

Every once in a blue moon you come across a study that kind of stops you in its tracks. This is that one for me. (Thanks to Tim Vaughn for pointing it out.) If the findings hold up it could transform our understanding and approach to fibromyalgia (FM). Plus, it has interesting possible implications for chronic fatigue syndrome (ME/CFS).
Is fibromyalgia actually an immune disease? (Image of antibodies)
In 2019, the Goebel team behind this study replicated the symptoms of CRPS (Complex Regional Pain Syndrome) in mice simply by giving them IgG antibodies from people with CRPS. What they did in CRPS a couple of years ago, they did in fibromyalgia this year.
IgG antibodies are the most common type of antibody found in the blood. Their ability to bind to pathogens means they make up an important part of our immune defense. These powerful factors also neutralize toxins, activate the complement system, etc. When they go wrong, though, they can also trigger autoimmune attacks on human tissues.
That’s what the authors of this study believe has happened in fibromyalgia. The autoimmune attack, though, hasn’t targeted just any tissues. Their study suggests that an autoimmune attack has targeted the actual nerves in fibromyalgia… No wonder it’s such a nasty disease.
Transformational Fibromyalgia Study (?)
“IgG is the principal pronociceptive, pathological serum component in FMS patients.” The authors
Goebel et. al started off their study by noting that along with the central nervous system problems found in FM, researchers have also found evidence of “peripheral” problems; i.e. problems affecting the body. These include hyperactive sensory nerves, disappearing small nerve fibers, and some immune issues. Those, plus the high rate of FM found in some autoimmune disorders, led the Goebel team to suspect an autoimmune basis for FM.
This is despite the lack of consistent and overt inflammation findings in fibromyalgia and ME/CFS. Researchers have looked for consistent evidence of the inflammation they’ve believed must be there almost since day 1. In 2016, Goebel proposed an alternative. While referencing chronic fatigue syndrome (ME/CFS), he proposed that autoantibodies may be causing pain without producing inflammation by tweaking the receptors on the pain-producing nerves.
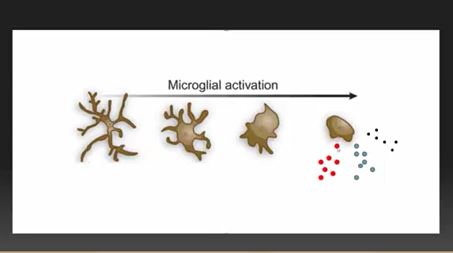
Activated microglial cells in the body play a key role in this hypothesis
One of the main culprits in this new scenario is our old “friend” – the glial cells. Presumably, to protect them, glial cells wrap themselves around the nerve bodies found in the dorsal root ganglia (DRG).
The DRG, which are found just outside the spinal cord, provide central processing gateways through which the sensory and autonomic signals from the body pass on their way to the spinal cord. The DRG then, determines which and how many pain, sensory, and autonomic nervous system signals make it to the central nervous system. That, of course, potentially leaves them at ground zero in fibromyalgia, and indeed, Dr. Martinez-Lavin proposed that about ten years ago.
Like glial cells everywhere, the glial cells surrounding the DRG can produce a wide range of substances (ATP, glutamate, TNF, other cytokines, fractalkine) that can activate the nerves, causing them to produce pain and other symptoms.
The Study
In his past fibromyalgia studies, Goebel had mostly concentrated on temperature and sensitivity issues, but in his 2021 study, “Passive transfer of fibromyalgia symptoms from patients to mice “, he brought autoimmunity to the fore.
The study injected purified IgG from people with FM and healthy control subjects into mice. Within two days, the mice given the IgG from the FM patients had become hypersensitive to pressure, cold, and pain, and reduced their grip strength. The mice given the IgG from the healthy controls, on the other hand, had no problems).
The problems stopped at the dorsal root ganglia- found outside the central nervous system (Image from Wikimedia- Ganglia Category)
Further studies indicated that pain receptors on the nerves outside the spinal cord had become hyperactivated in the FM mice. Over the next couple of weeks small fiber neuropathy cropped up. Goebel had swiftly given mice fibromyalgia simply by giving them immune factors found in his FM patients’ blood.
Importantly, the IgG from the FM patients never made it to the spinal cord or the brain; instead, it mostly accumulated in the glial cells surrounding the dorsal root ganglia. While the glial cells just outside the spinal cord became hyperactive, the activity of the glial cells inside the spinal cord remained at normal levels.
