Trouble…

Patterson’s promotions have garnered him a lot of clients (@25,000) – and some pushback.
Bruce Patterson MD rolled into a most interesting Solve M.E. event like he didn’t have a care in the world. Patterson has aggressively pushed his diagnostic tests and treatment regimen in the media – including publishing the same opinion piece in the San Francisco Chronicle, the Des Moines Register, and the Tampa Bay Times, and being interviewed on many social media sites.
His purported successes quickly got around. In “Lab discovers root cause of confusion, fatigue experienced by Covid ‘long haulers,” an NBC affiliate last year basically reported that he’d solved long COVID!
It’s been too much for some critics who prefer that Patterson take the time-honored (and time-intensive) approach of publishing first and promoting later. Patterson, of course, has published about the diagnostic test, but the studies are relatively small and need to be replicated in larger cohorts. Crucially, no studies have assessed the most important part of his package – treatment effectiveness. Even so, Patterson with his Stanford research background and his good communication skills has captured the public’s attention.
His promotional style might have gotten Patterson, a former Stanford researcher, bounced out on his ear if he was still in academia, but he’s in industry now and apparently hasn’t crossed any legal red lines.
For his part, Patterson replies, “We’re in a pandemic. I don’t know that these patients can wait 18 months for the results of a trial.” One could make the argument that if he really has the goods he has the moral obligation to get them out to the public and reduce the suffering as quickly as he can.
There’s always, of course, been a to and fro between doctors who push the boundaries of the medical profession and those more comfortable remaining well within them. We’ve seen that story played out many times at different levels in underserved diseases like ME/CFS, fibromyalgia, and now long COVID.
With the medical establishment unwilling to provide the funding needed to understand these diseases and assess treatments, patients have to choose between relegating themselves to few treatment options and generally poor health, and taking a chance with treatments that haven’t received much study.
For their part, doctors who use treatment approaches that haven’t been validated – and couldn’t have been validated through no fault of their own – can run risks. One of my MDs whose office was raided and his practice temporarily shut down felt that that kind of stuff was the cost of helping his patients. (It wasn’t the first time it had happened and he quickly reopened).
So here’s Patterson promoting a treatment that he says his data indicates is working but for which he has provided no hard evidence. He may have the goods but, Stanford background or not, it was inevitable he was going to starting taking some hits for moving forward so quickly, and he has.
In “Long-haul Covid patients desperate for answers turn to a private company,” NBC questioned whether vulnerable patients were taken advantage of, and featured a former patient whose test results returned to normal under his treatment – but she didn’t. Another doctor stated that marketing the diagnostic test/treatment protocol before both had been assessed in a wide variety of patients constituted a red flag.
That was nothing, though, compared to the takedown Mother Jones did in January of this year in their “Big Feature”, “Desperate Patients Are Shelling Out Thousands for a Long COVID Cure. Is it for Real?“.

Mother Jones focused a “Big Feature” piece on Patterson’s long-COVID work.
The Gist
- Bruce Patterson MD seemed to stride into a recent Solve ME event with the wind at his back. Proclaiming that over 80% of the over 24,000 long COVID patients on his protocol had achieved “profound improvements”, all seemed good.
- Patterson, though, has taken some hits from researchers and medical ethicists who’ve questioned if he’s gotten too far over the tips of his skis and is promising more than his data can deliver.
- A Mother Jones feature piece ” “Desperate Patients Are Shelling Out Thousands for a Long COVID Cure. Is it for Real?” noted that in the past year or so Patterson has been reined in by the FDA for overstating the results of one study, and was taken to task by a judge for exhibiting either “reckless indifference or deliberate gamesmanship” in a court case.
- Mother Jones brought in an outside statistician who skewered the findings of Pattersons’ core long COVID paper stating that it lacked some fundamental statistics, was too good to be true, and was too small a study to draw conclusions from. A prominent figure in the long COVID opined that he was one of the “sketchiest” figures around.
- Mother Jones, however, appeared to have a clear mission – to portray Patterson in a negative light. No patients who had done well with his protocols were featured and its characterization of a patient group as ‘cultish”, and its characterization of a doctor’s bedside manner was cheap and unnecessary.
- For his part, Patterson argues that given all the suffering produced by long COVID he’s morally bound to move forward to relieve it. He might say that he built his hypothesis around the data he gathered and then used it to successfully treat many of his patients. Whatever else anyone says, he must be right – and is going to prove it.
- The proof of his pudding will come with the placebo-controlled long COVID trial slated to start in a couple of months.
- Meanwhile, Patterson announced that he’s validated his biological signature in long COVID using another approach, that he expects his diagnostic kit for long COVID to be approved soon, and that ME/CFS, post-treatment Lyme disease, and other diagnostic kits are coming.
- Patterson’s preprint classification paper was short, lacking in description, and rudimentary. It attempted to use his long COVID cytokine grouping to differentiate ME/CFS. The long COVID cytokine package was derived, though, from an analysis of a wide variety of cytokines in long COVID (not ME/CFS).
- The paper did suggest that Patterson got his long COVID cytokine signature correct but its mediocre results regarding ME/CFS and other conditions suggested he has a ways to go before he can confidently state he’s found the same for these other conditions.
- Still, the paper was the first to attempt to do a difficult thing – differentiate long COVID, ME/CFS, post-treatment Lyme disease, and post coronavirus vaccine syndrome from each other immunologically. As such it could be viewed as a much-needed first step to attempt to differentiate these post-infectious diseases.
