


New technologies and creative techniques are providing insights into long COVID and likely ME/CFS/FM and related diseases.
As we hoped, technologies are being used in COVID-19 and long-COVID research that would have taken years, if not decades, to show up in chronic fatigue syndrome (ME/CFS), fibromyalgia (FM) research. They’re accelerating insights into potentially critical issues in these diseases. None is more critical – particularly in ME/CFS – than the cause of the fatigue and lack of energy in these diseases.
Because blood vessels deliver oxygen to the mitochondria they need to provide free flows to them. It just makes sense that poor blood/oxygen flows to the cells is going to impede energy production. The interest in the role blood vessels and reduced oxygen delivery to the muscles plays in fibromyalgia, and particularly ME/CFS, dates back quite a ways. As far back as 2013, Health Rising was asking: “Are Oxygen Starved Tissues Causing Pain and Fatigue in Fibromyalgia and Chronic Fatigue Syndrome (ME/CFS)?”
Perhaps the strongest evidence that blood vessel problems exist in ME/CFS comes from David Systrom’s invasive exercise studies. Systrom’s ability to measure blood flows and oxygen levels before and after the blood gets to the muscles is the key.
The Gist
- Because blood vessels deliver the “juice” (oxygen in the blood) the mitochondria use to produce energy, it’s critical they flow free and clear.
- David Systrom’s invasive exercise studies in chronic fatigue syndrome (ME/CFS), though, suggest they may not be, and that the blood vessels are failing in two different ways: in one group of patients, the blood is being diverted away from the muscles shortly before it gets to them. In another group, blood is being lost after it’s gone through the muscles – leaving the heart with less blood to pump.
- Recent attention in long COVID and COVID-19 has focused on the endothelial cells lining the blood vessels and the damage the SARS-CoV-2 coronavirus may have done to them as it entered them via the ACE-2 receptor. These cells dilate the blood vessels when needed, protect the blood vessels from leaking, and produce pro-inflammatory cytokines.
- Enough endothelial cell damage has been found thus far for some researchers to assert that long COVID is an “enodothelioapthy”; i.e. a disease of the endothelial cells.
- Both ME/CFS and fibromyalgia studies have suggested that damage to the endothelial cells lining the blood vessels is present. One FM study even suggested that endothelial cell damage might be diagnostic for that disease.
- An Isreali study that used umbilical cords as a stand-in for blood vessels found that no less than 70 percent of the proteins that the SARS-CoV-2 coronavirus produces negatively impact the blood vessels in one way or another.
- Another that used something called “evolutionary rate correlation” suggested that many of the proteins the virus produces affects the coagulation/clotting process in the blood.
- Several studies suggest that the virus’s ability to produce micro clots in the blood may be why it’s so much better than the flu at producing ME/CFS states such as long COVID. One study found nine times the level of micro clots in COVID-19 patients compared to flu patients.
- Another study suggested that high levels of amyloid (misfolded proteins) were implicated in the increased clotting found in COVID-19.
- That was interesting given the results of a 2005 ME/CFS cerebrospinal fluid proteome study, which highlighted the presence of amyloid proteins. It proposed that misfolded proteins embedded in the blood vessels of ME/CFS patients’ brains were causing small leaks.
- Avindra Nath’s finding of punctate hyperintensities that appeared to be causing leaky blood vessels in the brains of autopsied COVID-19 patients, in turn, brought us back to a 1992 finding of widespread punctate hyperintensities in ME/CFS patients’ brains.
- Deformed red blood cells could also be impairing blood flows. They have been found in COVID-19, but it’s ME/CFS researchers at Ron Davis’s Collaborative ME/CFS Stanford Research Center that are leading the charge. In order to assess if deformed red blood cells are causing problems, they’re developing cheap and effective ways to assess blood flows through the microcapillaries.
- Blood vessel leaks in different areas, micro clots, narrowed blood vessels, and deformed red blood cells could all be impacting the flow of blood, and therefore oxygen, to the mitochondria in long COVID and ME/CFS.
- COVID-19 is doing precisely what we had hoped it would do: it’s jumpstarted research into areas of critical concern in ME/CFS/FM, and is producing new technologies, insights, and even treatment possibilities (e.g. Bruce Patterson’s attempts to solve the blood vessel problem).
His studies suggest two different blood vessel problems are occurring in ME/CFS:
- One group of ME/CFS patients has higher than expected oxygen levels in the venous blood. That suggests that a microcirculatory problem is shunting blood away from the muscles – depriving the mitochondria of oxygen. The inability of the mitochondria to get a hold of this oxygen during exercise results in higher-than-normal oxygen levels in the venous blood. It’s very easy to potentially explain the fatigue/PEM in this group – their muscles are simply not getting the fuel they need for their aerobic energy production system to kick in.
- Another group with normal venous oxygen levels, but with reduced amounts of blood returning to the heart (low preload), appears to have sprung some leaks in their venous circulatory system. This group simply doesn’t have enough blood reaching the heart for it to provide normal amounts of blood to the muscles.
While the cause is different in the two groups, the end result is the same – not enough blood (i.e. oxygen) is getting through to the muscles for the aerobic energy production system to fully kick in. Note that both problems feature leaky blood vessels – either right before the blood reaches the muscles or afterward.
Besides Systrom, Shungu’s reduced brain blood flows, Visser’s reduced oxygen uptake, ACE-2 problems in COVID and ME/CFS, and several hypotheses (Wirth/Scheibenbogen, Fluge/Mella, Shungu) all question whether blood vessel problems are present.
Similar questions are being raised in fibromyalgia as well. A 2019 study, in fact, proposed that a marker of endothelial cell dysfunction might prove to be diagnostic for FM. Other studies have found reduced oxygen uptake during exercise, and stiffened arteries (in both ME/CFS and FM).
Given all that it was interesting to see a recent COVID-19 study, “Effect of SARS-CoV-2 proteins on vascular permeability“, use an approach that took the blood vessel issue in COVID-19, long COVID, and perhaps even ME/CFS, to a whole new level.
Leaky Blood Vessels, Leaky Gut, Leaky Brains?
2020 saw the emergence of SARS-CoV-2 coronavirus, which just happens to attack the same endothelial cells lining the blood vessels that some researchers believe have gotten whacked in ME/CFS. Blood vessel problems are now believed to play such a big role in COVID and long COVID that some researchers believe it is as much a vascular disease as a respiratory disease.
In fact, as time goes on and the damage to the lungs clears up – but the fatigue, cognitive and other problems remain – more COVID-19 researchers are turning to the blood vessels to attempt to explain the mysterious long-term issues the long-COVID patients are dealing with.
Bruce Patterson’s long-COVID treatment protocol is a case in point. Patterson uncovered evidence of unusual clotting in long COVID a year ago. His treatment protocol banks on the idea that endothelial cell dysfunction is the be all and end all in long COVID. He believes a similar process may be occurring in ME/CFS/FM and post-Lyme disease.
An Endotheliopathy?
Is long COVID a disease of the blood vessels, and if it is, is ME/CFS/FM as well?
Endothelial cells keep the blood vessels from leaking, dilate the blood vessels when more blood flow is needed, and produce inflammatory factors that cause clotting and other issues.
