgia. But again thank you for the article because it just proves that this is not all in our imagination ☺️
P.s. after looking more closely at the graphs in the original article, I thought it was really cool that they were testing for gliadin antibodies. For those of you that may not know, gliadin is the protein component of wheat (aka gluten). The protein components of barley and rye are similar to that of wheat, (in amino acid structure recognized by the immune system as foreign protein or food protein) so they all get grouped together as what we call “gluten”, so if you’re on a gluten-free diet you’re not eating wheat, barley or rye.
So if your immune system is responding to gliadin, that means you likely have celiac disease, an autoimmune disease triggered by eating gluten. It is very undiagnosed and misdiagnosed here in the US, and causes a lot of problems.
I developed fibromyalgia after being gluten free for 4 years so I’m not saying that going gluten-free is a cure, but it’s interesting that so many of the FMS patients were having reactions to gliadin in wheat.
However eating gluten made me terribly sick, and if I was still eating it I wouldn’t be alive today. I got genetically tested last year through Enterolab and confirmed that I carry the genes for both celiac disease and gluten sensitivity.
Thanks Aimee – I hope everyone who is eating wheat will take the possibility that wheat is having a negative impact on their health to heart and see what removing it does. While I still have loads of ME/CFS/FM removing it from my diet had an huge impact on my pain levels.
(…)”Many medical professionals do not recognize LGS as a diagnosable condition…
https://www.medicalnewstoday.com/articles/326117#:~:text=Leaky%20gut%20syndrome%20is%20a%20proposed%20intestinal%20condition%20in%20which,mechanisms%20and%20implications%20in%20humans.
Is LGS real? How do we know?
There is vast scientific literature on LPS/endotoxin.
The scientific world and the medical establishment are two separate things.
So look up in scientific databases / journals. Learn how to read studies critically.
We’re back to the gut again.
It makes me wonder where we are at since the start of COVID, which supposedly led to a paradigm shift. I’d love to know in a contained, concise way what our status is now on the science. I read all the time but I can’t put it all together. Honestly, it seems like we’ve made very little progress.
The model to me looks like the one before, one step taken in fifty different directions.
Zinc L carnosine has been shown in many to reduce gut permeability in rodents and humans. It is also been shown to reduce blood brain barrier permeability in mice. Last time I did a literary view of this, I found no studies about the human blood-brain barrier and Zinc L carnosine . However, I’ve noticed that soon after something makes my gut go bad, my brain follows.
Google Scholar will show you many studies on Zinc L carnosine and intestinal permeability.
After years of chronic low level irritable bowel, I got on high doses of Zinc L carnosine and also Miyarisan probiotic and after a couple years, I noticed gut problems were gone. REcently, I’ve also noticed I’ve some gut problems again. I suspect some eating lots of sweet snacks that contain alcohol sugar. It used to be I couldn’t eat any of this stuff but it got so my gut didn’t care and everything was fine. However I think that might be something that is special treat once a month rather than having it most days of the week because after eating some sugar-free sweets with erythritol or other g for many months, I noticed that my gut starting to ache again. Thinking about it it’s likely to be too much alcohol sugar which can lead to dysbiosis.
Thank you for this article which raises questions for me.
do you think a poor detoxifying liver from an early age, can being engorged by all the very harmful anti-nutrients for him such as salycilates, phenols, oxalates as well as by environmental exposure or even mycotoxins can lead to this kind of problem and EM…!?
This study would appear to tie in with an earlier one showing a reduced response of the innate immune system to exercise in people with ME/CFS, compared to healthy controls: https://www.healthrising.org/blog/2023/06/17/exercise-immune-system-letdown-chronic-fatigue-syndrome/#comment-1068227
They didn’t invoke the leaky gut, but showed some of the white blood cells are just not reacting, but resulting in a longer-term overreaction of the immune system causing inflammation. I find this direct evidence of a faulty immune response to exercise really encouraging.
Finally, a study that seeks to highlight and define the inter-relationships between multiple systems (GI, immune, metabolic) in ME/CFS patients.
Metabolic dysfunction, intestinal dysfunction, immune dysfunction and nervous system dysfunction are all present in ME/CFS patients. And working to improve the functioning of multiple systems simultaneously is a key to recovery.
Thank you Cort for continuing to report on the most important research findings of the day.
Jon D. Kaiser, MD
Thank you, sir! Let’s hope they get more funding.
Brilliant article.
I have been trying to download this article to show to my GP and Cardiac Nurse tomorrow but not having much success. Can anyone advise please?
YeS! Endotoxin gets some attention in the Me/Cfs field.
I was reading the post on Prusty’s most recent findings. There was mention of the flu-like/immunity symptoms of PEM being signs of autoimmunity. I hope I’m paraphrasing that more or less right.
I was going to comment there, that if you don’t know about endotoxin, it could be under your nose and you wouldn’t see it. You might even have idiotic thoughts such as the body is attacking itself, when what it is reacting to is endotoxin.
Also, little is actually understood about the microbiome.
“Human microbiome myths and misconceptions”
https://www.nature.com/articles/s41564-023-01426-7
Read and think about this when you reach for a pre/probiotic…
Since stress (any source) increases gut permeability and endotoxin into the bloodstream, you start to understand why keeping to your energy envelope and even the mindset programs can be beneficial. Because they reduce stress, reduce endotoxin, reduce serotonin, reduce inflammation, etc.
This is probably true for ANYONE with a chronic disease, any and all of them, not just ME/CFS.
So people, stop pushing!
Go watch some of Toby’s free videos on youtube for figuring out your baseline / encouragement.
When I hear of people who get better just from mainly not overdoing things, I believe it. A recent post on a man that got better with a lot of deep rest, not just his body, his brain too – I believe it!
It was not my case. I did a bunch of things to get out of the severe bed/housebound states – none of it would have worked though if I had kept pushing, and not kept to my energy limits.
I first learned of endotoxin and its role in degeneration and disease from Ray Peat. I got ideas for what to do and try by reading his work and applying it to my situation.
(A first try to post this comment got nestled in a conversation by mistake. Could the admins kindly remove the one? Thank you)
Great line of research. When relapsing I feel worst after eating breakfast and have an energy surge when hungry again just before meals. However if I do too much before eating my blood sugar goes through the floor.
Any info is welcome after 20 years of nothing and no real help.