The big news in David Systrom’s “Neurovascular Dysregulation Underlies Exercise Intolerance in ME/CFS” talk at the CDC yesterday was that the $8 million mitochondrial drug trial had, unfortunately, failed.
The ME/CFS Mitochondrial Drug Trial Fails

David Systrom reported the Astellas mitochondrial drug trial in ME/CFS was ended early.
Mitochondrial disease is usually associated with genetic abnormalities, but Systrom emphasized that most mitochondrial diseases are not genetic but are “acquired”; that is, at some point, an autoimmune or other process has taken an axe to the mitochondria. As there’s no evidence of mitochondrial disease at the genetic level in ME/CFS, if the mitochondria are involved – and we know they are in at least some people with ME/CFS – the mitochondrial problems were acquired later in life.
One way we know the mitochondria are involved is that a muscle biopsy done at Baylor found citrate synthase deficiency in 10/11 patients. That was the result that got Astellas involved in their $8 million mitochondrial drug study in ME/CFS. Its drug seemed like a great fit for ME/CFS. The participants were carefully selected – all had low oxygen extraction rates – and the drug, from what I could tell, was designed to enhance peroxisome function and fatty acid metabolism – two rather prominent findings in ME/CFS.
An interim analysis of the results, however, indicated that the drug failed to meet its primary endpoint – increase VO2 max (energy production) during exercise – and the company halted the study. The good news is that 3 quarters of the patients had already gone through the study by that time, leaving Systrom and his team with plenty of valuable data to chew on.
If he can do some “omics” studies, he may be able to tell where the drug failed and where it may have helped – giving us a clue as to what’s going on. Did the drug, for instance, enhance fatty acid metabolism? If it did enhance it – but not enhance energy production – then we know that more than fatty acid metabolism is involved in that. If it didn’t enhance fatty acid metabolism, by understanding how the drug was supposed to enhance that, Systrom could potentially illuminate where the fatty acid metabolism failure point really is in ME/CFS. It’s also possible that the drug was effective in some people but not others.
There is a drug that increases energy production during exercise – at least temporarily – in ME/CFS though, and that’s Mestinon. When given prior to an invasive exercise test, Mestinon increased peak VO2 max in ME/CFS. Instead of directly affecting mitochondrial production, though, Mestinon appears to be increasing blood flows to the muscles by tightening up the veins and propelling more blood back to the heart, thus reducing the preload failure problem.
The results of the one-shot study were encouraging because the effects were immediate, and it usually takes several months for the full effects of Mestinon to show up. Now we need a longer-term trial.
Note that three of the five drugs Systrom reported he uses are designed to increase blood flows in one way or the other:
- Mestinon – tightens the veins, sending more blood back to the heart.
- Florinef – an aldosterone mimic that increases blood volume.
- Midodrine – tightens the veins, sending more blood back to the heart.
- Low dose naltrexone – reduces pain and possibly fatigue through its effects on opioid receptors.
- IVIG/Hizentra (preferred) – if none of the other drugs work and small fiber neuropathy is found – immune modulator.
Coming Full Circle
This time Systrom also brought some history into the equation. He talked briefly about the forerunner to his lab – the Harvard Fatigue Lab – which from 1927–1947 did pioneering work in exercise physiology – pioneering, as in it basically helped invent the field. Exercise physiology wasn’t a field unto itself when the lab started. 300 papers, and 20 years later, it was. It became known for its “self-experimentation”; i.e., the researchers often used themselves as subjects.
In one case, worried about the high death rate among the workers building the Hoover dam outside of Las Vegas, the researchers ran physiological and biochemical studies on themselves as they replicated the workers’ workload. They concluded that, as hot as it was during the day, what the workers really needed was a cooler place to sleep as well as a salt-rich diet.
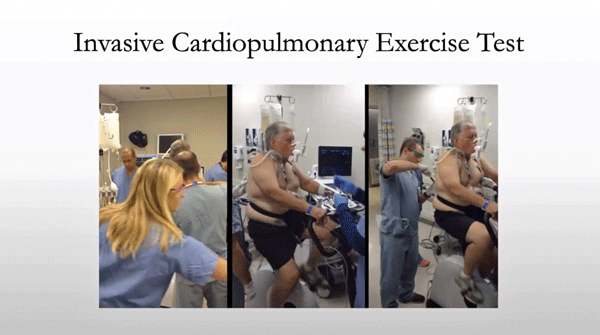
Better bike – better tools – exercise testing today at Harvard.