- So who is the real Bruce Patterson? Is he the former Stanford researcher who glommed onto long COVID early, quickly deciphered its secrets, and is hell-bent to do whatever it takes to help sick people? Is it just a matter of time before he gets all his ducks in a row and sets the long COVID, ME/CFS, and other diseases’ world afire? Or will the whole thing fall apart at some point?
- The rubber will meet the road with the clinical trial Patterson promised. A big, robust, and well-managed clinical trial will tell us if Patterson got too confident at some point or if he was right all along.
Mother Jones also reported that a judge concluded, in a suit that Patterson lost, that Patterson’s failure to disclose pertinent facts in a takeover bid for the company that produced leonlimab “reflected either reckless indifference or deliberate gamesmanship.” Mother Jones reported that Patterson then began disparaging the same drug he’d gotten in trouble for promoting on social media.
Following a long-COVID patient named Owen, the Mother Jones piece described a potentially costly treatment regime featuring tests and consults running about $600 and treatments up to $1,000 a month or more.
A Stanford bioethicist told Mother Jones, “The red flags are in full force on this one.” The piece noted one prominent member in the long-COVID community, Diana Berrent, founder of the big long-COVID Facebook group Survivor Corps, was not a supporter. She apparently hasn’t seen many good results from Patterson’s protocol, stating that “Bruce Patterson and Dr. Yo are, to me, the sketchiest guys out there.”
The biggest hit, though, came when Mother Jones took a whack at Patterson’s central finding – the cytokine results which underlie his entire diagnostic and treatment regimen. Maarten van Smeden, a statistician from University Medical Center Utrecht in the Netherlands, reported that the sample size – 224 patients was too small for a machine-learning study.
The fact that no-confidence intervals were provided and even the study’s sterling results raised question marks for Van Smeden. (He believed they were simply “too good to be true”.) He concluded that, “Even if we’d ignore the many methodological issues with this study…(it) is a long way from proving there is a cytokine storm going on.”.
Van Smeden presents a cautionary tale for us. While we do know that all studies need to be validated, hopefully by another research group, as patients we don’t have the statistical chops to properly assess these complicated studies. Nor is it clear that we should take one statistician’s word for it. With no consensus present, we’re stuck in the middle.
Patterson, for instance, might say, I have a good reputation. I got my data and developed a hypothesis and treatment plan around it. I even showed that the most potentially most problematic part of the hypothesis – that long-lived monocytes were carrying bits of the coronavirus around with them – was viable. Mostly importantly, my personal data indicates that my patients responded. Therefore I must be right. I’m not going mess around validating my diagnostic signature in an even larger group of patients – I’m going to treat people and and clinical trial will eventually show that I was right all along
The red flags didn’t only apply to Patterson. Mother Jones clearly spent little time finding and interviewing patients who had improved and, if Patterson is right, there are many of them. Describing a Facebook group of Patterson’s long-COVID patients as “weird and cultish” and a patient who got a “strange feeling” from Patterson’s partner Dr. Yogendra didn’t inspire confidence in the magazine’s objectivity.
The fact that Patterson said a randomized clinical trial will begin in a couple of months is a good sign that: a) he’s confident in the efficacy of his treatment protocol; and b) and he’s willing to subject it to a clinical trial. The important thing is that it actually happens and is big enough and rigorous enough to dispel any question about whether his protocol is effective or not.
While the Mother Jones piece raised some red flags, it didn’t indicate that Patterson is wrong. Anecdotal reports are close to useless. Only well-designed trials can provide us with any real certainty about the efficacy of his treatment approach. It’s simply too early to tell whether he’s on the right track or not.
The Solve M.E. Webinar
Beware! This part of the blog was taken from notes…

Patterson again used a machine learning approach to try to differentiate long COVID, ME/CFS, post-Lyme disease and post-vaccination syndrome from each other.
If Patterson had taken some hits from the media, you couldn’t tell at the Solve M.E. webinar – which provided our first update on his work in 6 months.
Getting at the Heart of Post-Infectious Illnesses? Bruce Patterson Talks on Long COVID and ME/CFS/FM
He breezed into the Solve M.E. webinar and confidently started announcing some impressive results. With his database now up to a staggering 24,000 patients, Patterson said his treatment protocol had produced “profound improvements” in over 80% of them.
Patterson stated that, using his machine learning technique, he’s been able to identify distinct cytokine signatures for people with chronic fatigue syndrome (ME/CFS), post-treatment Lyme disease, and post-coronavirus vaccination syndrome. If Patterson’s actually been able to do that, he’s just made a profound discovery in each of these diseases – none of which have a biological biomarker.
High VEGF levels (if my notes are right) are seen across the board in both ME/CFS, long COVID, etc. Produced by monocytes, they cause peripheral neuropathy. If you decrease VEGF, Patterson said, the neuropathy goes away, and so does the vasodilation and the headaches, tinnitus, brain fog, and heat and cold sensitivity, that comes with it.
Patterson is not the only one who believes the blood vessels play a major role in these diseases.
Reducing VEGF also resolves a drop in blood pressure, which increases the heart rate in POTS. (That’s not exactly true. POTS is sometimes associated with a significant drop in blood pressure but is also sometimes associated with a significant increase in blood pressure, or no obvious change in it.)
Post-treatment Lyme disease is more confusing and, oddly enough, shares some commonalities with people with post-vaccination coronavirus syndrome. A cytokine node centered around elevated IL-13 characterizes these patients. That and an elevated IL-8 agree with past Lyme study findings.
In a good sign, Patterson stated that a totally different algorithm and classification system highlighted the same diagnostic factors in long COVID. It too suggests that inflammation of the blood vessels (IFN-y, IL-2) plays a key role.