In October, Irish researchers proposed that a persistent “endotheliopathy” – a disease of the endothelial cells – is causing breathlessness, fatigue, and exercise intolerance in long COVID.
COVID-19 researchers believe they’ve already identified three sequential stages in which endothelial cell damage occurs in that disease.
First, the direct hit from the virus on the endothelial cells (it enters them through the ACE-2 receptor) causes an immune response that interferes with their functioning. The subsequent death of those cells causes the angiotensin I-converting enzyme 2 (hACE2) to activate the kallikrein–bradykinin, and renin–angiotensin pathways, which then results in increased vascular permeability; i.e; leaky blood vessels.
That’s a really intriguing model as a similar model for ME/CFS was proposed by Wirth and Scheibenbogen hypothesis pre-COVID.
Finally, an immune system overreaction damages the junctions between the endothelial cells, allowing the blood vessels to leak and a hypercoagulable state to form.
While the authors don’t mention it, note that leaky gut linings are also an issue in long COVID, ME/CFS, and fibromyalgia, and that the same ACE-2 receptor implicated in COVID-19, ME/CFS, and POTS is also found in the gut. This could conceivably be causing or contributing to the IBS-like symptoms often found in COVID-19, long COVID as well as ME/CFS, FM, and POTS.
Could problems with this receptor be causing problems in the blood vessels, the gut, and the brain in all these diseases? Except for a few small studies and the recent attention given it by Wirth/Scheibenbogen, this receptor has received very little study or interest in ME/CFS and POTS.
Endothelial Cell Breakdown Explained?
The endothelial cells, though, are receiving a lot of interest in COVID-19, – so much so that in “Effect of SARS-CoV-2 proteins on vascular permeability“, an Israeli research group turned to umbilical cords to study them. Using umbilical cords as a blood vessel model, the Israelis introduced the SARS-CoV-2 (the coronavirus) proteins into the model to see how they affected it.
A 2005 ME/CFS cerebrospinal proteome study suggested that misfolded proteins (amyloids) could be puncturing blood vessels in the brain. A COVID-19 study also found evidence of amyloid proteins.
Remarkably, almost 70% of the coronavirus proteins negatively impacted the endothelial cells lining the blood vessels. The Israeli researchers proposed that a system-wide problem of leaky blood vessels might be causing the multiplicity of symptoms and conditions associated with the virus. In their latest hypothesis paper, “An attempt to explain the neurological symptoms of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome“, Wirth and Scheibenbogen reported that no less than 5 studies have found damage to the endothelial cells in long COVID.
Another study went so far as to use something called “evolutionary rate correlation” to uncover proteins that co-evolved with the ACE-2 receptor. The goal was to uncover proteins that were interacting with that receptor. The study distinguished several coagulation pathway proteins, the immune response proteins, and proteins associated with the androgen receptor.
If you’ve never heard of “evolutionary rate correlation”, or using umbilical cords to study the effects of proteins on the blood vessels, don’t feel bad. No ME/CFS/FM studies have attempted to do anything like that – and aren’t likely to in the near future. In fact, we’re going to have to get used to new technologies popping up that we’ve never heard of or even conceived of before. It’s a coronavirus thing.
Micro Clots and Amyloidic Proteins, Oh My…
Speaking of clotting, The Irish study mentioned above, which went far beyond any assessment of clotting thus far done in ME/CFS/FM, found a variety of “pro-thrombotic changes” had occurred; i.e. it suggested that long-COVID patients are predisposed to produce blood clots.
The coronavirus’s propensity for producing micro clots could also help explain why COVID-19 is so much better at producing ME/CFS-like states such as long COVID than the flu is at producing them. Back in July 2020, a study that found 9 times the level of small blood clots in the lungs of people with COVID-19 compared to people with the flu may have stumbled upon a key feature in lingering, post-infectious disease states.
In August a South African study, “Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin“, found that people with both acute and long COVID were having difficulty breaking down the plasma proteins in their blood.
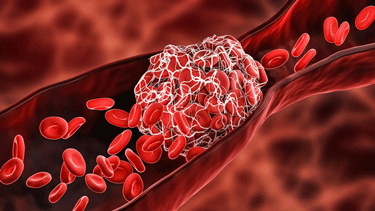
Studies suggest that micro blood clots may be more common in mild COVID-19 cases than severe ones.
This “pathological fibrinolytic system” appeared to result in “persistent anomalous (amyloid) micro clots”. The apparent inability of long-COVID patients to get rid of their misfolded folded proteins (i.e. micro clots) appeared to be gumming up their blood. The authors speculated that this might have been caused by a prolonged inflammatory state. Unfortunately, it didn’t seem to be going away over time.
It did, however, make Baraniuk’s 2005 cerebrospinal proteome study finding that much more interesting. Baraniuk reported that the ME/CFS “proteome was remarkable for the number of proteins associated with protein misfolding and cerebrovascular amyloidosis syndromes.” Evidence of amyloid deposition in the blood vessels, and a weakening of the blood vessels walls, was found. Baraniuk speculated that localized bleeding caused by amyloid deposition into the blood vessels might be occurring throughout ME/CFS patients’ brains.
The micro clot findings and the breakdown in the fibrinolytic system designed to get rid of them are intriguing given the hypercoagulation found in both ME/CFS and fibromyalgia about 20 years ago.
Back to the Future? A 1992 ME/CFS Study
The amyloid finding, though, brought us back to an even earlier finding in ME/CFS – the 1992 finding of widespread punctate hyperintensities in the brains of people with ME/CFS. These hyperintensities are thought to be the result of chronic microvascular disease.
Back then the researchers didn’t know what to do with that finding, but in 2020 Avindra Nath found evidence of similar thinned, leaking blood vessels (“punctate hyperintensities”) in the brains of COVID-19 patients.
Nath suggested that a series of small strokes had occurred across the brains of COVID-19 patients and even proposed that these lesions could be producing problems with heart rate, breathing, etc. Nath subsequently undertook a neurological study of long-COVID patients that could tell us much.
Deformed Red Blood Cells?
Then there’s red blood cell deformability – ME/CFS’s potentially unique contribution to the long-COVID field. If an inflammatory milieu around the red blood cells exists, it could have consequences for the red blood cells themselves. It doesn’t appear that red blood cell deformability is gathering much interest in long COVID.
A Russian study, though, found massive damage to the red blood cells, stacked, aggregated erythrocytes (red blood cells) capable of clogging the “microvascular bed” and diminishing the oxygen supply to the tissues in, get this, mild COVID-19 patients. It suggested that anti-thrombotic drugs be used. Dr. Holtorf has remarked that he’s found heparin to be useful in some people with ME/CFS.
The red blood cell deformability project funded by the Open Medicine Foundation at the ME/CFS Collaborative Research Center at Stanford University, though, began before long COVID reared its head, and it continues to evolve.
The RBC deformability saga in ME/CFS began with a finding by San Jose State University researchers, and moved over to Stanford. Recently, the University of California at Davis researchers have joined the show.

If deformed red blood cells are present in ME/CFS, they might be present in long COVID as well.
The project’s continued evolution suggests that deformed red blood cells could be impeding blood flows in that last, most crucial juncture – the microcapillaries that deliver blood to the muscles.