Systrom said that Harvard had come full circle – fatigue has once again become a prominent subject. Systrom came to ME/CFS because of fatigue, or more accurately, exercise intolerance, after a close chart review of 600 patients who’d taken the invasive exercise test revealed almost 10% exhibited a problem (impaired VO2 peak, i.e., impaired ability to produce energy) which couldn’t be explained by heart and lung problems.
Systrom and colleagues had zeroed in on a unique set of individuals that other exercise physiologists had either missed or had simply passed over. The big clue in this group’s case was the lower filling pressures on either side of their hearts – indicating that their hearts weren’t filling up with normal amounts of blood.
Over time, they came to understand more and more that – undoubtedly unbeknownst to their patients – many of them met the criteria for ME/CFS – and so a new research effort was borne. Systrom first showed up on the ME/CFS scene with a poster at the 2014 IACFS/ME Conference in San Francisco.
The invasive exercise test’s ability to assess both the arterial blood before it gets to the muscles, and the venous blood after it leaves them, is what has made it a game-changer. It’s an expensive and rather arduous test, but it only lasts 5-6 minutes and Systrom reported that most patients do not report a crash afterward.
It’s also the only exercise test that will reliably pick up significant abnormalities during a single exercise session. This is a critical point because a substantial number of people with ME/CFS and long COVID appear to pass a single cardiopulmonary exercise test with flying colors.
As he did with Mestinon, Systrom hopes to put the invasive CPET’s unique properties to use for proof of concept trials to test repurposed drugs. The next step would be to move into clinical trials doing less invasive tests, exercise stressors such as the SHAPE 3-minute step test (perhaps this?) and blood draws.
The Elephant in the Room – Preload Failure
Preload failure – which is caused by an inability to move blood from the lower part of the body up to the heart – is the signature invasive exercise finding in ME/CFS. It’s not only ME/CFS though. It’s also found extensively in POTS and in small fiber neuropathy. It appears to be a tie that binds many people who have these diseases together.
Preload failure means the heart doesn’t have enough blood to move it efficiently to the muscle during exercise. Virtually everyone with ME/CFS has it, but it comes in different flavors.
There’s the preload failure, for instance, that occurs alongside problems with oxygen extraction. Since the mitochondria are fueled by low oxygen extraction, it means they haven’t received all the oxygen they need. This results in higher-than-normal oxygen levels in the blood leaving the muscles, and showing up the venous blood.
This could be caused by mitochondrial problems that keep the mitochondria from taking up the oxygen that’s been delivered to them or a breakdown in the delivery process by something called left-right shunting, which shunts the blood past the capillaries that should be feeding the muscles. Either way, reduced oxygen extraction is found in a significant subset of people with ME/CFS.
Shunting
Medical Definition of Shunt. Shunt: 1) to turn to one side; to divert; to bypass. To move a body fluid, such as cerebrospinal fluid, from one place to another.
They know mitochondrial problems are present and they suspect that left/right shunting occurs. A fibromyalgia study that Health Rising reported on in 2014 demonstrated shunting occurs in the hands of some people with FM.
Systrom noted that an interesting fibromyalgia (FM) connection is present. In 2013, Health Rising reviewed a study, and in 2014 interviewed Frank Rice – a nerve fiber specialist and lead author of that study who had seemingly found an opposite situation – an unusual buildup of small nerve fibers in the hands of people with FM. Those nerve fibers, however, were doing just what Systrom was suggesting in ME/CFS – they were shunting blood in an unusual but different manner.
Instead of the blood being shunted away from the hands it was being shunted into them – and not out of them. Since our hands essentially function as reservoirs of blood that can be quickly redirected to our muscles when we exercise, or our brain as we think, the one-way reservoirs Rice found in FM could imperil their ability to exercise, or think, or even sleep well.
This problem has an interesting possible add-on effect. If enough blood is not getting to the muscles or brain during exercise or thinking, the body will try to squeeze more out of the blood vessels – thus producing the constricted blood vessels Wirth and Scheibenbogen, and others, have speculated is causing problems in ME/CFS.
The Gist
- The failure of the mitochondrial drug trial was unfortunate, but the data gained during the trial could help explain a lot about key problems such as fatty acid metabolism in ME/CFS.
- While the Astellas drug failed, the recent one-off Mestinon study succeeded in increasing energy production (VO2 max) during exercise in ME/CFS patients, apparently by affecting blood flows. In fact, three of the five drugs Systrom reported he uses in ME/CFS affect blood flows in one way or the other.
