

“No prescriptions, no medicines, no injections. That’s the future. That’s what gets me out of bed in the morning.” Kevin Tracey, MD.
Dysautonomia International really scored when they got Kevin Tracey MD to be the keynote speaker at their 2020 Virtual Conference. He’s not involved in dysautonomia and knows nothing about postural orthostatic tachycardia syndrome (POTS), but his work has real implications for people with dysautonomia, ME/CFS and/or fibromyalgia.
Tracey has got credentials up the wazoo. He’s a neurosurgeon, a groundbreaking researcher and an inventor and author. He’s the president and CEO of the Feinstein Institute for Medical Research, where he helped create the Center for Bioelectronic Medicine, and which New York State recently gifted with a $30 million grant. He’s also a professor of neurosurgery and molecular medicine at Hofstra/Northwell, and president of the Elmezzi Graduate School of Molecular Medicine.
He’s been recognized as one of the most cited researchers in the world. He’s also the author of “The Fatal Sequence: the Killer Within“, a book about his experience with a badly burned girl with sepsis which changed him profoundly.
A clear and engaging speaker, Tracey’s focus was on a topic which interests us all – inflammation. Inflammation, Tracey said, was the leading the cause of death and disease in the world today, and pointed to the major killers of our age – heart disease, diabetes, autoimmune diseases, etc.
The Surprise of His Life
Back in 1996, Tracey said he got the surprise of his life. He knew that a biological drug called CNI-1493 (Semapimod) was able to reduce the levels of the pro-inflammatory cytokine TNF-a in the body. Tracey hoped that if he inserted Semapimod into the brain, it would reduce TNF-a levels there as well – and it did – but to his utter shock, putting Semapimod in the brain also reduced TNF-a levels in the body (lung, liver, kidney, spleen) as well.
That didn’t make sense. Given the small amount of Semapimod he’d injected, it was clearly not working its way down to the body. Since everyone knew that the brain and body had their own separate and independent immune systems, how reducing inflammation in the brain could simultaneously reduce inflammation in the body was a mystery.
There was this thing called the vagus nerve, though. They knew that signals travelled up the vagus nerve to the body. Could they also be traveling down it?
They cut the vagus nerve – and low and behold, the inflammation crept up again. With the vagus nerve connection cut, Semapimod wasn’t having any effect on the body. With that, a new understanding of the immune system was borne, and the budding field of neuroimmunology took a big step forward.
The next big question was: how was the vagus nerve doing it?
The Vagus Nerve
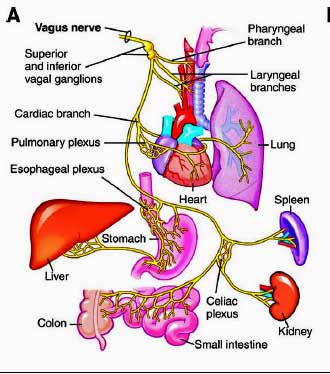
The vagus nerve has an incredible reach. The largest neural network in the body, the vagus nerves densely innervate every ‘sensing area’ of the body. Tracey cautioned against viewing the vagus nerve as a single nerve. It’s actually a complex bundle of nerves that contain over 100,000 fibers. Eighty percent of those fibers carry signals to the brain, and 20% send signals back down to the body.
It affects virtually every part of the body. In the cardiovascular system, the vagus nerve regulates our heart and breathing rates, blood pressure and blood flows. In the gut, it’s responsible for gut motility (the passage of food through the gut), gut secretions, inflammatory responses, the integrity of the gut lining and even appetite.
Information on touch, heat/cold, pain and chemical, metabolic, and hormonal operations of the organs are all transmitted via the vagus nerve to the brain.
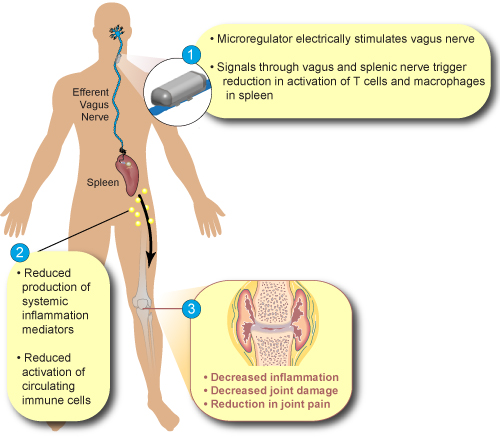
No one had suspected, though, that it might regulate the immune system. It took over 20 years of work for Kidder and others to understand what was happening. Signals flowing down the vagus nerve to the spleen are converted into norepinephrine, which then interacts with T-cells to produce acetycholine – which then turns off immune cells called monocytes/macrophages. The vagus nerve, it turns out, plays a key role in enhancing the anti-inflammatory response. Poor vagus nerve functioning results in an overactive immune response and inflammation.
