

Anyone who’s been around the scientific literature on chronic fatigue syndrome (ME/CFS) knows that dysautonomia – a dysregulation of the autonomic nervous system – is common. Two different kinds of orthostatic intolerance – postural orthostatic tachycardia syndrome (POTS) and neurally mediated hypotension (NMH) have been found. Virtually everyone with ME/CFS has reduced blood flows to the brain, and low heart rate variability is ubiquitous as well.
If long COVID is anything like ME/CFS, then dysautonomia is going to show up in the medical literature – and it’s starting to. Recent long-COVID studies are starting to validate the dysautonomia link, and as they do that, they’re validating the connection between long COVID and ME/CFS. Because they’re coming from around the world, they also open the possibility – so long as ME/CFS and long COVID are firmly linked – of creating a new global understanding of ME/CFS.
Small Fiber Neuropathy
A recent Indian/French study is a good example. I can’t remember the last Indian or French study on ME/CFS, but this long COVID study found evidence of small fiber neuropathy (SFN) in about 25% of long-Covid patients. Because the study measured just one possible facet of SFN (sudomotor dysfunction), it’s possible that even more people had SFN. It stated, interestingly, that infections such as mumps, HIV, Hepatitis C, Epstein-Barr, and Coxsackie B often produce dysautonomia (who knew?). Interestingly, people who had been treated at home instead of the hospital (i.e. had a less severe illness) were more likely to show evidence of SFN.
The only real disappointment from the study was that it neglected to mention fibromyalgia (!).
Last month, in “Autonomic dysfunction in ‘long COVID’: rationale, physiology and management strategies“, Hammersmith and Imperial College researchers in London raised the specter of widespread dysautonomia – a subject one suspects many doctors have little knowledge of.
ME/CFS was not mentioned but postural orthostatic tachycardia syndrome (POTS), of course, were. The authors reviewed symptoms, tests and drug and other treatments that might be helpful and told doctors to be on the lookout.
“We suggest that all physicians should be equipped to recognize such cases, appreciate the symptom burden and provide supportive management.”
They also posited a cause that was right up POTS and ME/CFS’s alley – an autoimmune-induced hurt to the autonomic nervous system. This idea will hopefully be fully explored in the long-COVID studies.
“We posit that this condition may be related to a virus- or immune-mediated disruption of the autonomic nervous system resulting in orthostatic intolerance syndromes. ….we speculate that there is an underlying autoimmune component to the post-COVID syndromes that we report.”
Next, in a letter to the editor, “COVID-19, fatigue, and dysautonomia“, a Singapore researcher reported increased fatigue was found in 50% of recovered COVID-19 patients after 6 months – a fatigue that he believed was likely due to autonomic dysfunction and problems with the neuro-cardiac axis.
Brainstem
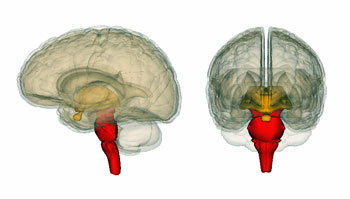
Nath found small blood vessel damage in the brainstems of long COVID patients.
But why are these autonomic nervous system problems occurring? One answer might lie in the brainstem. The brainstem is the first recipient of all the sensory and “motor” signals from the spinal cord. It regulates very basic functions – like breathing, heart rate, blood pressure, digestion, alertness, sleep/wake – a lot of which has gone wrong in chronic fatigue syndrome (ME/CFS).
In “The Vagal Autonomic Pathway of COVID-19 at the Crossroad of Alzheimer’s Disease and Aging: A Review of Knowledge“, French researchers report that the SARS-CoV-2 coronavirus has a predilection for nestling itself within the vagus nerve. They propose that a failure of the vagus nerve to institute an anti-inflammatory response may lay behind the oft-noted cytokine storm that can accompany COVID-19. (Could an inhibited ant-inflammatory response also explain why people with more severe symptoms are more likely to come down with ME/CFS in the wake of an infection?)