- Preload failure – the inability to return normal amounts of blood back to the heart – is the signature invasive exercise finding in ME/CFS – and is also commonly found in POTS and people with small fiber neuropathy.
- Preload failure is also accompanied by problems with oxygen extraction by the mitochondria and/or right/left shunting of blood away from the muscles. In one, mitochondrial failure is the cause; in the other, blood vessel problems are – and the treatments for each are completely different.
- While it’s not clear that shunting is present in ME/CFS and long COVID, a fibromyalgia study found evidence that more blood than usual was being shunted into the hands of FM patients and was getting stuck there. Since the hands serve as reservoirs of blood for the rest of the body, getting blood stuck in the hands could negatively affect the blood flows needed for exercise, concentration, etc.
- While attempts to link small fiber neuropathy to the exercise problems in ME/CFS have thus far failed, more extensive assessments of the small nerve fibers are underway and could yield different results.
- A trend towards excessive activation of the TRAIL inflammatory pathway, and inflammatory activity, in general, was upregulated in ME/CFS during exercise. Interestingly, Nancy Klimas’s supercomputer studies plucked out a drug called etanercept that knocks down the end of the TRAIL pathway line – a powerful inflammatory cytokine called TNF-a. A case report of a severe long-COVID patient found that drug to be helpful.
Small Fiber Neuropathy
Small fiber neuropathy (SFN) could potentially explain a lot. If something is damaging the small nerve fibers that direct the blood flows to muscles, then the poor oxygen uptake seen in at least a subset of patients is explained. Systrom has found that about 45% of ME/CFS patients with preload failure had SFN.
Unfortunately, attempts to show that SFN was associated with reduced exercise findings failed. Still, there’s hope as the current biopsy regimen probably does not get at the deeper autonomic nervous system fibers that would be involved anyway. Peter Novak at Harvard is looking into these in ME/CFS.
A TRAIL Trend
Systrom has talked before about an inflammatory pathway called TRAIL (TNF- related apoptosis-inducing ligand) that turns on a powerful inflammatory cytokine called TNF-a but came armed with some new information. He came in contact with a research group that’s interested in TRAIL and ME/CFS. They’re not the only ones interested in a TNF-a/ME/CFS connection, though. TNF-a is the cytokine Dr. Klimas’s supercomputer models told her to target in Gulf War Illness (GWI) and ME/CFS with etanercept. Plus, a spinal infusion of etanercept produced remarkable results in a patient with severe long COVID. It’s nice to see different research groups barking up the same tree.
Systrom said the TRAIL pathway is provoked by a lot of things that cause crashes in ME/CFS; It activates an inflammasome – a thicket of proteins produced by the innate or early immune system response that turn on inflammatory cascades. Because neurons are more susceptible to TRAIL, Systrom wondered if it was causing the small fiber neuropathy found in ME/CFS, POTS, and long COVID.
Systrom assessed TRAIL levels during exercise and found a “trend” for increased TRAIL levels in ME/CFS. (A “trend” is a term for a statistical outcome that approaches “significance” but doesn’t quite get there. Sometimes trends turn into statistically significant findings when a study gets more participants.)
A network analysis of the cytokines produced during exercise found unusually tight connections between the inflammatory cytokines in ME/CFS – suggesting that exercise had elicited a strong inflammatory response – something that Dr. Klimas has found as well. Systrom suggested this very early inflammatory response during exercise could signal the beginning of PEM.
The Long-COVID Difference
The long-COVID study looks remarkably like ME/CFS regarding their exercise physiology, with every long-COVID patient demonstrating preload failure – the signature finding in ME/CFS.
Systrom reported, though, that dyspnea/breathlessness during exercise appears to be more common in long COVID because of higher levels of hyperventilation. We think of hyperventilation in connection with low levels of CO2, but hyperventilation can also impair oxygen delivery to the muscles during exercise. As acidity climbs during exercise, oxygen gets offloaded from the red blood cells more easily – unless hyperventilation is present.
Some differences are bound to appear at some point and right now it appears that two differences between long COVID and ME/CFS may be more coagulation and more blood vessel dysfunction in long COVID.
Health Rising’s BIG (Little) End-of-the-Year Donation Drive Update
Thanks to the over 190 people who have donated over $16,000 to Health Rising thus far.

If analyses that go beyond the headlines float your boat, please support us!
HR was probably the first to bring the news of Systrom’s drug trial and may have been the first to bring the bad news as well. The news wasn’t all bad, though. About 3 quarters of the participants finished the trial, giving Systrom plenty of data he can use to tease out what didn’t work – thus laying the groundwork for understanding what might work. One possibility disappears only to open up a different one.