Unfortunately, the idea that this might be a problem seems to have escaped medical researchers before now and the technology to properly assess them was lacking. So, Ron Davis did what Ron Davis does – he, in collaboration with the Stanford and UC Davis teams, began to develop a microfluidic device that can do that.
Davis expects that this instrument will fit a couple of nice parameters: it’ll be inexpensive, easy to operate in different clinical settings, and produce results in a short time.
With the increased emphasis on clotting and oxidative stress, the red blood cells, and the microcirculation, this project comes at an excellent time – not just for ME/CFS but potentially for long COVID as well.
Different Roads Leading to Rome…

Many pathways may result in a fatigued, exercise intolerant state. Right now it looks like some of them may involve the blood vessels.
The saying went “all roads lead to Rome” simply proposes many different ways exist to get to your location; in this case to the fatiguing, exercise-challenged states of ME/CFS/FM, long COVID, etc. Leaky blood vessels found before or after the blood vessels, micro clots, amyloidal deposits, massive vasodilator release, and/or deformed red blood cells could all contribute.
These studies – coming from the U.S., Ireland, South Africa, and other countries – display a remarkable array of new technologies that have been pressed into service to understand COVID-19 and long COVID. More than anything, it’s new technologies that will likely get at the genesis of complex, multi-systemic diseases like ME/CFS.
If a multisystem origin is what you want, the blood vessels are a great place to start. Before the long-COVID saga is done, it’s clear that much will be learned about the endothelial cells, the blood vessels, the red blood cells, and the microcirculation – all potentially vital issues in ME/CFS. If long COVID turns out to be an endothelial disease, these studies will surely provide roadmaps that will be used in ME/CFS/FM.
Long COVID, then, is already doing what people with ME/CFS/FM wanted it to do. It’s bringing new technologies and new researchers to the study of post-infectious disease states, as well as an increased focus on factors that we already believe may play a significant role in these diseases, and is already providing potential insights into them.
BIG (little) Drive Update
Thanks to the 225 + people who have contributed to Health Rising’s year-end fundraising drive.

If that kind of long-term memory and knowledge base floats your boat – please support us!



 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



I have deformed RBC ( actually at one time they thought displasti) and ME/CFS!
Would blood thinners – even aspirin help the delivery system?
Well, Dr. Holtorf does find heparin useful at times. I assume he does clotting tests, though, to determine which patients it works in. Other things could be happening that don’t involve clotting and aspirin can have negative effects as well.
Like so much in ME/CFS there’s no clear answer yet – hopefully, we are getting closer. Time will tell.
Hi Cort and all, especially cort about this article.
Having read Wirth & Scheinenbogen among others I would like to affirm some of your comments but challenge others.
The endothelials at “peace” are anti-clotting and anti-inflammatory. ACE2 gets blocked by covid which affects angiotensin 1-7 which normally helps mitichondria to neutralize Reactive Oxygen Species.
It is natural to start a fever or inflammation when Covid starts bursting cells and that pushes the mitichondria into anaerobic metabolism to make it tough on covid . But its attacking edothelial cells and they govern inflammation and clotting. Tons of them are killed, the alert is raised to be anaerobic and emit cytokines. In anaerobic even, Ang 1-7 should limit ROS production in the mitochondria, but no ACE2 means no Ang 1-7 and the storm begins. So many endithelials are killed that atherosclerosis begins –body-wide. You mentioned increased permeability and that it provokes clotting. So im not surprised in other reader comments to have read that their platelet counts were high.
While SARS 1 did prefer ACE-2 receptor, as covid does, all previous flus and oandemics preferred sialic acids (receptirs) a-2,3 and later a-2,6 and in the middle of the history… both.
But not ACE2.
Please view this Pulmonologist video. He is saving covid patients in icu as a clinician and his major was chemistry. He explains what I just said and how covid must be an Endothelial dysfunction disease.
https://youtu.be/Aj2vB_VITXQ
I stiil dont understand why the blood is shunted away from exercising muscles, but I believe the same is true for cognitive brain cells, motor signal brain, limbic (emotions etc) system. I believe ischemia is occurring in me/cfs. I believe that forces anaerobic.
And its hard to express, but resting in a chair is no-stress. Walking is positive stress. But a body with chronuc high cortisol will take all stress as bad and will inflame, and I believe starve these typically exhausted cfs/me systems all at once. So exercise, strong emotion, difficult mental processing will each bring down the others when then inflammation/capillary ischemiA/hypoxia hits. And the PEM is for a different but related reason in my opinion.
W&S said the low (1/13th) production if ATP in anaerobic requires more sodium to be pumped into the muscle cells. Its more important to pump out potassium though. ATPs are “squandered” in removing the K , further reducing energy toward exhaustion, until the amount if sodium is SO high (bad) that now the NCX transporter also overpumps in lots if calcium.
They said the mitochondria are so used in cfs/me to anaerobic that they are hyoersensitive to calcium. So this damages the mitochondrial cycle. That would be your PEM while myscles Slllloooowwwwly rid the sodium and calcium and with the mito handicapped as well by all that calcium
What I neglected to mention is that the feverish cytokine storm, which I expect killed people in all pandemics, isnt sustainable.
If the body needs to continue inflammation it must switch to hypothermia as in sepsis. This continues some inflammatory chemicals, but no more pyrokines for example.
So the process in covid attack will not be the same as the process in an abnormally low temperature state (chronic).
However, once covid is over, and long covid begins, yes it would become the same process.
Cfs/me is a central nervous system inflammation by definition. Myelin? Not sure . Myelitis does not mean myelin. But endothelial cells are everywhere and they remain the mediators of inflammation
I take two 300 mg aspirins after my main meal each day and find a definite reduction i
n my paresthesia (numbness and tingling in arms, hands and feet). I also take quercetin, Vit C, Vit D3 & k2 an antihistamine and zinc and find the combination greatly helps.
What doses are you taking those vitamins and other pills, could you please write?
I remember that the retired Canadian specialist, Dr. Byron Hyde, suggested towards the end of his book, Missed Diagnoses: Myalgic Encephalomyelitis and Chronic Fatigue Syndrome, that he thought this would turn out to be a vascular disease.
He did all those early PET scans, I believe they were, which showed reduced blood flows through the brain.
That’s what I have thought; that ME/CFS may fall under the vasculitides.
Has anyone checked to see if the microclots exist in ME/CFS patients?
One or two studies from David Berg, I think it was, in the early 2000’s found evidence of hypercoagulation – which I assume means something like clotting – in ME/CFS. It was a big thing for awhile – then a small British study found no evidence of it. I think Berg may have retired sometime around that time as well but it was never taken up after that.
Rouleaux are stacks or aggregations of red blood cells that form because of the unique discoid shape of the cells in vertebrates. They occur when the plasma protein concentration is high, and, because of them, the ESR (erythrocyte sedimentation rate) is also increased. What do the new RNA vaccinations do? They inclease plasma ptrotein concentraion with all the spike proteins they cause to be generated!
