



A ‘Novel’ Anti-inflammatory
“We discuss the concept of using low-dose naltrexone (LDN) as a novel anti-inflammatory treatment for chronic pain conditions that are suspected to be associated with inflammatory processes.”

The Stanford researchers believe the action in LDN is in its central nervous system inflammation reducing properties not it’s effects on opioid receptors.
Low dose Naltrexone is one of the more intriguing treatment options to emerge for Fibromyalgia and pain patients in recent years. Not only is it dirt cheap compared to many drugs but side effects are usually minimal.
Because any compounding pharmacy can produce LDN, however, drug companies – the major source of drug innovation in the U.S. – have largely ignored it. LDN’s ‘outsider status’ has hampered research efforts, but the drug simply appears to be too effective and too intriguing to be kept down for too long.
A recent review article by Stanford researchers on LDN and pain published in the Journal of Clinical Rheumatology, suggests that LDN’s effectiveness in Fibromyalgia could open the door for a new conception of chronic pain founded on neuroinflammation. They anticipate the development of an entirely new class of nervous system inflammation reducing pain drugs. The review article should also help expand recognition of LDN as option for people with Fibromyalgia and other pain conditions.
We cover the review article, other microglial inhibitors and LDN news in this blog.
Low Dose Naltrexone, Inflammation and Pain: A Different Look at Fibromyalgia and Chronic Pain
The FDA approved naltrexone HCL in 1984 to treat opioid addiction. Low-dose naltrexone is typically given at about 1/10th the typical dose of naltrexone. By blocking opioid receptors naltrexone can increase pain, but at very low doses naltrexone has both pain-reducing (analgesic) and anti-inflammatory properties.
Tests with another drug, called ‘dextro-naltrexone’, which does not affect opioid receptors but does affect microglial receptors, suggest LDN’s effects in FM do not involve the opioid receptors.
Fibromyalgia – A ‘Central’ Inflammatory Disorder?
“LDN may emerge as the first of many glial cell modulators that could be used to treat chronic conditions, with more specifically targeted medications developed in the future. As conventional anti-inflammatories have poor blood brain-barrier permeability, we expect centrally active immune modulators to be an area of interest in the future.”
The authors introduced LDN as a pain drug that is able to modulate ‘glial cell’ activity. When I asked Dr. Younger why he’s focused on the microglia in Fibromyalgia he stated,
“First, a growing collection of basic science studies (by investigators such as Drs. Linda Watkins and Mark Hutchinson) suggested that naltrexone can reduce the excitability of microglia in the brain, preventing the release of inflammatory and neurotoxic chemicals.”
Animal studies indicate LDN has neuroprotective properties that are directly tied to its ability to suppress microglial activation and possibly reduce inflammatory activity in the body as well.
Glial cells provide structural support for neurons in the brain, relay oxygen and nutrients to them, and defend them from pathogen attack. Once activated, the glia (or microglia) spew a wide range of inflammatory and excitatory factors (substance P, cytokines, NO, etc.) that cause such symptoms as pain, fatigue, fever, cognitive and sleep problems; in short, just about every symptom associated with Fibromyalgia and Chronic Fatigue Syndrome.
Dr. Younger believes microglial cell activation may be causing the kind of central sensitization found in Fibromyalgia (and, we assume, Chronic Fatigue Syndrome patients) which causes a wide variety of symptoms:
“I think microglia activation is the best explanation for central sensitization when the symptoms are widespread (e.g., involves more than one of pain, fatigue, sleep problems, cognitive disruption, etc.). There are other types of central sensitization, but they would not lead to all-over-body symptoms and multi-symptom problems.”
Younger’s small Chronic Fatigue Syndrome leptin study suggested to him that the microglia had become sensitized to inflammatory factors. All it took were small fluctuations in leptin, which can cross the blood-brain barrier, for ‘sickness behavior’ (fluey feelings, fatigue, etc.) to be evoked.
“The microglia have been sensitized, and therefore small swings in cytokines and adipokines are sufficient to provoke a large sickness response.”
Fibromyalgia has not been thought of as an inflammatory disorder and anti-inflammatories are generally not very helpful, but LDN’s effectiveness in two small fibromyalgia studies (50% of participants improved) suggest a specific type of inflammation affecting the glial cells of the brain may be involved. If this is true then FM is probably best classified as a ‘central immune disorder’; i.e., an immune disorder of the brain, with lower levels of inflammation in the body.
A Novel Anti-inflammatory
The Stanford lab’s finding that higher erythrocyte sedimentation rate (ESR) readings at baseline were associated with greater reductions in pain in Fibromyalgia suggested a) inflammation played an important role in FM patients’ pain, and b) LDN was reducing that inflammation.