Despite all the CNS (central nervous system) findings in FM, Goebel was able to push the small pain nerve fibers found outside the CNS into a state of hyperactivity without any central nervous system involvement at all.
At no time did systemic cytokine levels increase. Whatever the IgG was doing, it was doing it very locally. Goebel’s findings could explain one of the great mysteries in FM and ME/CFS – why the cytokines often associated with pain and fatigue haven’t been found to be consistently up-regulated in these diseases. Well aware of the implications of their findings, the authors wrote their results may
“transform future research and facilitate development of mechanism-based therapeutic interventions (in fibromyalgia)”.
In 2013, Goebel proposed that chronic regional pain syndrome constitutes “a new kind of autoimmunity”, and that other “functional disorders” like FM, ME/CFS, IBS, interstitial cystitis, and chronic pelvic pain syndrome may be autoimmune as well.
Similarly, in 2017 ME/CFS researchers including Shoenfeld, Scheibenbogen, and Martinez-Lavin proposed that chronic fatigue syndrome (ME/CFS), POTS, and complex regional pain syndrome (CRPS) belong to a new category of autoimmune diseases they called “autoimmune neurosensory dysautonomias“.
While we don’t know how the central nervous system ties into these findings, they suggested that one may not need to access that complex system to make a difference. All that may be needed is to damp down the immune response and reduce the levels of those IgG autoantibodies.
Brief attempts to discover the suspected autoantigen – the protein the authors believed the FM patients’ IgG was binding to – were fruitless but suggested it was not a common one.
The idea that something in plasma may be causing FM brings up, of course, the intriguing question, given the similar findings, of whether the same process is occurring in ME/CFS. One wonders as well whether the adrenergic autoantibody findings from Scheibenbogen’s ME/CFS work could apply.
The Gist
- The study filtered IgG antibodies out of the blood of people with fibromyalgia and healthy controls and then introduced them to mice.
- The mice receiving the IgG antibodies from the FM patients quickly developed fibromyalgia-like symptoms; i.e. they became hypersensitive to pain, pressure, and heat, and their grip strength declined. The mice receiving the IgG antibodies from the healthy controls remained unchanged.
- Further study indicated that the pain-sensing nerves have become hypersensitive and that the mice over time developed small fiber neuropathy.
- No evidence of inflammation was found. Instead, it appeared that the IgG had activated the glial cells during the dorsal root ganglia or DRG. The DRG contains bundles of nerves that serve as the last sensory signal processing center before the signals reach the spinal cord and central nervous system.
- While the study found a high uptake of fibromyalgia IgG in the DRB and microglial cells it found no IgG uptake in the spinal cord or central nervous system indicating that the pain hypersensitivity found in the mice was all produced outside the central nervous system.
- The authors believe that a new kind of autoimmunity – one which does not produce inflammation – is present in fibromyalgia. Instead, the autoimmune processes directly target pain-producing nerves.
- The fact that the mice returned to normal when the FM IgG levels declined suggested that the illness is not permanent and could be reversed by removing the autoantibodies from the FM patients. They suggest that therapies like plasmapheresis and immunoadsorption – which is being assessed in ME/CFS – could help. Other therapies which target the suspect autoantigens (when and if they are found), as well as therapies like IVIG, are possibilities.
- Kevin Tracey lauded the study calling it a “tour de force” and “a shining example of how and why clinical translational research can and should be done”. He suggested it could provide insights that will lead to new and effective therapies for FM.
Next Up
The author’s next steps involve further delineating how these “autoreactive” antibodies are tweaking the glial cells and/or dorsal root ganglia. The authors didn’t downplay the central nervous system problems that have been found in FM but did state that future studies will determine if the process they’ve uncovered could be causing them.
Treatment Possibilities
As the authors note, the current treatment options for FM, which rely mainly “on lifestyle changes, physical exercise, and drug therapy with antidepressants and anticonvulsants”, leave “an enormous unmet clinical need”.
The fact that the mice returned to normal when the FM IgG levels declined suggested that the illness is not permanent and could be reversed by removing the autoantibodies from the FM patients. They pointed to IgG-reducing approaches such as plasmapheresis or immunoadsorption. Intriguingly, Scheibenbogen has found some success in small studies with immunoadsorption in ME/CFS, and the BC 007 aptamer is a candidate for both ME/CFS and long COVID. They also suggested that approaches that specifically target the autoreactive IgGs could be effective.
