


Suzanne Vernon PhD. has been working on ME/CFS for over 20 years.
Suzanne Vernon’s interest in pathogens and post-infectious diseases goes back to when, as a 20-year-old, she landed a work-study job at the CDC’s Division of Vector-Borne Diseases while an undergraduate at Colorado State University.
Ten years later with her Ph.D. in virology, she was a research microbiologist at CDC Atlanta, leading a team of scientists on the hunt for pathogens and molecular markers in ME/CFS. Her first papers for the CDC’s ME/CFS program in 2002 – searching through cell-free plasma DNA for the sign of a pathogen – and her pioneering use of new gene expression techniques – was emblematic of her focus on using cutting-edge technologies to peer deeply into ME/CFS.
It was during Suzanne Vernon’s time the CDC funded the groundbreaking Dubbo post-infectious studies led by Andrew Lloyd, and Gordon Broderick’s modeling efforts that would later come to define much of Nancy Klimas’s present work and set the stage for her treatment trials in GWI and ME/CFS (and her ample GWI funding.)
One of Vernon’s most creative endeavors came in 2006 when she gathered almost 2 dozen researchers to use cutting-edge statistical analyses to analyze a large data set gathered from over 100 ME/CFS patients and controls during a 2-day hospital stay. The 14 studies that resulted – the largest and most complex of their time – filled up an entire issue of the Future Medicine journal Pharmacogenomics.
The tests were state of the art for that time and arrived at some familiar conclusions. A “supergenes” study plucked out estrogen receptor, TNF-a (which Dr. Klimas is trying to suppress in ME/CFS), three genes involved in calcium and/or sodium ion channel function (channelopathy), two magnesium binding genes, a heat shock gene involved in protein misfolding (amyloidosis), two metabolic genes (acyl-coenzyme binding) and several signaling genes. Another study highlighted genes associated with lipid metabolism – a hot field of study right now – energy production, and the immune system.
The CDC data set was enticing enough to be used by the International Conference on Critical Assessment of Massive Data Analysis in 2006. This time, ten papers were produced, several of which highlighted tryptophan and cortisol, and produced one of the first ME/CFS systems biology papers.
After leaving the CDC, Suzanne was the scientific director of the Solve ME/CFS Initiative from 2008-15. Since 2017 she’s been the research director for the Bateman Horne Center. I was eager to get her take at this crucial juncture in ME/CFS after long COVID had leaped onto the scene.
First, I wanted to know if she thought the ME/CFS field had missed any opportunities. Was there anything the field could have done to better prepare it for when long COVID unexpectedly popped up?
She felt not but noted that she and Kim McCleary – Solve ME’s longtime leader – used to talk about the missed opportunities that the “one and done” studies in ME/CFS presented. Time and again, a study would get done – show some promise – but then, for whatever reason, get dropped. It was all, she thought, the result of poor funding – not enough funding to do large studies or to follow up on small studies; not enough funding ultimately to produce the bang that would really move the field forward.
Combine small studies with the heterogenous illness ME/CFS and you have a recipe for muddled results and slow movement. Citing a few heterogenous factors – people with different triggers, and people with different durations of the illness – she focused on people who use the criteria differently.
She said she was struck by Andrew Lloyd saying years ago that, as a physician, he could recognize a patient with ME/CFS the moment they walked into an exam room. Not all studies have an ME/CFS expert like Andrew Lloyd or Dr. Bateman on board who can ensure that only people with ME/CFS get into a study. Those that do – like Lipkin’s recent peroxisome study (which she co-authored) – are going to be that much more effective, she thought.
The RECOVER Project – “A Thing of Beauty”

Suzanne called the NIH’s RECOVER project “a thing of beauty”.
The NIH’s $1.15 billion RECOVER (Researching COVID to Enhance Recovery) program has rolled out slowly. Still, Suzanne felt it was a massive, well-thought-out program that will ultimately deliver the goods. In fact, the only program she could think of that compared to it was the fight to conquer HIV. Decades later, we have much more sophisticated technology we can bring to bear on long COVID.
Dr. Vernon said that from a research scientist’s perspective, the program is a thing of beauty.
Instead of being a top-down project, the study is designed to get inputs from many different people – many very smart people she said – and is designed to change over time. Standardized research protocols will ensure all the studies can “talk” to each other. All the data goes into central databases and is managed by the Data Resource Core. Hoarding data – an occupational hazard in the research world – is not allowed.
As big and as slow-moving as the RECOVER project has been, it appears to be a nimble project that will be able to change in response to new findings and is receiving input from a variety of sources.