It’s now believed that vagus nerve regulation of the immune system may play a role in asthma, allergies, arthritis, ischemia, hypertension, gut disorders, sepsis, inflammatory and autoimmune diseases and pain states.
Research indicated that the immune regulation goes both ways: signals traveling down the vagus nerve regulate the immune system in the body, while signals traveling up the vagus nerve send inflammatory signals to the brainstem and then up to the brain. This up and down signaling came to be called the “the inflammatory reflex”.
Bioelectronic Medicine – A New Frontier?
Biologic drugs which target cytokines and tone down the immune response have proved very helpful for some, but are incredibly expensive and don’t help everyone. Tracey had uncovered, however, a very different pathway to ramping down overt immune expression – manipulating, or rather stimulating, the vagus nerve.
It was not until 2007 that Tracey dared to produce a device which could electronically regulate the immune system. Founding a company called SetPoint, he got to work. Finding a lead researcher to take on the clinical trial was difficult given the novelty of the effort.
Tracey’s first surgically implanted vagus nerve stimulator (VNS) had immediate effects in a rheumatoid arthritis (RA) patient who’d been living on his couch in pain for 8 years. The stimulator was so effective that the patient started playing tennis while the trial was still in progress. Another woman who, after 16 years of severe RA, was so sick that that she was unable to lift a pencil, was able to ride a bicycle again within months. (Read her story in the New York Times.) Another young woman who’d spent 15 years in and out of her wheelchair had fruitlessly tried every drug in the book. A VNS worked – and she now works in his office.
Successful pilot trials in Crohn’s disease and rheumatoid arthritis followed and recruitment is expected to begin soon in a larger, “pivotal” trial in 250 moderate to severe RA patients who have not responded to other therapies. If the trial is successful, electrical modulation of the immune system could be available for RA patients in just a few years.
Tracey also helped to create The Center for Bioelectronic Medicine to map the vagus nerve in detail so that devices could be built to specifically calm specific nerves. Tracey, who at 63 was full of enthusiasm, believes the bioelectrical revolution may ultimately be able to help just about any kind of inflammation-associated disease. Stimulate the right nerve in the vagus nerve and you may be able to tone down inflammation in virtually any part of the body.
The most recent device, which is about the size of a penny, has a battery, computer chip, antenna and electrode and is controlled via an iPad.
Non-Invasive Vagus Nerve Stimulation
Non-invasive vagus nerve stimulation is cheaper and easier. Instead of being implanted in the neck, the non-invasive vagus nerve stimulator simply clips onto the ear. Lauren Stiles of Dysautonomia International reported that DI is funding no less that 3 trials on non-invasive vagus nerve stimulation in POTS.
While noting studies show that non-invasive vagus nerve stimulation can work, Tracey noted that the vagus nerve branch in the ear relays sensory signals, not autonomic ones. Given that, he doesn’t understand how non-invasive vagus nerve stimulation is working when it does.
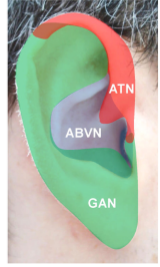
Given that only sensory vagus nerves permeate the ear, it’s not clear how non-invasive VNS works – but sometimes it does work.
With implanted devices, on the other hand, he knows what the device is doing to the nerve and how much charge to give it. The holy grail for Tracey over the next 10 or 20 years is to be able to develop devices which stimulate specific fibers in the vagus nerve.
In the Q&A section of his talk, Tracey suggested an interesting hypothesis – a broken vagus nerve is behind POTS. A balky vagus nerve that’s not doing its part to tone down inflammation could lead to the autoimmunity which appears to be present in POTS.
When asked what treatments people with dysautonomia could do now to support vagal tone, Tracey provided the usual suspects: meditation, prayer, fish oil, cold showers, ice water splashed on the face, aerobic exercise – all increase vagus nerve activity and decrease inflammation.