From there, it moves into the dorsal vagal center of the brainstem – an “integrative center” which regulates both respiration and inflammation. Mice experiments indicate that past SARS viruses have a propensity to attach to the brainstem. Plus, the brainstem has high levels of the ACE2 receptor that the virus binds to.
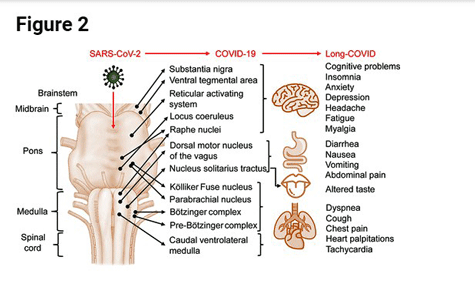
Possible implications of brainstem damage – from Yong (https://pubs.acs.org/doi/pdf/10.1021/acschemneuro.0c00793)
When the virus gets to this part of the brainstem, they believe it begins affecting the “autonomic nervous centers” that regulate breathing and other cardiovascular factors. A recent paper, “Computing the Effects of SARS-CoV-2 on Respiration Regulatory Mechanisms in COVID-19“, stated that “overwhelming evidence” linked acute respiratory failure in COVID-19 to viral entry into the brainstem. The virus produces a “neurogenic switch” which causes, among other things, hypoventilation – which has been found in abundance in ME/CFS.
Another potential consequence involves increased sympathetic (“fight or flight”) and decreased parasympathetic (“rest and digest”) tone – exactly what is found in ME/CFS and fibromyalgia (FM).
The brainstem popped up again this month in “Persistent Brainstem Dysfunction in Long-COVID: A Hypothesis“, by Shin Jie Yong, a Malaysian researcher. Yong noted that most explanations for long COVID involve some sort of tissue damage, persistence of the virus, and chronic, unresolved inflammation, but none of those, he thinks are correct. The problem, Yong proposes, is “persistent, low-grade brainstem dysfunction” driven by a virus which loves to inhabit the brainstem.
Autopsy results, Yong reports, have found evidence of inflammation and neurodegeneration in the brainstem area. SARS-CoV-2 genes and proteins were detected in 50% and 40% of the brainstem samples in other studies. Still others have found evidence of microglial activation (neuroinflammation) and small blood vessel bleeds.
Avindra Nath found numerous small blood vessel bleeds in the brainstems of the COVID patients he autopsied. Interestingly, most apparently initially had a mild illness presentation.
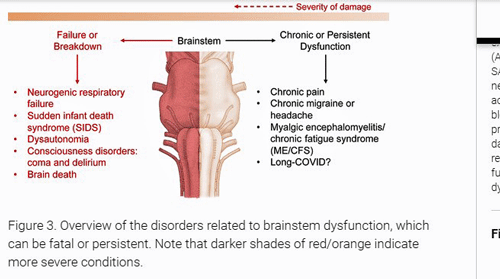
Diseases of long-term brainstem damage. (Yong-https://pubs.acs.org/doi/10.1021/acschemneuro.0c00793)
Since the brainstem is an important source of serotonin, norepinephrine and dopamine producing neurons, it could produce a vast array of symptoms including fatigue, pain, headache, depression, anxiety, sleep and cognitive impairments.
Yong believes damage to the brainstem could also be responsible for the gastrointestinal symptoms (vomiting, diarrhea, and abdominal pain) seen in long COVID. The ventral tegmental area and substantia nigra also reside in the brainstem’s midbrain, which supplies dopaminergic neurons to the higher brain regions.
Then comes the clincher. After noting that brainstem damage has been found in migraine, Yong writes:
“Notably, long-COVID resembles and is closely associated with myalgic encephalomyelitis or chronic fatigue syndrome (ME/CFS)… Interestingly, brain imaging research has found that symptom severity of ME/CFS associates and correlates with brainstem dysfunction, particularly at the reticular activating system (RAS).”