Three tiers of patients receive a standard testing protocol and produce a baseline data set. Everyone in Tier I gets a clinical examination (including an active standing test), their blood drawn and analyzed (19 lab tests including EBV and coagulation tests), and receives questionnaires every 3 months for 4 years.
Of that group, some will go on to Tier Two which includes things like a 6-minute walk test, home sleep test, cognitive tests, several antibody tests, cortisol, cytokine panel, pulmonary function tests and others.
About 20% of the entire group will move onto Tier 3 where they will get catecholamine, protein, MRI, gastric emptying, nerve conduction, skin and muscle biopsy, lumbar puncture, cardiovagal, tilt table, upper endoscopy, colonoscopy, an in-facility sleep study and several others I had never heard of.
Strong ME/CFS Connection
Plus, a strong chronic fatigue syndrome (ME/CFS) connection is present within the effort. RECOVER includes twelve Task Force Committees that provide in-depth scientific reviews and recommendations to the RECOVER leadership.
The Commonalities with Other Post-viral Syndromes Task Force Committee informs the RECOVER leadership of the similarities between long COVID and post-infectious ME/CFS. Several ME/CFS experts are active on this committee including Lenny Jason, Benjamin Natelson, Hector Bonilla, and Suzanne.
Recently, several of this task force published an Op-Ed in the Chicago Tribune underscoring what ME/CFS could teach us about long COVID. Noting the similarities found thus far between the diseases such as preload failure and EBV reactivation, the authors asserted that having ME/CFS cohorts embedded in the Initiative was critical for understanding both long COVID and ME/CFS.
The Bateman Horne Institute Makes It Into the RECOVER Initiative

Outreach resulted in the BHC becoming part of the NIH RECOVER Initiative
I’m always interested in how things happen, so I was very interested in learning how the Bateman Horne Center came to be part of the RECOVER Initiative.
It turned out that they didn’t just get asked in – it took some initiative. Early in the pandemic, Suzanne reached out to Drs. Will Dere and Rachel Hess at the University of Utah to let them know that the Bateman Horne Center was interested in collaborating on post-COVID-19 research. Her ace in the hole in her presentation was the 2006 Dubbo ME/CFS post-infectious study.
There’s no getting away from the gut which produces the lipids that protect the cellular membranes. (For those who don’t know, the Dubbo study followed people with an acute infection of three different pathogens (either Epstein-Barr virus (glandular fever/infectious mononucleosis), Coxsackie burnettii (Q fever), or Ross River virus (epidemic polyarthritis)) over time to see how many people came down with ME/CFS-like illness.
The Gist
- As head of the CDC’s molecular explorations into ME/CFS, the Solve ME/CFS’s research effort, and now as the Bateman Horne Center’s research director, Suzanne Vernon has been employing a cutting-edge approach to ME/CFS for 2 decades.
- She felt the ME/CFS field has been hampered over the last couple of decades by a couple of things: poor funding and small studies have left the field littered with “one and done” studies that showed promise but which for one reason or another were never followed up on.
- Long COVID and the emergence of the NIH’s RECOVER Initiative is changing almost everything. Calling the Initiative a “thing of beauty” from a research perspective, the only similar project of its scale that she could think of was the work done on HIV.
- She was sanguine about its short-term effects on ME/CFS funding but called the exposure to ME/CFS the long COVID research community is receiving “invaluable”, noted that ME/CFS researchers are embedded in the program, was excited about the potential for RECOVER research to impact ME/CFS, and reported that treatment trials will begin shortly.
- The Bateman Horne Center (BHC) became part of the RECOVER Initiative throughout outreach which highlighted the ground-breaking work the post-infectious Dubbo studies did in ME/CFS 15 years ago. The BHC and NIH Research Centers it collaborates with have now incorporated long COVID patients into their sophisticated immune and metabolic studies.
- Given the key role the peroxisomes play in energy production and cell membrane integrity the peroxisomal problems found recently in ME/CFS could be, if further research reveals them to be so, a core problem in ME/CFS.
- Problems with cell membrane integrity, for instance, could render cells inert and unresponsive – particularly in stressful situations like infections and exercise. Recent studies, interestingly, suggest that, in contrast to healthy controls, that the protein response to exercise is almost nil.
- The gut shows up again! Call it the center of metabolism, Suzanne noted that the lipids that make our cell membranes are manufactured in the gut. The microbiome findings in ME/CFS suggest that the butyrate gut bacteria that produce those lipids are, in fact, deficient in ME/CFS.