Also, without naming them, they proposed therapies that “interfere with the binding of autoreactive antibodies or prevent their functional consequences”. These therapies presumably include IVIG.
In his review, “From human to mouse and back offers hope for patients with fibromyalgia” Kevin Tracey, MD, proposed that therapies that interfere with IgG binding to the glial cells/DRG, or which stop those cells from reacting, will also be developed. Tracey, the President, and CEO of the Feinstein Institutes has published hundreds of papers and is a leader in inflammation, vagus nerve, and bioelectronics research.
Check out Tracey’s stirring talk at the 2020 Dysautonomia International Conference about the bioelectronics revolution.
Tracey called the Goebel paper:
“a shining, impactful example of how and why clinical translational research can and should be done” and stated, “This tour de force study at once provides insight into a mysterious disease affecting millions of people and offers a solid and promising insight into just how to begin developing effective therapeutic strategies for a severe, chronic, uncurable illness.”
Let’s hope!
Update – FM researchers score a major grant to contain their antibody studies.
Like This Blog?
Keep the Information Flowing by Supporting Health Rising


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!


thank you Cort! not that i have FM, i think because no specialist has followed me up and lonterm to bedridden to go to someone, but it gives hope for me/cfs to! i hope they do the study for me/cfs to. just read that syström says every patient with ME he sees has sfn and preload failure and snf is treatable with ivig. wish i lived in the US!
but is it not the same with bloodtransfusions? my dad had in his 80’s need on 7 or 8 bloodtransfusions. otherwise he was immediatly death. but i know, everything they do in medicine has his pro’s and cons. that is the hard thing on medicine. i respect totally your decision but if your life is in danger? same with for excample cancer therapys and so many therapys.
It gives me hope for ME/CFS too. This was a painstakingly done study and I hope similar studies are done in ME/CFS. I find the idea that something in the blood is contributing to both these diseases really intriguing…
And, off the subject a bit, what a role Systrom is playing in ME/CFS and how nice that he’s paired up with Ron Tompkins at the ME/CFS Collaborative Center in Harvard.
Plus what a bunch of fascinating hypotheses – from the Metabolic Hypothesis to the second part of Wirth and Scheibenbogen’s ME/CFS hypothesis (a blog on that soon) to the hypothalamic/limbic injury ideas – that have shown up recently.
What do you make of another recent article from a German scientist re BC007 drug that helped someone with long COVID – I read it works on B adrenergic receptors?
@ Julie
I have linked this study in a comment below. The drug B 007 is an aptamer (DNA or RNA oligonucleotide which can bind all sorts of molecules in its 3 D structure). It has been suggested for the removal of all sorts of proteins – including viral proteins of SARS-CoV-2: https://berlincures.de/new-paper-aptamer-bc-007-efficient-binder-of-spreading-crucial-sars-cov-2-proteins/
They have now tried it for the removal of GPCR-autoantibodies – apparently successfully (see my comment).
I read your article with great interest. I would like to tell you something interesting. I have had fibromyalgia since my early 30’s. I am now 70 years old and have suffered severely with this. I also now have osteoarthritis and lupus. In 2018 my rheumatologist tested my Immunoglobulins. I have IgG deficiency. I don’t make these antibodies. I don’t know how long this has been happening in me. But, oddly, my fibromyalgia pain symptoms are practically nil for the last few years. I still have severe chronic fatigue but little to no muscle pain. I had moved to Las Vegas 20 years ago for dry weather in hopes of feeling better but still had flare ups. Last year I returned to the east coast. I lived through an ice cold, snowy winter and an enormous amount of rain and dampness with no muscle pain, no muscle relaxants and no meds for fibro. I have done very heavy yard work with very low level soreness the next day. I have other health issues but I don’t even think about fibromyalgia any more. I see it this as a miracle in my life.
Is this a coincidence or could this be related to the info in your article??? I am very curious to understand this.
This is for Judy 7/9/2021. In 2018 what did you do that stopped the FM pain. If that could happen to me I’d think I’d died and gone to heaven. I was also diagnosed in my early 30’s and I am now 73. I am on a number of painkillers and muscle relaxants but I can honestly say that in the last 40 years there has not been one single day without chronic pain. If there’s any advice out there I’d love to hear it.