- Check out a Free Online Health Summit on alternative health and functional medicine ways to enhance parasympathetic nervous system/vagal nerve functioning starting Nov 9th.
The Vagus Nerve, Chronic Fatigue Syndrome (ME/CFS) and Fibromyalgia
It bears noting that the B2 adrenergic receptors being explored with regard to autoimmunity in ME/CFS are the same receptors that produce anti-inflammatory effects via the vagus nerve. The alpha adrenergic sympathetic nervous system receptors, on the other hand, that help to produce the fight/flight response that is amped up in ME/CFS, also stimulate the production of pro-inflammatory cytokines such as TNF-a.
The low heart rate variability (HRV) findings in ME/CFS and FM indicate that parasympathetic nervous system/vagal nerve activity is inhibited – suggesting that the stage has been set for increased inflammation.
In a 2011 article, “The Pulse of Inflammation: Heart Rate Variability, the Cholinergic Anti-Inflammatory Pathway, and Implications for Therapy“, that linked low HRV with inflammation, Tracey proposed that low HRVs could be used to target people who would benefit from vagus nerve therapies. Note as well that one of the drugs Dr. Klimas’s modeling efforts predicted would work in ME/CFS was a TNF-a blocker. It was TNF-a that Tracey blocked in the rheumatoid arthritis patients with his implants.
With regard to fibromyalgia and pain, the first hints that VNS might be helpful in reducing pain came when epilepsy patients reported experiencing reduced pain. VNS may be effective with chronic pain because it appears to be able to affect many of the factors known to contribute to it including: inflammation, oxidative stress, autonomic nervous system activity, the opioid response, central sensitization, and pain perception.
Check out a remarkable story of woman who recovered from severe fibromyalgia after having a vagus nerve stimulator implanted in her neck.
The Future
It’s not clear if non-invasive or invasive VNS will work in ME/CFS or FM. The low HRV readings, the increased fight/flight response, and the immune issues suggest that it could.
Implanted vagus nerve stimulators, though, are extremely expensive, are not covered by insurance and are hard to come by. No studies appear to be assessing them in either disease.
Non-invasive VNS provide a much more affordable option and have been shown in at least one small study to be potentially effective, but are tricky. It’s not clear what the proper signal strength should be, how long they should be used, which devices work the best, etc.
The Electroceutical Revolution

Could harnessing the electrical currents in the body be the future in medicine? (Image by Gerd Altmann from Pixabay )
The future is where the real opportunity for diseases like ME/CFS, FM, and POTS lies. An enormous amount of money has quietly gone into sparking an electroceutical/bioelectronic revolution medicine. The possibilities are enormous. Instead of flooding the body with drugs, bioelectronics could conceivably precisely target the nerves involved – reducing side effects and increasing efficacy.
Tracey estimated that $800 million has been invested in bioelectronic medicine and listed a slew of major companies and institutions that have jumped into the field. In 2018, the state of New York invested $30 million in Tracey’s Center for Bioelectronic Medicine as part of its innovative, economic investments program. From the Center:
“Our two primary discoveries are first, that inflammation is the central phenomenon in virtually all disease, from cancer to diabetes, and second, inflammation can be controlled through the vagus nerve, the body’s main “highway” of neural information that prompts and curbs inflammation. The institute’s work in molecular biology to fully understand the vagus system and other nerve networks has led to a means to both monitor the neural signals produced by the body and decode them, not only to control inflammation, but to anticipate incipient disease before it takes hold.”
In “A Spark in the Periphery”, Emily Walz reported that GlaxoSmithKline and Verily Life Sciences (formerly Google Life Sciences) are pumping more than $700 million into a new company called Galvani Bioelectronics, which will develop miniaturized electronic devices to stimulate nerves in the body. (Aloisio Galvani was an 18th century Italian scientist who discovered he could make frogs’ legs twitch with electricity.)
Recently, Astellas Pharma snapped up Iota Biosciences to capitalize on its ability to develop tiny millimetre-sized implantable devices to treat disease. After participating in an R&D agreement with Iota, Astella decided not to tread water any longer and plunked down $304 million to buy the entire company. Astella pledged to spend $125 million over the next five years to “aggressively” expand Iota’s work. (Iota was formed in 2017!)