- She hearkened back to a Solve ME 2015 study that found exercise disrupted the gut microbiome more, triggered more leaky gut, and left more gut bacteria in the blood for longer periods in ME/CFS patients than healthy controls.
- The NIH refused to fund further studies but the team – puzzled by the unusual immune response seen in ME/CFS – with the addition of Armin Allaedini persevered. A soon-to-be-published paper will explain the immune response and suggest that the gut-exercise reaction could play a major role in the post-exertional malaise seen in ME/CFS.
The presentation worked. Once the RECOVER Initiative funding was announced, the University of Utah investigators invited the Bateman Horne Center to be part of their application. When the application was approved, the BHC became the recruiting center for what is now called the Mountain States PASC Consortium (MSPC).
ME/CFS / Long Covid Cohort Studies Underway
The Bateman Horne Center didn’t wait for that to happen, however, to start their own long-COVID clinical research. By January 2021, several months before RECOVER started, they were enrolling long-COVID patients in their own studies. By the time RECOVER launched, the BHC was able to reach its enrollment target within a couple of months.
Plus, if the NIH hasn’t allowed ME/CFS patients into their studies (yet), the BHC certainly has. They’re conducting their own research comparing the two groups. In addition, they worked with Derya Unutmaz, of The Jackson Laboratory, and Ian Lipkin, of Columbia, to amend their Collaborative Research Center grants to include a long-COVID cohort.
Samples and data collected from these long-COVID patients are being analyzed by both centers and will provide a unique and in-depth look at comparing the immune and metabolomic perturbations in long COVID and ME/CFS.
That’s a potentially very significant effort given the insights metabolomic studies have produced in ME/CFS and the dearth of metabolomic studies thus far in long COVID. It could, one would hope, help convince the RECOVER Initiative to include an ME/CFS cohort in its studies.
The team is already finding important similarities and differences between long COVID and ME/CFS, as reported in this recent paper.
(That study found that a 10-minute NASA lean test produced a narrowing in the pulse pressure (the difference between systolic and diastolic BP) in the long COVD and ME/CFS patients but not in the healthy controls. While more narrowing in the pulse pressure was found in the long-COVID patients, the effects of the NASA lean test on cognition lasted longer in the ME/CFS patients. The authors proposed that reduced blood flows to the brain were causing the cognitive difficulties.)
The Tricky ME/CFS Long-COVID Connection
The ME/CFS long-COVID connection is a tricky one. Suzanne noted that because the researchers studying long COVID are learning about the similarities with ME/CFS, ME/CFS is getting massive exposure in the scientific community. That, she thought, was invaluable.
She is not certain, though, that NIH will increase ME/CFS funding because of the pandemic. Researchers are going where the funding is and right now, the funding is for post-COVID-19 disorders, including long COVID. While some ME/CFS cohorts may be included in some RECOVER studies, the focus now is on the understanding, prevention, and treatment of post-COVID conditions, including long COVID.
Long-COVID Treatment Trials Coming Soon

Treatment trials will begin soon. We may more treatment options for long COVID and possibly ME/CFS before we know their cause.
She also believes, though, that long-COVID studies will open up many avenues for ME/CFS, including new treatments. The NIH isn’t waiting to understand long COVID before they begin treatment trials: they’re moving rapidly with treatment trials – more rapidly than most of us would have thought.
The RECOVER Initiative put out a funding opportunity for clinical studies and named Duke University as the Clinical Trial Management Center. A short list of trials has already been created and trials are slated to begin by the last quarter of this year.
At the recent NIH Telebriefing for ME/CFS, Walter Koroshetz, the director of the National Institute of Neurological Disorders and Stroke, seemed rather excited for a change, and for good reason – he had some good news to report. He reported that the steering committee for the long-COVID trials was bulging with ME/CFS (15) and POTS (11) experts.
Koroshetz stated that the clinical trials are going to focus on eliminating persistent viral reservoirs and impacting immune functioning, as well as helping with specific symptoms like fatigue, sleep, cognition, exercise intolerance, and autonomic nervous system functioning.
The really nice thing about these treatment trials is that they’re not going to end up being slotted into the “one and done category”. They’ll be rigorously produced, and if positive findings result, they will be followed up on.
Suzanne believes new treatment possibilities will show up before we know how long COVID is caused.
- Update on the RECOVER Initiative: Enrollment has been picking up. RECOVER now has enrolled almost half (8,637) of the adults it wants (17,680). They are low, though, in people who are coming down with COVID-19 now.
