In 2011 the CFIDS Association (now the Solve ME/CFS Initiative) engaged a “drug repositioning” company called Biovista to search through thousands of drugs to find a new approach to ME/CFS.

Biovista threw information on ME/CFS into its huge database to look for new drug possibilities for the disease.
Biovista’s algorithm identified “every known gene, pathway, disease, anatomical location, cell structure and other components of potential drugs, including why and how they succeeded or failed, as well as potential side effects and drug/drug interactions”. They threw that, along with every bit of information on ME/CFS symptoms, pathophysiology, and treatment, they could find, into one pot.
Biovista’s apparently had a pretty good track record. It had previously identified two possible new drugs for progressive multiple sclerosis as well as drugs for brain cancer, thyroid cancer, and melanoma. In 2012 the firm boasted a 70 percent success rate in finding drugs that turned out to be efficacious in diseases.
It was exciting stuff and a good gamble.
In 2012 the Solve ME/CFS Initiative announced that a new drug combination had been found and that Biovista would attempt to find partners to finance drug trials. Three years later, (no drug trials in sight) and the Solve ME/CFS Initiative’s contractual obligations of secrecy over, the SMCI announced the results.
Biovista’s Drug Combo for Chronic Fatigue Syndrome (ME/CFS)
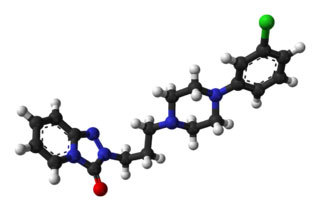
Biovista stated its platform enabled it to find “non-obvious correlations between drugs, molecular targets, pathways, adverse events and diseases.” The two drugs it came up with – low dose naltrexone and trazodone – ended up looking pretty obvious. That wasn’t necessarily the case in 2011, however, when Biovista’s search started.
Only one small study on low-dose naltrexone in FM had been done by then and LDN was probably not used much in ME/CFS. Trazodone was probably used more but only one study – involving a mouse model of ME/CFS – had been done on it.
While we didn’t get the out-of-box drug combination that made us think of ME/CFS in new ways or a drug trial the drugs Biovista targeted did have one really interesting thing in common – they both targeted glial cells – ground zero for neuroinflammation.
Low Dose Naltrexone – Glial Cell Inhibitor #1
Naltrexone is an opioid receptor antagonist that’s used in opioid withdrawal. Besides blocking opioid receptors it has the nice side-effect of increasing levels of the feel-good chemicals called endorphins.
The low-dose form of naltrexone (LDN), on the other hand, is believed to reduce inflammation by blocking TLR4 receptors on the microglia. LDN has been proposed to be useful in many diseases but most reports are anecdotal and few studies have been done. Much remains to be learned about this intriguing compound.
The fact that LDN popped out of Biovista’s search suggests, however, that Jarred Younger may be on the right track with his focus on microglial inhibitors for ME/CFS.
Trazodone – Glial Cell Inhibitor #2
Trazodone is not your average antidepressant. Structurally very different from other antidepressants, this triazolopyridine derivative has complex effects on serotonin not seen in other antidepressants. It also reduces arousal and has some anti-histamine effects.
Sleep Aid
Trazodone – not your average antidepressant – can be useful in low doses with sleep and may be able to reduce neuroinflammation.
Trazodone is used more as a sleep aid than as an antidepressant in chronic fatigue syndrome. (As with LDN, lower doses than usual are used to improve sleep.) It’s one of the few drugs (Zyrem is another) able to reduce the activity of the alpha waves known to hamper sleep in fibromyalgia.
A 2011 open-label FM study found that Trazodone significantly improved sleep quality, duration, and efficiency. The authors called the increase in sleep quality ‘striking’. Another 2011 fibromyalgia trial study found that Trazodone in combination with Lyrica (pregabalin) improved pain, anxiety, and morning stiffness.
The IACFS/ME Treatment Primer reported that Trazodone might be able to maintain its effects over time better than any other sleep drug. Dr. Bell and Dr. Lapp both promoted Trazodone use, with Dr. Bell stating Trazodone was one of his favorite sleep medications. Trazodone may be particularly effective in reducing the “hyperarousal” that appears to be present during sleep (and other times) in ME/CFS and FM.
Neuroinflammation Reducer
A 2015 study suggested that Trazodone may be producing its effects in depression by reducing neuroinflammation. Neuroinflammation is common not just in neurodegenerative diseases like Parkinson’s, Alzheimer’s, and multiple sclerosis but in depression as well.
In a mouse study, Trazodone upregulated levels of BDNF – a brain growth factor. BDNF appears to play a critical role in pain sensitization and neuroplasticity. BDNF levels appear to be high in FM but low in ME/CFS.
Trazodone prevented glial cells called astrocytes, from spewing out pro-inflammatory cytokines when confronted with an inflammatory stressor. If Younger and Miller are right about microglial cells over-reacting to small amounts of inflammation, a drug like Trazodone could be beneficial.
Trazodone also appears to affect astrocyte metabolism as well. The glia cells in the brains of mice respond to inflammatory triggers by pumping out lactate – a substance found elevated in the brains of ME/CFS patients. When given 72 hours prior to an inflammatory insult Trazodone enhanced lactate release. (Lactate may be produced in response to stress/pathogens, but the substance itself is believed to have neuroprotective effects.)
One of drug repurposing lessons is the different effects drugs can have in the body. Antidepressants aren’t just antidepressants anymore. The same drugs that relieve depression can relieve pain in people who are not depressed.
Nor is depression a purely psychological disorder either. A significant subset of people with depression are believed to have inflammation-driven depression. Given the possible immune involvement in ME/CFS and FM, it’s possible that much of the depression is, in fact, immune-based. If that’s true then perhaps two treatments targeting the microglial could be effective both in depressed and non-depressed ME/CFS patients.
Unfortunately, we don’t know why Biovista’s results plucked out these two possible glial cell inhibiting treatments. (Biovista did not return queries.) They may synergize in ways we can’t imagine.
Conclusion

If neuroinflammation is found in ME/CFS the two-drug combination might be an option for a treatment trial
The results teamed together two drugs that may target a hot topic in ME/CFS – glial cells in the central nervous system.
Although Biovista failed to get a clinical trial going, if imaging studies over the next year or two find evidence of neuroinflammation, we may have a drug combo backed by solid evidence that the NIH could use to get a study underway. A successful LDN/trazodone study demonstrating improvement which was correlated with reductions in neuroinflammation wouldn’t be the answer to ME/CFS, but it could be a good first step.






LDN at 4.5 has made the pain change from an 8 to a 2, dome days a 1 for my teenage daughter diagnosed with CFS
Cort I got a good and much needed laugh from your description of Viagra giving men “uplifting results.” Thanks!
Do you know why some folks are getting generic avatars in the comment section? Since I take trazadone and tried LDN to no avail, I’d really like to read them! Thanks.
I have been taking a low dose of naltrexone for about three months now. It does seem to reduce the frequency of pain, for which I am thankful. On the other hand, unpleasant dreams and night sweats are a side effect for me. I stopped taking it for a couple of weeks to check out this correlation and it is empirically true.
For those of you on LDN, who, please is prescribing this for you?
The CFS specialists I have contacted do not prescribe it.
Specific clinicians, specialities and names would be helpful.
Thank you, each.
Hi Galia, I got a prescription for LDN from my CFS doctor. He didn’t suggest it, because he thought it was just for FM, but he wrote the prescription. He is Dr. Barry Elson at Northampton Wellness Associates in Northampton, MA. He’s not perfect, but he is very nice and knows a lot about CFS and FM and has been treating these chronic illnesses for decades. He does a lot of testing and treating, but I have to prompt him to keep trying things for me.
Galia, the other thing about Dr. Elson is that he practices integrative medicine, meaning he does a lot with nutritional supplements, with less emphasis on drugs though he prescribes those, too. And he has an IV department. People come to his clinic from all over New England to get IV treatments for various things.
I have been taking LDN for a couple years now. I am using it to control my autoimmune diseases which it is doing quite well. It isn’t curing my CFS or FM but my quality of life is much better.
There are some things CFS patients need to know aobut LDN. AS it moves the upregulated TH2 immune system pathway back towards a more balanced state between TH1 and TH2, if you have candida it can become much worse and can even interfer with the LDN. You need to be ready to treat for candida when that happens or better yet, treat candida before or when you start LDN. If LDN was working and then it stops, first look at possible candida infection as the cause. Also the sleep issues in CFS can become even more of a problem with LDN. There is an LDN group on Yahoo that offers alot of really good information as well as some too generalized or bad information so be careful. Using LDN on CFS or Fibro patients requires more knowledge. My PCP has learned to start these patients at much lower doses of LDN and increase the dose more slowly. In my experience, the Yahoo LDN group as a whole doesnt have the right knowledge to advise CFS/FM patients about LDN and some of members blame the patients when they have problems with LDN. But the goup can offer a list of treating physicians and a list of pharmacies to compound the LDN or where to get it without a script. And they offer information and a perspective that I hadn’t found elsewhere. I have used information from the group to educate my doctors on LDN. On thir advise, I finally started taking LDN in the morning to correct sleep problems compounded by LDN. I was surprised but morning dosing is working for me.
A side note, I wonder if this company was going to a combo approach because while LDN is off patent, a combo could be patented.