Hi Jay, I believe Professor Resia Pretorius, who was one of the authors of the South African study Cort mentions above, is going to be looking at whether there are micro clots in ME/CFS. Prof Pretorius went over to Mulheim, Germany, recently to study Long Covid patients undergoing the Help Apheresis treatment with Dr Beate Jaeger. I was following the developments, mainly through the Tweets from Dr Asad Kahn, but Prof Pretorius Tweeted too. They are very well aware of the neglect of ME/CFS patients.
Amy Proal PhD interviewed Prof Resia Pretorius and the interview is available on YouTube here: https://youtu.be/C8tzTmVwEpM
I am currently investigating microclots in ME/CFS! We will publish within the next month or two.
Yah! Very much looking forward to that 🙂
Great article, again 🙂 Did any of this work point to what the post-exertional malaise reaction is, why it takes about 24 hours to really get started, and why it takes so fricking long to stop? Took two weeks to get better after my annual Thanksgiving relapse started. It feels like a signaling thing, telling my body to get sick, then waiting a long time to stop the signal.
I discuss one hypothesis for how leakages from blood vessels contributes to post-exertional malaise in my recent Health Rising post: https://www.healthrising.org/blog/2021/12/05/lipedema-lymphatic-fibrosis-ehlers-danlos-syndrome-fibromyalgia-chronic-fatigue-syndrome/. Essentially, the hypothesis is that fluid leaking from the blood vessels floods affected tissue, causing hypoxic conditions that give rise to inflammation. Fluid leakage could increase with exercise that raises blood pressure. I am not sure why there is a delay. The delay may be related to the slow accumulation of increased fluid or the time it takes the tissues to feel the feel effects of the hypoxic conditions? The accumulated fluid then leads to the formation of adipose tissue which becomes fibrotic, leading to lipedema that is painful to the touch and lipomas
@Jeffrey
I think any explanation of PEM needs to take into account that PEM can equally happen from physical exercise, cognitve exercise, mental stress, sleep deprivation and sensory overload. I therefore doubt that peripheral blood pressure elevations are relevant. Just something to think about.
@Herbert. You raise a fair point. I believe that the inflammatory mediators from mast cell reactions also interfere with endothelial function or cause endothelial damage, leading to increased fluid leakage. Increased fluid volume from elevated estrogen, cortisol and meds/supplements (salt, fludrocortisone, etc.) can also increase fluid leakages. There may be other things that cause increased leakage, but the core of my hypothesis is that the poisoned feeling that some experience during post-exertional malaise is the flooding of tissue with lymphatic / interstitial fluid. In my daughter, at least, it can be ameliorated to some extent by manual lymphatic drainage, though until you address the endothelial damage / dysfunction, the problem will continue to recur.
It’s possible the sympathetic nervous system comes on line in order to get the “thing” done and stay online for a bit before shutting off. But while it’s online, acts as a temporary stop gap / painkiller measure in order to do things it thinks maybe necessary for survival. Obviously via affecting the blood vessels in some way to temporarily get the thing done. Depending on how much sympathetic activation is necessary, longer you’ll wait for PEM
Even when my wife and I ran marathons we had what we called the two day lag rule. It took two days for the real fatigue and lactic acid pain after a race.
That’s true – with strenuous exercise that exceeds our boundaries, even when we were healthy there was often a day lag before the aches set in. I’ve often kind of wondered how that relates if it does.
I have the same experience from drinking alcohol, doing Yoga and acupuncture. There is always a days lag before the tiredness sets in.
With 44 years of ME/CSF I now attend an hyperbaric oxygen chamber when I can. It keeps me alive , helps brain function and neuropathy but hasn’t done much for fatigue.
Long covid patients are turning up, and most are getting better. Proteolytic enzymes are generally used and help. Nattokinase and serrapeptase are available at eBay. ,I’ve used them for a few years now as I’ve got high fibrin and family history of heart attacks and strokes from an early age.
I so hope they can help people with M-E we have suffered for years and years and still being gaslighted now by gps we are also suffering with the vaccine because some of us have over active immune systems not for days months some of us are unprotected and house bound after taking it we really needed looking into now we’ve suffered long enough and now suffering more with the vaccine s
I am one of those impacted by vaccine. I had issues before vax, but it has taken me to new lows with pain and neuropathy and no signs of improvement, months later. My Dr will not even acknowledge that vax could be the problem
Does BC007 fit into this picture? How excited should we be about this as a potential treatment?
Awesome job again Mr. Johnson. Thank you so much for providing this information
It sounds like they’re making some great discoveries and getting to the heart of what’s going on. I’m curious how mast cells and harmful antibodies come into play. Maybe all of the inflammation from the leakage sets off an immune reaction? Or is the immune reaction causing the leaking? What do you think?
It will be important to get to the heart of what’s causing this. It’s great that they’re making new discoveries and treatments related to leakage, but I wonder if that’s a downstream effect from something else. In this disease we are so often prescribed things that only treat symptoms (obviously, since we don’t yet know what the cause is). But I just hope that we can get to what’s causing this and find a way to treat or even cure it. Well, I’m sure we all are.
I don’t know much about mast cells but it would seem to make sense that they would be in the midst of this leakage. I wonder if endothelial cell damage could be triggering mast cell production?
I would hypothesize that the inflammatory mediators released by mast cell reactions could cause endothelial damage / interference in blood and lymphatic vessels, leading to increased leakage from microangiographic blood vessels, decreased lymphatic system effeciency, and increased build-up of lymphatic / interstitial fluid and corresponding post-exertional malaise and formation of adipose tissue. As the adipose tissue becomes fibrotic, it causes increased inflammation that in turn releases inflammatory mediators that perpetuate the cycle.
This would explain why I gained immense weight when I got long covid some are either gaining extreme weight or losing extreme weight. Luckily this all looks treatable as when I took the vaccine for a few days I felt back to normal.
You just blew my head clean off. I was diagnosed with Dercum’s Disease in 2013. It causes my body to grow painful lipomas (benign fatty tumors) and painful fatty tissue. The painful fatty tissue becomes fibrotic over time. I have since been diagnosed with ME/CFS, hEDS, MCAS, POTS, and Fibromyalgia. I am very Ill now—housebound and 90% bedbound. I had to stop working in 2916. I’m pretty sure that you just summarized the cycle that is happening inside my body that is making me so sick.
Way, way, way before Stanford a researcher here in little New Zealand found evidence of deformed red blood cells in ME/CFS in the late 1980s/early 1990s.
Thanks Matthias! I always hate to not acknowledge somebody. I do remember this. The name is on the tip of my tongue. It will come to me. Thanks for pointing this out 😉
Les Simpson.
Super interesting summary, Cort! I think you pulled it very well together, and this is very important now.
I think everyone is now quite convinced that endotheliopathy/endothelitis is ONE part of the picture of LongCovid/ME/CFS. As you have mentioned, it can go along with coagulation problems/microclot in some patients, possibly also depending on stage of disease. You have also mentioned that endothelial dysfunction may explain why the microcirculation doesn´t work – with all the cellular metabolic problems that can arise (mitochondrial dysfunction, poor oxygen utilization, red cell deformation etc…) In some patients there may also be fluid loss to the interstitium, which can add to the perfusion problems (possibly also to cardiovascular problems in some). Of course, the resulting blood flow problems may cause (or add to) problems in brain perfusion.