As Yong noted, there are papers by Barnden in Australia that have been painstakingly showing that the brainstem is affected in ME/CFS. Jeff and Jen Brea’s craniocervical stories demonstrate that a damaged brainstem can produce all the symptoms of ME/CFS. The French researchers believe this organ is at particularly high risk in a coronavirus infection.
ACE2
ACE2 presents another intriguing possible connection between ME/CFS and the coronavirus. As was noted, the virus binds to the ACE2 receptor, and the ACE2 protein is densely found in the brainstem (and the amygdala). The first thing that happens once the virus gets into the dorsal vagal nerve is that ACE2 levels drop, Ang II levels rise, the anti-inflammatory component of the renin-aldosterone-angiotensin (RAS) Ang (1-7), declines, and problems with baroreflex sensitivity occur.
Compare that to what appears to be happening in ME/CFS. The ACE2 inhibition found in ME/CFS also appears to result in high levels of Ang II and low levels of the anti-inflammatory Ang (1-7). The high Ang II levels in ME/CFS could be contributing to a host of problems including a ramped-up fight/flight system, narrowed blood vessels, inflammation, heart rate and blood pressure issues, and possibly reduced blood flows to the brain.
Hypoventilation and ventilation problems during exertion, in particular, and shortness of breath have been found in ME/CFS. Low CO2 levels may be common. Time will tell if the many respiratory problems found in COVID-19 are cousins to those found in ME/CFS. Given what we know so far, it would be very interesting to see what happens when people with long COVID are put on an exercise bike. Avindra Nath reportedly is going to do just that.
Conclusions
It’s nice to see so many possible fruitful connections pop up between chronic fatigue syndrome and long COVID. It’s good, as well, to see long-COVID researchers heading to the autonomic nervous system, the brainstem, and the renin-angiotensin-aldosterone – three areas one suspects not many doctors and researchers are all that aware of.
It would also make sense that the brainstem might have been missed in ME/CFS and other diseases. VanElzakker has pointed out that most brain imaging studies don’t produce a clear picture of the bottom of the brain. With the massive funding long COVID is about to get, though, hopefully, the brainstem and the autonomic nervous system will get their shot. It would be shocking if the potential autoimmunity issue wasn’t fully addressed as well. The presence of small fiber neuropathy adds another intriguing factor.
Are the stars lining up? Time will tell. Note that if these findings are validated in long-COVID – they all showed up first in ME/CFS and fibromyalgia.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



I had long COVID for almost 5 months. I got rid of it after trying a tretment for Lyme called Ozone 10 Pass IV. After a few sessions my symptoms, trouble breathing and chills went away. I still have CFS that I have since childhood. I would like to know what other people have tried.
I’ve been taking herbal treatments which increase blood flow to the brain, reduce the histamine response and help calm the fight/flight response. I drink nourishing herbal infusions to increase resilience. Also Stanley Rosenberg’s basic exercises to stimulate the dorsal branch of the vagus nerve have helped too.
can you share the herbal treatments?
Hi Laura, can you share what herbal treatments you have been taking to increase brain flow?
What are the herbs and exercises please?
The brainstem theory is very interesting!
And –
“… so long as ME/CFS and long COVID are firmly linked …”
Yes, so true – the connection is so very obvious. I hope the long covid & ME studies, particularly Avindra Nath’s, will make the case, irrefutably.
That’s what I’m looking for. Something irrefutable…Maybe I’m being too optimistic but I don’t see why that wouldn’t happen…
Your optimism is encouraging though!! Brain stem involvement makes so much sense and I hope the researchers continue to pursue this with vigour…
When is Nath’s CFS study coming out?
I’m wondering what this means in terms of a cure. Anything aside from surgery?