To find local doctors using LDN, you can try calling your local compounding pharmacy to ask if they make LDN and if yes, what doctors are prescribing it.
I order mine online from a pharmacy based in Israel that also donates to LDN research.
I have a rheumatologist who gave me an rx for it, but my sister and a friend with RA have doctors who refuse. ldnscience.org has the info.
Any doctor can prescribe LDN. Start with only a tiny amount!
Galia I have been seeing Dr. Irma Rey In Kendall Florida since 2011 at Nova Southeastern University,Institute for Neuro-Immune Medicine. She is an awesome doctor. She started me on LDN 1mg. going up slowly per 1/2 a mg. and I am currently at 3.4 mg. She also has me taking a anti-depressant, Remron or generic is Mirtazapine also low dose 7 mg. at night as a sleep aid.Megan
I believe LDN Research Trust has a list of physicians who will prescribe. Also check out the resource page on Health Rising mentioned in the blog. Lots of stuff on there.
Start here http://www.ldnresearchtrust.org/LDN_Prescribing_Doctors-OLD
If you can’t find a doctor in your area to prescribe this is an option, albeit an expensive on for a lot of people. https://ldndoctor.com
I have been taking 4.5mg Naltrexone at night before bed for about three years now. I am treated through Holtorf Medical Group in Torrance, CA. The physician I see there presently is Dr. Wayne Wightman, however all the docs there treat CFS and FM patients. I live in South Dakota, and was very surprised that my health insurance actually covers the full cost of the drug!
I’m astonished as well. How the heck did that happen? 🙂
Ihave been on LDN 4 mg first year now 8 mg oo. My Rheumatology (FM) doc manages for me. Stops the painful inflammational deep pain Of al four extremities from top to end of phalangies. Still have joint pain osteo and brainy fog and Dysautonomia. Having k née surgery Feb 15 . Home this makes a big fifference
Lindsey –
Skip’s Pharmacy is a compounding pharmacist. You can order on line & they will ship to you. Skip is highly educated with LDN and they DO return phone calls! He is highly recommended in the LDN community and I highly recommend him as well. You get a script from your doctor, mail, fax or upload the script & email it to them. They will get back to you and get your script off to you. Prices are great, if I recall it was about $25 per month. Here is the contact info…
21000 Boca Rio Rd
Suite A-29
Boca Raton, Florida
33433
561-218-0111
800-553-7429
Fax: 561-218-8873
Also, they have a FB page and their website is, http://www.skipspharmacy.com/
Galia, I just saw Dr. Montoya’s PA at Stanford a few days ago and she prescribed LDN. she has me starting on 1mg in the morning for two weeks, then 2mg for two weeks continuing to titrate up until I reach 4mg daily.
When they receive the results of my labs in 3 weeks they will choose an antiviral medication based on that – but I won’t start it for 12 weeks, until it’s been established that I’m doing well on the LDN.
I would love to hear from other patients of DR. MONTOYA about how this treatment protocol is working for them and what sort of timeline I should expect for results!
Hi Rachel,
I saw the PA last September — you were smart to get the labs done there, since I’m in an HMO I wanted to have my doctor order them and long story short, after 2 blood draws, am still lacking 3 and tired of messing with them. The PA had enough though and went ahead and put me on an antiviral 2 weeks ago. Be sure and check prices, the first one she ordered was going to cost me 100 bucks a month so I asked if there was any other she’d consider and there was – at 2 bucks a month. I don’t understand why you have to wait 3 months to see how the LDN is affecting you – maybe she’d give you less of the antiviral if the LDN is helping? I’d already tried it to no avail so it didn’t play into my treatment. I don’t hold out much hope antivirals will help but they do for some, so it’s worth a try. I don’t understand what people mean who post that they’re trying to get rid of viruses in their body — how do they know they have a chronic issue with viruses in their body, if that was possible wouldn’t the mystery of CFIDS be solved? From what I understand it’s the antibodies that are measured, but they only tell if you’ve had a certain virus in the pas so these can show up high in anybody. If anyone reads this and can explain this to me, please do. She did say that one test showed my Epstein Barr had been reactivated but I don’t think that even means it’s acting up now, but I don’t know. Maybe we should start a Stanford group on the forum, or maybe there already is one, I haven’t been on the site for awhile. Good luck and really hope you’re helped with these new treatments you’re trying.
Thanks, Betsy – yes it would be great to have a Stanford forum, I will check HR for one! I have a lot of questions too and am making a list for the PA – we have a March 3rd phone consult. Something curious, bewildering even – Montoya’s PA had NEVER heard of Ron Davis. Huh?!?
Does anyone who uses LDN, have pain apart from their Fibromyalgia, such as I have severe pain problems due to degenerative disc disease. I take a very low dose of hydrocodone 7.5, which just knocks the edge off barely, because anything higher knocks my socks off and I am useless. That’s why I want my state to recognize Medical Marijuana for this condition so badly, CBD without a little THC has little effect on DD. So any one have experience with FIBROMYALGIA WITH MAJOR SPINE PAIN AND LDN USE? Not shouting just getting attention.
Deborah – I was on LDN for fibro & was doing very well until i was diagnosed with several herniated discs in both my neck & lower spine. For me LDN worked great for fibro until the disc issues. Then i went off of it & went on oxycodone & a fentanyl patch. Right now I’m weaning off the patch because it came to a point where the pain doc wasn’t comfortable in increasing my monthly dosage anymore as they had been doing monthly. And my body got use to the dosage. So i didn’t feel as though i was benefiting from it anymore. I’m finding now that the stiffness from fibro is the most obvious pain. I no longer go to bed at night. I just nap sitting up on the couch because my pain & stiffness sets in after being still sleeping for more than 3 hours. This way i get up every couple hours to move around to avoid the excrutiating pain of laying down for 5 or 6 hours that brings me to tears upon waking. I’m appealing my disability denial. Id give my left arm to be healthy.
Hi Rachel, That’s really odd that the PA didn’t know Ron Davis. I did see some discussions on antivirals but no Stanford discussion group on the forum, unless I missed it. My email is girl42@comcast.net if you ever want to compare notes…..
Galia – pls e-mail me jmbjar@Hotmail.com for help in the question u ask
My neurologist prescribed it for me. Call compounding pharmacies in your area and see who the top docs are that are prescribing LDN. See if any match up to docs you are using it can use. I did this and found one of my docs is a prescriber.
I get it from my Functional Need doc but I’ve read that you can get it from a doc on http://www.ldndoctor.com. Or by calling a compounding pharmacy who sells it, like Skips in Florida or Belmar in Colorado. They may be able to direct you to a prescribing doc.
Go to a rheumatologist or naturopath
Take it in the morning.
The symptoms do go away, if you can hold out a few more months, i have been on it 4.5mg. for 8 months, and no more night sweats or weird dreams.
Can I take LDN in the morning ?
Katie, maybe look into 5-HTP – it’s known to prevent bad dreams and promote restful sleep. I am trying to find examples of where people have successfully used LDN and 5-HTP together as I’m considering that approach as well.
Did you night sweats ever go away? I have same issue and doctor hasnt heard of this issue.
Does trazodone have a withdrawal syndrome?
What low dose have people found most effective?
I started at 50 mg and went up to 100mg, which I’ve taken for years, maybe 15. I believe the antidepressant dose is 3-400 mg. I’ve gone off if trazadone several times over the years with no noticeable withdrawal symptoms, other than more problems sleeping.
Remy – I have been taking trazadone for about 10 years. I take 150 mg at night. I used to take it every night because the fear of not sleeping was so great but now I have gotten in the habit of not taking it if I am adequately tired/sleepy. i have no withdrawal effects (unlike Nortriptyline, which I take for Fibromyalgia, which leaves me feeling like I was kicked in the head by a mule if I skip one night) and sometimes I go a month without taking any. I was in Dr. Younger’s study on low dose naltrexone and I KNEW the second day that I was receiving the drug not placebo. It was the first weekend that I had gotten up off the couch in years. I had to stop taking it when I had surgery and I was really hurting during that time.
Yes, trazadone is very hard to withdraw from. I had been on a 100 mg dose for sleep when we determined that it was worsening balance problems and it was a bear weaning off of it. In fact, I never got below 25 mg and then went back up to 50 mg in hopes it would once again help with sleep problems.
So what kind of “study” was done? Did they actually combine the drugs in some sort of way and give them in a trial such as the Ritalin + Immune Stimulant trial done by Bateman etc?
No study – It was big data mining project using computer algorhithms to come up with what appeared to be the best match. I imagine that Biovista came up with a bunch of candidates and they thought this was the best one.
LDN made me so nauseous. I really wish it worked for me but I had to stop. I even started on a very low dose. I hope it can help some people.
Not that most people with CFS are drinking but make sure you are not consuming alcohol in any form as there have been reports in patient forums of vomiting in some people when the two are combined. You might also try a different compounding pharmacy as different ones can use different fillers and/or try a lower dose to start (as little as 0.5mg for ME/CFS patients).
I too had stomach problems but I knew the ldn was working so I asked my naturopath if I could have it made in gelatin sublingual form called a troche. It worked like a charm and I am delighted with the results.
Hi Debbie,
Thanks for the suggestion. Did your naturopath write you a prescription?