The erythrocyte sedimentation rate (ESR), commonly called a ‘sed rate’, is the rate at which red blood cells fall to the bottom of a tall thin tube in a period of one hour. It is a common hematology test, and is a non-specific measure of inflammation. When an inflammatory process is present, the high proportion of fibrinogen in the blood causes red blood cells to stick to each other.
The ESR findings suggest that people with inflammatory and autoimmune disorders such as lupus and rheumatoid arthritis could also benefit from low dose naltrexone.
LDN may be more effective in Crohn’s disorder (80% effectiveness rate thus far) than in FM, and may be helpful in multiple sclerosis and chronic regional pain syndrome. The authors recommended that future LDN trials include a variety of inflammatory measures such as ESR, c-reactive protein, cytokines, growth factors, etc. to explore its anti-inflammatory effects.
A Person with Fibromyalgia Describes Their Experience With LDN
Opioid Receptor Blocker
It’s also possible that LDN’s opioid receptor blockage could provide relief by inducing the body to produce opioids and opioid receptors.
Lost Cost and Low Side Effects
LDN can be obtained from compounding pharmacies for from $35-$55 a month
Calling the low side-effects one of the most exciting aspects of LDN, the authors emphasized they have seen no serious side-effects in their patients and contrasted that with the increased risk of ulcers, kidney problems, clotting, heart attacks, etc. resulting from use of the current crop of anti-inflammatories. The most common side effect they’ve seen has been vivid dreams.
Low Dose Naltrexone and …SEX!

Start me up, baby! The reports are all anecdotal but some people report having better sex after using LDN. Could it be true?
Although not mentioned in this review, several LDN patients have mentioned dramatically increased libido. Although Younger wasn’t clear why this would be so and hadn’t noticed it in his patients, one ME/CFS and FM doctor has noticed its effects in her patients. If it is happening, Younger speculated that lifting inflammation-induced anhedonia could be in play.
Conclusions
Research into LDN has been limited, but with the first human drug trial only being published in 2007 , LDN is new to the medical community. Despite the limited research, strong clinical results and word of mouth among patients is making LDN a relatively well-known opioid alternative. LDN is the first choice for pain for Dr. Nancy Klimas, a Chronic Fatigue Syndrome researcher and physician at Nova Southeastern in Miami.
LDN’s mode of action is still unclear, but the authors encouragingly noted in the paper that several laboratories are ‘vigorously’ engaged in determining exactly how it works.
LDN Projects On Tap
Meanwhile stating “there will definitely be followup studies” Younger has some projects lined up.
- Do we see inflammatory markers in the blood decrease when someone improves with LDN?
- What is the proper dosage? (We will be conducting a dose-ranging study to see if lower or higher dosages work better for some people).
- Does LDN help in other conditions such as rheumatoid arthritis, osteoarthritis, CFS, and maybe even depression?
- We want to do a large (200+ person) clinical trial of LDN and fibromyalgia
(It was not clear which projects have been funded. The 200 person trial has not been funded.)
Drug Approval
An FDA-approved LDN drug would increase interest in LDN dramatically, making it available to many more patients and increasing research interest dramatically. The ability of compounding pharmacies to produce LDN would seem to remove it from the interest of drug companies, but Younger noted that one drug company, TNI Biotech, is interested and is trying to commercialize a 4.5 mg version of LDN for around $1/day.
Touting LDN’s effectiveness and safety in pediatric Crohn’s, TNI Biotech hopes to use that disease to bring an FDA-approved version of LDN to market. They did not respond to a request for an interview.
LDN News
International LDN Awareness week, this coming November 1-8, will coincide with a live streamed conference in Las Vegas.
The LDN Research Trust
LDN Research Trust is looking for case studies from LDN prescribing doctors, pharmacists, researchers, and LDN users around the world. To have case studies included you must provide
- Name
- Country
- Condition(s) LDN is used for
- When LDN was started
- Up to 40 words telling what LDN has done to help
- A photo
Professionals List
Doctors who prescribe or pharmacists who compound LDN can add themselves to the LDN Researach Trust’s lists at the links below..
- LDN Prescribing Doctors: http://www.ldnresearchtrust.org/node/147
- LDN Pharmacists: http://www.ldnresearchtrust.org/node/148
If you know of someone who should be added, send the Trust their details and they will contact them.