If going beyond the headlines to try to understand the full picture is something that you appreciate, then please support us.
;







when will Klimas finally begin her study on ME/cfs treatment? it has now been years of waiting.. do you maybe know Cort? thanks!!!
I think its started. It was delayed because of the pandemic but I believe the funding was there and it was going to start up this year. Really eager to hear how it goes in ME/CFS and GWI. It was interesting and encouraging to me that Klimas found different issues in GWI and ME/CFS – but the model spit out the same treatment plan! 🙂
Yes frustratingly slow.
But Good to see progress towards putting the mitochondria theory to bed, another distraction from the immune-inflammatory causation of this illness.
I think all the problem s including autoimmunity are related to tissue type. We got the eds crappy end of the stick.
Everything else is downstream. Everything is weaker in us
So something is whacking the tissues – some autoimmune reaction – certainly a possibility.
To me, we weren’t born with the sane kind of tissue.
It’s more prone.
It’s just that eds research is not understanding what are the different tissue types in humans.
I think, as this thing runs in families, it is genetic.
Autoimmunity, in my opinion is downstream of tissue type.
Whose going to get leaky gut, have more excitable glial cells etc. Someone with weaker tissue.
We are tissue divergent from birth and prone to these things affecting us.
I know Jen Brea feels similar
I’ve always thought there should be an arm of research devoted entirely to regeneration in m.e
Even using crispr , once these tissue matrices differences are. Found out.
Ron Davies has eds. We all know that he got sick with a virus at a young age that went to his heart.
Same thing happened to my mum. Both sons ended up with CFS.
Why? The starting block was some kind of aberration in the tissues. We were never normal, although we have some kind of eds that disguises itself.
I sincerely hope I’m wrong. But I don’t think I am. Of course, I’m a no one but I’ve always noticed the difference between siblings . You can see the tissue differences from early on. My brother inherited my dad’s line, long livers , thicker connective tissue that doesn’t melt if he gets a virus, abd is not prone to autoimmunity
Like I said , I don’t think they’re rewinding this disease to ground zero. Las with my mum and Ron Davies with eds, virus invaded their tissue more easily.
Obviously there will be other factors st play but ground zero is the tissue at birth. I’m sure of that.
I hope crispr or stem cell therapy help. We’re getting rubbed out like a bad drawing
Interesting observations, Oliver. I believe the illness that Ron Davis had you are referring to is Rheumatic Fever, which my father had as a child, as did I and my youngest brother. That same brother had to have an adrenal gland removed last year. Although I have never been diagnosed with EDS, I have had some issues with my knees trying to go backwards on me slightly. My eldest brother is ‘double-jointed’. I do think there is a genetic component that makes us more ‘susceptible’ to ME/CFS. Besides having Rheumatic Fever, I also had Scarlet Fever, Chicken Pox, and Rubella as a child and was exposed to HSV 1 as an adult. When I got sick with ME/CFS 15 years ago, EBV titers were very high. I remember kidding with a friend that I was a ‘virus magnet’. Not so funny now… I now feel I have a ‘virus stew’ simmering inside me. Throw in candida plus gastric issues and some questionable meds through the years… Ye gads!
Do you know all forms of EDS are a collagen mutation?
So I read the link about hands posted in 2013. I wasn’t reading Health Rising back then as I was immeshed in learning about EDS. When I came across the comment about raising ones hands to think better I decided to try it. I’m another one who has cold hands and feet while sitting and some Renaud’s–and also have sweating menopausal hot flashes about once an hour. If I happen to vigorously exercise, my hands get hot and red too…
Anyway, I was sitting here with cold hands (and problems with word finding), so I held up my hands and by God, they started to tingle and became warmer! Weird! And here I thought the blood draining downward would cause them to become even colder.
My little N=1 has convinced me there really is something wrong with my oxygen/blood flow. Food for thought. Thank you Cort! And thanks for the new addition of e-mail notification if one gets a response to their posting.
I really wish that someone would do a deep dive into tissue structure in eds. I have a friend diagnosed with type three eds. I’m slightly tougher, bodywise but basically have all his symptoms, but I didn’t make the diagnosis.
That’s what I think m.e. is. Sone kind ofsubtler type of eds at ground zero.
The blood flow issues etc stem from there.
You can SEE the tissue in other pt. I always recognised a fellow sufferer if I’m well enough to be out.