Have you tried low dose naltrexone? We have a resource page on it on Health Rising = https://www.healthrising.org/treating-chronic-fatigue-syndrome/drugs/low-dose-naltrexone-ldn-fibromyalgia-chronic-fatigue-syndrom/How about cannabis? Really helpful for some people. We have a series of blogs on it.
IVIG has the same warnings as a blood transfusion. It is not without risk or some serious potential side effects. You are getting thousands of other peoples IGG antibodies.
I was offered it because of my Hypogammaglobulinemia and decided not to get it.
yes, i know. i read a comment on that with a link to an artikle that i already had read but was ofcource forgotten.
https://www.karger.com/Article/Fulltext/498858?fbclid=IwAR1jTE-7cwaXfw7d29Uxz2Lrk41iOGdQwZxmYUjBiun40nGMMrMwXw4cujQ#menu
maybe it can help someone,i hope so!
While IVIG may help some I don’t think it’s going to be the answer for this. After some good small trial results, Goebel’s large IVIG trial in CRPS – which probably took years to get started – most unfortunately failed. Totally reminded me of Rituximab.
https://pubmed.ncbi.nlm.nih.gov/29144634/
Immunoadsorption – which is available in some European countries, I believe, seems like a better bet. Even better, I would think, would be identifying the autoantigen and blocking the antibodies from attaching it. First, of course, they are going to have to find it. That search will be interesting.
Hopefully, this kind of research will get a boost from this fascinating study.
you are so smart and i have no brain anymore, thanks
can only try to survive…
Hang in there Konjin! Don’t miss out on what I know we’re going to learn from long COVID and stuff like this.
I did one IVIG treatment about 12 years back and developed aseptic meningitis the next day. My neurologist had her own IVIG clinic and had a number of patients with lupus, ms, polymyositis, etc. having good results. I never did another IVIG treatment after that one. I suffered for 2 months with severe headaches and pain. I have lupus, intracranial hypertension, Graves, mild hypermobility and chronic exhaustion. I’m a recently retired pharmacist. I appreciate reading these interesting articles by Cort and everyone’s comments. Thanks, Shari
I wonder if they are comparing their findings to the 2018 finding of Birnbaum et al of anti-calponin-3 autoantibodies which bind to the satellite glial cells of the dorsal root ganglia. These autoantibodies were discovered to be notably higher in Sjogren’s disease patients with neuropahty (and now we know some fibromyalgia patients have small fibre neuropathy too). Has Birnbaum et al identified the antibdoy target already? Given the overlap in symptoms and onset (often post viral) it would not be crazy to investigate this as a possible target? Of course there are likely many targets, but would be interesting to know
@ issie may i ask what you did instead or was there no insted? do you also have sfn from it and if, how do you treat it? thanks and best wishes!
Can you please explain what immunoadsorption is?
Good question – I looked it up and immunoadsorption is really interesting. In immunoadsorption specific immunoglobulins (antibodies) are removed from the plasma (blood) and then the plasma is returned to the patient. It was developed in the 1990s. It’s used in various autoimmune-mediated neurological diseases but is expensive.
https://en.wikipedia.org/wiki/Immunoadsorption
Lynn P., for more reading on immunoadsorption see
https://www.sciencedirect.com/topics/immunology-and-microbiology/immunoadsorption
Very interesting! What do you think are the chances of another group coming along to try to replicate the study?
I don’t know but Goebel’s is well published and Tracey is a hot researcher. It was good to see him lauding this study. Hopefully the study will get some good attention, Goebel will get some good funding, and we’ll see more interest in this general field in ME/CFS.
I personally believe that if we receive a transfusion (blood other than what we were originally born with) then it would be a foreign substance to our body. I had a transfusion as a child, also I’m an RH neg baby (me +/ mom-), had transfusion due to anemia, very tired at that time as well. At age 25, gave birth, needed transfusion due to complications. Growing up, had the fatigue and sleeplessness. It was not until this second transfusion that I really noticed the many nerve, muscle and bone pains. Diagnosed with Fibro in my 40’s.
What would happen if we all were given the anti-rejection medication that a person would get with an organ transplant. Blood transfused is foreign and should carry cells from the donor. Has this been tested for the Fibro/ME/CFS, etc. in any studies? I am almost inclined to ask my doctor to give me the shot so I can see if it works.
This IS a replication of the original study by David Anderssen from Kings College London. I reported the original study here many months ago. Anderssen was also a lead researcher in this replication but done in a different lab in Sweden.