Some researchers think autoimmunity is involved. If so there might be drugs that could help. It could also be a problem of blood flows to the brainstem or blood vessel bleeds or inflammation…Lots of ways to impact the brainstem! .lf this turns out to be real I imagine there will be quite a few options other than surgery
There’s also GHK peptides that may address the structural/mechanical aspect without requiring surgery. The first, and pretty much only, time I heard of this was last September when Jen Brea shared Mallory Tomaro’s full recovery story:
https://medium.com/@mecfsmedicalrecovery/me-cfs-spinal-instability-tethered-cord-recovery-protocol-3e1eefffac8c
Peptides therapy in general is interesting. Some functional MDs treating LongCOVID report success using Thymosins for immune regulation, BPC-157 to speed tissue healing, and Mots-C to boost mitochondrial function.
Thanks Cort for all your hard work (as usual)!
Thanks for providing the link to his story. I had not heard of it. 🙂 I haven’t read it yet but it’s so interesting how the brainstem shows up in both CCI and ME/CFS…
Thank you so much for collecting and posting this survey. I have been on the fence whether it is safe for me to get the vaccine. I’m concerned it is going to flair up my symptoms. And the survey was exactly what I was trying to find, people with my disease and how they reacted to the vaccine thus far.
🙂
Pursue with vigor…and find a treatment.
Thanks for these updates. Even after 17 years there is still a little voice in my head that says it’s all in my mind. This helps me to know what I’ve always known that something is definitly wrong physically.
Yes….It’s amazing how we doubt ourselves – even after time and time again busting our butts to work or exercise or get better.I think that is just everyone’s default when we don’t know: it must be me!
That’s exactly how I’ve always felt. It’s been 24 years for me off and on. I get some good years and then i get bad months or years. It always seems to hit me after a virus and hang around a long time. Then it goes semi dormant and is mild for some time
Aripiprazole for ME/CFS?
https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-021-02721-9
I was probably represented in this study–and was one of the ones that got worse. It was so bad that I was forgetting to take home my groceries after purchasing them or being ‘spaced out’ much of the time–and so on. I eventually gave up, so IMO, not a panacea.
Also the response ‘record keeping’ was rather rushed and not so carefully recorded. Questions about improvement usually happened at the end of the appointment and the responses, I felt, were hurried along. Far better to have people make daily records–which was not done.
Also after several months of Apipirazole ‘failure’ I was told by Dr. Bonilla that I just needed to take it longer. He spoke of ‘someone’ who finally improved after taking it for close to a year. Sounds hinky to me…
That’s not good. The majority seemed to get good benefits though.
It’s Stanford so I would have thought it should be credible.
Would be interested in Cort’s view, given it looks at face value to be very promising.
Hi Cort, This is a very interesting blog. From the beginning i was sick i was thinking about the brainstem. I always said that the parasympathetic nervous system (PSNS) is broken. I know you’re also be a fan of the autonomic nervous dysfuncion. I don’t think the brainstem is damaged but attacked by ……….? I still think that our disease is reversible or treatable. Thanks Cort for your always optimistic view on things.
From what I read could be inflammation, autoantibodies or could even be clogged blood vessels that are not getting the blood through. I imagine a autoimmune attack would be a way to produce the ligament problems found in craniocervical instability
Hi Cort,
I am a Covid Long-hauler. I believe that I most likely have ME, but have not been diagnosed. I very much hope the association between ME and Long-haul Covid continues to grow. You may know this, but I wanted to let you know that the Mount Sinai COVID center in NYC has focused on Dysautonomia for many months. Unfortunately, they were making recommendations that were not ideal for many of us. (I did not visit them, but I saw their recommendations groups). The heart rates they said were okay were too high, and they recommended physical activity as a treatment :(. They recommended fixing sleep to fix PEM and not laying down during the day. The recommendations were helpful for POTS patients without PEM, I guess, but not for those of us with ME symptoms, which months later, looks to be the majority of us who got last spring and are still sick now. Anyway, in the beginning, they considered MCAS, ME/CFS, and Dysautonomia. They picked Dysautonomia. Not sure what they are saying now. Thanks!
