

Problems with the thyroid – the “gas pedal of the body”, as Dr. Teitelbaum calls it – seem to make sense given the fatigue, exercise and other problems found in ME/CFS and FM. Assessing and treating the thyroid, though, is one area where the practices of chronic fatigue syndrome (ME/CFS) and fibromyalgia (FM) doctors may distinctly diverge from other doctors.
Standard thyroid tests are often negative in ME/CFS and FM but Dr. Bateman has stated that about a third of her ME/CFS patients are hypothyroid. Dr Holtorf – who has published on thyroid issues – believes the standard TSH tests are looking in the wrong place.

Do problems with the gas pedal of the body – the thyroid – play more of a role in ME/CFS than most doctors think?
Those tests assess the pituitary’s role in triggering thyroid hormone production. Holtorf asserts, though, that pituitary thyroid hormone levels often do not reflect the profound thyroid hormone reductions in other parts of the body in many people with ME/CFS and FM.
T4 – the hormone produced by the thyroid – is not the most active form of thyroid hormone. It’s only when T4 is broken down by non-thyroid tissues, such as the liver, that T3 – the biologically active form of thyroid hormone – is produced.
Holtorf believes that thyroid tests that more directly measure thyroid levels (free T3) or thyroid hormone inhibitors (reverse T3; free T3/reverse T3) as well as other metabolic and inflammatory tests (basal metabolic rate, SHBG, leptin, ferritin, tendon reflexes) should be incorporated into standard ME/CFS and FM testing.
Last year, a large (n=197) Dutch study explored the thyroid question in ME/CFS in greater depth than ever before, and in doing so, may have uncovered a new thyroid illness subset.
Higher Prevalence of “Low T3 Syndrome” in Patients With Chronic Fatigue Syndrome: A Case–Control Study Begoña Ruiz-Núñez1,2*, Rabab Tarasse1, Emar F. Vogelaar3, D. A. Janneke Dijck-Brouwer1 and Frits A. J. Muskiet1. Front. Endocrinol., 20 March 2018 | https://doi.org/10.3389/fendo.2018.00097
Hypothesizing that the “subclinical hypothyroidism” (based on normal TSH levels) found in ME/CFS is caused by low-grade inflammation, these researchers dug deeper than most. Not only did they examine a full array of thyroid measures, but they also assessed traditional markers of inflammation, metabolic inflammation (insulin resistance-mediated, de novo lipogenesis (DNL), and HDL-cholesterol (HDL-C). (Metabolic inflammation refers to conditions where metabolic and inflammatory pathways contribute to atherosclerosis, insulin resistance (IR) and type 2 diabetes.)
Plus, thinking of the leaky gut/inflammation question, they assessed gut wall integrity and the status of nutrients that can influence thyroid function (iodine and selenium) and inflammation (fish oil fatty acids, Vit. D, kyrnurenine, tryptophan, etc.).
With 99 people with ME/CFS and 98 healthy controls, the study appeared quite robust.
Results
The results bore Dr. Holtorf’s conclusions out that TSH tests are insufficient to assess thyroid functioning in chronic fatigue syndrome and fibromyalgia.

Except for TSH – which was normal – and reverse T3 – which was higher – almost every thyroid measure was lower in the ME/CFS group.
These Dutch and Spanish researchers found that TSH levels were indeed similar between ME/CFS patients and healthy controls, but virtually every other thyroid measure was significantly lower in the ME/CFS group (free triiodothyronine (FT3) (difference of medians 0.1%), total thyroxine (TT4) (11.9%), total triiodothyronine (TT3) (12.5%), %TT3 (4.7%), sum activity of deiodinases (14.4%), secretory capacity of the thyroid gland (14.9%), and 24-h urinary iodine (27.6%). Plus, higher percentages of reverse T3 (rT3) (13.3%) suggested that increased levels of thyroid inhibition may be present in ME/CFS.
The authors called the lowered 24-h urine iodine output of ME/CFS patients “remarkable” (which may simply mean “notable”). Both the ME/CFS group and the healthy controls also had about half the optimum omega-3 indexes believed needed to protect against cardiovascular and neuropsychiatric diseases.
Documenting evidence of widespread thyroid problems not picked up by standard TSH tests was progress indeed, but the finding that really stood out concerned the 16% of ME/CFS patients who fit the criteria for “low T3 syndrome”. (Seven percent of the healthy controls did too.)
A New Thyroid Subset for ME/CFS?
Low T3 syndrome or nonthyroidal illness syndrome (NTIS) (or euthyroid sick syndrome ESS) is a debated illness found in some serious illness states including sepsis, starvation, surgery, heart attack, and others. Dr. Leslie J DeGroot, M.D., a top endocrinologist, reported in a 2015 review “The Non-Thyroidal Illness Syndrome” that this syndrome probably occurs in “any severe illness”.
DeGroot starts off his review stating that NTIS, “refers to a syndrome found in seriously ill or starving patients” who have low free T3, increased reverse T3, normal or low TSH, and, if the condition becomes chronic, low free T4. DeGroot asserts that these findings indicate, as Dr. Holtorf asserts, that low tissue thyroid hormone levels are present, and thus tissue hypothyroidism is present. (Note that low total T4 was not found in the ME/CFS group.)