We have to remember, we are not all equal are we? Some people just aren’t prone to leaky gut etc and it’s because they have tight epithelial structure in their gut.
I think Jen Brea is the only one looking into this with a tiny company.
Crispr, if they find the fault would be ideal. Hook you up to a bag and voila, fixed.
I know it’ll never be that easy but you gotta have hope
Oliver, the medical community hasn’t referred to Hypermobile EDS as Type 3 for years. There was a huge Symposium around 2017 and they recognized that EDS is often a continuum and people who did not quite meet the formal criteria of Hypermobile Ehlers-Danlos (hEDS) could be classified as having Hypermobility Spectrum Disorders (HSD). For a while, researchers argued that the two were separate, but now, I think, most say that they are just versions of the same thing. But then again, if your doctor doesn’t recognize that, well, that can be a disadvantage…
I’ll bet a skin study like the one Rice did would be fascinating. Unfortunately, it appears he was not able to get an NIH grant to continue his work in FM.
A lot of research looks at the end products of our problems, far less about where it all starts. Blood delivers oxygen and nutrients and removes waste.
When 90 % of ME/CFS patients have orthostatic intolerance (OI) (van Campen, Rowe, Vissser 2018), then they also must have venous pooling. Many patients also have lowered blood volumes. If the blood is thick and sticky (Gijs, deze is voor u) and red blood cells are less flexible (Saha, 2019) that could be the starting point of a whole lot of trouble. Pumping up thick and sticky blood leads to smaller pre-load of the heart, lower heart output, lowered cerebral blood flow, which leads to brainfog.
Comments are welcome!
Have you ever tried pumping up ketchup in a plant spray bottle. No spray or stream, just a mess.
Is low blood volume messing up a lot of research? Dr. David Bell (Cort’s blog 30 sept 2015 noticed that 42% of hematocrit in ME/CFS is a lot lower than 42% in healthy people. (page 2).
Does that mean that ALL lab results are lower then they look?.
When I ask a doctor about this he\she says, “That’s concentrate”. But most concentrates are written down with “/L” at the end, which means per liter. When a liter or more is missing I also miss out on what would have been in that liter. When all looks well in ME/CFS lab results, we always come up “healthy”, but is that true? If all results have to be recalculated after blood volume testing, who knows what that could mean for researchers: They might find things a whole lot faster.
For me, I don’t think the vascular system is strong. The epithelium relies on smooth muscle, which I feel will not be like a strong individuals cardio. We just drew a bad lot.
I’m hoping there’s treatments and even cures but this illness is so systemic, it has to be at tissue level where this is occurring
I meant the vascular system, it’s collagen it’s muscle tone is weak to begin with. From birth. It only be ones apparent as we age and life’s trials hit us, stress, viruses, glyphosate etc. Then an already not so strong structure gets weajend. What you’re seeing in orthostatic intolerance is a deregulation to the point if the vascular system being broken. The cell danger response kicks in, shuts down nitric oxide as a protective mechanism. Again. I hope I’m wrong
Have you ever seen jack Kruse’s work on CFS. He’s an arrogant son of a gun but he says m.e is a loss of electrons which eventually leads to whole body dehydration.
I sent that work to the open medicine foundation and they said ” how do you know about this, were just studying this loss of electrons and subsequent dehydration and loss of blood volume.
It was jack Kruse’s ideas not mine!!!
It’s some downstream metabolic consequence of dysregulating the circadian rhythm amongst other things such as lack of sunlight etc creating the charge we need. Leaks the energy out of the mitochondria.
I was glad omf were looking at it.anyway.
ME/CFS = an extremely reduced state
My faith lies with Open Medicine Foundation…Ron Davis and his stellar team. 🙂
🙂 and note that Systrom is now co-leading the OMF-funded Harvard ME/CFS Research Center – so he’s part of the team 🙂
Thank you Anneke, (goed verwoord!), well said!
I’m so sorry Gijs, I messed up. I meant to mention Konijn, from Belgium. Thanks for the compliment anyway. If you compliment me about how I said it does that imply that the content doesn’t appeal to you. If it’s nonsense, in your eyes, you’re allowed to say so. I’m only 1,65 m but also a big girl.
Haha no problem -:) Ook medisch inhoudelijk vind ik het erg goed.
That’s an even better compliment. Thanks!! I hope more people will think about it thoroughly. Especially researchers like you. And specialists. Met one recently at a chronic fatigue department (university hospital). The specialist knew zilch about ME/CFS. The only test he offered me was hematocrit.