Thanks for the update Jennifer! It sounds like they’re going through a learning process. I noticed that POTS has in some places gotten more attention that ME/CFS with regard to long COVID which astonished me. I should try and get in touch with them.
POTS is easier and more objectively to diagnose. It’s hard to argue with a tilt table test. With ME tests things get either more challenging to do the test or more controversial. The “barrier to entry” for “non ME doctors and even former non-believers” is lower I’d say.
Hi Cort, I think initially POTS was getting more attention. Part of it, I think, was that a lot of early COVID people developed those symptoms at a certain month mark. It is a learning process but it looks like they are really learning. I came back because I just saw this.
https://www.autoimmuneinstitute.org/understanding-long-covid/?fbclid=IwAR3-cEaDwXJgMysCyFewv599qVwQd0mj6rf7x6HA9CEVoGmIHH4aFUggAjo
Thanks Jennifer for the link. I signed up 🙂
According to the author of “The Post Polio Paradox”, brainstem damage is also thought to lie at the center of post polio syndrome. (To tie the conversation all the way back to some of the oldest roots of ME.)
And autonomic nervous system imbalance and/or dysfunction is increasingly thought to lie at the center of trauma and chronic stress. (But through mechanisms quite different than the kind of organic damage caused by viruses, autoimmunities, etc.)
(…and POTS…)
One can see the possibility for all of the confusion around the “mind” or “body” nature of such symptoms right there, in that overlap.
(Though trauma, too, is increasingly thought not to lie in the mind…but mostly the body. But still, wreaking it’s havoc through very different mechanisms than things like viral damage and autoimmunity.)
I hope this new long hauler research will lay a strong foundation for recognizing the specific routes of such damage invoked by illness, separate than those of stress and trauma. Maybe then there could – finally – be a clear conversation and distinction about the overlaps – and differences – between illnesses like ME/CFS and POTS, and the symptoms/pathologies of trauma and stress. I suspect that core confusion is what has waylaid the entire conversation/research/treatment around ME/CFS for so long…always…slipping…over into the realm of psychology…and away from physiological (over psychological and behavioral) research.
Just a big thank you for that comment, MarthaLauren.
And to Cort, again.
Talk about coming full circle Martha! I had no idea. The brainstem certainly could have been more easily missed in ME/CFS because most MRI’s can’t see it clearly. From what I’ve read the ANS could be at the center of so much. Hopefully -the long haulers will have an easier time than we have had. I would be shocked if they won’t – particularly with this explosion of funding coming. That brings up the NIH – the money was allocated to them almost two months ago – and we’re still waiting to hear how they will spend it.
Yes, I feel sure that the brainstem is key! Lots of money available for research will not help, however, unless it is channelled into useful studies that are well informed by the people most knowledgable in the field. In this case, those who have been working on the brainstem and its potential involvement ought to be consulted and employed. Big money has often just gone to the same old people/places and the same old thinking that results in nothing for us. Remember the decades of foolish research based on the wrong notion that ME/CFS and FM were psychiatric. Let’s advocate for good brain research, especially focussing on the brainstem.
I just watched a very interesting video by a society who posts Covid treatment reviews and one of the drugs they are recommending is Ivermectin–which is usually used to treat internal parasites. It has been shown to be efficacious for treating active Covid infections.
https://www.youtube.com/watch?v=xy85K1hENn4
Now at 20.07 minutes into this presentation, they mention a small study of using Ivermectin on Covid long haulers with very positive results. Could this be a clue to help us with ME/CFS?
My wife and I were both prescribed Ivermectin in December 2020 three days after testing positive for Covid. Neither of us noticed any difference afterwards. My wife recovered but I am nearing my 4th month as a long hauler.