DARPA, the Defense Advanced Research Projects Agency, is throwing $90 million into the hunt for better electroceuticals.
SPARC
In 2014 Tracey reported that the NIH played a leading role in supporting work on bioelectronic medicine when no one else would. Three years later the NIH announced that its “Stimulating Peripheral Activity to Relieve Conditions (SPARC)” Initiative would spend $250 million in an attempt to learn how to manipulate nerves to reduce pain, relieve inflammation, heal heart problems, fix gut disorders and more.
Understanding the neural anatomy in exquisite detail is key. Many of the funded grants are attempting to map exactly which nerves control what. Working off a DARPA grant, a University of Wisconsin team recently had a breakthrough in that.
They found that the 100,000 nerves in the vagus nerve were organized in very specific ways that allowed the researchers to predict where they would end up. That should allow researchers to move forward more quickly on the mapping problem. Other studies include:
- A SPARC research team at Duke University is determining that different nerve fibers can be activated by different patterns of electrical pulses.
- Purdue University is mapping the autonomic circuitry of the gut – a task of interest to ME/CFS/FM and POTS patients, given the problems with gut motility – which can impact gut flora and leaky gut – present.
- A Mass General study aims to make non-invasive vagus nerve stimulation more effective by mapping how the nerves in the ear impact the cardiovascular functioning. They’ll be tracing signal production all the way to the brainstem.
- The Icahn School of Medicine at Mt. Sinai is using non-invasive radiowave and magnetic field signals, nanoparticles and bioengineered ion channels, which can be used to remotely activate and inhibit central nervous system activity.
Update!
A study which finds the master immune regulator at the connection between the vagus nerve and the brainstem opens up new possibilities for treating autoimmune/inflammatory diseases like ME/CFS and long COVID.
Conclusion
While nothing is going to happen tomorrow, the large amounts of money being poured into the “bioelectronics revolution” are encouraging. Just 25 years after we learned that the brain, through the vagus nerve, is impacting inflammation, researchers are tracking down in exquisite detail which of the 100,000 nerves in the vagus nerve impact which organs and body tissues.
In 2014, Tracey wrote in a piece called The Body Electric:
“The promise of bioelectronic medicine is that by targeting a specific organ, at a specific time, in a specific amount, many side effects can be eliminated. Prescription refills can be accomplished through a wireless interface. There will be no more daily injections, and no interruption to daily life. The potential exists for developing treatments for ailments ranging from rheumatoid arthritis to Parkinson’s to diabetes to cancer; the immune system contributes to all of these.”
Tracey has predicted that bioelectronics will eventually replace the drug industry. As was noted – this is not going to happen tomorrow. Whether the major breakthroughs in bioelectronic medicine that seem likely to occur will happen in time for people like me it is unclear, that but what is clear is that an era of bioelectronic medicine is coming, and younger people, at least, should reap its rewards.
With the autonomic nervous system issues present in diseases like ME/CFS, FM and POTS, we should ultimately be able to benefit from these advances. Tracey suggests that diseases characterized by the low heart rate variability found in ME/CFS, FM and POTS are good potential candidates for vagus nerve manipulation.
Plus, emerging evidence of neuroinflammation in ME/CFS and FM provides an intriguing possibility – tamp down the inflammation in the brain – and reductions in inflammation in the body may follow.




 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



Check out Apollo neuroscience. https://apolloneuro.com/ It’s a vibrating device created by trauma researchers, quite a few research papers supporting its use for conditions such as PTSD, anxiety. They market it as improving one’s resilience to stress, which is improving vagus nerve or parasympathetic function in a nutshell. Less stress = less inflammation = less pain and other FM/ME/CFS symptoms. I’ve had their device for a couple weeks now and I’ve had pain reduction and a significant increase in my HRV (from 30’s to 61!!) Sleep is a work in progress, but that’s a really chronic problem for me. They have an energy program which I’ve only used a couple times because I’m wary of over-doing it and crashing the next day but it did help me finish packing for a trip on a crash day and I wasn’t too bad the next day.
For me – this is definitely true – Less stress = less inflammation = less pain and other FM/ME/CFS symptoms.
That is a huge increase in HRV! That’s very encouraging!
Good luck with it and thanks for passing it along. I will check it out.
It’s nice to see so many possibilities. This is definitely a growth field.