He was paid for “hot air”. (dutch: gebakken lucht)
Things are moving, great! CPET, however, seems to be an awefully laborious and expensive research tool with significant side effects. Also, it excludes those patients from which a lot could be learned – the very sick and bedbound. So I would be very interested to know if Systrom may have found an easier proxy value in his studies which may be a fair alternative to CPET?
Venous oxygen saturation may come to one´s mind, and there have been a few reports of this test being helpful. For ex. here:
https://xdrx.substack.com/p/how-i-recovered-from-long-covid-using
Fascinating stuff – thanks!
Voici une jolie histoire ; C’est un ancien animateur de télévision Française de plus de 70 ans qui a fait une chute à vélo . Blessé légèrement, il est pourtant paralysé des doigts et des jambes. Il aurait, semble-t-il contracté un virus, et développé une maladie auto-immune. Il s’est retrouvé allongé sans pouvoir marché pendant 2 ans. Il va à Paris toutes les 6 semaines pratiquer une PLASMAPHERESE, et maintenant il remarche et n’attends qu’une chose c’est de reprendre le vélo…
Il a eu de la chance d’être bien orienté et d’avoir trouvé un traitement qui lui convient.
j’ai aussi un problème avec l’oxygène/sang. Je fais des petites ischémies (micro-caillots qui se forment) à l’effort., et j’ai souvent la respiration coupée , comme si l ‘oxygène n’arrivait pas jusqu’au coeur et poumons. Surtout quand je suis debout ou que je fais des efforts. Maladie de Raynaud également.
If you consider the work of Bupesch Prusty (mitochondrial fragmentation, factor X in blood of pwme, and now MiRNA hhv6 EBV in brain and spinal cord of patients with ME but not controls), I’m not really surprised tbh.
If Mitochondria “fall apart” in ME, you can try anything to push energy in/out cells/tissue, but it won’t work.
Then there are the “GPCR auto-antibodies” found in ME patients.
Some of which lead directly to vasoconstriction.
I tried mestinon. Couldn’t tolerate it.
Now i know why.
I test positive for GPCR auto-antibodies expressed mainly in the CNS and directly leading to vasoconstriction.
Mestinon constricts even more.
I of course wished this trial would lead to a solution or success.
I wonder if this perhaps would fit for people with POTS / OI issues NOT having ME?
ME patients have ‘all-over’ mitochondrial fragmentation and (virally/immune induced?) brain/CNS dysfunction at the ‘core’ of their illness.
Pots/oi/heart/… (logically) seem to be consequences. Not ’drivers’ of the disease.
The positive?
Even when a trial doesn’t hold up, it’s still valuable information.
You can go back to the drawing board knowing ‘this isn’t it’.
I admire each and every scientist/doctor trying to get to the bottom of this cruel illness.
Whether they succeed of fail.
They try.
They are exceptional white ravens in a sea of scientific & medical “ME-nothingness”.
Interesting study.
(…)Here in this paper, we attempted to analyze active HHV-6 transcripts in postmortem tissue biopsies from a small cohort of ME/CFS patients and matched controls by fluorescence in situ hybridization using a probe against HHV-6 microRNA (miRNA), miR-aU14. Our results show abundant viral miRNA in various regions of the human brain and associated neuronal tissues including the spinal cord that is only detected in ME/CFS patients and not in controls.
Our findings provide evidence of tissue-specific active HHV-6 and EBV infection in ME/CFS, which along with recent work demonstrating a possible relationship between herpesvirus infection and ME/CFS, provide grounds for renewed discussion on the role of herpesviruses in ME/CFS.
https://ammes.org/2022/12/05/tissue-specific-signature-of-hhv-6-infection-in-me-cfs/
Prusty published a paper in Nature in May showing that hhv6 miRNA inhibits host miRNA to screw up the latent/lytic cycle which screws up mitochondrial architecture . The paper is behind a paywall unfortunately so I’ve only seen the abstract. This fits with your interpretation of the medication trial failure: meds wouldn’t work on physically damaged mitochondria.
https://pubmed.ncbi.nlm.nih.gov/35508655/
The Nature paper is behind a paywall, however there is a preprint that is available:
https://assets.researchsquare.com/files/rs-820696/v1/b132da27-3109-46c4-832a-34ede3829dce.pdf?c=1651751711
Selective inhibition of microRNA processing by a
herpesvirus-encoded microRNA triggers virus
reactivation from latency
Published under Creative Commons. On a cursory review, the images look identical to the ones in the peer-reviewed Nature paper, but remember that preprints may contain errors fixed in the published final version.