In some ways, this syndrome is no surprise: serum thyroid hormone levels are known to drop during starvation and illness. Starvation (carbohydrate deprivation) inhibits the conversion of T4 to T3 (the active form of thyroid hormone) and prevents the metabolism of reverse T3 (which blocks the receptors for T3, thus reducing the production of T3.)
The reduction of active thyroid hormone levels seems to make sense, given a similar reduction in the basal metabolic rate induced by starvation: the body appears to be hunkering down and attempting to conserve its resources until food is present.
Starvation isn’t the only inducer of this process, though. DeGroot reports that a large proportion of people in intensive care also exhibit low T3 and T4 levels. They also tend to have increased sympathetic nervous system response – which is, of course, typical in ME/CFS.
Various hypotheses have been put forward which propose that NTIS is beneficial or inconsequential and should not be messed with. They include the possibilities that the abnormalities are artifacts, that they do not reflect true free hormone findings, and that they reflect the body’s way of dealing with the situation.
DeGroot, however, believes the findings reflect a pathological state. He does not appear to believe that pituitary thyroid hormone levels differ from those of the body in NTIS, but suggests that cortisol / cytokine levels / low O2 levels (hypoxia) / reduced leptin, etc. are likely responsible for the initiation of NTIS.
The T4 Paradox – When Treatments Make Things Worse
DeGroot’s personal experience is that treating patients with NTIS with T4 (unpublished) often results in significantly elevated rT3 levels (and T3 inhibition).
Nunez-Ruiz et. al. also suggest that standard thyroid therapy (T4) in the NTIS-like ME/CFS subset could actually induce a state of NTIS, and point out that T3, recommended by Holtorf for ME/CFS and FM, has been suggested for severe NTIS. They referred to the NHANES cohort, which found that T4 administration resulted in higher T4 levels but 5–10% lower T3 and FT3 levels, and a 15–20% lower T3/T4 ratio. They believe tissue levels of those factors are probably far lower.
Low “T3 syndrome” in our study resembles the thyroid hormone profile of a subgroup of hypothyroid patients receiving T4 monotherapy. The authors
Holtorf also has found T4 supplementation unproductive and even harmful at times in his ME/CFS and fibromyalgia population. When high levels of rT3 (>150) are found, or free T3/reverse T3 ratio is greater than 0.2 (measured in picograms per milliliter (pg/mL), instead of T4 (Synthroid and Levoxyl or Amour thyroid – a pig glandular product) Holtorf will prescribe free T3 (compounded) instead.
The Worm (Dauer) Rides Again
Ruiz-Nunez et. al. believe the metabolomic studies in ME/CFS may be providing a clue. The impaired mitochondrial production and hypometabolic state they suggest appears to fit with their findings of NTIS in a subset of ME/CFS patients (and one would think the general state of reduced thyroid activity in the group as a whole). Hypothyroidism is, after all, associated with decreases in metabolic and heart rates, oxygen consumption, body temperature and oxidation of glucose, fatty acids, and amino acids.
They throw the whole kit and kaboodle into the mix (inflammation, infection, gut issues, psychological trauma (particularly during childhood) as they suggest that the cell danger response (aka Naviaux) could be behind the NTIS-like findings in some ME/CFS patients. Inflammation may or may not be the issue.
The authors believe that ME/CFS probably represents a common pathophysiological state that can be reached in any number of ways. In a refreshingly direct manner, they reported that their study findings presented one possible way of reaching that state, “but do not get us closer to the cause(s).”
Herpesviruses to the Fore?
Could one answer – HHV-6 – be hiding in plain sight?
Back in 2001, Wikiland reported in The Lancet that fine needle biopsies demonstrated that 40% of people with ME/CFS have lymphocytic thyroiditis. The fact that half of those found with diseased thyroid tissues had normal thyroid lab values suggests that lab values, as Holtorf believes, may not always tell the whole story.
Hashimoto’s thyroiditis, the most common cause of hypothyroidism, occurs as a result of an autoimmune attack. Casselli’s 2012 study finding of a high incidence of HHV-6A DNA in Hashimoto’s thyroid tissues but not in controls (82% vs. 10%, p≤0.001), suggests that HHV-6 could be triggering Hashimoto’s. (HHV-6A was also active in the Hashimoto’s patients, but not in the healthy controls.) If HHV-6A is triggering Hashimoto’s, it might also be triggering the hypothyroidism that seems almost endemic in ME/CFS.
The fact that Caselli found HHV – 6A, not HHV- 6B was notable because while virtually everyone is infected with HHV-6B early in life not everyone is infected with HHV-6A – and when they are it tends to be later in life, which is when most cases of ME/CFS begin.
Kristin Loomis of the HHV-6 Foundation has tried for years to interest someone in examining the thyroid tissue of people with ME/CFS for pathogens. She’s noted that many of the suspected viral culprits in ME/CFS (HHV-6, parvovirus B19) can cause “smoldering” infections in the thyroid.
Now enter Bhupesh Prusty, a Solve ME/CFS Initiative-funded researcher who found HHV-6 in about 60% of a small group of ME/CFS patients’ cells. Prusty found that one HHV-6 infected ME/CFS cell was able to alter the mitochondrial functioning of neighboring, or even distant cells – apparently by secreting mitochondrial inhibitors. Prusty will be speaking at the NIH ME/CFS conference in April.