- Find a study site near you
The New Organelle in Town – the Peroxisomes
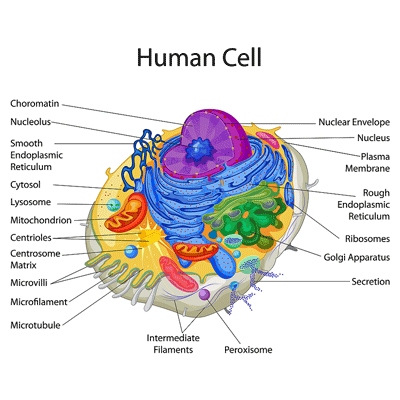
Peroxisomes may be small – see the little purple circle at the bottom of the cell- but they affect major processes in our cells. Could peroxisomal dysfunction be the smoking gun in ME/CFS?
We’d never heard of peroxisomes prior to the last year or so, but Suzanne was clearly jazzed at the result of the recent Lipkin study (of which she was a co-author). The peroxisomes’ small size belies the impact they have on cell functioning. Not only do they break down the long-chain fatty acids that are used to power the mitochondria, but they also produce phospholipids and other components that make up the cellular membranes.
That cell membrane connection could be critical. Since cells communicate with their environment via receptors embedded in their cellular membranes, damaged membranes could leave cells inert and unresponsive – a particular problem during stressful times such as infections and exercise. (We recently saw evidence of an enormous slump in protein activity after exercise in ME/CFS. Did exercise whack the cell membranes so hard that they were unable to respond?).
Vernon remembered the strange – at the time – signaling issues that kept cropping up in her early gene expression work. She couldn’t explain them at the time – now with these findings suggesting multiple ways the lipid membranes of the cells in ME/CFS are damaged, maybe they make sense.
The recent Lipkin study also brought back memories of her two hypotheses that loomed large in the patient community back in the early 2000s.
People who’ve been around for a while may remember Rich Van Konynenburg and Martin Pall. Rich Van Konynenburg was a kind of citizen researcher who championed the idea that depletions in the master antioxidant in the body (glutathione) played a significant role in ME/CFS. Martin Pall was a Washington State University biochemistry professor who, after coming down with ME/CFS, originated the NO/OHNOO- hypothesis which proposed that chronic elevations of nitric oxide and the peroxynitrite radical were causing ME/CFS.
Massive forum threads were devoted to their ideas and treatment implications, but both made little headway with the mainstream research community. Rich died an untimely death and Pall seems to have faded away. Now, given the focus on lipid breakdowns, oxidative stress, and peroxisomes, Suzanne wondered if they may have been right all the time.
With peroxisomal dysfunction, you’re potentially getting at some fundamental factors: the ability to produce energy and the ability of cells to communicate and function. Could peroxisomal dysfunction be the smoking gun in ME/CFS? Time will tell.
Viral persistence?
The Long COVID Research Initiative recently launched a $15 million effort to determine if the coronavirus was alive and well, or in pieces and creating problems in the tissues of long-COVID patients. As an infectious diseases specialist, I asked her if pieces of the virus, or the virus itself, might remain.
She thinks that’s possible. She noted that a couple of publications report persistent viral shedding has been found. Having viruses, bacteria, and protozoans hanging around is nothing new for humans – they’re all basically part of the human package – but she wondered if people with ME/CFS and long COVID now had trouble tolerating them.
“The Root of All Metabolism” – The Gut

There’s no getting away from the gut which produces the lipids that protect the cellular membranes.
The fact that it’s the gut flora or microbiome that produces the lipids that make up our cellular membranes makes the gut all the more interesting. If the microbiome is off – and every study indicates it is in ME/CFS – problems in the gut could be affecting the lipid membranes of our cells. Throw in the fact that it’s the butyrate-producing bacteria – which play an important role in lipid metabolism – that appear to be deficient in ME/CFS – and the potential linkages build up.
Dr. Vernon led one of the most interesting “one and done”, or as we’ll see, “almost one and done” studies in 2015. The study, conceived and funded by Solve M.E., examined the effects of exercise on leaky gut in ME/CFS. It found that exercise altered the gut microbiome more, and induced significantly greater amounts of gut leakage, in people with ME/CFS than healthy controls. People with ME/CFS also had more trouble clearing the gut bacteria from their blood: their bacterial levels were still high 24 hours later.
It was a fascinating study that potentially showed how exercise can affect an area we generally don’t associate with exercise – the gut – and produce a result – inflammation – that is likely associated with post-exertional malaise (PEM). Despite the positive result – and the high p values in this small study – the NIH declined to fund a larger study.