People with the post-vaccination syndrome have the same symptoms as long-COVID patients. Noting that their prevalence was low compared to the chance of getting long COVID from having COVID-19, Patterson did not suggest staying away from the coronavirus vaccines.
A profound decrease in CD8 T-cells in long COVID and Lyme produces similarly profound immunosuppression, which, in turn, allows herpesviruses as well as two tick-borne pathogens Borrelia (Lyme) and Babesia to become reactivated in some patients.
Long-COVID patients for whom the virus is still active 3 months later can benefit from antivirals. After viral replication stops – leaving perhaps viral ghosts behind – drugs that stop viral replication won’t help.
Patterson also stated he’s seeing a lot of people with “long COVID” who have prior cases of bad EBV or Lyme disease. Even ten years afterward, the old Lyme cases still have distinct cytokines (high IL-13).
Patterson stated he’s producing the first diagnostic kit for long COVID and will produce similar kits for ME/CFS, Lyme, etc. In the middle of May, Patterson stated he was 1-2 weeks away from the first regulatorily approved diagnostic for long COVID. If my notes are correct, he expected European approval for his long-COVID diagnostic test on May 26th. (That doesn’t appear to have happened.)
Treatment
Patterson reported that a 20-patient clinical trial of pure long COVID (unpublished) using maraviroc and statins (to bring down clotting) found that autonomic symptom scores (including fatigue and shortness of breath) decreased significantly and were associated with changes in cytokine levels. Reductions in TNF-a and IL-1 were also associated with improvements in fatigue.
Patterson noted that cytokine hub-directed therapeutics have been proposed for autoimmune diseases. He said his long-COVID patients get almost completely better. Those who come down with long COVID after Omicron are a bit different: he sees dramatic increases in IL-6 in them.

Patterson’s focus on using precision medicine to combat these conditions was welcome indeed.
Focus on Precision Medicine
Patterson’s focus on precision medicine to inform treatments provided an inspiring ending. Stating, “It’s so critical to have precision medicine” in these diseases, in particular, Patterson noted that in his career “there has never been a more complicated disease”. Noting that, “We don’t make a move in cancer without precision medicine,” he asked, “Why not with long COVID?” Why do we stop wit “stupid markers like CRP (c-reactive protein)? Hear-hear!
Patterson’s Classification Preprint Study
Patterson’s preprint paper, “Cytokine Hub Classification of PASC, ME-CFS and other PASC-like Conditions,” was recently published. Note that a warning on the paper stated:
“preprints are preliminary reports that have not undergone peer review. They should not be considered to be conclusive, used to inform clinical practice, or referenced by the media as validated information”.
It’s a very short paper. It’s short in every way. Documentation is sparse (no mention of disease criteria), and the results and discussion sections are rudimentary. Even though the paper is focused on using cytokines as diagnostic tools, it doesn’t state which cytokines were highlighted in ME/CFS. (It also misspelled ME/CFS (as “ME/CSF”) five times). In short, everything in this paper shouts “rush job”. I can’t imagine that it’s going to mollify Patterson’s detractors – quite the opposite. It’s a bit puzzling as Patterson has generally produced full-fledged papers.
The study used a machine learning approach and the same group of cytokines that Patterson used to produce his cytokine signature in long-COVID patients.
64 people with post-acute sequelae of COVID-19 (PASC/long COVID), 26 with mild/moderate acute COVID-19 (MM), 25 with severe acute COVID-19, 50 with myalgic encephalomyelitis /chronic fatigue syndrome (ME-CFS), 29 with post-treatment Lyme disease (PTLD), and 42 post-vaccine individuals with PASC-like symptoms (POVIP).
Note that Patterson first assessed a wide range of cytokines to come up with his cytokine signature for long COVID. He didn’t do that here. Instead, he assumed that the same cytokines would tell a similar tale in these other diseases and plugged in the same cytokine set. The model did a good job at identifying the long-COVID, or PASC, patients. It wasn’t as effective with the other diseases.
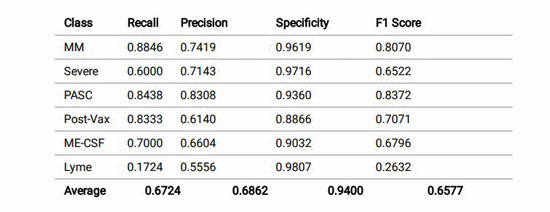
The model was effective at finding the long COVID (PASC) patients; the others not so much.
The study used an F1 performance metric (0-1.00) to assess the quality of his cytokine-based machine learning model. The F1 score is a convenient score because it combines two different factors: precision and recall. For that reason, though, it’s not considered a particularly rigorous score. One website stated the F1 scores are best used for a “quick, hi-level, comparison“.
A 1.00 score means that every person was put in the correct classification. The score Patterson’s model received (.61) was described as being below “good” and on the lower end of “OK” (with OK ranging from .5-.8) by one site. The authors acknowledged this, though, noting that some conditions were more difficult to correctly classify. Post-treatment Lyme disease was particularly problematic.
How effective the model was at predicting who did and did not have ME/CFS can be seen in the “recall” and “precision” scores:
- Precision refers to “what proportion of predicted Positives are truly Positive?”; i.e. what proportion of the patients in the model predicted to have ME/CFS actually had it? Table I in the paper indicated that 66% of the patients that the model predicted to have ME/CFS actually had it.
- Recall refers to “what proportion of actual Positives are correctly classified?”; i.e. what proportions of the people with ME/CFS were classified correctly? The model identified 70% of the people who had ME/CFS as having ME/CFS.
The model did have high specificity though. That is, while the model missed identifying a significant portion of people who had ME/CF – and when it sometimes incorrectly reported that someone had ME/CFS – when it stated that someone did not have ME/CFS, it was generally spot-on (.90).