Kristin Loomis speculates that if HHV-6 is present in the thyroid, it may have found its way to the brainstem as well, where it could help produce the autonomic problems in ME/CFS. At the 2016 IACFS/ME Conference, Dr. Klimas presented evidence that indices of HHV-6 activation are correlated with symptom severity in ME/CFS. Learn more about HHV-6 here.
Conclusion
A fairly large Dutch study found evidence of low thyroid functioning in ME/CFS. The study needs to be replicated, but about 15% of the ME/CFS group met the criteria for Non-Thyroidal Illness Syndrome (NTIS) which occurs in starvation, sepsis and probably other serious illnesses. While most people with ME/CFS did not meet the criteria for NTIS, the overall findings – normal TSH levels, increased reverse T3, and reductions in a variety of thyroid tests – suggest the group as a whole trended in the direction of NTIS.
The findings may call for the use of T3 instead of T4 for some people with ME/CFS/FM. While studies assessing the effectiveness of T3 are lacking, both Leslie DeGroot, a prominent endocrinologist, and Dr. Kent Holtorf, an ME/CFS/FM specialist, support using T3 (in Dr. Holtorf’s case, compounded T3) instead of the usual T4 preparation (such as Synthroid) in patients with NTIS (DeGroot) or ME/CFS/FM (Holtorf).
Learn more about what some doctors say about using T3 in ME/CFS/FM:
The cause of atypical thyroid issues (normal TSH/low T3/high rT3) found in some people with ME/CFS/FM is unknown but could reflect an attempt by the body to conserve resources (Dauer state), could be produced by cytokines, cortisol, hypoxia, etc.) or possibly a pathogen such as HHV-6.
Check out Dr. Holtorf’s approach to ME/CFS and his thyroid testing regiment:
The Low T3 Series on Health RIsing
- The Atypical Thyroid Issues in Chronic Fatigue Syndrome (ME/CFS), Plus a New Thyroid Subset?
- Pure T3 Thyroid and Stories of Recovery from Chronic Fatigue Syndrome (ME/CFS) and Fibromyalgia: An Overview.
Check out the Critical Illness Series as Well
- Neither dying, nor recovering”: Learning from ICUs to Solve ME/CFS and Fibromyalgia – A Synopsis (Nov. 2019)
Notes
Support Health Rising in Getting the News Out!


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!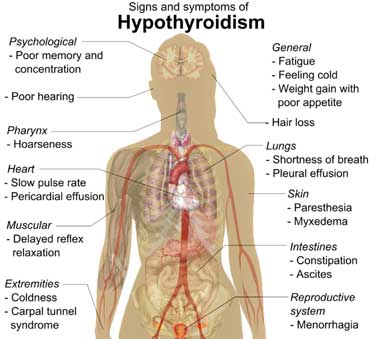



I really don’t find the HHV-6 ideas credible. We have been down the viral road many times….
Viruses can trigger CFS but they do not perpetuate it.
I really hope researchers don’t waste time on viruses.
I see good reason to believe that the body *must* reduce energy output by the mitochondria in order to protect against oxidative stress when oxidative stress gets rampant.
Oxidative stress (actually molecular hydrogen peroxide) has been mentioned in the Naviaux metabolic paper as a signal molecule that triggers the mitochondria to go in a hibernation state. That points to its important role.
Now many retroviruses like HHV-6 and EBV both remain permanently present in the body after infection and increase ROS production.
For HHV-6: https://hhv-6foundation.org/latest-scientific-news/hhv-6-induces-oxidative-stress-and-chlamydial-persistence
“They report that HHV-6 caused NADPH accumulation, decreased formation of glutathione, and increased oxidative stress”
Note of myself: excessive NADPH accumulation can cause oxidative stress just as much as too few of it.
“This work may provide a window into further understanding the role played by HHV-6 in several clinical conditions associated with the glutathione/oxidative stress balance. Several studies, such as a recent study by Shungu et al, have shown that increased oxidative stress plays a key role in the development of CFS (Shungu 2012). Others have indicated that the depletion of glutathione, which helps counteract oxidative stress, may be a key cog in the mechanism that leads to conditions characterized by drug hypersensitivity such as DIHS/DRESS (Moling 2012). Increased oxidative stress due to decreased glutathione synthetase activity has also been observed in cases of encephalitis (Castegna 2011).”
and https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4177780/
“Our results suggest that oxidative DNA damage is involved in acute encephalopathy associated with HHV-6 infection.”
“These results suggest that oxidative DNA damage in the brain caused by HHV-6 infection may be independent of inflammatory reactions and subsequent axonal damage.”
“In the present study, we only examined levels of CSF biomarkers in the acute phase of the diseases. Further investigation is required to clarify whether lipid peroxidation may be involved in the chronic phase.”
Note of importance: the combined decrease in T3 and increase in reverse T3 may indicate there is more going on then a “lack of conversion to T3” as this does not explain the increased reversed T3 well enough IMO. It may be a protective reaction of the body trying to actively induce a state of hibernation. Correcting this hypothyroid situation may be a double edged sword if the cause of it is not well investigated.