But I think this is not the whole picture, and I am thankful that you pointed this out. Indeed, there is another big entity besides endothelial dysfunction that we should include in any “unifying” hypothesis, and this is CNS inflammation (neuroinflammation). You have mentioned the vascular leaks that have shown up on MRI in ME/CFS and Long Covid which of course could affect CNS function, and we´ll probably hear more of these. Yet we need to understand that any endothelial dysfunction at the blood-brain-barrier (be it from leaks, lack of oxygen, microclots or oxidative stress) will have profound effects on the defensive immune system of the brain (microglia), a very delicate and powerful system which easily becomes reactive, i.e will spring into action and cause neuroinflammation and dysfunction on many levels of the brain (and, unfortunately the whole body). Also, if the blood-brain barrier does not work properly, you will have immigration of immune cells into the CNS, which usually should not happen and then adds to inflammation in certain areas of the brain. As I understand, Jarred Younger is currently examining this for ME/CFS and has first results. A British team has recently identified a marker in LongCovid patients that may indicate T-lymphocyte migration across the blood brain barrier in Long Covid patients (CD70). Mike vanElzakker has just shared first results of neuroinflammation in the cingulate cortex.
So I think there are now good reasons to start to think endothelial dysfunction, problems in microcirculation and neuroinflammation TOGETHER. Here I am in contrast to the Wirth/Scheibenbogen hypothesis which claims that neuroinflammation does not exist in ME/CFS and that the CNS dysfunction seen in ME/CFS can be sufficiently explained from cerebral blood flow problems alone. Personally I think that this underestimates the CNS defense by a wide margin – but as these are all testable hypotheses, they will be answered sooner or later (I think: sooner, as indeed study results are coming in at an unprecedented rate).
In the meantime I would suggest to look for the bigger picture whereever possible. Yes, some Long Covid patients have microclots in their blood – but this is only ONE part of the LC-picture – more relevant in some patients than in others and possibly more relevant in some stages of the disease development. So is neuroinflammation, it´s ONE part of the picture. The same may apply to GPCR-dysfunction (although this may well be a central leg and possibly one very closely related to the inceptional process, at least it appears that the sensitive bioassay that Berlin Cures uses (the BC-007 company) identifies GPCR in virtually all LongCovid (and apparently also all ME/CFS) patients.
Cort, thanks so much for what you are doing, your presentations of the cutting edge of research are tremendously valuable!
Good points. I feel the unifying factor is tissue. It’s all eds related. It’s how they interact with the environment and viruses.
I don’t think tissue is studied enough in this disease
Got my Antibodies meassured from berlin cures. Removed them, nothing happened. The end. Berlin cures tries to sell their product / BC007 for 20 years now. And changed the diseases about every 5 years. Just now their heart failure study failed for antibodies and BC007. Its a scam. They want the product on the market at all cost. Nothing to see there then another money making company on the backs of desperate patients.
I heard from someone who should know that the product BC 007 will cause no harm but it will not help either
Hello Badpack,
in Germany, Berlin Cures are partnering with a respectable university clinic (Erlangen) and a lBC007 LongCovid research project has been funded by the German government. It will start in the beginning of 2022, and Bettina Hohberger, the Dr from Erlangen university clinic, will be tweeting about it.
My feeling is that autoimmunity is an important component in many ME/CFS and Long Covid cases, but is probably not the whole story about ME/CFS. So it would not suprise me if BC007 helps some but does not help others.
Therefore I think your “negative data” is very valuable too for understanding ME/CFS. It would be great if you coud share 2 more details to copmlete the picture:
– Which antibodies were elevated in your case?
– Which treatment did you remove them with?
Here is a German patient summary of iknowledge about BC007 from Sept 2021: https://www.me-cfs.net/aktuelles/einschaetzung-des-wertes-von-bc-007-fuer-me-cfs-erkrankte-aus-derzeitigem-wissensstand-heraus, which also mentions baseline values for antibodies to distinguish healthy from sick.
One topic I meant to research but am currently too sick to do myself is that someone mentioned on twitter a presentation from the 2021 dysautonomia conference that discussed the CellTrend antibodies test and the topic of reliably distinguishing healthy from sick. Unfortunately the tweet did not link to the presentation.
Does anybody remember this presentation?
Cort, would you know more about the differences between CellTrend and Berlin Cures antibody testing, and the abovementioned presentation? Thank you!
Thanks Herbert – I hadn’t thought about the BBB and very much looking forward (with some trepidation, I must admit) to Younger’s results. Good for MERUK for funding that fascinating study.
I’m really wondering about this ACE-2 receptor! Could it be causing or contributing to leaks in the blood vessels, the gut, and the blood-brain barrier in both long COVID and ME/CFS? Check this out:
SARS-COV2 Alters Blood Brain Barrier Integrity Contributing to Neuro-Inflammation
Very helpful comment, thank you! I would add one possible connection between these two areas. Increased leakage from blood vessels due to endothelial damage / dysfunction could cause a build-up of lymphatic fluid and the formation and fibrosis of adipose tissue, as is theorized to happen in lipedema. In my daughter, I have seen that fibrotic adipose tissue blocks the free movement of lymph, and I would propose that the same process may be at work in blocking fluid from exiting the brain in the glymphatic system, contributing to increased intracranial hypertension, which could lead to neuroinflammation. Some new research has found a connection between intracranial hypertension and glymphatic system dysfunction. Links to relevant research included here: https://www.healthrising.org/blog/2021/12/05/lipedema-lymphatic-fibrosis-ehlers-danlos-syndrome-fibromyalgia-chronic-fatigue-syndrome/
I had my Pfizer booster and it has left me with the worst cfs I have ever had. I sometimes get cfs after a viral infection . The first AZ jabs I had didn’t cause me any problems but the Pfizer has floored me.
Sorry to hear. Wish you the best! The pro vaccine fixers don’t want to hear this kind of news,. Negative experiences are ignored by the media and experts. In Europe there is an excess mortality not due to corona but something else. What that is is not yet known. There is a statistical relationship with taking vaccines. But not yet proven. Nobody wants to prove this, of course. The interests are too great for that. Also, 5 times as many athletes died in 2021 from heart problems. How come?
Did you read how many people died from blood clots from Johnson’s and Johnson’s vaccines? I think it was 9 out of 1 1/12 million. The highest risk group is middle-aged women in which 1/100,000 may have a clotting issue.
Those are very low numbers Gijs and yet they are enough for the CDC to recommend against taking the Johnson & Johnson. I would think the ability to discern that 1/100,000 women in a certain age range are at risk – and then act on it – might make one question whether a massive coverup is going on.
I know two healthy men in their mid 50’s who died from myocarditis from the evil Covid JAB. The Covid Jab is just a needle full of poison.
They died from the first Jab. They took the first Jab and developed heart problems the very next day and were dead within weeks. Their doctors said it was from the JAB.
Judy, the huge studies designed to test the efficacy and safety of the vaccine showed no signs of an increase in myocarditis. They did show signs of a very rare clotting issues in adolescents I believe. The fact that they were able to pick up that indicated how thorough the studies were.
You can choose to ignore the science or not but science itself was developed so that we would not go by anecdotes – the reason for that is that anecdotal reports don’t work. Science, on the other hand, has proved to work very well – which is why all serious endeavors use it.