Hi Cort! I really appreciate you and all you write. I really like your writing style. I am trained in science and am trying to keep up with the Fibromyalgia/ME literature. With fatigue worsening I am finding it difficult. I was wondering if there is a spreadsheet anywhere with a synopsis of the current hypothesis with scientific evidence, treatments that have worked for some, labs to have done to narrow down etiology & treatments, treatment centers to go to, etc. I can’t keep up with what tests and treatments to ask my doctors for. I know this would be a huge undertaking. Does one exist? Thanks so much.
I wish….we will have something to help with treatment centers and treatments soon, hopefully.
Diana-I got the Apollo at about the same time as you and I would be very interested to hear further about how you are using the device, what device you are using to measure your HRV and how it’s going for you now. I have also been hesitant to use the energy side of it for fear of a crash. I’m keeping my fingers crossed that it might be helping with migraines and pain from trigeminal neuralgia. I also have FM/ME/CFS. If you could e-mail me at debbie.griffis@mac.com I would love to talk with you about the device. Thanks, Debbie
I know this is supposed to be encouraging, but I find myself somewhat despondent over the amount of time it will take to develop these treatments.
I agree, and worse is our health systems will take another decade to say ME/CFS sufferers can use these devices too, but prior to that they will allow a host of other diseases first access in the queue. Just like Covid long haulers who more than likely are developing ME/CFS have preferential treatment and get to go to Covid rehabilitation centres in the UK, yet ME/CFS sufferers have the same problem but have waited over30 years and still nothing.
It’s a travesty of exclusion by the health system. A form of class bigotry on disease type
Yes, the lack of interest in ME/CFS/FM/ES has been absolutely frustrating & deeply unfair.
However, if we could advocate for focus on these diseases (especially getting across the severity aspect, which most don’t get) to someone like Dr. Tracey, we might get on their list of diseases on which to research these advances.
It’s also possible the long haulers, by dint of their sheer numbers, will light the way for us (decades) long-term patients.
I agree. It is incredible how long it takes to get new treatments out. In Tracey’s case it was even worse because he had fight dogma multiple times. Nobody believed back then that the vagus nerve affected the immune system. Then few believed that you could electronically manipulate the immune system. This is truly revolutionary stuff..
The good news is that the medical research is moving in this direction and there appears quite a bit of funding. If the RA trial works I imagine funding will jump dramatically. It’ll be fascinating to see how that works.
Anyway – now that more of the research community is involved hopefully things will move faster. We live in really interesting times. Hopefully things will move fast enough to even benefit old geezers like me.
Cort, is there an RA trial? My friend just got RA and is in severe pain and nothing is helping. She is a strong woman and not a crybaby but this rheumatoid arthritis has her on her knees. Sincerely, hipjaven@gmail.com
I wonder if the lymph drainage massage (that has when I’ve had it done improved me) is actually stimulating the vagus nerve instead?
Unfortunately for me I don’t have the energy to massage each of those areas 50 to 100 repetitions. But if you have a partner or can pay someone maybe give it a go.
Anyway could gently massaging the vagus nerve be something we can do while we wait years for an electronic device?
https://youtu.be/QA-wi0d7-Ro
I crazily used one of those electrode massage TNS machines a few years ago in an attempt to stimulate the vagus nerve, and my frontal lobes but I kept bumping the sensitive dial and the zaps were painfully powerful on my head. Like being repeatedly being punched. So powerful once that I couldn’t coordinate my hands to to turn off device for about 20 seconds of horror, lol
I also found out that the TNS devices alternate their polarity with each pulse, so you can’t send a single one way. If I could have set the polarity one direction I would have continued with it, but also more careful at a much lower current
Hi Brendan Rob,
I have a “bed of nails” for quite some time. I learned to know it in https://www.healthrising.org/forums/threads/a-bed-of-nails-for-better-sleep-acupressure-mats-fibromyalgia-and-chronic-fatigue-syndrome.5221/
It helped sleep a bit and I was happy-ish with it. It helps but for me it still is a sort of torture device to lay on. So I hadn’t used it for like over a year since I start to sleep better even without using the torture tool.
Some time ago, a friend video called and told about her ongoing sleeping problems. I could offer few help until I thought back about this bed of needles. I got it, showed her and explained her how to use it. Then we kept on chatting. By chance and lack of will to put it away straight away, I laid the matress on my belly with the needles pointing to it and my hands on the other flat side acting as a small weight.