Hey Cort, Will there be an article coming on nasal glutathione or did I miss it?
Oxidative stress as well. Very interesting! So many potential pathways… Thanks DeJurgen
Yes, there will. I want to get the CBD oil blog out first and then will turn to that. Thanks for the reminder 🙂
I wasn’t aware of “They report that HHV-6 caused NADPH accumulation, decreased formation of glutathione, and increased oxidative stress” until today. It leaves some intriguing options.
NADH and NADPH bear a lot of resemblance. Both can create free electrons in the electron transport chain. These free electrons in turn are known sources of oxidative stress. The higher the amount of NADH and NADPH is, the more free electrons are created.
This does not contradict with NADPH’s role in the battle against oxidative stress. NADPH is the “fuel” for the immune system to create rapid bursts of hydrogen peroxide in order to kill pathogens. So its capacity to create oxidative stress is not new. It is glutathion that is superb in fighting oxidative stress, not NADPH itself. And in order to recycle oxidized glutathion you need NADPH.
Now if a virus has learned the trick to partially block the recycling of glutathion then glutathion amounts will decrease and NADPH in the cells will increase. It’s in fact a bit more complex: to get the same glutathion recycling rate as before NADPH in the cells needs to be higher if there is less enzyme available. For the same amount of NADPH less glutathion recycling will be done if there is less enzyme available and so on…
This has a triple effect (compared to the same person not having a latent Herpes infection):
* less glutathion will be available; this will lead to more oxidative stress
* more NADPH will be available in the cells, this will lead to more oxidative stress
* as more NADPH is available the immune system has more “fuel” to create its “weapons” and *may* become more active
Both more oxidative stress and more immune activation are common to ME. These viruses may not be needed to have ME, but they seem to “facilitate/empower” it or in other words make a person more susceptible to it.
Now increased NADPH levels may sound confusing and in contradiction with my believe that NADPH levels are reduced in ME. So let me explain.
Consider a healthy person having 100% (of “normal”) glutathion, NADPH, ROS and immune “strength modifier”.
A Healthy person with a latent Herpes infection may then have for example 90% (of “normal”) glutathion, 110% NADPH, 110% ROS and 110% immune “strength modifier”.
A person with an “average” case of ME may then have for example 60% (of “normal”) glutathion, 75% NADPH, 140% ROS and 140% immune “strength modifier”.
A person with an “average” case of ME combined with latent Herpes infection may then have for example 50% (of “normal”) glutathion, 85% NADPH, 150% ROS and 150% immune “strength modifier”.
While the later person would still have lower NADPH values then a healthy person, they would be increased compared to the “non-Herpes” ME patient and the lower glutathion and higher ROS and immune “strength modifier” values would make him sicker then the average “non-Herpes” ME patient.
At the same time, the altered setting of the healthy “Herpes” person would get him closer to “ME values” and by that likely more susceptible to ME in the case of a strong “hit and run attack”.
Theoretical but it kind of makes sense to me.
I would not give up on HHV-6A just yet. It’s been implicated in other neurological diseases and Nancy Klimas found that indices of HHV-6 reactivation are associated with symptom severity in ME/CFS and (as I just added) Bhupesh Prusty has some evidence indicating that it may be able to turn off the mitochondria in ME/cFS.
I would think sampling thyroid biopsies for that and other bugs would be a good idea given huge (and unexplained) increase in hypothyroidism in ME/CFS.
This is very timely. I have had HHV6, EBV, and parvo. After being on T3 for 8 years, my thyroid is being biopsied tomorrow. Does anyone want to look at the tissue?
Mmmmmm…. not convinced! We have been down the viral path many a time, for nothing. I will be very very surprised if a virus has a role in perpetuating the illness.
Hi Cort.
I’ve wondered whether ME was/had a sub-set of people with a thyroid problem. However, a couple of things. One, I can’t see a new biomarker for thyroid problems here. You mentioned a paper I need to review (Dutch?) which recently suggested thyroid problems in ME; however, I’m not clear what new biomarker/technique they used. Two, as you are aware, Ron Davis’s colleague referred to the bloodstream as the body’s sewer – conveys waste products. So the theory regarding a virus would have to account for the fact that virus’s/breakdown products from virus’s don’t turn up in the bloodstream – Ron Davis. Also, would hypothyroidism not turn up in a metabolomics study? I.e. if ME has a similar metabolomic “fingerprint” and there’s been a metabolomics study on hypothyroidism. This is still a theory I like; however, I think we’d need a better biomarker for hypothyroidism – suggestion to OMF/NIH?
Here’s the Dutch study: Higher Prevalence of “Low T3 Syndrome” in Patients With Chronic Fatigue Syndrome: A Case–Control Study Begoña Ruiz-Núñez1,2*, Rabab Tarasse1, Emar F. Vogelaar3, D. A. Janneke Dijck-Brouwer1 and Frits A. J. Muskiet1. Front. Endocrinol., 20 March 2018 https://doi.org/10.3389/fendo.2018.00097
You ask the big question. Neither Davis nor Lipkin or an SMCI funded study were able to find any evidence of pathogens. Davis believes the techniques are so precise that they should be able to pick evidence of even a smoldering infection.