I looked up both of your claims (mysterious excess mortality in Europe statistically related to vaccines; 5x as many athletes died from heart problems) and was not able to substantiate them with good sources. I was able to find some garbled phone chain type rumors that claimed people were dead who are still living. Do you have better sources that I was not able to find?
As bad as a ME/CFS flare up is, ME/CFS flare up and “death” are extremely different outcomes when doing a cost/benefit analysis. Usually the rationale to vaccinate patients at risk of flare up is that the virus would confer even greater risk of flare up, as well as the risk of mortality, in a patient unable to shelter and avoid the virus indefinitely.
Yes there’s been a massive politicisation of the vaccine. You can’t even question the efficacy and safety of the vaccines without being part of the anti Vax brigade. It’s fear of course but it’s also a dangerous mindset.
@Girl
According to Wikipedia, under “List of association footballers who died while playing”, in 2001-2020 there was an average of 4.2 deaths per year attributed to SCD or SUD, the vast majority being SCD. In contrast, in 2021, according to Real-Time News‘ list, there were 21 cases of SCD/SUD among FIFA players. In other words, instead of 4 SCD/SUD deaths per year (according to Wikipedia data), or 5 cases per year (calculated according to the BMJ) during 2001-2020, 21 players have died so far this year.
https://americasfrontlinedoctors.org/2/frontlinenews/500-increase-in-sudden-cardiac-and-unexplained-deaths-among-fifa-athletes-in-2021/
There is a significant excess mortality in European countries such as England, the Netherlands, etc… In the Netherlands 25% a week. Not because of corona. How is this possible? Nobody knows yet. There is a trend with vaccinations, but as yet unproven. Maybe it’s because of the virus itself, which seems to disappear like HIV but then strikes at a later stage. We do not know.
As for ME and vaccination: In survey, 19% of people with ME/CFS said health worsened after vaccination compared to 4% reported by controls.
Where are your data on ME patients who have died or are seriously ill after corona? Compared to a control group?
Only then can we see whether vaccination is so much better than not vaccinating. Now they are unscientific statements based on fear. Many ME patients have endured corona fine. With which I do not want to say that corona can be harmless. It is a decision that everyone has to make for themselves
Good luck Paul. I hope it resolves soon. It’s amazing how differently the vaccines are affecting different people. The Astra Zeneca vaccine actually produced the most symptoms in our polls.
Hi Cort, Of course the CDC don’t recommend J and J. America makes more money from Pfizer. Pfizer promotes almost all media programs. Don’t close your eyes to that. About 15 out of every 100,000 young men develop a heart infection after vaccination. J and J does not work against omicron also 2 vaccinations with Pfizer do not help against omicron. Also astra zeneca does not help against omicron. In England, the number of infections with omicron is rising. But Omicron is a milder variant, according to the South African experts. Hardly anyone goes to the hospital. Many people I know have had corona. Both vaccinated and unvaccinated. They all got just as sick. No one came to the hospital. The chance that you will die from corona when you are 50 is 0.05 percent. So your chance of surviving is 99.5%. Above the age of 60, 70 and 80, the number of deaths increases significantly. This also applies to people with diabetes and overweight. And other underlying diseases.
One of the first symptoms, beside fatigue, that I went to a doctor for was swelling in my hands and feet. It finally makes sense.
hi cort,
i could not read the text or the comments, only the summery. a bit.
if (my brain is verry bad) but they found also things in the brain of ME sufferers, allready forgot what you wrote, sorry, verry il.
it scared me…is there something to do about it or is it brain permanent “gone”
thaks so much!!!
It wasnt actually in the brains – it was in the cerebrospinal fluid – and yes, the suggestion that amyloidal (misfolded) proteins were found in there was rather shocking given the Alzheimer’s connection. We know, though, that people can have lots of amyloid tangles in their brains and not have Alzheimer’s. Something else is going on as well.
Systrom’s exercise dyspnea sounds more like exercise inability than exercise intolerance. Intolerance to me means PEM the next day, not breathing hard after exercise.
I always come back to PEM, perhaps because that has been the biggest problem throughout my CFS career. And the problem I see with cardiovascular or metabolic hypotheses is that they don’t adequately explain the time delayed PEM.
The dyspnea term threw me off too. If I remember correctly, the term was used because it’s traditionally used in that setting to refer to exercise intolerance – not because that’s the kind of exercise intolerance present. Systrom’s cohorts have contained a range of ME/CFS, FM, POTS, etc. patients and he hasn’t broken out people with ME/CFS until recently – https://pubmed.ncbi.nlm.nih.gov/33577778/ . I’ve never worried about that melange of patients as his patients fit the mold so well.
Systrom’s been deeply interested in ME/CFS for quite some time. He first presented an abstract at a San Francisco IACFS/ME conference seven years ago. He’s been embedded in ME/CFS research for years now and is working with ME/CFS doctors and other researchers and has been funded by Solve ME and I imagine others. (He has also found breathing problems during exercise in ME/CFS.)
Cort, I have read every post and comment going on 7-8 years now. I am certain that you were an angel sent by God. Over these past many years, there are certain researchers (and clinical MD’s) who seem particularly dedicated toward solving the ME/CFS nightmare. I “zero in” on any comments from: Klimas, Ron or Mark Davis, Fluge, Bateman, Patterson, Lapp, Dantini, Pridgen, Lipken, Younger, and the Lights. I chose to finally post today, because you mentioned both UC Davis and glycocalyx breakdown in blood vessels. While working on my Master’s in Molecular Biology at Davis, I contracted ME/CFS. I was accepted into an MD/Ph.D. Program on the east coast, and had intended to spend my life researching ME/CFS (and other neuro-immune diseases.)
Unfortunately, like so many others, the Fibro fog, daily headaches, and fatigue knocked me out of the program 25 years ago. However, I did learn enough during that time to ask some intelligent questions. The leaky blood vessel and gut idea does make sense, in that this is exactly what happens with Dengue Fever. The Glycocalyx of the blood vessels is broken down by an immune system reaction to the virus, and plasma leaks into the tissue. But, those patients show an increasing hematocrit as the disease progresses. I am confident that I do NOT have excess plasma form my blood circulating in my lymphatic tissue. I also do not believe that the chronic fatigue patients would have such a profound and extended reaction to exercise, if a lack of health erythrocytes at the cellular level was the sole cause of the disease. They would recover sooner. Dr. Lipken’s data on the gut microbiology is most likely a RESULT of the disease, as opposed to the cause of the disease. Pridgen. Dantini, and Lerner (RIP) are correct in that this is a virally mediated disease. Patterson may be running down the right path!
Anyhow, I feel as if I know many of you personally. Especially you Cort! I am confident there are many more who follow the thread, but do not post. Who are the UC Davis researchers that you mentioned? And, Thank You !!!
Thanks Richard – good to hear from you :). What a shame that the ME/CFS research field lost you back then… Interesting that another virus interfers with the blood vessels. With ME/CFS there is this possible problem with the ACE-2 receptor – the same receptor, ironically, the coronavirus enters the cells in. Only a little study has been done in ME/CFS and POTS, though. I very much wish someone would really dig into that receptor in these diseases so we could know if this bizarre and uncanny coincidence really is real.