I was in my frequent “hang-sitting” posure so the matress stayed neatly on my belly. I sort of intuitively got my shirt up a bit so the needles were in contact with the lower part of my belly. I just had a sort of flash in my mind saying “if your gut is in so bad shape, maybe this will help it relax a bit”. My belly wasn’t in view as I could check on the small window with my own video on it so it couldn’t bother my caller.
Then something really unexpected happened. I started to relax in a way I hadn’t for ages in less then 5 minutes. It was like a wave coming over me. My caller just saw it happening by the clear change in my face and expression relaxing so quickly. She asked “what’s happening, you are getting so mellow?”. It was the matress! And this way, it hurts WAY less then laying down on it and works WAY better for me.
I also tried the small neck piece and gently pushed it to the front of my neck: same reaction but less strong. That may be because my dysfunctional gut is in worse shape then my tense neck muscles?
Since then I use the thing often after a meal to get my gut and myself in a better rest-and-digest state. I feel it clearly helps me. My gut keeps slowly improving (I do other things for it to I must say, but this does help).
If it would work for others too: have a cheap bed of needles now rather then an expensive bioelectronic device next decade :-).
Fascinating!
My ME/CFS stems (essentially) from contracting the Ross River Virus. There was a myth from years past that farmers who were shocked by electric fences had been cured of their post viral symptoms. With a support crew and borrowed defibrillator, I went around the area I lived in (acreage that floods, so lots of horse paddocks) and tried shocks from a lot of electric fences. The only thing it confirmed was that a lot of electric fences required maintenance, and those that did work weren’t rated to enough joules to bother a horse too much. I had an uncle who managed to get a good jolt from an “big as it gets” bull paddock electric fence, on his head once. His description of blacking out then coming to and wondering how many teeth he had just cracked makes me think that might be more the level of energy/application method involved in the farmers’ stories.
Reading this article, I wonder if there might be something to that famers’ myth though.
Getting utterly off topic, I wonder about the prevalence of people who believe their mobile phone has messed with their health having big ears. The non-invasive VNS certainly sounds like the sort of thing you couldn’t make up. I’m just kidding by the way.
Thank you Lono for the laugh-out-loud moment! They are hard to come by these days 😀
There is a microcurrent device available at the moment that isn’t classified as a vagus nerve stimulator but I do know it’s calming Alpha-Waves have improve my deep sleep, reduced my pain and has totally taken away my fibro-fog. I have seen recent research that alpha-waves can reduce central sensitisation. I have to use my device daily but not everyone needs that. I have been using my device for 11 years now and it has lowered my fibromyalgia symptoms by 40%. I’m sure in the future bioelectronics will be better able to target their treatment and I look forward to that. At least at the moment there is a well researched device available called the Alpha-Stim. I suggest try and find a practitioner who loans out these devices to trial. Most people need a 3 to 4 week trail to see if it will work. For those with all over-body pain and /or non-restorative sleep the ear-clip only device called the AID should be sufficient but please remember those of us with FM and CFS are very sensitive so please start at the lowest setting possible, for a minimum of an hour daily and use water on the felts.
I got the Alpha Stim through Brain Harmony, who also provided me with a vagus nerve audio program developed by Dr Porges. Also currently doing the iLs program. Brain Harmony is a practice run by occupational therapists who guide you through the entire program. It created enough of a shift in my son with NDPH to make a difference in his life.
Thanks for the mention of alpha-stim. I’ve heard of it before – looking forward to checking it out.
People have been using the AlphaStim with great success for almost two decades. Acupuncturist inventor Daniel Kirsch with his wife Tracey has done great work for humanity with this multipurpose device. Takes a little experimentation for sensitive people. Deep slow breathing while using it always helps. Sensitives are advised to use the very lowest setting for only ten minutes per session when starting.
Others get trained on the very sophisticated Tennant Biomodulator from Senergy Medical. It is a much more complex and expensive device with technology bought from the Russian space program after perestroika. They even carry a super pricey scalar wave device said to be designed with Nicola Tesla technology.
I think I have heard about both devices improving vagus nerve tone. This makes sense for those familiar with acupuncture and yoga body technologies and energy medicine. The Biomodulator is specifically designed for balancing the right and left meridian channels while delivering electron boosts that awaken cell mitochondria healing. Both the AS and Biomod need a doctor’s prescription, and these are easy enough to get.