Interviews
If you’ve tried LDN, you can also be interviewed over the phone. Your interview will be made into a video for the Trust’s YouTube Channel. They have over 400 videos listed.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!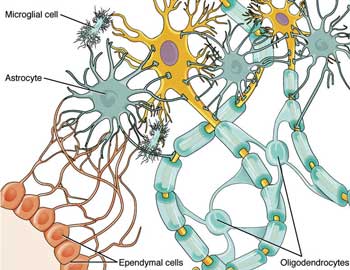




I’ve been on LDN at various doses for almost 4 months and it’s not helping. Does anyone know about taking ultram with it? I’ve cut down to just 75 mg in the morning and take the LDN in late afternoon. I read that Dr. Teitelbaum thought a small amount of ultram would be ok? Also, even if it doesn’t improve symptoms, is it good to take for general immune function? And is there really a best time for it? I’ve read conflicting info on that too. Would be grateful if anyone could clear up this confusion.
On facebook I have learned a lot from a few different groups. My favorite so far is called: GOT ENDORPHINS? LDN (Low Dose Naltrexone) You can read there and learn from others. I am not on LDN due to the fact that I am limited by what doctors I can see. So far I haven’t found one who takes my insurance who is willing to prescribe it. I am currently seeing a new Doc in Palm Coast FL named Dr. Daniel Dantini and he does anti-viral testing so that is the direction I am going in currently. I know this group is familiar with that doc. If anyone has feedback on his approach I would be interested in hearing it. I am reluctant to do anti-viral meds but am probably going to do it. I will be seeing the doc for my blood test results in a few weeks.
Hi there, if you got to the Facebook page LDN Research Trust or contact the website for them that Cort has put on this article, they can help to put you in touch with a doctor who will prescribe LDN.
I just went to a meeting a few weeks ago where the pharmacist who compounds it in the UK spoke…he said ‘it is safer than taking aspirin’!
We just gotta get the word out and get doctors up to speed with this medication and how it is helping people!
A.
Good luck jsuzor, sure hope the anti vitals help you. I’m considering them too but also hesitant. Still……
Thanks Jsozor for the props on the group!! We do keep it nice and friendly and supportive! I added the website link https://www.facebook.com/groups/GotEndorphins/
Betty,
I take LDN at 9pm. Sometimes that is right at bed time, sometimes I stay awake for another couple hours. I can take instant release morphine during the day if I really need it for pain, because it does get out of your system in a couple hours. I recommend you try taking it at night and see if that makes a difference. You should be able to take ultram in the day if you need it. And hopefully you’ll get better results letting it work at night. A good site for reading about LDN is LDNScience.org. Good luck!
Hi there, you need to leave a 4 hour window on either side of taking Ultram with LDN. ie. take the Ultram up to 7pm and then take the LDN after 11pm.
Everyone seems to be different in regards to the best time to take it – the best suggestion is to try it in the morning if you have been taking it at night or vice versa. Also, some people actually take 2 doses (divided of course), morning and night.
There is a lot of great support, help and advice on the Facebook page called LDN Research Trust.
A.x
Naltrexone is available at alldaychemist on line, without prescription but at 50mg, it needs to be diluted in water to get the “low dose”, 1 to 4.5 mg. Cheap but must be ordered as Naltrexone, not lDN. RP
Sorry to hear it is not effective for you. I’ve been trying to figure out how to get an LDN trial for my FM pain, getting much worse over the years. Maybe subtypes of FM with those high inflammation markers are the only responders. I don’t have any inflammation marker tests on my last blood tests and my PCP would not prescribe this off-label anyway. This recent study may push me to look into traveling out of state to find a prescriber. It’s odd that one standard prescription naltrexone tab, titrated at home by a careful FM sufferer, could provide him with a lot of med. Too bad I don’t know any addicts. The risk/reward for this med looks pretty good to me.
Hi there, contact the links that Cort has put on this page for LDN Research Trust – they can put you in touch with doctors (and pharmacists) that will prescribe for you. (They work with 154 different LDN Member Countries – and have excellent access in order to help you!).
A.x
Wow, you posted while I was writing, thanks, will check it out.
My dr. is in longevity medicine. I tried ldn on 2 different occassions and found no success with it. I have had fibromyalgia/cfids for 26 years.
I just want to point out that LDN can take up to 6 months for people to notice a marked difference. In saying this, unfortunately, it doesn’t work for everyone, but equally, it is important to give it time.
For me, although my pain is still here, I am sleeping!!! Full nights!!! Which hasn’t happened in 20 + years! And I also have more energy and my fibrofog has cleared quite a bit.
I would also say that if you aren’t tracking your daily pain levels, sleep, mood, energy etc then sometimes it is hard to see a slow but positive change until you look back at your records.
A.x
I’d never heard it could take up to 6 months. Thanks for all the info Alyssa.