For some reason even when the model produced horrible results, otherwise it still did well with regards to specificity. The model, for instance, was simply terrible at classifying people with Lyme disease correctly (recall .17) and was poor (.55) at predicting who had Lyme disease, but it rarely suggested that someone who did not have Lyme disease had it (.98!)
Trying to differentiate these very similar occurring diseases using just 14 cytokines was not an easy task, and Patterson’s the first one I know of to even attempt it. Rather than seeing this as a victory or a defeat I prefer to view it as a good first step and applaud Patterson for taking on this knotty problem. I hope we’ll see many more studies like this as researchers take on post-infectious illnesses.
The model’s lower F1 score and the 30% mistake rate in the model’s recall and precision figures suggest the model was perhaps a start but wasn’t particularly effective either. That wasn’t the message I got from the webinar.
Conclusions

Has Patterson gotten too far out over the tips of his skis – and is going to take a fall? Or does he have the answer to long COVID (and other conditions?). A promised clinical trial slated to begin in a couple of months should tell us much.
The Patterson saga reminds me just how much at sea we are with all this and how little firm ground there is to stand on. Take the critique the statistician in Mother Jones leveled on Patterson’s first paper. Would another statistician have another opinion? We don’t know.
Patterson has a good pedigree, is confident and convincing, is gathering more patients all the time, and must have tons of data. He’s got a fascinating hypothesis, appears to have validated an important part of it (monocytes with coronavirus proteins), and is planning to do a clinical trial.
Over the past year or so, he’s also gotten in trouble with the FDA, exhibited, according to a judge, “reckless indifference or deliberate gamesmanship” in a legal case, had his core study skewered by an outside statistician, and some researchers and medical ethicists have called his promotional style into question.
Who is the real Bruce Patterson? Is he the former Stanford researcher who glommed onto long COVID early, quickly deciphered its secrets, and is hell-bent to do whatever it takes to help sick people? Is it just a matter of time before he gets all his ducks in a row and sets the long COVID, ME/CFS, and other diseases’ world afire? Or is it all going to fall apart at some point?
Time will tell. The most important step in this saga for all of us will be the clinical trial that Patterson stated will begin in a couple of months. Let’s hope it happens and is big enough and done well enough that we’re finally left with some certainty.
Health Risings Hot, Hot Summer Drive Update

Thanks to everyone who’s helped our summer drive blast off so quickly.
It’s still early summer but it’s gotten blistering hot over at Health Rising as the donations continue to roll in – leaving us 2/3rds the way to our goal after just five days of our 3-week (3-week?) fundraising drive. Thanks to the over 100 people who have contributed over $10,000 thus far.
The Patterson blog exemplifies a core commitment of Health Rising – to try to be as balanced and objective as possible – even in a situation where we would above all love to have good news. If that kind of approach makes sense to you please support us.






‘Right to try’ is on a lot of minds, perhaps more so than ‘publish first’.
This should remind one of Montoya’s treatment that looked great with 13 patients but when put to a larger test became a huge failure. That’s why clinical trials are done.
Interesting article Cort, thanks for bringing this info to us.
I look forward to reading more when the results of the clinical trial come in.
Seems he has not made any progress since your July 2021 article which had 117 comments that apply here also. In those 11 months, he could have made a significant difference with a peer-reviewed study. Anyway, I don’t have the luxury to afford his treatment.
I will say this – he has a lot of his plate. I’m not too surprised that things are taking some time. I was actually surprised he moved forward on ME/CFS given everything else that’s going on.
Thanks for this article.
I have taken their tests and they have recommended drugs for me. I am wavering because the drugs are expensive, have liver toxicity warnings and I read the mother Jones article.
On the other hand, when I sent my blood they didn’t know I had ME/CFS. When I got my results, my cytokines put me firmly in the me/cfs category according to his paper. This did impress me.
That’s pretty impressive! Not bad at all. That’s encouraging.
I forked out the dough and it has worked for me. Next is to get another cytokine panel. Thank You Dr. Patterson for sticking your neck out.
Hi – can you please advise how and where you got the test?
The Bruce Patterson saga seems eerily similar to another saga, in which another Bruce figures prominently: Bruce Gillis – another reputed academic who “invented” a test to heal suffering patients (here: fibromyalgia patients):
https://www.statnews.com/2021/10/20/selling-certainty-epicgenetics-physician-was-one-of-fibromyalgia-patients-few-true-allies-or-was-he/
Worth reading – you´ll notice a nearly identical script (including: we´ll do a real study – later)
I had forgotten about them. I did a blog on them. It sure sounded great at the time. I was quite excited.
“Buoyed by demand for their FM/a blood test, the Los Angeles firm recently quadrupled their footprint, moving their labs and offices into a 50,000 sq. ft, facility. It’s all coming to a head, though. Their blood test, genomic test and treatment effort are all of a piece and will likely all rise or fall together. With their treatment trial underway, we should know over the next year if EpicGenetics is the next big thing – more accurately, the first big thing in FM – or not.”
https://www.healthrising.org/blog/2019/12/26/epigenetics-fibromyalgia-fma-diagnostics-vaccine-treatment-chronic-fatigue/
Three years we are still waiting for that clinical trial! Let’s hope that doesn’t happen with this. It would start with we need more time….Let’s hope not.
We’ll see what happens with Pridgens FM clinical trial but he’s been so impressive just steadily moving things a long – the Phase 2 trial, the animal studies – gathering steam – and now he’s a big Phase 2/phase 3 trial going. It’s taken about ten years – he had to create his own startup company – for sure – but he’s hasn’t put a foot wrong that I can tell.