As you note there are so many possibilities with these diseases and yes, it would seem that the viral attack is key for many people. (Maureen Hanson wonders if everyone’s ME/CFS, even those who did have an obvious infection,is post-infectious. What a fascinating idea that is.
I think these may be the UC Davis researchers – “This effort is led by Jiandi Wan and Sitong Zhou, in collaboration with myself and my colleague, Mohsen Nemat-Gorgan of the ME/CFS Collaborative Research Center at Stanford University.” from Ron Davis
This resonates – a lot. No matter if the reason for blood flow problems to the brain are caused by structural problems (Eagles, instability, vascular malformations), endothelial problems (inflammation or faulty connective tissue formation) or a clotting disorder (like antiphospolipid syndrome) the result would look similar and be devastating. It feels like brain damage and it looks like brain damage. Ouch.
Yep, stem cells and crispr will eventually be the way out. In the meantime, anything for the relief would be good
Cort, Has anyone reported on how the Ampligen treatments in Argentina are progressing with ME/CFS patients?
In one of your posts, Cort, you asked whether MCAS was the process that tied together ME/CFS/FM and Long Covid. I am convinced that it is. Virally mediated changes in mast cells could be the physical mechanism that causes all of these reactions. Remember, sometimes mast cells will react as a local group and not systemically.
It may turn out that ME/CFS/FM and Long Covid and some others are symptoms of MCAS, as well as conditions in and of themselves. When I cascaded from fibromyalgia into MCAS this year, I treated the MCAS and found that my fibro symtoms went away.
Treating for MCAS got rid of my fibro stiffness and muscle pain. It got rid of that sour poisoned feeling that you can almost taste. Exercise intolerance is almost gone. Depression and anxiety almost gone. IBS symptoms resolving. Whole body inflammation receding. Brain fog is taking longer, but I can see a difference. On the heredity front, I can now identify my mother’s symptoms from 60 years ago when no one talked about these things. Now, I can see my daughter’s symptoms as well, all fitting with MCAS when her official diagnosis is fibromyalgia. She is undergoing chemo for breast cancer right now and I told her that her symptoms would probably multiply. I hope I’m wrong, but I’m scared I’m right.
How did you treat mcas issues? I have tried different supplements, nothing seems to help. I am guessing I need more intense treatment, but can’t get much help from Dr
Here’s what you said in your blog, Cort:
“Could activated mast cells be tweaking the nerves in FM? Something is doing that, after all, and that something has not been identified. Nor does that something, similar to IBS, appear to be producing much of a systemic reaction or causing any tissue swelling or damage. Instead, in its hidden way it’s going straight for the nerves. It’ll be interesting to see if this IBS study sparks studies that start looking for mast cell activation in FM.”
Mast cells tweaking the nerves. Mast cells tweaking the microcirculation. Mast cells causing leaky gut. Mast cells causing neuroinflammation. Since mast cells are everywhere, and can respond as a local group, this may account for the varied symptoms of MCAS:
IBS
Fibromyalgia
ME/CFS
Ehlers-Danlos syndrome
and probably others
The mast cell activation may stay local, or may escalate and add other localities to the whole syndrome, depending on triggers like infection, or wildfire smoke, or trauma, physical or mental.
Yes, Les Simpson, possibly decades ahead of his time:
https://me-pedia.org/wiki/Leslie_Simpson
Thanks
Me too. I was so wary of getting the jab but was convinced by my (ex) GP and the desire to see my grandma before she passed.
Here I am 14 months later, still dealing with persistent symptoms brought on by the vax: incessant muscle twitching, muscle weakness, blurry vision, cognitive issues, tics, and what I assume is some kind of autoimmune condition attacking me at night, etc etc.
As someone with long-term (thankfully mild) CFS, I already felt let down by the medical system. Now I feel completely betrayed by a society I had a hand in building.
Anne, j’aimerais connaître ton traitement du MCAS s’il te plaît ?
Sinon, en France, les médecins spécialistes de l’EM/SFC donnent des antihistaminiques, genre CETIRIZINE, AERUS… A part cela, ils ne préconisent pas grand chose, à part des compléments alimentaires tels que le Chardon Marie pour protéger le foie, et un complexe regroupé dans une même gélule ( acide alpha lipoïque, huile de poisson, NAC,Q10, Vit C, resvératrol, glutathion, L-cystéine, vit B1, B2, B3,B5, zinc.
Ce qui m aide le plus, c’est le Pacing. Et REPOS +++
Je remercie Cort pour son travail. Ca demande beaucoup d’énergie. Je remercie aussi toutes les personnes qui laissent des commentaires, c’est très important pour les malades.
Et je remercie aussi Google traduction qui me permet de suivre le site.
Bonnes fêtes de fin d’année à vous tous
Merci and thanks for the questions. Here is what I take every day:
Vitamins:
A, 10,000 qd
B, Balanced B-100 bid
C, 2000mg tid
D, 3000 units qd
E, 400 units qd
Cal-Mag-Zinc 250mg, 250mg, 5mg qd
Copper 2mg qd
NAC N-acetyl-L-cysteine 600mg tid
Quercetin, liposomal 500mg qid
PEA Palmitoylethanolamide 400mg tid
DAO Diamine Oxydase 1,000,000 HDU before each meal
LDN Low Dose Naltrexone 6mg topically at bedtime
CBD 30mg qid
Microdose of 100mg psilosybin twice a week
Glutathione nasal spray 5-6 times per day
Resveratrol 250mg qd
CoEnzyme Q10 200mg qd
Oxaloacetate 100mg qd
R+Alpha Lipoic Acid 200mg qd
Green Tea Extract 300mg qd
Rutin 250mg qd
SAMe 400mg bid
Melatonin 6mg sustained release at bedtime
Green bananas when I can get them
Low histamine diet
(coming soon: Luteolin, Fisetin, nasalcrom)
Benadryl for anaphylaxis symptoms – lips swelling and asthmatic breathing
I have sent for herbal remedies for dysbiosis (Dysbiocide and FC-Cidal) and when they arrive I will do the full course for 4 weeks and then after that I will begin probiotic therapy with different anti-histamine strains and butyric acid producing strains at high doses. Thanks for asking.
Oh, and LED red and near-infrared light therapy 8 minutes twice a day, at 630, 660, 810, 830 and 850 nanometers making 153 megawatts per square meter at a distance of 6 inches.
https://platinumtherapylights.com/products/biomax-rlt?variant=15601444487234#biomax-product-biomax-specification-section-1
C’est moi qui te remercie Anne, oui, j’avais oublié de parler des probiotiques, très important d’après mon immunologue spécialiste d’EM/SFC. J’ai fait tester mes selles, j’attend le résultat pour savoir quels probiotiques prendre, car chacun a son propre microbiote…
J’ai aussi une lampe infra rouge 1/2 h , 2fois par semaine, soit sur les douleurs soit sur le ventre ou sur les reins.
J’ai également de la cortisone en pulvérisation nasale pour un nez bouché en permanence.
On vient de me découvrir un léger emphysème pulmonaire, mais d’après mon pneumologue il n’y a rien d’inquiétant (pour l’instant).