You may have been reacting to a filler. If you are generally sensitive you may want to get the version made with pure Naltrexone powder (NOT crushed tablets) and distilled water with nothing else. This will eliminate any possible filler reaction. Check with your pharmacist about the life in the fridge. Since this version doesn’t have preservatives it may only be good for 30 days (but check). OR you could get the sublingual drops or transdermal cream to bypass GI issues.
Many have reported headaches or nausea when the filler Avicel (Microcrystaline Cellulose) was used or when the starting dose was too high.
More info at Low dose Naltrexone (LDN) for chronic illness & infections…. https://www.facebook.com/groups/108424385861883
I have tried LDN myself (@3.5 mg doses and 5mg dosages). There does seem to be an awesome effect with either dose for my various “pains”. However, I (have, and will continually) propose to concentrate all efforts on ridding the body of latest residing pathogens: Lyme bacteria and all Herpes viruses!!! Then let’s talk on fixing other things…
For Albert can you please comment on your efforts to rid the body of Lyme, Bartonella possibly, and Herpes viruses? what treatment are you using? is it helping? do you think that this is the main cause of all of the symptoms of ME/CFS. I was biten by a tick 2 years ago and my health has never been the same. neurological symptoms and cardio muscle weakness. sleep disturbance, etc I often wonder what is really wrong with my health and what treatment I should be pursuing. Thanks
Mark S. please get tested at Igenex in Palo Alto CA you def have Lyme disease. I too have it you need to get on treatment ASAP
LDN is believed to have anti-viral properties. Celebrex, too.
No reason you can’t continue LDN while you work on these other issues. LDN supports correct functioning of the immune system so it can help with your Primary issues also.
From my own research & experience, I discovered that apparently LDN can’t work as well as it should if the patient has any underlying undiagnosed or untreated, infections (ie. viral, bacterial, fungal, parasitic, whatever). I tried LDN a few years ago having been prescribed 4.5 mg has (at bedtime). For about 3-4 months, I felt like a whole new person & had my lofe back but sadly after awhile, it failed to help anymore at all & I found myself back at square one in pain, no energy & unable to sleep:-( I’ve never tried Trazasone but recently have found Nabilone (synthetic THC) @ 1 mg taken between 5-6 pm is helping to reduce my overall daily pain as it’s helping me to finally get some decent sleep. My pain level, as it is for many similarly afflicted, is always higher when I don’t sleep.
Mare, I have had the same experience with LDN. The first five months of taking LDN I felt like a teenager – a lot of energy, no pain and no brain fog but then no energy and poor stamina returned to the same low level I had before LDN.
Like you I’m still taking it if I stopped I believe I would be back in bed most of the time. I sure would like to know why it stopped working.
I take trazodone 50mg for sleep and it has been a life saver I get a solid 8 hours of sleep.
Thanks for the good info. Cort and thanks to all who commented.
Judi, during the time you “felt like a teenager,” did you increase your activity? (Dumb question?)
I love having more energy, on those rare occasions that I do–but no matter how well I’m feeling, if I overdo, I always pay for it.
Did you “overdo” while taking the LDN?
Responding to Tami.
Yes I did over-do when I was feeling so good but I haven’t felt well in 6 or 7 months now. A couple of days ago I decreased my dosage to 1mg and I felt much better the next morning but the feel-good only lasted one day 🙁 I’m becoming discouraged. I’m sure it’s finding the right dose and they do say less is more with LDN.
See if you think this article might apply
LDN Rebound effect…. https://www.facebook.com/notes/ldnnow/ldn-rebound-effect/10152209009858391
I so wanted LDN to work for me, but side effects were too scary. I started out on 3 mg. The next morning I was on top of the world – felt like a million bucks even though I could hardly sleep. The second night I woke up at 3 a.m. feeling as if my chest were EXPLODING. It was alarming, but the weird feeling went away after a half an hour. A couple of weeks later I tried a much, much lower dose – 0.5 mg. No endorphin high the next day, but I got chest pain. I’ve gotten this peculiar kind of “atypical angina” after taking other meds – Armour thyroid, Cytomel, 5-HTP, tyrosine, Ioderol, Lugol’s solution – all stuff that makes me feel better. No one has ever offered me a theory about what’s going on with me. I think that feel-good neurotransmitters might be causing cardiac vasoconstriction. Has anyone else gotten this side effect from LDN?
I have used LDN for about 1 year now… It really helped with my FMS, took pain level down about 60%. If you are starting it, start very slow, at maybe 0.5mg and then increase weekly by about 0.5mg at a time. Top dose for FMS is usually 4.5mg. If you are having vivid dreams, you need to decrease the dose by one degree until that goes away. Too many people are starting on too high a dose… drs are not well enough educated.
Also, some do better with taking it in the morning, I do. Doesn’t have to be taken at night.
Right – the word on LDN used to be that it had to be taken at night to take advantage of the opioid blocking and the endorphrin kick in I believe but that’s apparently not true at all. There are some things I can’t take at night – like Kombucha – which leads to waking up and being unable to sleep – in contrast to just waking up multiple times – which I can handle during the day.
Regarding dose timing:
In a presentation made by Dr. Bernard Bihari in 2002, he gave this perspective on LDN dosing:
The reason the [timing of the LDN dose] is important is that 90% of the endorphins are made in the middle of the night, between 2 and 4 in the morning. If a small dose of naltrexone is taken in the late evening, generally at bedtime, generally endorphin production is boosted as much as threefold, 300%. The naltrexone itself is gone in about 3 hours, but the endorphins remain elevated all the next day. So the naltrexone doesn’t significantly block the endorphins but does cause them to rise.
So the initial directions were for people to dose at bedtime. However in a recent study from Penn State, the presenting doctors said: “At present, the timing of administration of LDN is a patient preference, and there are no basic science studies that conclude morning or evening consumption is either harmful or better.”
This issue still seems somewhat up in the air. Here is a link to a thread about the subject:
http://ldn.proboards.com/thread/2681
And from that link a comment by Dr. Skip [a knowledgeable LDN pharmacist and user who has been a speaker at LDN conferences]:
“I really hate to get involved with this question, and the he said,he said responses. Who is right, Zagon or Bihari, well they both are right. One has to look at the diurnal nature of endorphin secretion to understand HOW they are both right. You get your zenith peak during your normal sleep cycle. You get your penultimate peak during your wake cycle. Taking LDN prior to your zenith peak will give you more bang for your buck than prior to your wake cycle peak.
Therefore you will get some increase taking LDN at times other than prior to bedtime but they will not be the most you can get. Now if you take it indiscriminately temporally, then depending of which peak is next in the cycle, you will get a fraction of the response you would get if taken at an appropriate time frame. This explains why both docs are right, for those taking it in the am, you get the PM peak to skew upwards, those taking the PM dose gets the am peak to skew upwards.”
Dr.Skip
It seems like many members of the FB LDN groups use morning dosing and still benefit. However based on what Dr. Skip is saying above it may be that they would benefit more with bedtime dosing. Overall it seems like it’s a good idea to try bedtime dosing to start with and maybe to re-try at some point if you have to switch, but if you are having too much trouble sleeping you may have to take a mild sleep-aid or do a period of morning dosing. When you can try bedtime again. Sometimes it will work for people the second time around. This is especially important to try if you have been morning dosing and feel that you are not getting adequate healing from your LDN protocol.
From the document “Side Effects and Dosing” in the Files of the facebook group GotEndorphinsLDN
I have been taking Trazodone for sleep for 3 years. My dose is 100mg at bedtime. I just started LDN for autoimmune disease. The worst side effect is that I can’t sleep and it causes all my muscles to stiffen/joint pain. Can these two drugs not be taken together?
Particularly for the first couple of weeks LDN can cause problems with sleep. They usually disappear, though. You might want to start at a lower dose. It’s not uncommon that that’s needed.
Ironically, one project actually suggested that LDN and Trazodone might be a really GOOD combination. It’s neer been tried out in studies though.
https://www.healthrising.org/blog/2016/02/04/low-dose-naltrexone-combo-chronic-fatigue/
Good luck!
Hello Rachel, i know this is old. But I felt I should tell you what I learned with ldn. I read if you take thyroid medication once you start ldn you will need to lower your dosage of thyroid medication. It’s been a while but it has to do with your thyroid regulating itself on ldn, so the thyroid medication becomes too high. I experienced this. While taking ldn I got horrible insomnia like I was wired (wired and tired feeling) and my heart was racing. When I had my blood drawn my thyroid levels were too high. I had gone from hypothyroid to HYPERthyroid. So my Armour Thyroid had to be decreased. I started reading about it and found that was why. I have Hashimotos diseases. The ldn lowered my antibodies significantly and my thyroid was functioning better so the Armour Thyroid was causing Hyperthyroidism and making me feel wired like my heart was gonna pump out of my chest. My father has had 2 heart attacks and had heart valve disease so this concerned me. Once the Armour was lowered the insomnia decreased and no more tachycardia. I wish I remembered the websites but it was about a year ago. I’m sure I saved them. If I
I find them I will post for you.
After 5 days on .5mg LDN, I had an episode of super ventricular tachycardia (svt) 250bpm. I was able to to resume a normal heartbeat by using breathing techniques I’d been taught. I also get this when taking 5-htp. I stopped taking it.