Did anyone have sob cured chest tightness and fast heart rate
Research into cytokines and profiles in ME/CFS has led nowhere in recent decades. When researching Long Covid, you see that researchers follow the same path. A dead end in my opinion.
https://bmcneurol.biomedcentral.com/articles/10.1186/s12883-019-1433-0
What has happened to research into NK cells and how they work? The only consistent finding in ME/CFS. Does this also apply for Long Covid?
A critical remark: in England the definition of Long Covid is if your complaints have not disappeared after 2 to 4 weeks. This is really much lacking. It should be at least after half a year and better after a year. Then you can really talk about Long Covid.
I agree – cytokine studies have surely had the worst return on investment in all of ME/CFS research. Many have been done and few consistencies have been found – and none which point a finger at anything really actionable I don’t thank. VanElzakker wrote a paper bashing the consistency of cytokine studies. The only thing I wonder about is the machine learning aspect – could that make a difference?
Encouragingly, the cytokine signature for long COVID did seem to hold in the second study.
I think machine learning is an important way to tease out the meaning of all the cytokines. There are too many that exist to begin with, and study results seem to be conflicting. But you can’t compare 1 to 1 cytokines in CFS & controls. Has anyone taken into account a profile of cytokines like a profile analyzed together at the same time? Far to complex to do by humans.
Patterson’s first analysis of ~240 cytokines with machine learning to see which matter & to get a “index measure” was the most innovative thing. I’m shocked that no other CFS researcher thought to take that approach: measure ALL cytokines & use ML, AI, PC to figure out a meaningful cytokine constellation. Unfortunately, Patterson didn’t redo that 240 cytokine analysis for CFS, Lyme, Vax, etc. I thought that would be a no brainer. But maybe he wanted to find out if his original 13 cytokine test would be enough to differentiate CFS, to save time & money.
Yes I wonder about this machine-learning stuff. I thought that trying to mimic his long COVID results in ME/CFS was a reasonable try and it was partly successful. Hopefully, he can go back and redo the analysis. His long COVID signature did seem to hold up.
Cytokines have been looked at before and tried in ME/CFS. There isn’t much difference between cytokines in peME and healthy controls if I remember correctly. I think SOME people have low NK cell cytotoxicity though. That is why ME is called Low NK Cell disease in Japan. But Paterson’s protocol does not just focus on NK cells. Imagine if a British doctor made up a cure all in the UK and started selling it. The GMC would strike him off the doctor’s register before you could say Boo to a goose. Why is Paterson even allowed to sell an unproven protocol in America( and around the world) when there have been no clinical trials and approval by the FDA???? He should be struck off the doctor’s register in all honesty. He is a QUACK selling potentially dangerous and QUACK treatments (yes MARAVIROC is a HIV drug) and someone on Youtube who did his protocol for Long Covid said the pharmacy charged him $3000 for Maraviroc drug. Go figure how much Paterson has made out of this. Probably millions of dollars by now and maybe a few people have recovered, even if they were going to recover anyway. QUACK
I’m on his protocol and it’s working. It has helped me immensely, especially when no one else was willing to help.
What is the protocol?
Can you share the protocol?
Maraviroc, Plavix and Pravastatin for me. Disregard all the naysayers in the comments section. If they did the research and background for Dr. Patterson you would find quite a bit. Yes, this is research but again, I couldn’t find any other help. Absolutely nothing in Canada since I have never tested positive for Covid. My affliction was from the vaccine. And try getting any physician to belive you. Good Luck!
Did Dr Patterson’s protocol cure you in the end? Just wondering
Hi! I thought I had reached the end. However, I am on my third rebound and am presently back on the protocol. I believe it’s a “viral reservoir “ that is reinfecting me from the vaccine. To my knowledge , I have not been exposed to Covid and the symptoms are the same but lessening with each rebound. Each time on the protocol, it does work with a few months reprieve. Damn Canadian mandates!
I am in NH and we only have 1 long haul clinic and its an hour drive. I too am vaccine injured and have had long covid symptoms since 4/8/22.
I have received no help from doctors and was just recently referred to the clinic, whether any help will come out of it is anyo
ne’s guess.
What is said in this article about Dr Patterson’s treatment can be said about the vaccine…even worst as the vaccine is given to healthy individuals…why judge with different standards?
Dr. Patterson and his team do not prescribe or sell drugs. They offer the testing (of 14 cytokines) for which you pay $400, similar to the cost of other advanced tests. From their lab results they generate some recommendations but you have to go to a local doctor to get these prescriptions. You can request a consult for a fee, and if you are following the treatment no doubt you’d want to do follow-up testing at additional cost. But this does not constitute quackery, in my opinion. Nor are they getting rich at this point.
I don’t think it’s quackery either. I disagree about the getting rich point though. The company exists to sell diagnostic tests – that’s how it makes its money. It must be doing very, very well
Sillia, I agree with Cort here that Dr Patterson is making an awful amount of money from this game. I paid myself just to see what happened here in the UK and the results came back with various high cytokines and I am not even ill anymore with ME/CFS. So go figure why my cytokine profiling would show up some abnormally high cytokines if I am not ill anymore if this is the answer everyone is looking for. I was offered to speak to a consultant about my results but I did not go that far as it would cost me another £250 to do so. I had CFS for 16 years BTW before it cleared up for no real reason.
@Martin, at least him and others on the “fringe” are trying, rather than waiting for studies and published results. As a patient with LC since last August, I’m still witnessing the mainstream medical community completely deny the existence of vaccine injury, while they promote the same tried and failed long covid treatments. This whole thing has made me lose respect for the entire profession.