On the other hand a doctor just told me that doctors have had a lot of trouble identifying the virus in the spinal fluid of the cases of acute flaccid myelitis found recently in children. This is a disease which directly effects the spinal column. http://theconversation.com/acute-flaccid-myelitis-what-is-the-polio-like-illness-paralyzing-us-children-109551
It’s quite confusing!
While I am not a believer in HHV-6 being a major cause of the Somatic Symptom disorders, your assumption that viruses do not contribute to the chronic it is incorrect. Innovative Med Concepts.’ Research has completed research that an ongoing HSV-1 appears to be the actual engine that maintains the abnormal stress response and the central sensitization seen in fibromyalgia and to a lesser degree in MECFS. What is the basis of your bias?
Reference for their research? Ratified by others?
We’ve seen many ‘promising’ leads on viral influences over the last 30 years but they never stand up to future scrutiny.
I have no bias, only deep skepticism.
If a viral factor was found tomorrow to conclusively perpetuate this illness then I would be delighted to be wrong, especially if there was a clear treatment path…
But that can be said of any non-viral theory as well. No theory has been proven
Ah I see you are the man behind Innovative Med Concepts. I would suggest you have the bias / vested interest.
Or…. will we see some results, and ones that are replicated, that strongly back up your claims? I am very happy if your research proves me wrong.
Btw, I am talking cfs here, not FM.
Dr. Pridgen, I would like to look into the HSV-1 issue you mentioned. I am currently seeing a virologist/neurologist who is primarily a researcher. Testing showed that I can’t fight off HSV-1 (an antigen challenge I believe it was) and she told me that she believes my autonomic ganglia are inflamed. These nerve cells are directly tied to the stress response/fight-or-flight. I have been diagnosed with both FM and ME/CFS and have been ill for 32 years.
HHV6 may not be THE cause, but it may be a contributor and/or a co morbidity.
I know that I tested positive for HHV6 and super low T3 and that I was treated with several super anti-virals and valcyclovyr and with T3 for 4 years.
I got much better (60 percent I’d say) and have been able to function in the world for the past 10 years, _and_ I don’t get every virus that passes thru town.
Maybe that is just a bunch of unrelated hooey. Maybe not. I vote not. ?
Sadly, I am in a medical group with a Dr that doesn’t ‘do’ T3. Sigh.
With respect, I don’t think you were having a day to give the article a critical read. Thyroid related issues are a consequence (symptom) /process gone awry. It’s not the illness itself. So to find this incredible link between a virus and that an entire physiological system is awry in many of us despit normal testing is incredible. This illness is coming to affect so many of our others physiological systems. I think your comment was a bit simplified into the illness and a cause. This is excellent research.
You have a hypo and hyper group of ME patiënts. Slow heart rate etc.. fast heart rate pots like patiënts.
It’s actually possible to have the fast heart rate, etc from orthostatic intolerance and still be hypo. What happens is that low blood volume results in not enough oxygen getting to the brain, esp when standing. That causes the body to go into a state of panic (with a purely physical cause). That panicked state increases sympathetic nervous system responses, inc heart rate in order to try to get sufficient oxygen to the brain. And, the parasympathetic nervous system does not have enough energy to calm the SNS back down, so the adrenaline & rapid heart rate, etc continue until the SNS runs out of steam. This drains the adrenal glands, which in turn interact very strongly with the thyroid, making hypothyroid symptoms even worse. (Anytime you are treating the thyroid, you also have to make sure the adrenals are working right, and vice versa or treatment is ineffective.)
Thank you for your explanation! I did not know that this was possible with hypo too 🙂 So the short of oxygen in the brain explanes also some panic attacks without fear for something not psycholocial at all 🙂
You’re welcome….and yeah, the panic attacks are very much physical when this happens, not psychological
omega-3 the fishy stuff and not some unknown-to-me hormone?
HHV6 was just one of many in me, whenever we got rid of it another one took its place, drove me and my alt. healer nuts
Omega 3 fats are from fish oil, flax seed oil, & various other foods sources, or supplements.
Hashimoto’s is fairly common in EDS if it gets diagnosed but a majority who get treated with T3 are still Sick so it is not any cause or answer. Even a top World thyroid Endocrinologist at Southampton General Hospital said to me that the results do not add up with treating this & something else is going on
who was endo at southampton ? thanks
I was found to have low T3, high antibodies and a goitre. NHS didn’t want to treat so I got natural thyroid on recommendation from a private doctor but it made me worse and starving hungry all the time.
Hi Victoria, did you get the test at your GP? Why wouldn’t the NHS treat you?
I tried naturethyroid and became super hungry on it as well…I could not tolerate it even though my t3, and t4 are very low.
Dr. Paul Cheney talked about his theory for why the T3 level is low in this 2013 presentation.
https://paradigmchange.me/wp/cheney/
He seems to be suggesting that this is a functional state, to protect the body from “something worse” happening.
My own experience has been that although I can force my body temperature (which often has been 97F or lower) to be fairly normal with enough T3, I never have felt that good about doing that.