Has anyone else had stomach problems when taking Trazadone? Stomach pain and nausea slowly crept up and got worse and worse and I had to stop taking it after 3 weeks even though I was on a low dose.
I’ve taken LDN for 3 years – I tolerate it well. I don’t know how much it helps now, as I continue to decline, but I keep taking it assuming I would be worse off pain and sleep wise without it.
Thanks for the report Cort. I must admit I find it quite disappointing. But glad to hear what really happened with all of that.
I had Trazodone Mylan described by doctor, immediately had stomach pain, nausea and suicidal thoughts. after swimming through those bad side effects for 3 weeks, I finally experienced some nights with 7hrs of sleep for 3 months, then had to go fm 50 mg to 100 mg for about a year. But from day 1 I felt very ‘drugged’ all day, no nice feeling. And the insomnia nights were never completely gone and came back. still at least once or twice a week; tried several doses but the side effects didn’t really improve ‘quality’cof life. I stopped taking it.
I have been taking 4.5 mg LDN at bedtime for 32 months. It may be helping. I still have quite a bit of pain. When first starting, I titrated up at 1.5 mg increments. I think it is worth trying.
I tried Trazadone for sleep. One 50 mg tablet did not work. I tried 1.5 tablets (75 mg). Here are my notes on the bottle: Woke several time during night; Woke with very tight muscles and tight jaw; Vision is very blurry in AM; DON’T USE ANY MORE!
I agree with Albert C. These diseases are being caused by infectious pathogens, and these pathogens need to be destroyed.
Does anyone know how LDN would affect medical marijuana use?
My understanding is LDN and marijuana can be taken together. The only drug that should not be used with LDN is opiates.
I tried LDN at a dose of 1.5 mg. I took it only one night. It made my pain skyrocket, made me dizzy, and caused heart palpatations. Felt I was way overmedicated.
I would go lower – .5mgs ? At the last LDN conference I was really struck by the wide variety of responses to doses. Some people who have horrible responses at a higher dose at first can actually work themselves up to from and benefit from that dose if they go really slowly. It’s weird how the body works…
I’ve been on LDN for a couple years. I’ve started out at a very low dose (0.2 mg) and am now at 0.8 mg/day.
Yes, it does cause some nausea but the benefit of reduced pain has been worth it. When I feel nauseated, I drink a little ginger ale, and it goes away.
I have tried LDN for over a year and it didn’t seem to make any difference at all.
Katvil, that’s a bummer. As I understand it, LDN is supposed to retune your immune system away from autoimmune reactions toward fighting infections. Maybe it would help if you increased the dose.
I have tried Trazadone also without help for sleep. Instead I became very swollen with blurred vision and felt more sick with heart palpitations. I tried several dosages for 2 months as prescribed without success
I do know several people trying LDN with great success but you cannot take it with any other pain medicine or alcohol as Cort said. Most patients start in very small amounts such as .05 or even liquid. There are several Facebook groups where people discuss their use of LDN.
One of the Facebook groups is called Got Endorphins.
I was on it for over a year and felt no effect, good or bad. I’ve just stopped.
I would like to find a specialist in ME/CFS in the Naples, FL or Ft. Myers area. If anyone knows of a doc who is really well-studied in the complications of CFS, that’s who I need. I am a write and researcher on the subject of pain management, and have a wonderful PM doc here in Naples, but he’s not into really digging in and doing testing, etc. Thanks, Krissy
Krissy,
I have gone to Dr. Jeffrey Dach in Davie, FL. His website is JeffreyDachMD.com. He works in integrative medicine and did extensive testing to sort out my situation. He was the doctor who first suggested LDN to me. I started with an office visit and then followed up long distance, coordinating care with my family doctor. Good luck!
I used to live in that area. I see Dr. Irma Rey at Nova Southeastern in Ft. Lauderdale. Very knowledgeable and does extensive testing.
Hi Cort, Thank you for writing about Biovista’s drug repurposing project that was one of 5 grants made by the CAA in 2012. You can read about it here: http://solvecfs.org/breaking-ground/.
I’d like to make a few clarifications. As noted above, Biovista wasn’t “engaged”, they submitted an application to a competitive grant process. There application scored well and the grant was awarded. Grantees adhere to the organizations grant policies (grants are quite different from contracts).
The grants made by the CAA were intended as seed funding for projects that were riskier or had little to no preliminary data. It was a great mechanism for enticing new scientists into ME/CFS research. The fact that Biovista’s bioinformatic platform identified 2 drugs that are used (off-label) in ME/CFS was actually quite exciting!
We also conducted survey of treatments used by ME/CFS clinical experts. Their responses indicated that LDN and trazadone were effective in treating ME/CFS symptoms. This was important because it validated what Biovista’s bioinformatic platform found! A paper describing this survey was just published (http://www.tandfonline.com/doi/full/10.1080/21641846.2015.1126025).
Biovista prepared an Investigational New Drug application and met with the FDA to discuss the combination and the study design requirements. This FDA meeting occurred on the heels of the 2013 FDA Patient-focused drug initiative meeting – where ME/CFS was the first workshop held by the FDA. The FDA was helpful and interested in doing what they could to make ME/CFS clinical trials happen. It was now up to Biovista to raise the funds to conduct this trial.
I don’t have to tell you that raising the $2 million to conduct a 150 person randomized controlled clinical trial is no cake walk. Since the combination has been disclosed it less attractive to potential private and pharmaceutical funders.
It could potentially be funded by NIH. Drs. Deftereos and Persidis remain committed to using their repurposing platform to identify drugs that can be used for ME/CFS treatment and I am hopeful that we will find funding to pursue further repurposed drug clinical trials.
Your analysis of the mechanisms of action of trazadone and LDN are spot on. It is entirely plausible that when combined the mechanisms of these 2 drugs would target sleep, pain and immune modulation. This is why a controlled clinical trial for this combination was and still is important. And we all know that ME/CFS clinical trials and treatments are desperately needed.
In closing, far from underwhelming, Biovista’s identification of this novel drug combination demonstrates that targeted treatments are within reach for ME/CFS and every effort should be made to ensure studies like this and clinical trials occur.
Two million dollars – ouch!
I think this – along with Ampligen and Rituximab trials – would be a great way for the NIH to demonstrate its new found commitment to ME/CFS.
Thanks for filling in some of the blanks. It was good to hear that the FDA was enthusiastic about a trial like this. Somehow a way has to be found to assist diseases like ME/CFS in which the pharmaceutical companies show little interest. Ditto with drugs like LDN which show real promise in helping people but which big pharma is just not interested. If the NIH and FDA is really serious about doing everything they can to help everybody who is ill these challenges need to be addressed.
Hi Rachel,
I’m on the Cape but will note down Elson. (Thought of driving there for MJ but they never have any cbd strains). Virtually no mainstream doctor will prescribe LDN in my experience so I got it from India and Israel for my FM. Unfortunately at night I had two terrifying nightmares and when I switched to AM use, going up from .5 to 2.5 gradually with no benefit, I suddenly experienced unbelievable intensification of my FM pain in areas not activated for decades. Went back to “normal” pain in several days after I discontinued. There is an LDN cheerleader group on Facebook that reco’s it for everyone for everything.
Hi Steve, LDN seems to be very tricky. Dr. Elson is not great at micromanaging doses based on side effects. He will prescribe, but it’s not one of his main things. There are other practitioners in his clinic – Elson now works only two days a week. Dr. Lynch is full time and has been there a long time. I hear that he is very smart. And there are a couple of nurse practitioners. Check out the website for Northampton Wellness Associates.
Thanks, Rachel, will check it out, but I do wish I had tried LDN again at much slower dose increase before my FM pain accelerated, would be tricky to manage with Tramadol and now Lyrica…too many variables!
Steve, I hear you about the problem of too many variables. It makes the whole thing confusing. But I was impressed with Dr. Jacob Teitelbaum’s philosophy – he thinks that you have to hit everything at once. For example, he thinks that for sleep it works better to take very small doses of a lot of soporific herbs rather than putting all your eggs in one basket.
I was not impressed with his book. Does not jibe with my scientific background. Up late this AM after first 50 mg Lyrica (not cheap), pretty dopey and headache. we’ll see.
Steve – In case you are not aware…
If you need financial assistance with paying for meds in the US, go to pparx.org and follow the prompts. Some meds are available for free …sponsored by the pharmaceutical company.
One of mine is sent to my doctor on a quarterly basis and I just go & pick them up.
Thanks, Donna, nice of you to post that, will tell some people I know. I actually have very good insurance and money is not an issue at the moment. I was just struck by a thirty buck co-payment for a month of 50 mg t.i.d. Lyrica, suggesting to me it is pretty expensive med without insurance or with weaker insurance. I had two wake up calls about meds this week. The pain doc told me no opiates (have never tried) anymore until I have failed a bunch of FM meds, they have tightened up the protocol with all of the ODs and people with real need are going to have to jump through hoops. The second moment was after I handed my scrip to the pharmacy and the tech said ” let’s see if your insurer will pay for it.” That’s when I realized that the people controlling our medical care are not our doctors, nor even the Feds, but the drug companies. I have read that Obamacare passed only because Obama caved to Big Parma about not negotiating prices. Reportedly, the aide who wrote most of the legislation later left and went to went for a major drug company I won’t mention.