Patterson is one of the few – may be the only one – to produce a study on post-vaccination syndrome. He’s certainly in the lead there and he’s been in the lead trying to get the medical establishment to pay more attention to long COVID. Of course, he does benefit from that as that’s what his work us about but whatever his reason – we can’t know what it is – it’s been good to see him out there saying it’s serious and it’s real.
I fear Paterson is just gaslighting Long Covid to make money. I don’t believe that he really is that interested in LC, ME and CFS. Where has he been for ME and CFS before LC came? NO WHERE.
John, I feel for you mate. Welcome to the world of dishonest researchers and doctors all on the gravy train but who hardly ever make real progress. Vaccines DO infact cause injury as well as causing ME (eg the New York ME epidemic in 1904 as described by Dr Byron Hyde in his book Understanding ME). I hope you can get the compensation you deserve,
Well why hasn’t anyone tried to replicate or disprove Patterson’s work?
His cytokine test panel doesn’t contain any cytokines anyone else couldn’t measure. Plenty of test subjects at LC clinics. Plenty of interested researcher. Plenty of AI people that could help.
Why hasn’t anyone run the data on previous CFS cytokine studies though a AI/machine learning algorithm? That would be a real cheap study.
ME and I doubt Long Covid have got nothing to do with cytokine profiling. ME is an enterovirus hitting your nervous system and causing a vasculitis injury. Long Covid is a vascular disease that affects the heart and lungs (and brain I think)
That’s a common question. Why not try to validate these results? Please???? We see findings being left hanging time and time again. Hopefully someone will. It would be so helpful if someone did that; ultimately, it’s critical that someone do that.
Anyone follow Creaky Joints/ GHLF podcasts? No where near detail of coverage of things here but interesting to hear The Health Advocates share several takeaways from attending a medical conference on rheumatology. One was a “bombshell finding” that chronic pain is now identified as its own disease. Seems like a major step in our area of unrecognized or understood suffering.
If that really got embedded that would be huge. Not just for fibromyalgia but I would be it would help with ME/CFS too. The science has certainly been trending that way. Thanks for the good news 🙂
Cort, would you consider posting a poll where people could put in their feedback regarding their experiences with Dr Patterson’s protocol?
It would be a valuable resource to hear the perspective of patients and get some idea of how successful his protocol has been and whether or not treatment successes have lasted.
Great idea! I will do that.
We could ask about diagnostics – did they seem to get it right – costs – general experience – treatment effectiveness —wow – great idea – thank you.
Awesome Cort! Thank you! I’m sure it will be a great resource!
Maybe this is my ignorance showing , but why not just let the patient decide. I don’t have a background in science, but I do understand the importance of clinical trials.
How is this any different that being on 35 different antibiotics. Being on 10 different drugs that are prescribed for other conditions.
Doing a 10 week series of blood ozone treatments. Doing another 10 week series of ozone sauna. Doing infrared treatments.
Taking numerous herbs and supplements that I have never heard of.
Doing magnetic therapy for 10 weeks.
Buying a WAVE machine that cost hundreds of dollars that is supposed to work at the cellular level.
I personally will try medication that is FDA approved even if it is used off label. Do I want my liver check or heart or anything else when I’m taking this medication, yes.
I personally think it is our right to try anything that we might think helps us.
Could it end up harming or killing us, yes. But the alternative is just lying in bed day after day not having the strength to shower. It’s like you are not alive but not dead.
I’m probably letting my ignorance speak but at this point I’m too sick to care.
I was diagnosed with lyme disease 4 years ago. The test showed 5 positive bands. Now chronic fatigue has been added to the diagnosis.
The patient is deciding. The question some people have is whether Dr. Patterson has the necessary evidence to be making the claims he is making; i.e. the 80% improvement rates. He is also financially benefitting from all the diagnostic tests done. There’s a difference between a doctor saying “let’s try this” and someone who is promoting and financially benefitting from a diagnostic test.
I’m not in any way saying that Dr. Patterson is wrong or that’s he’s doing the wrong thing. As I’ve said before, that I personally feels that if he has the goods, one could feel that he has moral obligation to find a way to get them out there as quickly as possible.
You’re certainly right that most of the drugs used in these conditions have never undergone clinical trials for them.
Hi Cort! I just did my consult with IncellDX and am considering doing the protocol. But definitely want to learn more & would love to check out this poll if it happened – any chance you can share a link?
Haven’t gotten to it yet – hopefully soon!
All good, thanks for letting me know!
I have followed this research since June 2020 (long covid sufferer since March 2020). I am now on the protocol (a statin and maraviroc) and it has been the only thing to make me feel normal again. A few things: Dr Patterson lives in a world of possibility and can be over optimistic in terms of what the team can do/when and get in trouble in some of his communications, etc. I understand people can question their motives but in my experience the team really want to help people and costs have been very reasonable (maraviroc is expensive and they are working towards producing a generic for this). I agree the statistics were dubious for the immune marker paper (sample size was too small) but I believe their hypothesis is compelling for the mechanisms and treatment does seem to be helping many many people. The Mother Jones piece comes off as a sloppy hit piece in many ways – as you say, they didn’t bother to interview the many people that have recovered on the protocol.
Can I call you because I want to make sure this is real please I’ve been suffering for 10 months
Katherine….Agreed! I too compiled alot of research with repect to IncellDx. I even sent his treatment protocol to a retired physician friend of mine who was employed at a pharmaceutical company and he agreed with the repurposing of the drugs. It has given my life back. I have had a bit of a rebound of my symptoms lately and submitted a S1 Immune Subset Panel blood test which confirmed the S1 is still causing issues. Back on Maraviroc and Statin to finally rid myself of the vaccine influence. Calgary, Alberta
So statin and maraviroc worked for you.