On the other hand, if I take quite a bit of iodine and other mineral supplements (such as zinc and selenium), my temperature has tended to be normal quite a lot of the time and I feel a lot better than I do when I am instead taking T3.
My vague theory about that at this point is that my body temperature is dropping to protect me from heavy metals or radiation poisoning or mold toxicity, and that the minerals are helping to detoxify those things and therefore allowing my body to move to a higher temperature and higher metabolic state without being harmed by it.
Interesting – I think Cheney came up with that protective response idea a long time ago! I imagine that he was first actually. I remember he said I would push the body towards health and it would pull back.
Interesting, though, how the protective response idea isn’t present just in ME/CFS but in NTIS -which occurs in all these severe disease states.
Cheney also believes that HHV-6A is associated with AIDS, MS and chronic fatigue syndrome.
I never thought of taking iodine although I have a particular affinity for seaweed 🙂
https://www.healthline.com/nutrition/iodine-rich-foods#section2
An endocrinologist once told me that with Hashimoto’s, taking iodine was like throwing gasoline on a fire…..so my Lugol’s is in the back of my cabinet.
Cort, ME folks should try the Iodine Patch test – a topical application is applied and then the hours needed for absorption reflects how much the body is in need. Start slow because it is an antiseptic as well as being the # 1 thyroid protector. Often not digested well from foods or supplementation, so topical is much better.
The functional medicine doctors tend to be way far ahead on issues like this, though of course since they are not treating based on solid research findings, they can be wrong about things.
Here is an article by Amy Myers on the topic of T3 and thyroid that discusses mineral status, for instance.
https://www.amymyersmd.com/2015/08/thyroid-health-part-vii-four-essential-nutrients-for-thyroid-function/
In this article, Dave Asprey talks about the importance of seeking out a non-toxic iodine supplement, especially when thyroid issues are a problem.
https://blog.bulletproof.com/thyroid-symptoms-problems/
I wonder if that endocrinologist might have been seeing a “gasoline on the fire” effect as a result of prescribing a toxic iodine supplement?
At present I have been using Bulletproof and Microbe Formulas supplements, and both of those companies seem super-concerned about toxicity issues.
And I do feel like I have gotten much better results with those than I have with iodine supplements I have used in the past (which didn’t seem to be of help at all).
I wonder if the toxicity issue is why.
This is our conundrum isn’t it? Particularly with hormonal issues. Lots of anecdotal reports from doctors and patients but few actual research studies.
Makes it hard to know what to do. Dominic’s blog will show lots of anecdotal evidence for using T3.
Other doctors are firmly against it or indeed assessing thyroid the way Holtorf does.
You can add me to your anecdotal evidence. I was tested in 2010 for thyroid by Dr Joanne Pizzino, a FM/CFS specialist in Cary, NC. She put me on Triiodinethyronine ( compounded) at that time and told me to take it til I die. Incidentally, I also have had a herpes simplex virus that showed up on my lip when I was around 30, and I’ve long wondered whether That has had anything to do with my FM/CFS. My virus testing in 2010 didn’t show anything but I’ve had two serious long term viral episodes since then that each took 6 weeks to clear. I’m thinking it’s time for more viral testing.
I remember Neil Nathan and Jacob Teitelbaum doing a radio broadcast several years ago discussing the T3 issue in a way that I thought was pretty good.
I wonder if it was this one.
https://www.voiceamerica.com/episode/79759/thyroid-and-adrenal-problems-what-you-dont-know-may-be-hurting-you
I always feel tired and also don’t sleep without medication.
I have had ME/CFS since 1986. Even though my thyroid levels were in the “normal” range, I had a thyroidectomy in 2000 because biopsies of numerous thyroid nodules were inconclusive. They did not find cancer, but what they did find was that one side of the thyroid was “totally blackened with autoimmune disease” and the other side was “already partially blackened.” They were unable to identify the type of autoimmune disease. After trying numerous thyroid replacement therapies, including taking Cytomel T3 along with Synthroid, I found that Armour worked best for me until they changed the formula a number of years ago. I then switched to Naturethroid which worked well until this year when they apparently changed the formula somewhat. I’ve been taking NP Thyroid for a couple of months now and have found it seems to be working well.
Low thyroid function was a big issue for me. Eventually I learned about Wilson’s Temperature Syndrome – a condition that consists of hypothyroid symptoms and low body temperature, a condition brought on typically by stress and is reversible by taking for a period of time incremental doses of T3. This therapy really helped. I recommend others with CFS look into this at http://www.wilsonssyndrome.com. Addressing low thyroid function however was only a part of puzzle of my multifaceted condition, which ultimately was diagnosed as copper toxicity and pyroluria now being treated successfully with Nutritional Balancing.
“which ultimately was diagnosed as copper toxicity and pyroluria now being treated successfully with Nutritional Balancing.”
Interesting, both go hand in hand with increased oxidative stress. And I believe low body temperature and these thyroid symptoms might too.
Good to hear T3 supplementation worked for you. Hope you further improve with this new Nutritional Balancing protocol and would love to read on your experiences on the forum if your up too!
Kind regards, dejurgen
Omg… hhv6, t3, rt3, t4, too much, not enough, oxidative stress, glutathione balance, hashimoto thingy whatsit,,!!