I’ve researched IVM ivermectin as treatment of Long Haul. Its higher doses. 0.6 mg/kg x3-5 days with other supplements. I’m on day 3. Sometimes better but this moment I’m not great. Dizzy, low blood pressure, ocular aura migraine that’s got me down, plus brain fog and continued diarrhea, fatigue, joint pain, general malaise.
https://covid19criticalcare.com/wp-content/uploads/2021/06/FLCCC-Alliance-I-RECOVER-Management-Protocol-for-Long-Haul-COVID-19-Syndrome.pdf
Lyme disease also causes dysautonomia, just to add to this of diseases implicated.
Yes there is a link in ME/CFS with the Renin- Angiotensin- Aldosterone system . Also Renin is high in Addison disease which is very low cortisol levels. With low levels of cortisol one has major problems with inflammation, also you would have problems with blood pressure as cortisol helps maintain blood pressure. The Renin- Angiotensin- Aldosterone system is then affected with a release of Renin from the kidneys which in turn acts on Angiotensin from the liver to form Angiotensin 1 then ACE (Angiotensin converting enzyme) is released from the lungs to act on Angiotensin 1 to form Angiotensin 11 which then acts on the adrenals to stimulate release of Aldosterone. Angiotensin 11 also acts directly on blood vessels stimulating vasoconstrition. As they say cortisol has possibly hundreds of affects on the human body. With continuing inflammation from viral infection the levels of cortisol would be severely compromised. This would be the reason people have a good response to low dose 10mg hydrocortisone. I also took the same for one month with good results and during that time noticed my blood pressure return to normal levels. Unfortunately this did not continue as after stopping for a short time and recommencing did not get the same effect. I actually think the viral load increased. There is a Renin inhibitor called Aliskiren would like to know if anyone has tried.
I have tried Ivermectin which was a three day course of 12mg /day (3mg×4). Unfortunately I took Doxycycline with it and although I thought I had some improvement was unable to know was it the Ivermectin or Doxycycline. I am not sure on side effects if was taken for a longer time as it’s use for parasitic infections is only for a limited time. Does anyone know more about this treatment for viral infection.
https://covid19criticalcare.com/wp-content/uploads/2020/11/FLCCC-Ivermectin-in-the-prophylaxis-and-treatment-of-COVID-19.pdf
I posted this on another blog, but it may get missed and I think people need to see it.
An EXCELLANT blog was just posted by Debbie Moon on her Geneticlifehacks blog site. Here is the link:
https://www.geneticlifehacks.com/genetics-chronic-fatigue-syndrome-and-long-haul-viruses/
There are genes that can be checked that can indicate if possibly some of us are genetically predisposed to ME/CFS. (I’m wonky on many of the genes listed here ) She also addresses how virus can affect gene expression and cause us to have epigenetic changes. Also has some more information, about what I posted, about EBV and it using a similar substance like IL10 to do a sort of “fake out” that it is there to evade the immune system.
Very well done Debbie. Thanks for allowing me to link it here! And thanks for writing it and helping us draw more awareness to ME/CFS and other connected illnesses like POTS.
Hi Issie, I had a look at Debbie Moon’s blog and I found it very interesting. She mentions NLRP3:
‘Another innate immune system component, NLRP3, is also implemented in fatigue syndromes including ME/CFS and MS.[ref][ref]
NLRP3 is a lynchpin at the start of the immune cascade.’
I’d never heard of NLRP3 until last year there was an announcement in the media here in Ireland:
‘Biotech firm Inflazome bought by Swiss giant Roche for €380m’
‘Headquartered in Dublin, Inflazome was founded in 2016 by Professor Luke O’Neill from Trinity College in Dublin and by Professor Matt Cooper from the University of Queensland in Australia.
The company develops orally available drugs to address clinical needs in inflammatory diseases by targeting inflammasomes.