Not to hijack the thread…I don’t want to get my hopes up but Lyrica, despite doping me up today for a few AM hours, also made me feel more normal painwise, even awful neuro pain, than I have in two years, astounding, held my AM Tramadol unti 2 PM. I don’t see how anyone can take this during the day so I’m going to open the capsule and weigh out half of the 20 mg and take tomorrow in a trial, assuming I again make it to 2 PM if I take at midnite. I also slept through three nightly 2.5 mg Ambien dosages, also astounding. Maybe this is something Big Pharma got right for me. I have had initial positive stuff before with stuff that did not continue or had adverse effects but we’ll see.
A book I reco everyone here read is Cure: A Journey into the Science of Mind over Body by Jo Marchant. This is a cutting edge book by a good scientist, both with research and anecdotes that provide actionable info. What color meds work best, can a placebo work even if you know it’s a placebo, etc. I’m going to try to use classical conditioning to see if I can use a placebo to extend my Lyrica benefits, if I find it works, or strong pain meds, if I end up on them. Good science, I’m a psychologist with good research background and speculated about some of this stuff in my head the last few years but this is convincing me to try it.
Just fyi…my 30 day Lyrica copay is $100. A 90 day supply is $250. I have “good” insurance and my husband even works for a large retail pharmacy chain!
Eek, Lois! I hope it works well for you! Day four…it’s not going to be anyone’s favorite med, if they can do it, is it! I hope the woman doing the trial with the improved version finds it loses a few side effects and ups the positive effects.
Wow! The price. Here is a clear example that Big Pharma will make more money on a symptom-treating medication than on a cure.
There is a Facebook Group called “Low Dose Naltrexone (LDN) for Fibromyalgia and (CFS) Fatigue” that has a lot of info. It’s more of an informational type of group vs a chat group. In their files they have a list of doctors that they have been compiling that will write a prescription for LDN.
I was on LDN and it did great for my Fibro. But I had to go off of it because it didn’t help the pain from the many herniated discs that I have. (It wasn’t intended to work on back pain). I started at 1.5 mg and didn’t do as well after increasing to 3 mg. The correct dose for each person tends to vary. The best thing to do is start at a low dose. Unlike most drugs, this one doesn’t work better as you increase the dose. Increasing the dose is rather intended to find out which dosage works best for you.
I have taken Trazedone 50 for many years for sleep. When I thought it wasn’t working well anymore it was upped to 100mg, Nothing happened – couldn’t sleep. So I went off of it for awhile and I slept. When I started back up (because I couldn’t sleep again), I went on 50mg and slept like a baby. Now if it stops working, I cut it in half and try that and it works the same. Or I go off for two to three nights and it will start working again. My point is that more may not be better, and it does cause a hangover.
I am encouraged by this article today, but because I have several pain problems I am on high doses of Oxycodone and Fentanyl. I wonder how it would work to go off those and try LDN. I wonder if it would be enough pain meds for me. Have to ask the PM doc but this is very interesting. I also take 800mg Ibuprofen every six hours and it helps me tremendously. Without the meds I wouldn’t be able to function at all. With them I get enough relief to be at home, but with the progression of RA and possible Lupus, DDD and ME/CFS, I still can’t go out and do anything reliably. All I need is an electric wheelchair in order to get some freedom. I have to sell my electric tricycle first. Bummer because I can ride it at times. Ok, rambling because I am tired. Good to read all these comments after a great and encouraging article!
Krissy A
I love it when less is more 🙂
Cort: I just looked at my comment and I have no idea where the word (my moniker) “Dock” came from. Where do I go to change that? Thanks- Krissy
Krissy – I’m on the same pain meds that you are on. Scroll up to read my comment. LDN worked for Fibro but not herniated disc pain. In the FB group that I mentioned there should also be a Youtube video available to learn more on how LDN works with Fibro. You can try it, everyone is different but I also know how difficult it is to wean off of these meds. I’m now weaning off of Fentanyl. And I know how difficult it can be to get these meds. (I don’t, but some people do). If these issues could potentially be a problem for you I would reconsider trying it. To avoid waking up in pain at an 8 or 9, I don’t sleep for more than 3 consecutive hours at a time. It’s not ideal but it works for me.
Thanks, Donna. I am just being curious for people in general I guess. I have no intention to changing my meds right now.
Thanks for the response.
Kristine, What jumped out at me was where you wrote “…with the progression of RA and possible Lupus…” I needed to encourage you of something that helped me press on. Mindset is very important and when my doctor said to me one time, “Well, your issues are just the PROGRESSION of the disease…” I really reacted!! They DON’T KNOW that the disease has to progress because they don’t know everything that causes it and what can reverse it! My mindset is to HALT or REVERSE it- not monitor it’s progression as they have been taught.
YOU are on your own most of the time in finding the latest information or protocol. That is unless you can spend unlimited $$ on the few medical professionals that will be able to help because none of them take insurance and also charge $$ because they can. I have found the occasional doctor that is in the system that helps as long as I do my own footwork.
So much starts in the gut. Taking Ibuprofen and other NSAIDs in really bad for the gut lining which will make it worse in the long run. We need to restore and heal that. DIET is everything- nightshades, lectins (NOT just GLUTEN) cause the inflammation. Take a good quality Curcumin (Turmeric extract)- amazing anti-inflammatory! Use ginger supplements.
Do research on your blood type foods (D’Adamo). Dr. Stephen Gundry has done NEW research. Google him. Get you genome done through 23andme BUT have it analyzed by another online source (because 23andme is really pathetic at doing so). I like Promethease, Nutrahacker, D’Adamo’s Swami…
If you have not tried LDN, I would encourage you to do so. Just be aware that the way it is formulated (fillers used) and the reputation of the compounding pharmacy is very important. I have taken it since 2010.
It has helped me so much with RA, Lupus, pain- even my Hashimotos BUT I have had bumps in the road recently in my dosage…It has helped my thyroid normalize so much that it went too high on the meds I was taking and I have had to cut them by 1/3…
I have been using different things over the years. I am going to try Trazadone again with this new info here…I remember over 20 years ago, a doctor prescribed Trazadone for sleep, suggesting it would help my Fibromyalgia symptoms. I don’t know the dose strength of the tablet but it was way too strong, making it hard to wake up fully. I kept cutting the tablet until I was taking a speck- maybe an 1/8th or less. When I found the right dose, It helped me sleep so well and so fast! At the small dose, I could wake up refreshed. My doctor laughed at the dose BUT he told me I was very sensitive to medications and that I should note that for the future. Those of us with these conditions are the “canaries in the coal mines”- it’s a toxic world but our bodies are on our side trying to manage and adjust to the onslaught- have the mind set that your body is NOT attacking itself, it is just trying to find homeostasis- sometimes that causes problems that we need to help with. All I write here is to encourage you so please don’t think it is a lecture- I am on the same journey. Please don’t give up because you are an encourager too! <3 Best!
Kat, you have a wealth of information! And I like your attitude about assuming that you will reverse the illness. About HRT I forgot to give you links last night. About the safety of using hormones cyclically, google Danish study cyclic HRT and you will get a bunch of hits. And about my psychiatrist’s approach to HRT, he got a lot of it from going to seminars given by a Dr. Neal Rouzier who has a website – http://www.hormonedoc.com. I found a series of videos he gave about how HRT done right is safer than no HRT.
I agree that getting well is a do-it-yourself project. I happen to have a doctor who has been treating CFS/ME and fibro for decades and he does take insurance. But I still have to do the research and suggest things to him. Then he says OK.
I agree about diet. Pain when almost all away when I gave up gluten and dairy. Then I found that oats, quinoa, and corn gave me inflammatory symptoms, too. I just tested allergic to beans – if I don’t eat beans, does that mean I’m getting rid of lectins? Nightshades – maybe they should be the next thing to drop. NSAIDS are bad for leaky gut – I used to take them all the time. I can’t take curcumin – it gives me joint pain for some weird reason.
I did 23 and Me on my doctor’s advice and got the data to Genetic Genie – confusing. I’ll look into the other outfits you suggested for interpretation of results. I wish that I hadn’t had such scary reactions to LDN – I avoid drugs that give me chest pain.
The latest thing I’m exploring is whether I have a genetic variation that makes it hard for my body to clear mold biotoxins and whether I might have Chronic Inflammatory Response Syndrome – CIRS. There is a Dr. Ritchie Shoemaker who discovered this syndrome which sounds a bit like CFS/ME. Last week I had 6 blood tests to check myself out for this. This problem has a quick and easy fix! A quick fix would be fantastic.
Thanks for your encouraging words, Kat! We all need encouragement.
Rachel & Kat,
I definitely agree with diet & with your other comments. Definitely on the right track. I have Mixed Connective Tissue Disease (RA, Lupus, Sjögrens & Raynauds) as well as Hashimoto Thyroiditis, which I got many years before MCTD. After trying a myriad of different auto immune medication I stopped all medication and went on just diet and supplements. I started with it Eat Right for Your Type & then tried other diets, such as Paleo, etc. Eat Right for Your Type worked best for me, but I had to tweak it with an elimination diet to find out what triggers my flareups. I was 99% flareup free until recently when I had a root canal which seem to trigger my Sjogren’s badly. My doctor put me on Plaquinil for awhile to stop the inflammation immediately. But I can attest to diet, supplements & mindset as being crucial to the road to recovery.