If the NIH wasn’t jazzed by the study findings, the researchers were. Armin Alaedini – a gluten and celiac disease expert – collaborated with Sanjay Shukla and Dane Cook to further analyze the before and after exercise samples. Alaedini didn’t find evidence of celiac or gluten problems but, struck by the strange immune response the team had found in the ME/CFS patients, he kept looking for ways to explain it.
He succeeded, and seven years after the last paper, this paper should be published soon. Suzanne said it demonstrates that the microbiome and immune changes that exercise produces may be key contributors to ME/CFS; i.e. the study could help explain why exercise is so problematic in ME/CFS. Columbia University thought the finding was important enough to list it as a potential therapeutic target or ME/CFS biomarker on their technology ventures website. She also noted that leaky gut is a cell membrane problem – which means that peroxisomes may be involved there as well.
Maybe the NIH will fund the team’s next grant application.
Conclusion
It was good to hear this longtime ME/CFS researcher express excitement about the RECOVER Initiative and its ultimate effect on ME/CFS. While Suzanne could not say the ME/CFS funding will increase in the short term, she believed the exposure ME/CFS was getting was invaluable, expected many insights to come out of the Initiative, noted that ME/CFS experts are embedded in the effort, that the Bateman Horne Center is part of the Initiative, and reported that treatment trials will begin shortly.
Peroxisomes have shown up big time recently in ME/CFS research. Noting the key roles they play in energy production and maintaining the cellular membranes, Suzanne suggested that damaged cellular membranes could leave the cells inert and unresponsive. Since ME/CFS patients’ guts are deficient in the bacteria that produce the metabolites needed for the cellular membranes, the gut may come into play in this equation as well. In fact, an upcoming gut study will show that exercise-triggered gut issues may play a key role in ME/CFS.






I’m interested in learning more about how they will be “eliminating persistent viral reservoirs.” How do they find these reservoirs?
I assume they will be looking in the gut and other tissues for them. How I don’t know but they’re certainly interested in the persistent virus or persistent viral proteins aspect.
I thought it was interesting that Suzanne noted that viruses can hang around – that’s not new – but that its possible that people with ME/CFS have trouble dealing with them. It’s possible – I’ve heard this mentioned – that our immune systems may be overreacting to them.
I am so glad that Dr. Vernon is looking back to these previous studies and making connections between those studies and the studies going on today with ME and Long Covid. It will be interesting to see what these new studies will turn up. I wonder how the gut and its possible connection to PEM, especially after exercise and inflammation, the perioxisomal dysfunction, and cellular membrane damage along with inert and nonresponsive cells, and microclots and thick blood connect together. I think that Amy Proal and her crew are really on to something with the microclots and Ron Davis is on to something with red blood cells not deforming. Could there be microclots in the gut possibly–leaky gut and all? Could there be viral material in the gut, even in the tissues? And could the cellular membrane damage and inert and nonresponsive cells be the reason why people worsen? Are there other factors and what are they? And what can be done to restore cellular function? I’m just throwing some questions out there and trying to figure out how things work here.
Do you know if Dr. Vernon is in contact with Dr. Van Elzakker and Dr. Amy Proal? Or Dr. Ron Davis and the OMF? It seems to me like her knowledge and ability to connect the dots between studies is what is needed to help crack this thing open and start getting some treatments going.
Cort,
Dr. Myhill has been referencing all of this for years just from her clinical observations of ME patients. Leaky gut caused by a modern high carb diet, Mitochondria membrane dysfunction, problems with persistent bacterial/yeast infections, EBV hiding like the “bogeyman” in our DNA, HPA axis issues, thyroid issues, Magnesium channel problems, etc. We all know that 80% of ME patients have IBS. Dr. Rich Von K. was/is correct about glutathione all along. “Butyrate producers” – Yes, nothing new there either. When are clinicians going to begin actually throwing “darts” at this thing in the form of “curative trials”? Maybe some Fecal Transplant work? Actually determining if the “fibro fog” is from reduced blood flow, or just constantly “guessing and wondering”. This disease is the “walking death”. So, someone dies from a rare form of E. coli infecting a fecal sample. Who cares at this point? Start bringing down the hammer on this thing. Enough talk. That is all that happens over at the NIH. There is no “sense of urgency”.
I was a researcher at U.C. Davis when the NIH, CDC, and the politicians brought the hammer down on AIDS. It was like The Manhattan Project at all of the U.C. Schools, and Stanford back then. This is still a joke considering how many people are suffering from ME/CFS. If they don’t care about the suffering, then maybe the staggering financial cost to the country might move them. I am at a loss: “one, and done research projects”. No serious Ph.D.’s work this way !!