I’m lying here in my dark room listening to my family get on with their lives with out me, struggling to read through my hideous brain fog just to pronounce the words let alone let put them into this theory or that context… you all are doing a wonderful job, but everything from what I see is a hypothesis, if you ask me it’s bloody voodoo. !
But seriously can anyone answer this please….
Can it really be this complicated? A friend of mine had his thyroid removed because of cancer, gone! Nothing there anymore! He takes 2 pills a day, gets up every morning and works his manual labour job, eats average food for lunch, gets home every night, grabs a cold beer, eats his wife’s average dinner, puts his kids on his knee, does thier homework with them, laughs at TV, kisses his kids good night, talks with his wife, off to bed (possibly a little nooky) has a great night sleep and wakes up and does it all again.. how is it that easy? When we all are discussing the multi faceted hormone function of the thyroid and he ain’t got one?
Good question. He’s clearly taking a thyroid supplement which works. I imagine it’s possible that other aspects of the thyroid pathway are not working in ME/CFS – whether it’s thyroid uptake, protein binding, thyroid metabolism, other HPA axis problems. It’s really quite complicated.
The pituitary tells the thyroid to make T4 but for the most part other parts of the body break down T4 to T3.
Since hypothyroidism is considered a co-morbid condition in ME/cFS/FM – which can usually be controlled – hypothyroidism is present in some people with ME/CFS but it is clearly more than that.
mostly replying because I liked your post ;o) got bugs removed somehow and all their former homes, brain/thyroid/adrenals, are trembling like crazy so if those were all affected then just replacing one of them wouldn’t make much of a difference or even just ‘upset’ the others more, just like what Cort says about the HPA-axis
My chronic fatigue followed Hashimoto’s. I then had 4 six month long episodes of viral thyroiditis over the next several years. I wasn’t clued in to the viral reactivation problem with chronic fatigue so at that point my thyroid was removed. I have tried various thyroid formulations without success.
Many ME/CFS patients may have undiagnosed liver disease. Please keep in mind that liver enzymes cannot rule out Liver disease.
“Therefore we can describe particular alterations of the thyroidal pattern typical of chronic liver diseases: low T3 syndrome, low T3 and T4 syndrome, high T4 syndrome, mixed forms. T3 and T4 diminish due to inefficient hepatic deiodination and defective hepatocellular uptake”
http://algogenomics.blogspot.com/2019/01/liver-bile-acids-and-thyroid.html
I’ve had problems with my liver for years and can always tell when the liver enzymes are elevated because I’m even more tired. Drugs thrown at me usually make me feel worse.
My first symptom that my liver is not working properly is usually anger and irritability. Taking Milk Thistle for two or three days seems to get me back to whatever my “normal” is with this illness!
HHV-6. Seriously? This sounds like Hilary Johnson’s ‘contagious’ theory all over again.
Newsflash: Almost everyone has HHV-6 and gets it as a young child.
ME/CFS isn’t infectious. If it were, a growing percentage of people would be sick as each year passes. That hasn’t happened.
Yes, Everyone gets HHV-6B but not HHV-6A which appears to more common in MS and ME/CFS and no one knows why. It could be present and then get reactivated when the immune system goes down I suppose.
Large doses of T3 and cortisol replacement therapy will improve any ME/CFS patient there is no argument . Dr Holtorf is helping people all over the world including my self. Standard endocronology has been leaving people ill for decades its a real shame. Things need to change the proof has been there for decades.
Most people with Lyme Disease also have Hashimoto’s Thyroiditis.
We also commonly have many coinfections like Hhsv6, EBV, parvovirus, Coxsackie, and other virus and bacterial infections.
I wish the study had looked at statistical relationships to Lyme, although that might be clouded by the fact that Lyme is often misdiagnosed as ME/CFS, MS, and other illnesses, or symptoms simply written off as all in our heads.
Another brand of iodine that seems to be non-toxic, at least to me, is Eidon.
Please keep in mind when taking iodine supplements that a goiter can be caused by either too little OR too much iodine. So it’s important to not overdose.
I was happily taking the same dose of iodine for years, but didn’t realize that as I got healthier it had become an overdose. My thyroid gland swelled up. I finally figured out what the problem was.
Standard Process makes a supplement called Thytrophin PMG which contains T1, T2 and T3 from cows. I take it in addition to Armour thyroid in order to create a good balance of T3 and T4.
Thanks for the interesting article, Cort. I am probably in this subgroup.
I have had CFS for 12 years. I went to the Holtorf clinic for several years. The T3 helped me for 6 months and then I relapsed. After this, nothing else worked. The clinic really pushes their overly priced supplements too. Overall, I don’t really trust their practice.
I have also gone to Stanford and tried several anti-virals at 3-6 month intervals. All they accomplished was making me feel worse.
The only thing that seems to help a little is Midayis but after several months of taking it, the medication seems to have lost a lot of it’s effectiveness as well.
Does anyone else have a pattern of responding at first to medication and then it stops working?
“Does anyone else have a pattern of responding at first to medication and then it stops working?”
Yes, it’s quite common. I’ve had it plenty of times. Far more medications did induce a change both when starting them and when quitting them, with not much difference while just taking them besides side effects. That’s far more then the number of medications that actually do work longtime.