Yes, I forgot to say that I have had my stool tested, as well, but I do not have the results back yet. Best of luck to us and Happy Holidays to you.
Could the 1992 study be named and linked to please?
I did try to look through COVID-19 fatalities in athletes (mostly believed to be down to myocardiatis or blood clots I believe) but could find surprisingly little information. Michael Ojo was one who died on the basketball court of heart problems after COVID. A few with long covid listed on https://me-pedia.org/wiki/List_of_famous_people_with_long_COVID
Research on the cause of COVID toe also shown to involve small blood vessels https://onlinelibrary.wiley.com/doi/10.1111/bjd.20707
Scheibenbogen and Wirth’s endothelial dysfunction and Unifying hypothesis is a revelation – but just seems to be suddenly appeared when I haven’t seen previous work from them before – it’s hard to grasp all of what they are saying and a name for their hypothesis would be helpful but it is encouraging to see the treatment trials that they have ongoing from this. They seem to be linking together many different pieces from other teams, which is encouraging.
A UK Long COVID consortium called STIMULATE-ICP has some drugs being tested including one addressing micro-clots which they say cause dead tissue (over time). Is that relevant to ME/CFS or endothelial dysfunction? Part of this suggested a drug for PEM which they (incorrectly) describe as possibly resulting from oxygen availability due to micro-clots.
The comment by the former marathon runner above I believe refers to Delayed Onset Muscle Soreness (DOMS) which can be really painful but isn’t like post-exertional malaise although I could not find clear comparisons in research. DOMS never caused me a sore throat, severe stabbing pain in a small joint, and certainly isn’t triggered by too much mental activity.
Also if the misinformation website link for the so-called “Frontline Doctors” could be removed from one of the the comments higher up that would be much appreciated since they have been banned from almost every social media platform and are considered a health risk. A quick search will establish their various anti-vaccine conflicts and lack of actual credibility.
thanks Cort
🙂 https://pubmed.ncbi.nlm.nih.gov/1309285/
Can you please add the name and link for the 1992 study Cort?
A comment from an ex-marathon runner above refers to PEM but is actually talking about Delayed Onset Muscle Soreness (DOMS). There’s not really any research comparing but DOMS never gave me a sore throat, can’t be caused from too much mental activity, and while it can cause severe muscle pain it does not to me feel similar to PEM (I don’t have fibromyalgia) but do have a nerve injury and DOMS is also nothing like that).
I checked into athletes after Covid-19 but could find very little info – mostly Michael Ojo’s death after collapsing while playing. A few athletes with long covid are listed on https://me-pedia.org/wiki/List_of_famous_people_with_long_COVID
Scheibenbogen and Wirth’s hypothesis could do with a name other than “Unifying hypothesis” – I am not aware of any of their previous research but the endothelial dysfunction work in particular seems to link many established pieces of knowledge together and makes a lot of sense. Also they are already starting a drug trial based on this.
Micro-clots from long covid – has any research looked for them in ME/CFS? I saw in the STIMULATE-ICP study website that they think this is linked to PEM (I’m not sure that they understand PEM though), they are saying that the clots cause small patches of dead tissue over time which reduces oxygen available to large muscles. I wondered your thoughts on this.
COVID toe – another recent thing – has two possible factors involved one is vascular – the other involves interferon I – while ME/CFS doesn’t involve covid toe might these factors be in the mix? https://onlinelibrary.wiley.com/doi/10.1111/bjd.20707
Also if you could remove the link to the misinformation website for the so-called “Frontline Doctors” from the comment much further up it would be much appreciated. That group have been banned from pretty much all social media for covid misinformation. Any quick search will reveal their anti-vaccine conflicts and lack of credibility.
I have long covid. It’s been a little over two months. I was admitted to hospital yesterday. Tested positive on PCR test still two months later.
Clearly, Ivermectin did not “cure me”. However, I became desperate because of ongoing debilitating shortness of breath. Contacted “Frontline Doctors” and went through that referral to get Ivermectin. The first day taking it, my breathing dramatically improved. I finished the course. I am no longer housebound. I am not fully resolved but amazingly, I can walk a same distance and only be winded where before I was gasping for breath heaving close to hyperventilating or passing out.
When my blood is drawn, 3 separate times and 3 different persons, all noted my blood does not come out right. Like “gummy blood” that just does not come out and fill the vial “like normal”. And I can remember pre-COVID blood draws where the blood came out thin and easily.
My blood has changed from COVID. Now, I do not have the extreme shortness of breath, but I still have problems with exertion in lightheadedness and things.
I only got the Ivermectin because I could not handle being so short of breath. I waited for 6 or 7 weeks. I respected the medical professionals who advised it, but I stayed with other medical professionals who did not. But, after 6 or 7 weeks of suffering – I figured it wasn’t going to kill me – so why not?
To be fair to the “Ivermectin crowd” – it was not early treatment. No one promised me it would “be the cure” of everything.
I had the problem with my blood being noted as thick and slow moving by persons drawing it that it was thick prior to taking the Ivermectin and after.
I do not see Ivermectin helping my blood in long COVID though it did help my breathing dramatically though it did not fully cure my breathing issues. I became stronger physically and able to breath better.
I am grateful not to be housebound as I was.
Yet, when I physically exert past a limit of standing and walking slowly combined for one hour – I am not getting enough oxygen bodily. It seems related to my blood so far to me.
It is only fair to be thankful to those who have aided in recovery – medical professionals who put their heart and soul into helping people like me in the face of being called terrible names by persons and being deplatformed.
I am sharing my personal testimony.
My personal testimony is not subject to the opinions of the CDC, FDA, or any other organization or its associates and representatives.
None of them are experts who know anything about or superior to what I know by my own life experience
I am thankful to those who helped me.
Thank you.
i have had fibromyalgia fatigue for over 30 yrs. exciting that there might be an explanation. do you know if there are any studies being done for micro clotting and cfs? i would love to be part of them if there are.
People on reddit are trialing themselves on nattokinase.
While I do believe there is vascular damage, I do not think that is the rate limiting factor across the board.
Many long covid sufferers are deriving great benefit from supplementation with Thiamine or Thiamine and Magnesium. Both are needed as co-factors for the dehydrogenase enzymes essential to aerobic energy metabolism.
The fact that such individuals, including myself, see an undeniable improvement in a very short time indicates, that in those cases, something else is going on.
Some have found such supplementation to be very close to a complete cure. Others (again like myself) have found it to be an important piece of an ongoing health puzzle.
Thanks Cort, for making the complex clearer! I’m wondering about the “leaky blood vessels” and particularly the idea that blood is getting “lost” before it gets back to the heart. Where would it be going? I’m curious because one of my kids with ME has chronic and recurring low iron and ferritin, which only resolves when he gets venofer infusions monthly. His hematologist says she has no idea where the blood and the iron are going – they seem to magically disappear. I wonder if he might be in that category of patients losing blood before it returns to the heart. What do you think?
Interesting! It sure potentially makes sense to me (a rank amateur). Have you seen this ME/CFS/POTS story – a young man who also really benefited from I think it was venofer infusions. I don’t think this is done very much (unfortunately). How did you find out about it?
https://www.healthrising.org/blog/2016/08/23/iron-man-young-persons-pots-mecfs-recovery-story-pt/