Richard, is it true that 80% of sufferers have IBS. Gut issues are not a compulsory diagnotic on the Canadian Criteria. I feel gut issues are a recent add on, and given the appalling diet many of us eat, huge diabetes rise, and obesity…..an almost non existent phenomenon when I was a teenager in the UK…..it doesn’t surprise me that so many people have gut problems. I’m not well informed as brain fog prevents, but why the link with ME/CFS.
Lots of great questions Fed Up. I don’t know if Suzanne is in touch with Ron Davis or VanElzaker. She’s certainly in touch with other researchers such as Ian Lipkin, Derya Unutmaz as well as others.
Hi Rich – there’s no evidence that the NIH feels any more urgency about ME/CFS than before but it certainly feels urgency about long COVID. I was astonished that they’re starting treatment trials so early. (They’ve long said no funding for treatment trials in ME/CFS until they know more about it – and, of course, have refrained from giving it much funding.)
The good news for ME/CFS is that ME/CFS experts are embedded in several parts of the Initiative and they appear to be making an impact.
Cort,
The NIH realizes that they have “dropped the ball”, because so many young PC physicians just recently learned in Med School that Fibro is predominately a “sleep disorder in women” who experienced a traumatic childhood. This is laughable at this point. Any urgency the NIH is showing toward long Covid is due to the number of potential patients, and the obvious overlap they now see with ME/CFS and long Covid. The NIH is going to be asked why they ignored all of the evidence of infectious triggers for ME/CFS, because now we need that data to deal with this massive long Covid problem. Dr. Younger and thousands of physicians are certain that LDN helps lower microglial inflammation. Dr. Younger wanted to extract out the non-active mirror image portion of the racemic mix of LDN, and then administer the more efficacious enantiomer. The NIH refused to fund this??? This is the only medication known to significantly reduce ME/CFS symptoms near the root cause: neuro-inflammation. (Lyrica, Adderall, and Savella just mask symptoms!) As for Jane’s question about the gut and fibro-fog. The Pituitary produces MSH which helps maintain tight junctions in the gut and the blood brain barrier. An inflamed Pituitary produces less MSH. And, brilliant physicians like Myhill and Heymen are certain that the Pituitary is inflamed in ME/CFS. I agree. It is interesting that Dr. Amy Proal is now being mentioned, because many Neurologists used to scoff at her ideas concerning cellular Vitamin D metabolism and viral infections. She may be wrong, but her theories are sound, and should be examined. Some Neurologists were even calling her a “Quack”. (Sara Myhill has endured the same treatment in the UK!!!) There is nothing “juicy” for big Pharma, or most of the MD/Ph.D’s to grab in researching ME/CFS. Heck, Dr.’s Davis and Bateman have a dog in this fight. So, they slog forward. But, when the NIH will not fund Dr. Younger’s work, all you are going to get is more LYRICA. Unfortunately, or fortunately, a bunch more physicians and/or politicians (Tim Kane) need to contract Long Covid, before ME/CFS research moves forward.
Jane,
Three people concerning the gut:
You need to start reading Dr. Sara Myhill’s excellent books on diet and chronic fatigue. Dr. Myhill left the PHS in the UK many years ago, as she felt that she could not treat her ME/CFS and Fibro patients, under the guidelines of the PHS. She’s brilliant, and truly does care about treating chronic fatigue. (“It’s Mitochondria, not Hypochondria” is her first book.)
Myhill believes that you must heal the gut first to manage ME/CFS.
Also Dr. Jacob Tietellbaum wrote a book that he constantly updates called “From Fatigued to Fantastic”. He, like Myhill is a genuine “healer”. Not sure on his spelling, but Dr. Jacob T. was forced to leave Medical School to deal with his ME/CFS. Nice man. Cares about you. Has a very fluid protocol that he has used help many thousands of patients.
Lastly, Dr. Andrew Heymen in Aldi Virginia. All three are brilliant biochemists, and cell biologists.
Just based on what we have learned in the past two, several clinical trials should already be in the works. Butyrate producing bacteria should have already been transplanted into the colons of 200 ME/CFS patients. Those patients should be on their tenth treatment by now. What? Only C. dif. patients are sick enough to receive fecal transplant therapy? Or, I need to travel to the UK to receive fecal transplant treatment?
At the UC Davis Vet school, if they have a horse in the Equine Research Center suffering from diarrhea, they hit him with a transplant of normal feces. In two days, that horse is usually back to normal gut function. But, It will take 5 years to get that study funded for ME/CFS patients. How screwed up is that? They don’t care. None of them have a Whitney Defoe at home just “waiting and praying”
It is screwed up. Two fecal transplant studies are underway in ME/CFS I believe – predictably none in the US but the Norwegian, I think it is, study is pretty large as I remember.