Donna Eden has some podcast energy boosting routines and a fantastic manual on acupoint stimulation that can help the lymphatic system and vagus nerve regenerate the body over time.
For those who love low tech (listening to binaural sounds while resting with a portable CD player in bed) for the moment or are on a budget, the Jim Donovan Sound System is a series of sound CDs for vagus nerve healing, improved sleep and other healing modes created by this sound therapist/engineer and professional musician and professor (M.Ed.). It costs about $60. for seven or so CDs and is well worth the investment for a moneyback guarantee. Even one CD is worth the price for the “pink noise” nightly sleep inducer.
One does not have to wait for another ten to twenty years to find relief.
Hi Beca
Thanks a lot for such interesting information. I use Donna’s general energy techniques but would be grateful if you could tell me where I can find the information/ manual on the vagal and lymphatic system. Many thanks. Evelyn
Thanks Cort, I find this really interesting because one of the symptoms I had with the virus that triggered my ME in 1998 was shooting pains up the left side of my neck. I never thought about it until early this year when I read “Accessing the Healing Power of the Vagus Nerve” by Stanley Rosenberg (prompted by something I read on the HR website). I realised that the shooting pains I had all those years ago were most likely my vagus nerve. Maybe the virus affected it in some way? Since January, I’ve been doing Rosenberg’s “basic exercise” which he says increases the blood flow to the brainstem and stimulates the ventral branch of the vagus nerve. I now have more energy and strength than I’ve had for 6 or 7 years and some nights I even get refreshing sleep, which hasn’t been the case for decades. I think there are other factors in my improvement too, but I do think that stimulating my vagus nerve with the basic exercise and another exercise he recommends has been hugely helpful for me. It’s such an easy thing to try – I do the basic exercise while lying in bed with my arms supported by my pillow so I think most people with ME could do it. It might be worth a try for just the cost of a book. I confess I wasn’t able to read the whole book but the exercises are all in part 2 at the back.
Thanks for the tip, Nicole. I look forward to checking it out.
I found Dr.Tracey’s talk fascinating. There is a lot of emerging research in the literature that portable transcutaneous devices are also effective, though possibly not with the same precision or power as an implanted device. I’m wondering if the Nemechek Protocol or the one that Sterling Cooley promotes would be a viable alternative. Has anyone here tried either of these?
My family has been doing a vagus nerve program through Brain Harmony, a program developed by Dr. Porges.
I was on Sterling Cooleys FB page for quite a while and asked him some questions whereupon he banned me from the group. This has happened to several people that I have personally seen, and my questions were not negative in any way so I would be careful of him and his products. I don’t necessarily think he’s a bad guy, but he’s definitely selling something and not interested in discussing the concepts. Caution is warrented. Just my 2 cents.
I too would be wary of Sterling Cooley’s program. I followed the group for quite a while and got a very bad feeling about the whole thing. The Nemechek protocol has a lot of rules and if you do anything “wrong”, you get blamed. I also did the Brain Harmony program and wrote about my experience here, https://9gurus.com/polyvagal-theorynot-the-disney-kind-of-frozen/
Interesting topic, Cort. Recently, the FDA issued an Emergency Use Authorization for the non-invasive ElectroCore Vagal Nerve Stimulator for asthmatics at risk for COVID-19 and there’s an Allegheny Health Network Research Institute clinical trial for it as a treatment to reduce respiratory distress, prevent the need for ventilation, and evaluate cytokine trends (cytokine storms, oxygen requirements) & reduce mortality.
https://clinicaltrials.gov/ct2/show/NCT04382391?term=ElectroCore&cond=COVID19&draw=2&rank=1.
How about that! I had no idea. Thanks for passing that on 🙂
Cort, this article indicates it’s already approved for migraines & headaches and that “anti-inflammatory” effects may be due to signals sent from the brain down through the vagal nerve regulating cytokine production.
https://spectrum.ieee.org/the-human…lator-gets-emergency-approval-for-covid19-use
Sorry, above link didn’t copy correctly. Hopefully this one works.
https://spectrum.ieee.org/the-human-os/biomedical/devices/handheld-vagus-nerve-stimulator-gets-emergency-approval-for-covid19-use
Just like Tracey said…..Anti-inflammatory, baby!