I hate to say it, but the pandemic was a great help to FM/ME/CFS research. I certainly don’t want people to die for my benefit, but the “long I haulers” are showing medicine that we aren’t crazy! I read about long I haulers that commit suicide and wonder why we don’t get published suicide reports?! I always knew this was an autoimmune issue. I had gastric bypass in 2004 and started experiencing symptoms in 2008-2009. I also had a baby in March of 2008 at 40. All these things stirred in a pot and FM soon surfaced. Also though my Sed Rate is never super high, I always have an elevated one which would indicate inflammation!
Amy Proal PhD @microbeminded2 has this response to the Goebel study on Twitter:
‘And the million dollar question is: what were the IgG antibodies in the blood of the #fibromyalgia patients created in response to? Certainly a broad range of pathogens/organisms induce IgG antibody production.’
@Tracey
Exactly! We should not forget this point.
If there are auto-antibodies there is some upstream process that induces their production. This most likely is an inflammatory process with an activated innate immune system. This activation of the innate immune system may be caused by a) active (viable) microbes, b) a microbial remnant (left-over from an infection which continues to stimulate the immune system (“viral ghosts”), c) reactivation of dormant endogenous microbes (e.g. herpesviruses like EBV – indeed shown to possibly happen in Long Covid patients: https://www.mdpi.com/2076-0817/10/6/763 or d) but also autoimmune processes that may have been triggered during an infection and have created immune targets within the body. There are also other processes that can activate the innate immune system, including mechanical strain/sheer stress on tissues.
All these processes are being discussed for ME/CFS (and now PASC).
I’m a LHer for the past 6 months.
Interestingly, Bruce Patterson et al has also published (pre-print)
https://www.biorxiv.org/content/10.1101/2021.06.25.449905v1
Showing that “A statistically significant number of non-classical monocytes contained SARS-CoV-2 S1 protein in both severe (P=0.004) and PASC patients (P=0.02) out to 15 months post-infection.”
I see some patterns here.
I did notice a noticeable downturn after my 2nd dose of Moderna.
The upstream process will be (like ME/CFS) an epigenetic process and the protein folding subsequent to this. I also think that the protein, which ends up being an antigen for the IgG binding, involved in receptors is not an uncommon one at all. I suspect a melastatin protein which is a co-effector on the receptors. However there are many possible other structural proteins. We also must take into account in finding this protein (if it is only one) the many other symptoms of FM (and ME) ie the protein/s will be occurring in other receptors beside pain receptors and the nervous system.
Herbert,
I believe the issues with FIBRO, ME/CFS are all EBV related.
Good to hear that Amy Proal is on this. I hadn’t thought of the pathogen angle. We must remember that, as in ME/CFS, an infection can trigger FM. Or perhaps an endogenous retrovirus could have been unleashed. Or as Marshall and Ariza are showing in ME/CFS – a smoldering infection could be leaking proteins into the body. Or come to think of it – perhaps a leaky gut could be leaking proteins (or viruses?) into the bloodstream that are triggering an immune response. Goebel was one of the first to identify leaky gut in FM. I assume but don’t know that IgG antibodies would respond. They also respond to toxins…
There’s the original trigger and then there’s the protein in the body that the antibodies are attacking. I think they’re going to focus on finding that protein…
Very interesting, one may speculate that the target tissue in FM may be the glial cells in the dorsal root ganglia, while in ME/CFS it may be the glial cells in the CNS ?
Here in Berlin at a private company (Berlin cures) they are already working on a drug (BC 007 – a polyspecific anti-autoantibody of sorts) that has been shown to effectively remove GPCR-autoantibodies (like adrenergic or muscarinergic autoantibodies) from the blood. (So it works a bit like the apheresis that Scheibenbogen has used on ME/CFS patients to remove autoantibodies but it is much less cumbersome, less expensive and more specific). The authors write:
“BC 007 is a powerful new drug for neutralizing GPCR-AAbs associated with various pathological constellations” ( https://berlincures.de/new-paper/
Today they report that they have treated a Long Covid patient with the drug – apparently the patient is free of symptoms after the treatment:
https://berlincures.de/long-covid-patient-succesfully-treated-with-bc-007-long-covid-patient-erfolgreich-mit-bc-007-behandelt/
Details on the experiment: https://www.fau.de/2021/07/news/medikament-gegen-autoantikoerper-hilft-bei-long-covid/ (only in German) – apparently the patient suffered from anosmia, fatigue and cognitive dysfunction after Covid-19. They gave him (or her) one infusion, within “a few hours” he/she improved, on discharge after a 3 day observation period he/she had regained her sense of taste, concentration difficulties had subsided, general performance had increased.” They confirmed this with a novel method of capillary angiography, in which one can visualize capillary perfusion (which increased after the drug was given).