If you read Dr Shoemakers book Surviving Mold, I tell the story of how a mystery illness hit our little town of Incline Village back in 1984.
Everyone in town was trying to heal their intestinal dysbiosis.
They tried every supplement, nutraceutical and probiotic they could get their hands on.
Five new health food stores opened up in a very small town.
Even the biggest supermarket in town got into the act. Set aside an entire aisle for “Health Food”
None of this seemed to do very much.
Then I stumbled over the peculiarity that camping out in the desert was like a miracle. All my food reactivities disappeared.
Ironically, I met another Incline Village “mystery illness” patient out there doing exactly the same thing I was.
We went into Carson City to celebrate our restored ability to eat by pigging out on beer and pizza.
We went back to town and told Dr Cheney and Dr Peterson but they weren’t interested.
No one else was, either. Which we thought was odd since they claimed to be looking for clues and anything that helped.
Hi, Cort–Martin Pall has by no means “faded away”–he has been very active in fighting against the damage done by RF stuff–which I hold responsible for my own ME, which began a few months after moving into a cell phone tower masquerading as a high-rise apartment building–look up “the tower of doom in London” for an analogous situation.
Thanks, Chris – glad to hear it. if I remember correctly, that was an issue that prompted him to produce his hypothesis in the first place.
Not to take away from this great article at all, but this morning I received an email from Solve ME, also a part of Dr. Suzanne Vernon’s professional resume. A new study found 14 genetic links to ME CFS in a group of 2400 diagnosed patients and identifies patient subgroups. I broke into tears, being validated for something I knew was always there. And yes folks, many possible versions of this horrendous affliction showing up in genetic SNPs. But this study got at 14 critical genes involved with illness and energy production, sleep, susceptibility to viruses and bacteria, etc. I stand today with my fellow community of sufferers, especially women, many of us who were seen as hysterical hypochondriacs, and I say never again.
How did you find this out? Are you in a study? I am participating in their gut microbiome study.
https://mecfsresearchreview.me/2022/09/15/new-study-links-14-genes-to-me-cfs/?fbclid=IwAR304vQctBnsmt3u4C_uIrb76zZCL4jwdkjWcta2TxRxIvk3BxNlIjJtwiA
not to forget the big UK decodeME study, 20.000 ME/cfs patients 20.000 controls 5000 long covid patients
I didn’t know that they’d gotten long COVID patients in there. They are also, of course, in Solve ME’s Patient Registry, which was initiated by Suzanne Vernon years ago. DecodeME brings us another important part of the research infrastructure diseases use to figure out what; going on. A big win for us!
Is it true that this study (UK decodeME) will take more than 10 years? By that time many here are already elderly or deceased.
Replying to Gijs – DecodeME have said initial results will be available in a year.
They will have so much data I’m sure it will take a long time to fully analyse, but we will get results along the way which will presumably guide where they go with the rest of it.
I heard about that – it sounds very promising. Since Suzanne really pioneered the use of gene expression in ME/CFS – she was doing it before others were in other diseases – I think she’d be happy that this was mentioned in this blog 🙂
I am sure Cort will be on this!!
I think Simon McGrath did a nice blog on this.. https://mecfsresearchreview.me/2022/09/15/new-study-links-14-genes-to-me-cfs/
Advocates have repeatedly been asking the RECOVER team for a list of the over 25 post viral experts that RECOVER says they’re utilizing in their studies. At this point, RECOVER say they don’t have the list readily available. I find that absurd given the nature of this initiative and $1.15B they have to work with.
That’s the way they’ve been. It’s rather puzzling how close mouthed they’ve been. They haven’t provided a list of any of their committee members. In fact, they haven’t even provided a list, to my knowledge, of what executive committees even exist. Nor have they provided a list of the grants they’ve funded. They have provided a list of the recruitment centers but much else is missing.
Glad to see someone linking gut issues to PEM. It has seemed like a lot of theorists have not attempted to explain the connection between the two. Now, can someone include brain fog in their theory? Is there one explanation out there that links them all, and with strong evidence?
I don’t think so. There is this thing called the gut-brain axis, though….
I wonder how microclots and thick blood play into PEM and the gut. I’m going to go out on a limb and ask–is it possible that there are microclots in the gut? Could the issues with the gut, the microclots and the perioxosomal dysfunction all be connected to PEM? And in what ways are they connected?