Inflammasomes are understood to drive many chronic inflammatory conditions, from Parkinson’s and Alzheimer’s to asthma, inflammatory bowel disease, chronic kidney disease, cardiovascular disease and arthritis.
Today’s deal gives Roche full rights to Inflazome’s entire portfolio which is composed of clinical and preclinical orally available small molecule NLRP3 inhibitors.’
https://www.rte.ie/news/business/2020/0921/1166446-inflazome-roche-deal/#:~:text=Irish%20biotech%20company%20Inflazome%20has,upfront%20payment%20of%20%E2%82%AC380m.
Interesting Tracey Ann.
I appreciated that Debbie has found connections to genetics that could indicate some issues that may be connected to us with ME/CFS. My genes show issues on many of these markers.
She also talked about TRMP3 and there is a study from Australia, called the Griffith study. It connects issues with calcium channels. We talked about this back in 2017 on Health Rising Forum. I was wonky on those markers too.
So there are some things we can look into in order to see if some of these possibly apply to us and we are predisposed to issues. That can give us more to look into in order to find our own WHYs.
I have been a member of the Genetic Lifehacks group for just about a year and find it very interesting and helpful. I also thought her article on viruses and ME/CFS very good. Like Issie I also had many of the SNPs putting one at a higher chance of developing ME/CFS especially the SNP involving the calcium channel TRMP3. However the NLRP3 one was normal in my case.
When she did an article on Autoimmunity I had many SNPs that are involved predisposing one to this problem especially Celiac so I have avoided wheat for several years now.
Pam
Hey Cort…Wow, that sounds like they’re on the right track!…Could this mean that any virus or maybe even a concussion in the past could cause the brain stem to disfunction and/or disrupt to some degree as to cause our ailments?…That would definitely narrow the band of trigger variables in most of us and give us some hope for finding a way to managing/possible curing FM/CFS(ME)!….
I found it interesting that when I spoke on the phone with InHouse Pharmacy when I requested they carry Ivabradine (Ivabeat) for POTS, which they will, stock arrives mid March, the subject of Ivermectin came up. He said that a great many of their customers ordered and have used Ivermectin for COVID. That was surprising as I was familiar it was an anti-parasitic.
This woman’s description of her long hauler symptoms seems very much a dysfunction of her autonomic serious system like so many ME patients know a lot about! Hopefully the research now being done will help to find answers for all of us!
https://www.kcra.com/article/covid-19-long-hauler-treated-in-sacramento-uc-davis-health/35192445#
Yes!
Yesterday in the news WHO warns for long Covid https://news.un.org/en/story/2021/02/1085772 :
“With one in 10 people still feeling ill 12 weeks after having COVID-19, authorities must do more to support them, a UN-backed policy brief issued on Thursday argues.”
With 28.4 million offical cases in the US so far, that is 2.84 million estimated long Covid cases so far (already and “in the making” by those infected now) in the US https://edition.cnn.com/interactive/2020/health/coronavirus-us-maps-and-cases/
Hopefully a large part of them will still recover over a longer time, but many wont.
Long Covid will be a lot harder for the medical establishment to deny then ME it seems. Also, these numbers make clear why social distancing measures and effective vaccination campaigns will be vital to the health of large populations: such very high rates of long Covid would be a huge burden on any country if the virus was let free to reach herd immunity by itself.
In addition, remember these high long Covid rates are reported after one infection per individual. With natural immunity seeming to be time limited in a large part of the population and the spread of new variants like the Brazillian one who infects large numbers of people who already had the orgiginal variant, that long Covid rate might be per each time an individual is infected…
Yes, I saw that from the WHO. In Ireland I’m hearing Long Covid/Long Haulers mentioned by some healthcare professionals on the radio. They usually bring it up in relation to the opening up of various parts of society – which is being presented as ‘safe’ to do – as in even if individuals are not severely affected by a Covid infection, they may suffer with Long Covid.