Can I ask what condition do you have ?
ME/CFS or long covid ?
How do you get on Dr. Patterson’s study as I would like to enroll.
You can find his long COVID center here – https://www.covidlonghaulers.com/
Thanks so much!
I’m on the protocol and it has helped me immensely. Will it totally rid my Covid Long Haul. I’ll find out in 8 weeks once I finish this set of drugs.
Thanks for the update – good luck!
My money is on “got consumed by his giant ego.” We’ve been here before folks. How often has the guy who took all kinds of shortcuts and copped all kinds of legitimate criticism actually turned out to be right? Like, ever? BP is an interesting entry into the hall of quacks given his background though, I’ll grant that.
And why is it always 80% when these guys quote percent of patients improved? That’s a big tell for me. It’s never 100% — that’s too unrealistic— but it’s never 50 or 60% either — not impressive enough. Always 80%, the Goldilocks number for these guys talking through their ego. 🤷🏻♂️
I don’t know if Dr. Patterson is right about his diagnostic and treatment protocols, but to say that the only protocols that can be tried are those with large, double-blind clinical studies is not helpful. To complain about the cost of his diagnosis and treatments ignores the fact that most ME/CFS patients have spent hundreds of thousands of dollars trying to get well, none of it covered by insurance.
I have had ME/CFS for 40 years. I cannot name a single treatment I have tried that has been proven effective in ME/CFS by large, double-blind studies. If I missed something, please let me know.
I watched a long and rather technical video on YouTube by Dr. Patterson describing his work. Where he is likely to get into trouble with the medical establishment is drawing parallels between ME/CFS, Long Covid and illness following vaccines.
You have to ask yourself who influenced Mother Jones to write this article and not interview any patients who had a good experience.
I am also concerned by the use of a powerful HIV drug that has some very serious side effects. If the use is short term and the expected relief is likely, it might be work the risk. But, once again using an AIDS drug to treat ME/CFS, Long Covid and illness following vaccines suggests some disturbing connections.
What does Maraviroc actually do? Anyone know?
Maraviroc is an inhibitor of a cytokine receptor that plays an important regulatory role in the immune system. The receptor is called CCR5. There are not many pharmaceutically developed CCR5 inhibitors around, and they are all very expensive. Some drugs used in the Chinese Medicine system do contain compounds now identified as CCR5 inhibitors. One is a compound called DT-13, described in about two dozen studies (https://www.sciencedirect.com/science/article/abs/pii/S0753332217338052). These studies point to comprehensive anti-inflammatory, immune modulatory, hepatoprotective and cardioprotective effects of DT-13 (https://www.worldscientific.com/doi/abs/10.1142/S0192415X05003417).
DT-13 is contained in the plant (root) dwarf lilyturf tuber which has been used in Traditional Chinese Medicine for ages (under the name MaiDong). Interestingly there is a planned trial for LongCovid in which MaiDong seems to be administered: https://clinicaltrials.gov/ct2/history/NCT04924881?V_1=View.
So, possibly, if someone can sort out the right way to extract this compound, we´d have a much cheaper herbal alternative to Maraviroc (no medical advice, just summarizing the literature).
I was signed up to take part in the UK. It turned out to be VERY unwieldy and I was offered a choice of 2 hotel bedrooms to attend at airports at 8.30am; from where my blood would be sent to Spain for analysis.
Results would then require my GP (NHS) to be on board in analysis, discussion and treatment….it was never going to happen was it. Could he have made it any more difficult for someone with severe ME/CFS. And all at a cost up front. I was advised to give it a wide berth
Jane. I had the test done and I no longer suffer from ME/CFS, although I had it for 16 years. My cytokine profile came back with a number of abnormally high cytokines yet I am pretty healthy now. So go figure. Dr Patterson is just making money from this due to Long Covid being in the news a lot. Cytokines have been studied before in ME/CFS and nothing specific has been found if I remember right. So you did well to avoid wasting your money.
Hello Cort,
How about picking up a phone to get some of these questions answered? The test received CE-IVD approval in the EU May 26. You ask the questions whether other statisticians would agree with the one you mentioned-well they don’t as the paper went through peer review. Last we have a 3000 patient analysis identical to the outcome study that is being peer reviewed to address the stereotypical rebuttal from those who have not published that the sample size is too small. A MAJOR correction-I never worked for nor associate with Cytodyn, the makers of leronlimab who are under investigation. I don’t mind a well balanced article as long as the facts are correct rather than a regurgitation of a poorly written hit piece.
🙂
Will do.
Peer reviews help but are not a panacea. I recently published a blog on a paper that went through peer review and was admonished later for the awful statistics on it. I did note with your study that that was just one statistician and that another one might have helped differently.
I think your characterization of this blog as simply a “regurgitation of a poorly written hit piece” (ouch!) is unfair. I do my own work. I try to think things though. That particular blog took quite a bit of time and effort as I tried to be as fair as I could.
As much as I hope that your protocol works out and that you continue to finetune your diagnostic algorithm for ME/CFS and other diseases, the questions posed by the Mother Jones article HAD to be included given our commitment to be thorough. That said, I didn’t feel that Mother Jones presented all sides and I clearly said that – and stated that the article did not mean that your treatment and testing protocols don’t work.
I promise I will reach out next time, though.
Thanks
Hi Cort. I appreciate your objectivity in this article. It’s only fair to bring up all perspectives when bringing information to chronically ill people. The last thing we need is to get our hopes up because of a lack of info.