Very interesting and somewhat linked to the FM-study you discuss! Thank you!
That is fascinating. I had no idea. How cool that it’s cheaper as well. Thanks for passing that along.
I was diagnosed with fibromyalgia Six years ago and with a rare allele of alpha-1 antitrypsin deficiency (AATD) three years ago. I’m heterozygous for the rare mutated allele so, though my alpha-1 antitrypsin level is low, I don’t qualify for plasma infusions.
There’s research that shows that people with AATD who have fibromyalgia and receive blood plasma infusions, their fibromyalgia symptoms resolve. The particular study involved two sisters who then weren’t able to have plasma infusions for a prolonged time due to a plasma shortage, and their fibromyalgia symptoms returned.
I wish I could get plasma infusions to see if my fibromyalgia symptoms would resolve. I wonder if people diagnosed with fibromyalgia are deficient in alpha 1 antitrypsin and aren’t aware of it.
Interesting. I’m also heterozygous for Alpha 1 Antitrypsin and mildly deficient. I have both ME/CFS and Fibro and a history of Hashimoto’s and Guillain-Barre. So I wouldn’t be surprised if some kind of autoantibody is discovered as the driver of the first two.
I think in that case it means their fibro was aatd then? By plasma infusion do you mean IVIg? Or infusion of aat
Auto immunity, auto immunity, auto immunity, auto immunity, …FM or ME/CFS
Doesn’t matter which. We’ve been saying it for years. It’s not until it manifests itself in an easily tangible form that we recognize it.
Took a spinal cord lesion for them to recognize it in me.
But is this treating symptoms and not the cause???
Yes – so far as I can tell immunoadsorption would be treating the symptoms. I assume, but don’t know, that treatments would need to be ongoing. If they find something that blocked the autoantibodies I imagine it would be the same. That’s kind of standard for chronic illnesses; they can be beaten back but are rarely totally resolved. I don’t know if it’s possible to turn off the production of specific antibodies or not.
The Goebel study is really impressive. I think we may be missing an important point, though. If circulating IgG contains elements responsible for ongoing symptomatology, this has important implications for therapy. Plasmapheresis* is a commonly available procedure with only modest risks. It has wide applications and most hospitals will have it available. If it can be shown that many/most people with fibromyalgia or ME get symptomatic relief from this procedure, it will redirect research efforts in these conditions. A clinical trial should be straightforward to organize.
Immunoabsorbtion, a related procedure, yielded encouraging results in small ME studies by Scheibenbogen (2018) and Tolle (2020).
* https://en.wikipedia.org/wiki/Plasmapheresis
Thanks Rich…Good to hear about plasmapheresis…:)
I wonder how many of us have a positive ana with a rare pattern. Or how many have a high Ch50 ( complement 50) like I do. Both of these are the only indications that there’s something wrong with me and none of my doctors want to answer why my Ch50 is high or what my pattern means.
Interesting findings and thanks for the good report Cort!
That may link to the large group of ME patients that nearly never suffer from infections like colds. It sort of can link to near all patients with a strong Dauer response. The Dauer response and / or mitochondrial fragmentation turn the mitochondria away from producing ATP and towards producing a chronic strong innate immune response.
A constant strong innate immune response will tank feeling healthy and energetic, but will fight many pathogens without the need to produce *and multiply (pre-existing)* antigens.
=> So: if the body knows you have trouble with “undefined” auto antigens, it can:
A) operate the immune system “as if you were healthy” and get you big chances for a “real” auto immune disease with clear observable damage each time the adaptive immune system is activated and multiplies (also pre-existing) antibodies?
B) turn up the innate immune response 24/7 preventing most pathogens ever becoming that numerous so that the adaptive antibody producing system does need very few and rare activation, reducing multiplication of wrong / auto antibodies that cause trouble (but at the cost of lack of a functional life)?
Issie sort of exemplifies that: diagnosed immune insufficiency that “should” make her unable to survive several common infections but having not only survived those but several other infections that “should” have killed many healthy people.