What can we do to heal brain stem if this is the cause?
Probably I would think by healing what caused the brainstem problems in the first place – reducing inflammation, stopping an autoimmune process, increasing blood flows or ????….
I have had ME since 1997, acquired following a severe viral respiratory infection. Autonomic dysfunction has been a key element and the brainstem is certainly implicated due to the key roles of dopamine and epinephrine.
the dysautonomia was largely controlled by a combination of Adderall and clonazepam. I also had chronic bad fibromyalgia pain and disabling PEM for at least a couple of days if I overexerted. Over time I had increasingly bad and frequent migraine headaches. A close friend, who was also a pediatric gastroenterologist suggested that the muscular problems seemed similar to a genetic problem that was inborn in some infants affecting the ability to digest Carnitine. In Infants this was treated by supplementation with Levocarnitine. A blood test verified I had this problem and I was prescribed Levocarnitine ~ 9MG per day. Amazingly, the migraines disappeared within a week of starting the Levocarnitine. I regard this as a genetic or “epigenetic” problem caused by the viral infection and unrelated to the brainstem.
In 2008 it was discovered that I had kidney cancer. I was lucky (blood in urine, unrelated CAT scan). I had endoscopic surgery in 2009 and the cancer was removed even spared the kidney. However the anesthesia was very intense and afterwards I had to increase my Adderall to 90MG per day. Back to the brainstem. I think that surgical anesthesia is just like a concussion and can do real damage.
I could go on but what I am getting at is that the brainstem is an important focus for research there are other significant problems such as with mitochondrial function that should not be overlooked.
That’s very interesting re the carnitine. I do seem to need to eat meat now, which I never did before. I used to live on muesli, apples, wholemeal bread, salad etc.
Thanks Gregory. I had never heard of migraines being treated by levocarnitine. That’s amazing.
I have often wondered about surgical anesthesia too. I’ve had a number of surgeries with the major ones being neck surgery during which three vertebrae were fused and, more recently, a shoulder replacement. I can only imagine the amount of anesthesia that would be needed to keep someone unconscious during these and other surgeries. I have read occasionally that ME/CFS may occur after surgery but nothing detailed or specific.
Interesting… I agree it would certainly be unwise at this point for a researcher to overlook this part of the body as being of possible relevance.
In the first year after onset, when I abruptly transitioned from mild to severe ( thanks ‘exercise therapy’ advice ) completely losing my ability to walk long distances coincided exactly with the first experience of an intense pain at the back of the neck
that had me in bed for days.
I had three vertebrae (c5, c6, and c7) fused in 2006. I have never been the same and have had a slow decline to the severe ME/CFS that I have now. I realize that these vertebrae are not in the area of the brain stem, but I’ve often wondered if that surgery was the beginning of my illness. Does that sound at all plausible?
Very interesting …!
And … v commonly prescribed #Psychmeds ACT v crudely ON the brainstem ….
Could they be ‘messing up’ functioning & homeostasis.. thereby contributing to dysautonomia?
Mark Solms recent talk? possible clues? (esp bit near middle of talk … 52.52 ..)
https://www.youtube.com/watch?v=W8ZSMK-XVy4
Interesting article. Most of the research has been focused on POTS and dysautonomia but I am wondering if you have a history of CFS and fibromyalgia and suffered from long hauler symptoms of post covid, whether there is a stronger correlation of auto immune dysfunction than auto nervous dysfunction as the cause for long covid symptoms? Wondering if any doctors or researchers in NJ or NY are studying ME/CFS as the cause for long covid?
https://academic.oup.com/qjmed/article/106/9/865/1530481 – Snakebite, dysautonomia and central nervous system signs Excerpt: In a study of common krait bites, 139 of 210 victims (66%) exhibited autonomic dysfunction (AD), which was more marked in those with severe envenomation. I would check into Bryan Ardis and the solutions he provides for this.