As for Dr Neal Rouzier, he was my HRT doctor for awhile when he had his practice in Palm Springs. I felt like a million bucks with his bio-identical formulations, however, he took me off of my normal thyroid meds & put me on “natural” thyroid & I became incredibly tired. I saw my rheumatologist & he tested my thyroid & it was if I was not taking any medication for my thyroid. My rheumatologist put me back on synthroid & Cytomel.
I went back to school to become an integrative nutritional health coach & I learned from a fellow student about a book called “The Emotion Code”. She also had Hashimoto thyroiditis and after doing the emotion code, she no longer needed any thyroid medication. I read the book and did it on myself often for about a month. Two months later I started having horrible heart palpitations, headaches, etc. Come to find out when I had my thyroid tested, I was told that I was on way too much thyroid medication. I was on 200 mg Synthroid for years. In one year my Endocrinologist has lowered my thyroid medication from 200 mg down to 75 mg and I’m still taking too much. I truly believe it is because of the emotion code.
I was researching LDN, as my brothers wife started on LDN for Hashimoto Thyroiditis & I have been researching it. It’s interesting, but I try to stick to natural remedies, unless I have no other choice at the moment.
If any of you have not tried an elimination diet, I highly recommend it, as well as supplements such as Black Seed Oil, Curcumin, NAD, L-Glutamine (to heal your gut), etc. As Kat said, “So much starts in the gut”. Most all disease come from an unhealthy gut. Try to be happy & take the focus off of your pain, no matter how hard that may be & find things to be grateful for. Believe me…I’ve been there. There were times I didn’t want to go on. I am a different person today. Eat clean. Be mindful. Believe that all things are possible! Because they are!
Two more supplements that are good to take, especially for Fibromyalgia, is d-Ribose (1 scoop 2-3 x a day, as well as Source Natural’s Fibro-Response, which is a multi vitamin for Fibromyalgia. Even though I don’t have fibromyalgia, both those helped me & I still take d-Ribose for energy.
Naltrexone ( I hope that’s right damn SPELLCHECKER keeps changing it) and this is probably redundant not low dose. Are we freaking Americans so afraid someone might get better and live a happy life? Or does the medical community need that much cash. But this drug has treated Canadians with Fibromyalgia successfully for years, because I checked into and found out I would need to have a Canadian daddy and could only get prescribed 3 months at a time. Too cold in Canada. We are so developmentally challenged in the greatest nation and it isn’t just the Bible belt where I live. It’s the DC Beltway! I would love to have the Congress tested since you can’t sell Marijuana for profit or grow it, especially without a DEA permit, and seeds are illegal to sell in the U.S. I guess you go to DC and they give out complimentary joints on your motel and hotel pillows and in bowls in Bars instead of Pretzels. Or maybe it’s like on Cops and it’s just laying on the ground. Hypocrites. You should have seen what I wrote to Georgia Legislators when I was asked if I felt my condition was left out and why? If you cannot understand Perocet and oxycontin are more addictive in treating pain then Medical Marijuana, but you afraid because then you may lose control of Marijuana as one of your arguments. Do you not know Percocet and oxycontin have been lost control of, too. Are still more addictive, and less effective? Are we dealing with knuckle draggers? I did not vote for these people. It’s been a very stressful day. And I am sick of the shit. We have played this game for decades. No stingy funding. Laughable care. Russian roulette prescriptions.Low dose is not enough, sorry if Canadians get the pill and are doing great, we American s will, too!
Minx on the Health Rising Forums just reported that it really matters what time she takes LDN
http://www.cortjohnson.org/forums/threads/ldn-low-dose-naltrexone-whats-the-latest.2873/#post-10413
When I tried it last year i was taking it at 8 a.m. (no sleep if I take it at night). This time I have experimented. I took it at 4 a.m. 6 a.m even 5 a.m. But the perfect time for me is 5:20-5:30 am. The difference in how I feel is huge.
Even if I don’t get back to sleep, which I usually don’t, I’m awake and up by 7:30. I used to lay in bed in a coma and have to drag myself out. I couldn’t function for hours.
I have more energy, mentally and physically. My pain has decreased a huge amount. Inflammation in my brain and lungs are down. Fog is better. It’s pretty incredible.
I have to be careful. If I overdo it I can feel things flare, brain fog gets worse, pain gets worse, I get wiped. Yesterday I had to go out at 9 a.m. I was gone 1 1/2 hours and spent the day on the phone, copying, faxing and dealing with a huge problem. I expected to be bad today. I wasn’t. I’ll see how tomorrow is.
Understand, I’m not going to run laps or even go shopping. But for me, this is pretty big.
I take LDN at bedtime. It’s supposed to work by blocking your opioid receptors for two to three hours per night, thereby causing your body to make more endorphins. Normally, your body would re-absorb the endorphins.
I started at 1.5 mg for three to four weeks in June 2013. I had 1.5 mg capsules compounded at a local pharmacy.
At first, I had some startling reactions like a new type of wakefulness and vivid nightmares; a definite impact on the brain. But after three or so days, those calmed down.
I increased the dose by 1.5 mg, and stayed on that for three or four weeks. Then I added a third 1.5 mg capsule, making the total 4.5 mg recommended by my doctor. 100 capsules @ 4.5 mg cost about $80.
In one sense, it would be worth a higher price if Big Pharma would actually do big dosing studies for our conditions, variable as individual responses are. My two terrifying LDN nightmares were of dying in a car crash and another situation I have forgotten, game enders for me for evening dosing. When I switched to AM, as I noted, my fibro pains went off the chart. I don’t know what could make me do another trial, a whole lot of great data maybe.
I started LDN a year ago. 4.5 mg made me too nauseated and caused insomnia, so I dropped down to 2.25 and it’s taken me a year to titrate up to 4.5, upping the dose every 90 days. I do get a little nausea and slight insomnia for about a week, but then I’m fine. I’ve found that LDN has increased my stamina. I rarely get tired any more, so now my main issue is fibro pain.
That’s so great. Which country are you in if you don’t mind telling? I am taking just cannabis oil without THC and that’s no real help with pain, like chronic pain, but has cleared nerve pain, insomnia, nausea 80%, IBS70%, and most everything but chronic pain and energy is not consistent, but have weird POTS, probably low blood volume, but live in Georgia, U.S., and Doctor, started looking November for someone who has even heard of that test near, still no answer.
You know after reading all the warnings, that it has been available since 1985, that it’s linked to so many “quirks” I already have. I can see why I was never sent tilting after that Wind Mill. So happy for the rest of you that you have found your relief till science catches up with the causation to this madness. I need just simple and natural, and the older drugs just work better, and the fewer the better, though minimal relief, the side effects are just down right deadly at times. But as I have said again and again, all us Immunologicals are warehouse under a few simple Names, might as well describe us: Sony, Panasonic, Vivatar, GE, Samsung, Sharp, LG, Vizio. But if I went into Best Buy and said I need to replace my Samsung and that’s all the information I had. I would get a whole lot of different and diverse products. That’s the way we are and different parts relieve. We are on different paths of our pathogens. We are at different stages. And we are all important, worthy and worth being fixed as well as be willed according to our individuality. We need our own specialist for our specifics, whatever we have. Whoever’s brave enough to admit we are more than a sum of symptoms, but individuals.
I tried taking a very small dose of LDN (1.5mg) after reading about how much it seemed to help others with ME/CFS & FMS. I was disappointed because it made me not sleep, at all. I took it for two nights, but I just couldn’t forego sleep for the third night. Sleep is so important to me and so fragile, I can’t take a medication that interferes with it, even for a few nights. Thanks for the info on trazodone. I may see if my GP will let me try that.
About 18 years ago I was put on Trazadone to help me sleep. It left me in a state almost of hallucination. (In the UK the only ME ‘treatment’ was Cognitive Behaviour Therapies. I had to see a psychiatrist first to determine if I was depressed. He considered me to be depressed and put me on Prozac. A month later he doubled the dose. A month later I complained of poor sleep so he halved it and added in Trazodone. After a few nights on Trazadone I cut it out completely and also stopped the Prozac. No side effects. My next visit to the psychiatrist I told him I was feeling great and he admitted I was the best he’d seen me – and then I told him I’d cut out all medication. Of course he could not deny my improvement but could not accept that perhaps I had never been depressed in the first place and it is just that the ME is worse at certain times of the year and he had seen me at my worst.)
A few years later I took part in a trial at a local hospital where the test drug was a breakdown product of Trazadone. My first experience was with the placebo. The second was the drug – and it was obvious! I could not even raise an arm. Afterwards I could not walk a straight line down the corridor to the taxi but had to be helped by a nurse. The doctor in charge gave me her phone number to call if I needed her at night. (I think I should probably have been put in hospital rather than sent home alone.) I did not sleep all night but also could hardly move. The nurse told me that everyone who had ME reacted badly whereas the control patients had no reaction. (I gathered that the idea was that it could be used as a diagnostic tool for ME. However, I have never been able to find out anything about the research.)
I have always said I would avoid Trazadone after that, but perhaps the problem was the quantity, not the drug itself.
Oh my! That’s the same “paralyzed but unable to sleep” reaction I had to Xyrem. It was horrible. Maybe I won’t run right out and see if my doc will give me trazodone….