That is really good to know because I tend to drop things quickly. I agree that it’s hard to tell sometimes that you’re making steady progress unless you keep track of things. Dr.Peterson has said the same thing; the patients sometimes are the last to notice. 🙂
I’ve taken LDN first at 4.5mg for a month or so then switching it over to am rather than pm. I also took Tramadol with the LDN which my doctor/pharmacist (compounding) said was ok to do. No results. So, I was bound and determined to start at a low dosage and work up until I found my “best working” dosage for me. I went back to 1.5mg for 1 month and jumped up .05 every month. Well, I got to 3.0mg and still nothing. I could no longer go on as my fibromyalgia/CFS required more pain medication (you cannot take opied(sp) pain meds (oxy, morphine, hydrocodone etc) along with the LDN as it is an antagonist. I still wonder why the allow you to take the Tramadol with it as it is listed as an opied too. Well, my doctor told me I could as long as I took any of my pain meds 4 hrs before or 4 hours after (4 hour window). There are conflicting theories with this as you will find. I was a member of GOT LDN website as well. I does seem to do wonders for many especially people with MS. I just never saw any results nor could I go any higher on the dosage due to my need for opied pain med relief. I was hoping to one of the people blessed by LDN. I guess not.
You gave it a great try. I’m going to have a post on other microglial inhibitors coming up. May be there will be something in there for you.
I now live in the UK (relocated from Finland) and have tried to get hold of LDN through national health service since I have little money to spare. They are unable to issue it because it hasn’t been approved by the government. My husband suggested trying to get it from my GP as a private patient. My doctor would have been quite willing but was unable to get hold of small doses. He also lamented the fact that it hasn’t really been proven effective (of course he had to say that…). It’s a disgrace. I understand that it works for many people with ME but was unsure of FM. This article was helpful in convincing me that I should try it. I can get it online but it’s going to cost a lot of money which I don’t have, as I survive on a disability pension. However, it does sound as if it could be worth a try. My insomnia and lack of libido are only getting worse in spite of my best efforts. Thank you.
You can get the regular 50mg tablets use water to dilute them. For example one 50mg tablet in 100ml of water would be 1mg/2ml of solution, so to take 4mg you would take 8ml of solution which can be kept in the fridge. Any of the online groups mentioned can confirm exactly how to do this. In any case the ldnresearchtrust.org mentioned above is in the UK, they should be able to help you. The tablets should be very cheap even if you have to pay for them yourself, just get your doctor to write the prescription.
It sounds like it’s pretty cheap in the US and if you have compounding pharmacies in the UK hopefully it will be there as well. I know when money is low ANYTHING is expensive.
Check out the LDN trust for prescribing doctors and pharmacies. Hopefully they have some. Good luck!
I tried LDN but had some side effects and had to quit taking it. I’ve heard that it takes a while to work but without knowing whether it def would or would not work I just couldn’t keep it up to find out.
I ran out of LDN last week after being on it for 2 years. Its taken me a week to get back to what was normal. I can only describe what it was like last week as suffering from “restless body”!! Itching, twitching, waking up and having to jump out of bed to get any kind of relief from restless legs etc… I’m now back on LDN and sleeping through the night. Thank god for LDN!!!
I just had my SED lab taken and it’s only a 1 three years ago before I started LDN it was 20! Halla!! https://www.facebook.com/groups/GotEndorphins/. Yes I agree got e Is a good group go join
Wow… Jarred Younger would love to hear that. 🙂
The original medical article by Younger et al doesn’t mention CFS/ME once. Why? Because there are no case reports in the medical literature of people with CFS/ME improving when they take LDN. (Our anecdotes on the web don’t count as far as doctors are concerned.) If researchers are to get funding for clinical trials of LDN in CFS/ME they need case reports first to build on.
I am willing to have my story written up – I’m one of the lucky ones who have responded well to LDN. Are there others who could also volunteer? Ideally you shouldn’t be using any other medication as that would confuse the picture, and if you had objective evidence of improvement, such as returning to work, that would be even better.
There’s also the problem that people who get better tend not to follow posts – they are back at work. So if you know anyone who would be suitable – can you ask them as well?
Here’s a link to the LDN Trust newsletter where I asked for volunteers.
http://ldnresearchtrust.org/sites/default/files/Jan%202014%20use.pdf
Thanks
Monica
I’ve been on LDN for about a year. I am very sensitive to any medication, so I started at the lowest dosage 0.2mg for a few weeks, then 0.4 then 0.6. I’m still at 0.6. Side effects include nausea. I hadn’t heard about that before taking it. It works very well for me for fibro. This winter in Chicago was brutal and without the LDN I would have been in a world of pain, but with the LDN, the pain was quite diminished.
I do not remember if I posted the results of my using LDN. I used the LDN for over 6 moths, first using the lower dose and then increasing it gradually over several weeks. Unfortunately, I received no relieve.