Thanks for alerting us to this research, Cort! I had CRPS, Neuropathy and Fibro- mylagia, in that order, between 1992-1997. I still have neuropathy and I developed POTS in 1994..and about 25 other conditions. since then. I pushed some into remission but I’m still trying to push all of them away, especially POTS. I also have elevated ANA. Over the last two years I’ve worked at ruling-out Sjogren’s Syndrome and Myasthenia Gravis but I don’t appear to have either of them. However, I still suspect an autoimmune condition is the underlying cause of my symptoms. Many POTS patients do have underlying autoimmune conditions. Lately, I have wondered if I have an unknown autoimmune condition that attacks nerves. So, this article really peaks my interest! And, I hope they continue to pull the thread on this discovery!
I have said for years that Fibromyalgia is Autoimmune, I’m so happy that research is going forward to help us all
Habe ich zu meinem Arzt auch immer gesagt. Als Antwort kam: Schweigen, weil er sich da nicht festlegen will.
I used IgG and IgE food testing to inform my choices about what to eat. I needed some form of objective information to make changes to diet. Guessing would have left me in the dark because I react to so many categories. These are not perfect tests, but has been high on the helpful tests for me.
Dejurgen: interesting comment because I (diagnosed with M.E. & FM) I had toxoplasmosis without knowing it, survived a brown snake bite without medical attention and although next to my husband did not get covid-19 when he and my daughter had it last year.
Thank you Cort for all you do, and helping us to cope & understand the best way we know how. To say the least, it hasn’t been easy, or that simple to have ME & fibromyalgia. Its hard to suffer alone, thank you for bringing us all together…. having this connection and showing us we aren’t all alone.
After reading your article, I came across a different one posted on Jul 6th, 2021….. not sure if you, or others have seen this???
>>> https://fibromyalgiaresources.com/fibromyalgia-autoimmune-problems/
We can only hope that through constant research we all can find answers, and some type of relief in suffering.
The hardest is the unrelenting pain. Luckily, my doctor prescribed LDN, and for me its been helping curb the exruciating pain even during sleep. It’s good to find an understanding doctor.
My previous primary care doctor couldn’t even figure out I had Pernicious Anemia!….. while my current doctor uncovered ME/cfs, fibromyalgia, PA, I’m also immunocompromised, etc…..
We look to you as our only hope! Again, thanks Cort for being there!
Thank goodness you’ve found an excellent doctor as it makes all the difference! I had not seen the study you linked us to and I only skimmed it for right now but it be interesting to hear what anyone else has to say about it.
Well said about Cort making this a community and the hope that scientific research brings. I’ve been ill for almost 25 years and it becomes really frustrating when nothing seems to come from the research but Long Haulers seem to have changed the game plus this potential treatment that is now an official study sure makes me more hopeful
Cheers!
You’ve reported on the mysterious X factor im the blood in dysautonomia as well.
So it’s in ME/CFS, and FMS and dysautonomia – and who knows what other disease processes too, because they either haven’t beem studied or we do not know about studies.
Here’s what I wonder:
What if the substance us easy ti detect it’s just that it’s not normally thought to be problematic, so it evades recognition?
What if it is hiding in plain sight
out of ignorance basically?
After taking 800mg of ibuprofen for three days I found my pain all over from fibromyalgia had pretty much receded significantly. I had an inflamed knee is why I was taking the ibuprofen per the doctor. After taking all three covid shots my body literally stiffened up big time along with gastro issues and the knee got stuck with inflammation and I could barely walk. So basically I have inflammation all over was my aha moment. My doctor looked at my labs and it turns out for at least 10 yrs I had slightly higher RBC, hemoglobin and hematocrit levels. I bet if I go back farther it will show the same slightly elevated numbers too. So basically I am holding onto too much iron. Hemochromatosis has many simular symptoms to fibromyalgia. I have lived with this pain for two decades and been to so many doctors and not one picked up on these higher levels until last week. They all just rubber stamped fibromyalgia diagnosis. They either don’t believe in fibromyalgia or they don’t know and just don’t want to go there so once you get that label from doctor any real help or investigation is pretty much nil from doctors after that is why I personally experienced. The pain started two decades ago and I have lived with this for that long before getting better answers.