I am taking two of the five drugs Dr.Systrom uses to increase blood flow, Midodrine and Low Dose Naltrexone. I have been taking the Midodrine since 2007 and definitely notice a rapid decline if I do not take it. I took the LDN on and off from 2010-2014, stopped taking it for a number of years, then started taking it again last year. I’m not really sure how much of a difference the LDN makes for me but, as long as I can tolerate it without a problem, will continue taking it as I see more and more doctors are prescribing it for their patients so it must be doing something good!
I feel that Orthostatic intolerance could be the whole deal for me, brought on by menopause. I have had vasovagal syncope (prodrome only) several times through my life, and had Raynauds mildly premenopause. so some blood flow issues prior to menopause, though healthy and sporty. Since menopause I’ve been exercise intolerant, alcohol intolerant. Slightly fatigued after exertion and brain foggy for a few years but much better now. I’m very interested in Systroms work. The only thing that hints at more than OI for me is that I don’t ever get colds anymore
Perhaps because of division of labor – that it is usually a cardiologist that prescribes the florinef to the POTS patient,
I’ve found it odd that this doesn’t make them stop for a minute and think about it being an endocrinological problem. Florinef is used for people with adrenal problems.
Cold hands = stress hormones running the show because of a lack of thyroid function.
Cold hands = hypothyroidism
Not rocket science…
We should consider the possibility that the trial failed because the hypothesis was wrong. I understand the desire to leave no stone unturned, but the sooner they move on from mitochondrial or vascular theories, the better, imo. As it is often the case in science with failed hypothesis. There of course are exceptions, like beta amyloid theory in Alzheimer’s. But those are hypotheses that explain everything about the disease. It might be reasonable to question in such cases if the trial was actually testing what they thought was testing. Mitochondrial hypothesis about MECFS isn’t one of them: it has to go on a limb to explain things like brain fog and PEM.
Exactly!!!
It’s important to remember that Dr. Systrom has stated that only 20-25% of ME/CFS patients (in his clinical practice/research studies) show strong indicators of mitochondrial dysfunction (i.e., impaired systemic oxygen extraction). So it’s true that the mitochondrial hypothesis doesn’t apply to everyone with ME/CFS, but it might play a role in a subset of people with ME/CFS.
Also, besides red blood cells, mitochondria are in every cell in our bodies, including immune cells and brain cells. So mitochondrial dysfunction might be able to explain other symptoms besides fatigue (in the 20-25% of ME/CFS patients who show indicators of mitochondrial dysfunction). For example, the common symptoms experienced by patients with mitochondrial diseases are multi-systemic (including brain fog-like symptoms) and are surprisingly similar to ME/CFS symptoms. In addition, a major part of the treatment plan for mitochondrial diseases includes pacing and energy management strategies because according to Mitoaction.org, “activities well beyond your baseline will drain your energy bucket and cause negative symptoms such as pain, fatigue, and even signs of organ dysfunction if severe.” That sounds very similar to PEM to me, but it’s describing what happens in people with mitochondrial diseases.
So even though the mitochondrial hypothesis might not apply to all people with ME/CFS, it’s seems like a worthwhile line of research for at least a subset of ME/CFS patients.
Agreed!
It sounded strange for a multinational pharma to spend $8m on a chance that 20-25% of patients may benefit, so I looked at the study. Turned out, it mentions nothing about MECFS; it’s a study about people with reduced maximum oxygen intake, no more no less. So, even if the study succeeded, it would have had no implication for MECFS till they repeat it with an applicable subset, if such subset exists. (Subset is another notion that I think needs to be dispelled, but that is another topic).
In any case, PEM that I’m familiar with, and I suspect most of MECFS patients are, is not a drainage/fatigue after an exertion that you quoted from mitocondria.org. It’s more like something that hit you like a bus after a 24-48 hour delay. I used to be amazed how punctually it arrived almost exactly at 24 hr mark after a slow walk that made me feel better.
Dont get swayed by the title of the study – it simply refers in technical terms to the physiological state of the participants. The participants all came from Systrom and he’s been referring to the low oxygen extraction subset of ME/CFS patients for years. Plus all the participants in the study presumably had ME/CFS. Systrom looked over the blog I wrote stating that this an ME/CFS clinical trial and found it was fine.