I have been diagnosed with CFS but have no pain or fibro. My doctor says that LDN is good for pain, and since I don’t have any, I don’t need it. What are your thoughts? Thank you for replying.
I would suggest to go back & read all of Cort’s posts on the subject. And watch the video on how LDN works with your body. LDN is said to work with improving your immune system. I would think that should be very appealing for anyone with CFS. The nice thing is if it doesn’t work you can simply stop & it’s affordable.
I started taking LDN 18 months ago when it was prescribed as a mild prokinetic for SIBO. I believe it works by decreasing gut inflammation. I started at 0.5mg and upped it very slowly over a year to 4.5mg. Honestly I’m not sure if it helps my SIBO directly but it totally zapped my fibromyalgia muscle pain! I barely have any bodily inflammation anymore. I plan to take LDN for the long-run if it keeps working its magic. It hasn’t helped my brain fog and low mental stamina as much, so somehow it doesn’t seem to quell the inflammation in my brain as obviously as it is in my body. Fortunately other things are helping me there. As I continue to unwind the tangled web, LDN has become one of a handful of potent solutions for me.
That’s very good. Probably a subset of LDN responders. Or maybe a subset of non-responders…
Indeed, Steve. One big thing I’ve learned on this journey is how differently we all respond to different therapies. There are many more things I’ve tried that haven’t worked than have, even though they work wonders for others. The art is in the experimenting! n=1
Jennifer….I have horrible inflammation in my brain, so your comment really caught my attention! I am seeing a ND, but have just started down this path. Might you share what you’re doing to help with brain inflammation? Thanks!!
I want a drug that is proven, in CFS, to reduce exhaustion and brain fog.
I don’t need its pain relief capabilities; I have that under control ‘Feel good’ is nice, but without dealing with the brain fog, it’s an illusion. And the extreme fatigue needs addressing. Period.
Does LDN do that? I can never get an answer.
I’m interested, at this point, ONLY in that (and that it not make anything else, like sleep, worse).
Alicia, I think we’d all like all like a drug that would reliably reduce extreme fatigue and brain fog for everyone with CFS. I think maybe it doesn’t exist yet. Like you, I have the pain aspect under control more or less. Years ago some part of my body hurt all the time. When I went gluten free and then dairy free, the persistent inflammations went away. The other thing that helped a lot with pain was magnesium – taking pills didn’t do it so I had to get magnesium shots every 3 weeks, which chased away my painful restless leg problem and the other aches and pains all over. But when I got rid of pain, fatigue took its place! Not fair.
A comment about sleep: I have been leery of taking meds like trazadone, so I’ve developed a concoction that works for me most of the time. My sleep medicine doctor told me that the most effective thing for postmenopausal women is hormone replacement. When I increased my dose of estradiol and of natural progesterone (200 mg 25 nights a month), I finally got good sleep for about 7 hours most of the time. In addition I take some calming amino acids – theanine, taurine, and glycine and 3 mg of sublingual melatonin before bed.
Rachel, Please tell me more about the estradiol and of natural progesterone. Is the 200mg a combined dose? Compounded?
I wish to resume my bioidenticals again as I feel I was better off. Please mention any links to info where I can find more info.
Your other suggestions are also helpful. Thank you!
Kat, I take a generic estradiol patch that I change twice a week. The dose is .075 mg. My health fell apart when my gynecologist reduced the dose to 0.05. Finally my PCP increased the dose back up and the hot flashes that had been waking me up 3 or 4 times a night went away. I think the patch is much safer than the pill – much lower dose and it doesn’t do a first pass through the liver. I saw a study that found that taking HRT cyclicly is much safer than if it’s daily, so I take a three day break once a month.
The progesterone I use is bio-identical natural progesterone. I had been taking one 100 mg pill 12 days a week. Then I went to an integrative medicine psychiatrist (not because I was having psychiatric problems) and he upped the dose to 200 mg 25 days out of 28. This made it much easier for me to go back to sleep if I woke up briefly. My PCP is horrified by this – she blames my fatigue on the progesterone because it can make you sleepy. The psychiatrist says that natural progesterone protects against breast cancer. At first I was taking the brand name Prometrium, but then it went generic. Now I’ve switched to one 100 mg pill and 100 mg of compounded bio-identical progesterone in a topical cream that I get from Life Extension. Since it doesn’t do a first pass through the liver, it provides a bigger actual dose. Also the psychiatrist says that the transdermal hormone will make me less sleepy. If I’m really in need of a good night’s sleep, I’ll take 300 mg of progesterone and sometimes I get a night when I don’t awaken.
Another doctor prescribed a cream form of an alternative estrogen called estriol. It’s supposed to be safer, but it’s much weaker and I started to get way too many hot flashes and awakenings so I gave up on that. Also I was supposed to apply it twice a day over a lot of skin, but not before a shower. Too much trouble.
I hope this is info is helpful, Kat. My HRT meds are my favorite meds. Sleep is so precious. One more thing – I think the progesterone makes me feel calm and relaxed. I had a couple of horrific eye surgeries last year and I didn’t freak out at all – just did what I had to do without worrying. This psychiatrist prescribes HRT because he finds that it helps people feel better psychologically.
Rachel,
Sleep is adequate, and I’m far enough past menopause that I have no problems there.
I overreact to almost all medications, and would never take hormones – with all their side effects – because the likelihood of help is small compared to the likelihood of problems.
I can’t afford the time it takes to try out new medicines – it stops my writing flat any time there is a problem. And that’s the only thing I have left of myself.
So I keep reading these things. I think we’re not going to get anywhere until they figure out how this illness works. I want to know – and then to have someone target the disease specifically.
For heavens’ sake – they got a vaccine for Ebola in record time! HIV/AIDS is no longer a death sentence. And they can’t even figure out what’s wrong with us, agree, and find a cure?
I don’t want half measures and comfort measures and pat-on-the-head measures. I want results.
Glad the hormones work for you.
Alicia, I’m glad you sleep OK. That’s huge. And I hear your frustration. I hope they figure out what’s causing CFS and how to fix it, but meanwhile I’m up for anything that helps somewhat. I’m frightened of most meds, but I take a lot of supplements, each of which helps a little bit in some area of distress. The thing that’s giving me the most noticeable help these days is hyperbaric oxygen treatments. I feel better after an hour breathing 100% oxygen under the pressure of 1.3 atmospheres for an hour. I’m thinking of renting a chamber to use at home because I feel so much worse on the weekends when I don’t do it. Money and trouble! But some relief. Let’s keep our fingers crossed about the medicos coming up with a treatment that actually works!
There is no magic bullet to eliminate/ reduce exhaustion and brain fog. There are many reasons and variables involved.
If you mean “proven” as in FDA and clinical trials, you won’t get those for LDN (which is Low Dose Naltrexone) because it will not make BigPharma any profits so there is not incentive.
Pain is an indicator of something not right in your body. You also say you have the pain “under control”, but not what you do to control it. Have you decided that you cannot eliminate it without a drug ? There is much you can do for yourself but it requires you to not totally put your life in the hands and control of the pharmaceutical world…
Alicia – I think we’d all like a specific protocol to follow for any of our symptoms. Each one of us with CFS and/or Fibro have a lot of primary symptoms that are very similar but at the same time there aren’t two people with the same exact symptoms as I’m sure you are aware. To add to that the majority of us have additional illnesses. For me, I heavily research each experience (med/supplement/avoidance of foods, etc) that is of interest to me. If the symptoms, results and expense of trying it is within my reach I take the risk of trying it. Each one works differently on everyone. With all the variables it is very difficult to get a direct answer on how it will work for you. I will tell you that I am not a patient person by any means and I’d love for a direct answer for any of this. I’ve been sick with EBV/CFS/Fibro for nearly 30 years. I’ve tried many things. My symptoms have also changed over the years. And if I do find something that helps with a specific symptom, another symptom will become my worst symptom. I hope you can find something to help.
Alicia, the answer is that LDN will do that for some people. You are welcome to ask your question at the Facebook group https://www.facebook.com/groups/GotEndorphins/ where we have members that take LDN for a variety of conditions including CFS. Results are very individual so we can’t say with certainty what will happen for a given person. But it is not expensive and the side effects are minimal so for many people it’s worth a try. If you tag me there I can direct you to a thread with people’s experiences.
Better group where I am now:
Low dose Naltrexone (LDN) for chronic illness & infections…. https://www.facebook.com/groups/108424385861883
After getting diagnosed with metastatic thyroid cancer, on top of my severe me/cfs at age 25 I nearly lost all hope of ever improving. However, I remembered something in my medicine cabinet, LDN. I had taken it one night during my first six months with cfs and it made my symptoms worse so I didn’t pursue. I decided to try after a thyroidectomy made my fatigue paralyzing. I can’t believe how much it has helped me. I’ve been able to leave the house everyday and even exercise mildly! (Ex Hour on a horse). This was completely impossible before LDN. I still have cfs but it’s a 5 not a 10. It’s a miracle for me. And just in case your wondering, my cfs doctors (dr Levine) and oncologists do not believe my fatigue is due to cancer. Thyroid cancer is almost always asymptomatic:)
So weird to read this now! I was just prescribed Trazodone last week for sleep and have been feeling much better. I wasn’t sure if it was because the sleep was much improved (I had a sleep study done once and there were not major issues) or perhaps that a subtle underlying depression was resolved. I ended up doing some research and found this study: http://www.ncbi.nlm.nih.gov/pubmed/25911310 which I was going to show my doctor, and incidentally, was also going to email Cort about it. Ah well, my work here was done for me lol.