During the few times I’ve had acupuncture, I believe I had heard that the ears are some of the most powerful places to place needles into. For some reason, the non-invasive techniques mentioned in this article immediately made me think of that. Perhaps the Ancient Chinese were onto something with acupuncture in the ears!
I have long been interested in VNS and have collected quite a few research articles about its use. It is possible to rig a TENS unit for non-invasive use although micro current devices are reputed to be easier to use. It is relatively easy to get all the components on-line.
Not being very knowledgable about electrical currents, I have been shy to try this. There also is not a lot of agreement on what voltages are most effective. The body contact is parts of the ear although one side of the body (can’t remember which) controls the heart and so best to avoid experimenting with that side!
There are some research papers reporting amazing results including one researcher who cured achalasia using something similar. I have achalasia and so tried to locate this paper but was unsuccessful–so far.
For less adventurous folks, various types of yogic breathing (not just belly breath) have been studied and do contribute to vagus nerve tone.
Thanks, Nancy. A blog on yogic breathing is actually coming up. Very interesting stuff….I’m surprised at how many different types of breathing techniques there are.
The Senergy Medical people (Dr. Jerry Tennant and family) have done a lot of specific research on voltage requirements for cellular healing, at least for their devices. They do not provide their devices without offering a week’s training whenever possible for the client.
Both Dr. Tennant and Donna Eden cured themselves of serious disabilities using their chosen methods. Their stories are available online.
Any benefits for Hyper-Adrenergic POTS?
Hyper-adrenergic POTS indicates really, really high sympathetic nervous system output. Since the goal in much of this is to increase parasympathetic nervous system activity in order to reign in the SNS, I would hazard a guess – without knowing too much about hyperadrenergic POTS – yes.
I’m so glad that the word may be getting out about this! I was able to regain quite a bit of functioning after beginning IV fluids (as long as I continue 2L over 2 hr every 2 weeks- after Cort’s previous article about ORT I tried to get off my IVs and only use ORT, but I started to go downhill without my IVs for some reason).
Anyway, I was able to start exercising again but still flaring about 20% is the time until I began alpha stim. I now have felt 100% normal for the past 6 weeks, which has never happened since I came down with CFS in 2016. I wonder how the treatment in this article is different from alpha stim? Either wayx the next huge step is to get these treatments covered by insurance and for universal healthcare coverage!
I read this site regularly but have never written before
I’ve been sick almost 40 years with CFS
This is very exciting and I’m wondering if it is possible to get the implanted device somewhere at this time?
I know it will be costly etc but does it exist?
thanks everyone
I don’t know. It is approved for some people with epilepsy I believe but I know that at least one person with fibromyalgia has had it done. I’m not sure how that happened.
Dysautonomia International has three studies underway on non-invasive vagus nerve stimulation, though, in POTS. They’re looking at three different kinds of stimulators. Once those studies we should know more about which types work best – and they will be much cheaper and much easier to get.
O.K. admittedly my comment belongs in another of your blogs Cort, but I think this tidbit warrants some consideration;
https://www.scientificamerican.com/article/what-we-know-so-far-about-how-covid-affects-the-nervous-system/
The most interesting part is that researchers are doing experiments with proteins present on the tip of Covid spikes to control pain! Maybe there is a silver lining to this pandemic…
Any blog is a good place to place a comment on COVID-19. Thanks! I think there will be several silver linings for ME/CFS to the pandemic.
I’ve just taken part in Niki Gratrix’s free Weekly Healing Circle. I really enjoyed it and even though it was my first time there, I found the experience calmed me and reminded me of a deeper connection to a natural energy source that I used to have but have unfortunately become disconnected from.
There were over 500 of us from all around the world. The webinar is then available to watch for free for 48 hrs. It might not be for everyone but I was pleasantly surprised by how friendly and accessible it was. Niki chats about a topic for the first 15 mins, or so – today was blood sugar balancing and then she has pre-recorded meditations with music and Niki talking. Niki mentioned that she will be discussing the Vagus Nerve at the Parasympathetic Summit in November.
Here are links to Niki’s Weekly Healing Circle and the Parasympathetic Summit, that Cort mentions above:
https://nikigratrix.com/weekly-healing-circle/
https://parasympatheticsummit.com/?idev_id=869
Thanks for passing that on.