Endotoxin and serotonin
@ Cort
has Andrew Lloyd ever spoken about what things he sees that helps him pick out FS/ME patients when they walk in? or is it pheromones given off?
Other doctors have actually said that. The presentation of a person with ME/CFS is apparently so unique that once they know what it looks like, they can quickly spot one.
@Cort
i dont recall who said this, but at one time it was reddish haired women with young looking faces for a person’s age.
i would like to hear more about what the researchers say causes them to “know” some is CFS/ME
It’s interesting that this article mentions Andrew Lloyd in it and Suzanne Vernon talking about him. Check out this article from the BMJ, written on September 21, 2022. It has Andrew Lloyd and his discussion on Long Covid and they talk about his 2006 Dubbo Study in there. here’s the link:
https://www.bmj.com/content/378/bmj.o2188
Very interesting article – thanks for sharing that. Good to hear this from a Lyme Disease expert
Good article. Although I am tired of the viral persistence theory, it’s long been a false lead.
(…) ‘Not all studies have an ME/CFS expert like Andrew Lloyd …. ‘
You mean the supporter and promoter of the PACE trial and cognitive behavioral therapy? And deny that ME/CFS is a physical illness? That expert? Lloyd.
https://me-pedia.org/wiki/Andrew_Lloyd
(…) ‘Andrew Lloyd saying years ago that, as a physician, he could recognize a patient with ME/CFS the moment they walked into an exam room….’
Wow , is he clairvoyant now?
I don’t remember Lloyd ever proposing that ME/CFS is a psychological disorder. He has been quite a CBT/GET proponent, though and I wrote a blog about that – one of the few negative blogs I’ve written about a researcher:
The Decline and Fall of an ME/CFS Researcher? the Case of Andrew Lloyd
https://www.healthrising.org/blog/2015/05/16/the-decline-and-fall-of-an-mecfs-researcher-the-case-of-andrew-lloyd/
Lloyd also, of course, created the Dubbo studies – which I still consider perhaps the most important studies ever done in ME/CFS. Two years ago he wrote this in a paper
https://pubmed.ncbi.nlm.nih.gov/32248536/
He’s overwhelmingly focused on physiological research but really hasn’t done much in the ME/CFS field recently. He’s a mixed bag for sure.
Other doctors say the same thing: that once they get to know what an ME/CFS patient looks like it’s easy to quickly identify them.
Whoa. I did not know that Andrew Lloyd was into CBT and GET. He definitely needs to check out the new NICE guidelines and understand why these therapies are bad for people with ME. Interesting how doctors can identify ME when they get to know what they look like. That reminds me of Osler and the importance of listening to the symptoms that patients report instead of focusing so much on tests. Wish there were more doctors like that. That being said, a good diagnostic test would be helpful, like Ron Davis’s nanoneedle. I sure hope that becomes ready for prime time soon.
https://www.virology.ws/2018/04/04/trial-by-error-a-post-about-andrew-lloyd/
Although I do believe in his theory that ME is a disease in which the brain (central nervous system) reacts abnormally (hyper) to every stimulus.
Can you ask him how ME/CFS patiënts look like? Sounds very interesting.
I know and have seen many patiënts but most of them looks completely normal. That is the point. To put it nicely, I think it’s bullshit. What he says.
Things had been quiet on the gut front for a bit, so it was interesting to hear the leaky gut result come back to the fore.
I’ve been thinking about leaky gut again recently in a new light: of low-dose naltrexone.
LDN antagonizes TLR4 which is sensitive to lipid polysaccharides (LPS) which are the byproducts of Gram-negative bacteria.
I start to wonder if that may be the biggest factor in how LDN benefits (some) patients.
I am wondering how many ME/CFS patients have had CD4 and CD8 cell measurements particularly at the beginning of their illness, also the 8 way multi-test that measures your body’s immune response to 8 common antigens.
This is such an interesting article. My Dr had me do what he called a gut shock, 500 calories a day and 1200mg of PEA (Palmitoylethanolamide) this gave me a new kind of normal, one I remembered pre 2008 after I became sick with EBV and RRV. I believe the gut plays a big role toward treatments and hopefully a cure.
Virginia,
Do you mind me asking who you dr is?
I need to find any dr that knows anything about this disease. Just have a GP.
Thanks.
FYI I got covid this past July and after taking paxlovid I felt like my old self again for two whole days. It was amazing. I have tested positive for EBV, Coxsackie and HHV6 in the past. Since Coxsackie is the only RNA virus of those three I assume it is the culprit for my CFS of 11+ years. I wish I could try a long term regimen of a low dose RNA antiviral but it’s unlikely any doc would prescribe it.