I do believe this temporary improvement is often due to medications “overriding” a safety mechanism that work shortly by blocking the safety mechanism until the body installs a new one in order to protect the body long term.
To me, that is a strong indication that the symptom the medication tried to correct might be the effect of the body trying to solve another problem. For example: exhaustion does cause plenty of long term damage to the body. The body does decrease thyroid function in order to make us exhausting ourselves a lot more difficult.
We perceive it as a very bad symptom, which it is, but it may be there to prevent strong permanent damage to our bodies.
A T3 medication or a T4/T3 combination medication would only be called for if TSH levels were outside the normal range, right? My wife has ME/CFS and is on levothyroxine (50mg/day) for hypothyroidism. Her TSH earlier this year was 1.64, which is the normal range. So, my read of this article is that she is not likely to benefit from switching to a combination therapy. Do I have this right? Any advice would be welcome.
Which is the best anti-viral for HSV-1 ? I recently had an outbreak on my lip that seems to later have gone into the throat and maybe along the optic nerve tissue. I think it may have all started with a flare up in the stomach brought on by some antibiotics. oddly. I new I had the hsv-1 lurking around and had gone off antivirals for unrelated reason. then had right side stomach pain for 2 months and after a coffee it flare up like a volcano then the new outbreak i described above . I started taking elderberry syrup and that cleared up the throat and I think is helping the stomach too. I also have been taking acyclovir but seems to be doing nothing. the elderberry seems to be miraculous though. this has not been a fun experience.
There is an animal model that has shown neurological effects from HHV6-A.
“Marmosets inoculated intravenously with HHV-6A and HHV-6B showed low-level DNA detection in CNS tissues, while only those inoculated intravenously with HHV-6A exhibited neurologic symptoms (including sensory and motor impairment, facial palsy, and diminished sensation in extremities”
In addition to thyroid tissue, there is a uterine biopsy test for HHV-6 A.
Perhaps, looking for HHV-6A in tissues may turn out to be the preferred way to find active infection rather than blood tests.
However, in the early 1990’s, I was positive for active viremia with HHV-6 (not sure A or B). The test was at a special lab doing DNA amplification testing for Dr. Paul Cheney. I was treated with Kutapressin shots for more than a year.
From the very beginning, my main symptoms were neurological and immunological.
Over the intervening years, I have relied on Immunopro, a whey based immune booster mentioned on the HHV-6 Foundation website and Cell Signaling Factors to stay at about 70-80%. Recently, I have added a mushroom mix that seems to help with fatigue and also glutathione patches.
Interrupted sleep is still a problem and I use a tiny dose of clonazepam (l/4 of a .5 mg tablet at night and 200 mg. of gabapentin. It took me a year to reduce the clonazepam from .5 mg to a quarter of that amount.
I think I felt better on the .5 mg, but it is a drug that you can’t run out of and many doctors will no longer prescribe it. I am always concerned they are going to take it off the market.
Thanks http://upicodabri.gq
“Note that low total T4 was not found in the ME/CFS group.”
Nonsense! See page 5:
“Chronic fatigue syndrome patients exhibited lower FT3, TT4, TT3, %TT3, SPINA-GD, and SPINA-GT, lower ratios of TT3/ TT4, FT3/FT4, TT3/FT3, and TT4/FT4; and higher %rT3 and rT3/TT3 ratio. There were no between-group differences in other thyroid hormone parameters, notably TSH, FT4, rT3, and %TT4 (Table 1). FT3 below the reference range was more frequently found in CFS patients (16/98) as compared to controls (7/99; p = 0.035) with an odds ratio of 2.56 (95% CI = 1.00–6.54).”
Also see table 1 and figure 2: Total T4 (TT4) was on average 12% lower in CFS patients compared to healthy controls.
This is an excellent useful article, Gratitude for sharing the extraordinary enlightening post and continue to share the article.
https://www.mid-day.com/brand-media/article/synogut-reviews-is-this-advanced-gut-health-formula-safe-customer-revealed-23262111
Oxidative pressure also. Exceptionally fascinating! Such countless possible pathways…
https://www.mid-day.com/brand-media/article/sharpear-reviews-scam-or-legit-safe-ingredients-or-risky-hearing-supplement-23284875
Thanks! I look forward to trying it
Thanks For Sharing this information to The user.
I have tried this and this is the best thing that worked for me and went along even better with this blog post I have checked out!
Thanks! I look forward to trying it
I have tried this and this is the best thing that worked for me and went along even better with this blog post I have checked out!
where can a person, with disabling me/cfs on medi-medi insurance get meaningful care in Los Angeles? or anywhere in California or Nevada?
I go to ucla and cedars but I can go to any md who takes my insurance.
Patients were identified based on Fukuda criteria, and therefore did not all have PEM. Therefore, these thyroid results are useless for ME/CFS.
I’m sorry but that is not a correct conclusion: just because Fukuda does not require PEM doesn’t mean that many or even most of the people did not experience it. They probably did.
Respectfully Cort, I disagree. We don’t know how many had PEM and how many didn’t, and what the thyroid values were for pwME. We are rightly critical of CBT/GET studies using Fukuda criteria, and we should be equally critical of biomedical studies using them.