Interesting. I’ve taken both for years, without side effects. Trazodone has been the most effective drug for me overall with ME/CFS. I don’t have problems getting to sleep and sleep for hours continuously but it still doesn’t make me refreshed. Still, a lot better than when I could not maintain continuous sleep at all; that period made me understand why sleep deprivation is a form of torture.
In my practice pre-CFS, I prescribed it regularly as, relative to other sleep drugs, it has a reasonably safe long-term use profile and is effective. It’s been used for decades to counteract the insomnia that some antidepressants cause but is also use just for insomnia. It’s also cheap (even without insurance). Interestingly, it was also the first sleep drug my own doc prescribed for me when I came down with ME/CFS so we think alike. You won’t see many younger docs prescribing it though because no pharma company is making mucho $$ off of it so no advertising to docs or patients. One major caveat: it can exacerbate orthostatic intolerance in some people so start with a low dose and take it at night not just for sleep but so you will be lying down soon.
LDN helps take the edge off my constant headaches so it helps somewhat.
Neither drug is a miracle worker — still very sick here — but some help via cheap drugs without major side effects is always good.
I’ve taken Trazodone 50mg for about 4 years to help with sleep. It helps me a ton, especially with reducing wakings and enabling me to sleep 8-9 hours most nights. 75mg gives me heart palpitations (as do many meds/supps.), but 50mg is fine. I also take melatonin and Benadryl to help with sleep induction, as Trazodone doesn’t work as well at putting me to sleep, but is great at keeping me asleep. I failed at LDN because it made my sleep worse, but will try taking it in the morning. Thank you!
Looks like the webmaster needs to do some work as the Avatars are blocking some of the text in comments. See http://screencast.com/t/RI6SD4rDfo0f
Thanks for mentioning this too Brian – I can’t read the comments on my ipad – seems they should be visible regardless of what device you’re on. I’m interested in these trazadone comments because after working famously for me at 12-25 mg for about 4 years it suddenly doesn’t work at 50 or even 75. And if I do get to sleep I wake up for good in about 5 hours. So weird as this happened virtually overnight.
On the topic of morning or evening doses being better (I currently take 4.5mg at bedtime) my doctor said she was happy for me to experiment with two doses daily. Adding a dose in the morning. Does anyone have any thoughts or experience with this idea?
Hi Elizabeth, at the Facebook group below we have recently published a document from the Norwegian LDN group that explores possible benefits of 2X/day dosing. If you join this group and tag me (Brian Haviland) I can guide you to the document.https://www.facebook.com/groups/GotEndorphins/
Hi Brian, I’m in the group… can you guide me to the 2x day dosing Info?
I am no longer in that group. Tag me at
Low dose Naltrexone (LDN) for chronic illness & infections…. https://www.facebook.com/groups/108424385861883/
Rachel, what are the lab tests for chronic inflamatory response syndrome? Did you get your results?
Hello, has anyone been to Dr. Lapp in Charlotte, NC? I have an appt with him tomorrow. I have Chronic horrible debilitating fatigue, cmv, ebv, lung and liver nodules, hx of cervical adenocarcinoma from HpV. So… a lot of viruses and other health problems. I am so fatigued (not a word stong enough) and my pain is so bad. No one has or will diagnose me with fM even though about 6 people at least on my paternal side have been diagnosed with pain syndromes. Specifically FM. Including my father. I have family on disability for similar symptoms (not that they used that diagnosis for their diagnosis to obtain disability). We (family) all have early onset degerative disc disease,insomnia, spinal Maternal side has lupus (great aunt) and Ms (great grandmother) and early onset dementia (my mom), pain unrelated to spine issues, many of then have Ra or other autoimmune disfunction. I am so nervous about going to a mystery illness specialist (Dr. Lapp) because it seems like my last ditch effort. I am afraid to hear, “we don’t know” and “it must be a rare genetic disorder that we cant detect or treat” and/or “your fatigue must be because of opiates (multilevel spinal stenosis)” even though fatigue, muscle stiffness and tenderness and other Fm type symptoms all were exhibited before I went on opiates. I don’t know if all of the viruses have caused my chronic fatigue. I have also had positive ana numerous times but no other autoimmune tests have turned positive yet. I live on a farm and I have had many ticks but traditional tests are not showing positive. I have all the symptoms of lyme dx, thyroid, CFS and fibro. Any advice would be helpful and if you have or haven’t seen Dr. Lapp let me know your thoughts and experiences. Who have you seen (doctors) that could help near or around South Carolina? Maybe Atlanta, GA, Charlotte, NC, Asheville, NC and/or Greenville, SC. I am also four hours away from Raliegh, NC. I am so sorry for all the suffering. I am glad I found this blog. Thanks again! Shannon my email is scarrollrnvp@gmail.com if you want to share experiemces or can offer ideas on non traditional treatment ideas that have been known to help.
You deserve some real help. Good luck with Dr. Lapp!
Shannon, you have so many challenges that I’m sure you will need to do a number of things but I hope you will ask the doctor about Low Dose Naltrexone as it is likely to be helpful.
Conditions treated with LDN……………… http://www.ldnresearchtrust.org/conditions
Here’s one doctor’s way of explaining LDN.
From Dr. David Gluck, MD: “LDN is absolutely unique. And that’s part of its problem, in that it’s a brand new paradigm, a new way of thinking of treatment. Instead of the medication actually doing the work, LDN goes into the body and essentially tricks the body by forcing it to double and triple its output of endorphins and metenkephalin, also known as opioid growth factor (OGF). Those endorphins and metenkephalin, in turn, cause the immune system to [balance itself]. A nice way to think about LDN is that it is not like any other medication whatsoever. It is a way to strengthen (in the sense of regulate/modulate/normalize) the immune system.”
intro video……….. https://www.youtube.com/watch?v=z0p0ykSzy9o
Kelly, if you are a member of the FB group you will be able to see the 2X/day dosing document here: ALTERNATIVE DOSING STRATEGY …… https://www.facebook.com/groups/GotEndorphins/946851798732935/
Wow this is interesting! I have been on LDN and Trazadone for several years and I still only function at about 30% of normal on a good day. Would love to know the ideal dose of each.
Hi Nar – I don’t know if they got that far or not. Suzanne Vernon might know.
Does someone has experience in combining Trazadone with LDN?
Dear folks who experience CFS and/or FM.
I’m not sure why patients want to pair those two syndromes. Yes, Cort reports having both, and yes, a friend of mine has both, plus 6 other diagnosed disorders.
Other rhan widespread pain, brain fog and non-restorative sleep, I dont see what links those two syndromes any more than either might get linked to MS or lupus. Those other two other disorders also have the pain, fatigue and brain fog.
I started trying to understand COVID in Mar. 2020 and then in August learned that my new friend had botb CFS and FM. After about 6 months of learning amd reserarch analysis I was able to conclude that rhe symptoms of my friend’s 8 diagnoses were locally: homeostasis knocked off kilter. When I researched how each homeostasis in question could be disrupted at once, I could see that vascular damage from pandemic flus was the common trouble.
Now I am reading the latest HealthRising reviews and I already understand what is at issue:
When a person does not endure 4-5 90-minute healthy sleep cycles in a night, each of which should have 4 stages, the delta low-brainwave stage being the restorative one,
their brain and body nerve receptors have not had enough time to be fully reset to baseline. That means they carry over overstimulation to the next day.
In FM, leading modern neurologists have shown that at the dorsal root ganglia at entry to the spine from the peripheral body nerves have produced twice and 3 times the pain receptors after pain did not subside before the deadline for becoming “chronic” pain.
With regard to light or interrupted sleep, I have seen Health Rising referring to the brain stem, whixh can be called the Reticular Formation. This area decides, based on signal intensity whether the brain should be vigiliant, or can ignore stimuli under a threshold.
Finally, it has been shown in early military experiments and later in sleep studies, that if one loses ine hour or more of the 4-5 90-minute cycles needed, The brain will be more active the entire next day. In such a way that it’s “alert, alert, alert” messages flitting around, but not useful cognitive thought.
It detracts from cognitive function
It leaves inadequate resources for adequate cognitive activity.
What it comes down to is that the patient needs to find a way to relax ir descend into delta phase sleep or else those receptors will not be returned to baseline, and the whole body/brain will be hypervigilant each day.
For this reason you all need to look seriously at your melatonin function,
to know that serotonin is converted at night to melatonin, and the reverse occurs upon waking, and that in FM studies have shown that the analgesia function of the brain is found to he deficient in the serotonin and norepinephrine that are designed to go back down to the dorsal root ganglia to turn down the amplitude of pain signals to the brain stem.
If this is failing, then the lasting exaggerated pain will keep the brain stem hypervigilant, sabotaging balanced sleep cycles. We can only blame, here, the melatonin delivery system in sleep that is not adequately silencing non-critical stimulus messages to the Retucular Formation.
Job 1 is to correct sleep cycles.
Many more systems are off-balance, but disturbed sleep is the most destructive, reviewing my own research review NIH and higher.
Chris
Find a functional medicine doctor.