I had been to a b.s. Kaiser seminar for patients about fibromalgia many years ago. Basically the female doctor talked for about 10 minutes and quickly left the room and they handed out this bibliography to patients of things they can chase for answers to keep us out of their waiting rooms. The conference room with about 4 dozen patients were shocked about how lame this was. We all came to learn more about it and what to do and they just gave us this multi-page handout of things to chase is all. Then some guy from Kaiser’s physical therapy in a Hawaiian shirt told us Kaiser will not provide any massage therapy for fibromaylgia patients and the whole room of mostly women groaned cause we all know it helps with pain. Then one women stood up just flabbergasted at how we were all duped at the seminar and she said “look around this room we are all over weight. That must mean something.” (Turns out from what I read is that hemochromatosis does impact the pancreas and insulin resistance too which translates to me packing on the pounds.)Then others began talking and then one woman asked if anyone had any pain meds on them and that started a whole lot of women talking about all the pain meds there were on per their Kaiser doctors. Well I did hospice for my father and I was hearing end of life type of drugs, hard core, being used by these fibromyalgia patients. The patients said it is a pain condition and this is the only way to deal with it. My jaw dropped hearing how they are being prescribed this heavy duty pain killers to keep them out of their waiting rooms. There have been studies showing how this condition costs healthcare sector big bucks and was the single most costly condition they have to deal with and that is saying a lot if you consider cancers etc. They say they would love to cure this but I wonder if it is the gift to their spreadsheets that keeps on giving and creating drug addicts. Not one of my Kaiser doctors caught the high levels of iron in my blood.
There were so many warnings in my blood labs but no one picked up on this.
I had donated blood 7 plus years ago and the nurse has to check the iron in the blood before hand. She said I had a higher iron level in my blood than men. My husband who is 6’4″ eats way more meat etc than I and my level was higher than his! That nurse didn’t say hey you might want to check on that. I hadn’t had a cold or flu for 4 yrs and I thought those high iron levels were part of the reason for that, which was unusual for me to avoid getting sick.
From what I have read you can still have hemochromatosis even with normal blood test results too. It is genetic. I wonder how many other people here really have other issues and have been diagnosed incorrectly. They say hemochromatosis is unusual but the stats I have read show it is more common than so many other conditions we hear about all the time! My labs were not super high so that may explain why I haven’t died from organ failure etc. but why I fly under the radar of doctors looking closer. What I did like with my new doctor and you may also have this as well now is the my chart online access. You can click on a tab that will bring up the last few years of labs so you can actually compare how many years your higher RBC, hemoglobin and hematocrits have been. You can see a pattern.
What I have found is it is hard to find a GP worth going to now a days. So many are older with one foot out for retirement and not really staying up to date and sleep walking or on the opposite side they are millennials who are unsympathetic and don’t want to listen and are also sleep walking and having to deal with too many patients during a day. This conveyor belt type of managed care is really not working well at all for many and can be costing folks organ failure, death or long term issues.
I have to go in for my blood tests but I now think fibromyalgia is more about an inflammatory expression from other known conditions that are not getting scrutinized and identified. Maybe this explains why doctors don’t believe in it? I now don’t.
Just as you can have multiple paths to why you vomit, so it is true with inflammatory responses. But vomiting is not a condition but a result of something else. Fibromyalgia is not a condition but can be a inflammatory result of Lyme disease or Homochromatosis or _____. But so many like to make money off of this by mudding the waters on this topic and selling snake oil ideas or treatments to keep this in play.
Before we get too excited and assume that “IgG antibodies are the cause” of fibromyalgia, let’s put this in context. There have been a number of studies that have found interesting biological imbalances in fibromyalgia.
For instance, various studies have found the following alterations in people with fibromyalgia: intestinal microbiome, serum cytokines, mitochondrial function, autonomic balance, vitamin D levels, hormone changes, and so on.
The current study, and those other studies do tell us that there are a variety of biological imbalances. But they don’t clearly tell us **cause** in a way that is practical yet.
It’s indeed compelling that the authors **caused** fibromyalgia by injecting IgG from affected animals. But we don’t know what caused the infected animals to have those nasty IgG’s in their system.
We also don’t know that those “fibromyalgic mice” have the same clinical picture as human beings with fibro. And we have some evidence telling us that different people may have a different overall signature of fibromyalgia, depending on their genetics and history and other factors.
In my experience, it is helpful for many people fibro, when they use safe means to address some of the biological imbalances. It will be interesting to see whether the current research study will lead to pragmatic treatment for human beings.
I see where Hep C patients commonly have Fibromyalgia. Not being detectable until the 1990s, Hep C infections had time to grow, and it is those patients who displayed Fibro symptoms.
Darn foggy brain, I can’t find the article but it was a recent
Web MD posting.