The description of the $8m clinical trial on clinicaltrials.gov only states that they’re studying the effects of the drug on “reduced maximum oxygen uptake due to poor systemic oxygen extraction,” and doesn’t mention ME/CFS. I’m not sure why they didn’t include ME/CFS in this description but instead described a major symptom of ME/CFS (i.e., almost all ME/CFS patients show “reduced maximum oxygen uptake” during cardiopulmonary exercise testing and is an objective measure of both fatigue and functional ability levels). According to Dr. Systrom, this clinical trial is only enrolling ME/CFS patients (and a few Long COVID patients) who showed indicators of mitochondrial dysfunction during an iCPET (i.e. their reduced maximum oxygen uptake was due to poor systemic oxygen extraction).
We know that at least a subset of ME/CFS patients have mitochondrial dysfunction because that was found in 10 of the 11 patient muscle biopsies Systrom sent to an outside lab.
Thanks Cort, that’s good to know. Then the result of the trial does have an implication on the mitochondrial hypothesis of MECFS as I first thought.
KLB, my guess is that this trial is really meant to test drug’s efficacy for mitochondrial dysfunction, and therefore no mention of MECFS in the title. The subjects just happened to come from MECFS cohorts with mitochondrial problem. That way, the company gets to test the drug for mitochondrial dysfunction, and Dr. Systrom gets to test his mitochondrial hypothesis of MECFS in the process. Kill 2 birds with one stone, so to speak.
I would encourage you to stay with LDN. Dr. Klimas’ group has noted that it is effective in increasing Natural Killer Cells in both quantity and function. This effect would not be identifiable to an individual unless NK cells were tested regularly as in annual exams.
Additionally, my doctor believes that LDN effects the HPA axis in a positive way. This came up when I no longer needed adrenal supplements after being on LDN for about 9 months.
Fibromyalgia patients have had some success with reducing pain with LDN.
Hi there and thanks for the reply. Yes sane here. Rheu atic fever for my mum.
I also noticed Whitney’s mum and a brother who died fairly early as did my mother’s fatger of heart disease at 46. All the make members of his side lived short lives. Of course I know times have changed but I’ve cone to the conclusion we have an inherent tissue weakness. It allows viruses in. It generates mast cell activation, synonymous with poor blood flow, less muscle mass ( which holds tissue to bobe)
Yes I think eds is so poorly understood at the moment. I think the really serious cases are noted early, and defined as eds type whatever, but there are loads of us who are subclinical, or a type not known to science yet and we break more easily.
Everyone has a breaking point but why are ourselves breached more easily. What is resilience. To me it’s tissue type. Thees even the phrase, thin skinned isn’t there.
To me, all problems are down stream from this ground zero and the bud out into different branches of expression.
I also, perhaps controversial ly have an inkling that this is part if nature’s way of creating an alarm system abd hierachy in one set if people for the tribe. We notice, we feel, we suffer more easily and generally we have less dopamine and ate more likely to be submissive in relation.
So what do we do? I think the best bet is gene editing, stem cell therapy.
There’s a woman working from Panama who has started exuting her own muscle mass and markersfor ageing, her name escapes me, but she is trying to find a cure for type one diabetes for her son. Part of that process was to reverse the ageing process metabolically.
As we’ve seen recently, have metabolic issues related to tissue. I wish someone would link up with her
Like I say, regeneration seems like the best way
There was a time when I was well, before my allostatic load, my limits were breached.
Hey I’m also similar to you in that I feel I’ve reactivated my ebv recently. It’s not fun is it.
,Less collagen always means less health. Surely there’s sone way to rebuild colkagen
Would one expect orthostatic diastolic hypertension to be an objective sign of ‘preload failure’? The diastolic phase is when the heart fills up with blood I believe. Thanks for any insights.
What if you don’t have ME/CFS? What if you have something else that IS treatable? Or what if your genetic makeup has made you more susceptible to develop ME/CFS or Long Covid?
Enter POSSUM. The POSSUM database includes information on over 5000 syndromes, including multiple malformations, chromosomal abnormalities, skeletal dysplasias, metabolic disorders, linked to over 30,000 images including photos, Xrays, scans, diagrams, and histology.
The diagnosing doctor enters your symptoms in POSSUM and the program churns out a report with the possible condition(s) you may have. https://www.possum.net.au/
POSSUM is a subscription service and may be found in some larger university settings, but you could contact the company to find out if it is available in your area.
All mitochondrial diseases are genetic. A genetic disease can be in-born, inherited, or acquired. If you have a mitochondrial disease that was acquired, that means you have an acquired, genetic disease. Genetic does not mean in-born. Genetic also does not mean hereditary. Genetic means that it has to do with genetic changes. Genetic changes can be acquired.