Hi Cort, Contact me if you are interested in knowing “my journey” and why I had to reach out to Dr. Patterson.
“If we simply lower VEGF…”
Well how on earth do we do that?!
It lowered mine and my brain inflammation has been reduced to approx. 1 out of 10. Used to be 9 out of 10. I’m on his protocol and it’s working for me.
I’m on his protocol and it’s working for me. So far, so good. Thanks Dr. Patterson!
I have never met Dr. Patterson, but after taking the blood tests from his lab and bringing his literature to my doctor she set me up on this protocol of Maraviroc and Statin and it is the only thing that has allowed me to sit up in bed after a year of disability. I think I would have died if I hadn’t found this.
Good to hear – thanks for passing that on, Jennifer.
I am also on the Patterson Protocols and it is the only thing that has helped after a year’s disability that lost me my business, my savings, and almost my life. While I can understand that there is a standard way of doing things- COVID has not been a standard disease. Millions of people are suffering and the standard has not offered any relief. Perhaps the “cultish” following of those who have benefited from Dr Patterson’s protocol is better explained by speaking to those afflicted, and listening to the horror stories of medical gaslighting and neglect that is endured as result from wilful ignorance surrounding this issue. Fear of legal repurcussions has replaced medicine’s commitment to patient care. I fired my doctor after he told me when I came to him with my lab diagnostics and print outs of Patterson’s papers because he literally told me “I’m not interested in the science”. If a doctor is not interested in emerging science to treat an unprecedented disease why continue to see patients? Luckily, I found a wonderful practitioner and we’ve made progress to the point that I can get out of bed, drive my car, and have hope that this nightmare will end.
Maraviroc, Statins, and Corlanor have been game changing for me. I know this bc I had to stop taking them in order to take a round of paxlovid to see if that can get me back the rest of the way to where I was pre COVID-19 and attempt to rebuild the life it destroyed.
Disclaimer: I believe part of Patterson’s theory is the existence of persistent live virus in hard to treat spaces in the body, but Paxlovid is not part of the protocol at this time. Just want to be clear- the antiviral is a suggestion from my doctor who *does believe in science and continues to familiarize themselves with research being done. Also, Corlanor for relief of POTs symptoms is not a part of the standard protocol.
Lastly, to address questions about why Patterson would take on ME/CFE or lime while working on the PASC Covid issues- and this is pure conjecture on my part- I would assume it has something to do with the similarities of symptoms and inability of the medical community to offer relief to those afflicted communities. Despite years of research and funding. The real pandemic is taking a look at how medicine treats those without the simple answers provided by the same 13 blood tests ran when a patient has symptoms. Medicine not having an answer yet does not mean the answer isn’t out there or patients complaints aren’t real.
I’ve had Long Covid for over 24 months now. Last year I tried Dr. Patterson’s protocol. While I certainly appreciate the he is trying to help people with Long Covid, and apparently has helped some, it did not work for me. I spent 6 months on it, and saw no improvement; in fact, according to the cytokine blood tests, I got worse. I’m still anxiously waiting for someone to crack this thing! I get NOTHING from my GP. Everyday is a struggle.
Hello all,
In order for this to work, do you have to take both pravastatin and maraviroc or just one of them? Also for how long and dose?
Thanks heaps
I’ve had experience with Mother Jones attacking a field I was expert in (agriculture) and knew all sides of the story. As far as I’m concerned, whatever Mother Jones advises to run from I will run towards.
That said, my 26-year-old daughter who was working as a Registered Dietitian had extreme fatigue, muscle pain, brain fog, and a host of other issues within days after getting the Moderna vaccine in January 2021. The symptoms have never subsided. She has never had COVID. She had to stop working and return to our home for care. We have been trying to get the Patterson protocol since the spring of 2022 but it requires working with a general practitioner. In California the GP’s have been afraid of it. We have been delayed months as GPs initially told us they would work with us and then cut and ran. In one case the GP said he would work with us and then two weeks later told us he had to check with his group’s legal department. Then it took five more weeks for the attorneys to answer and advise him that even though every drug on the protocol was FDA approved, the combination was not approved. It turns out that FDA doesn’t approve or disapprove protocols. It was obvious that he was just making things up to back away without alienating his patient (wrong!).
After almost six more months of my daughter continuing to live in a miserable state we finally found a doctor who would work with us. We will start it this week. Mother Jones be damned!
In the state of California this issue has destroyed the credibility of our medical system. They have ceased to be doctors or healers and have become pawns of a cynical state government that seeks to protect insurance companies and politicians over everyday citizens. I now say to friends, “My doctor is good as long as I don’t get sick.”
Mike,
Did your daughter begin the Patterson Protocol, and, If so, what had been the effect?
Hi Mike, I have been injured by the Moderna and have been bedridden almost 3 years. I am wondering if maraviroc helped your daughter. Thank you , Sarah
Dr. Patterson helped my son SUBSTANTIALLY by perscribing Maraviroc to him. After almost three years of unbelievable suffering, about 50 doctors who either did nothing or had him try stuff that didn’t work, and hundreds of thousands of dollars on doctor bills and medications that didn’t help, Dr. Patterson was the only person able to give my son something that is actually making him feel better. And not just a little better- a LOT better. I understand the skeptism and the complaints about promoting before fully testing. However, I strongly suggest you give this guy a try.
-Tracey Ganesh
Alpharetta, GA
Good to hear Tracey – thanks for sharing your son’s progress. 🙂
I have had Chronic fatigue and brain fog ever since I got COVID twice. I have seen all kinds of specialist and now I’m seeing an integrative specialist who I’m hoping will help. This has been a hellish few years but I still got up to work full time regardless.