

Metformin – energy regulator, anti-inflammatory, blood vessel protector, antiviral, anti-cancer agent – has a lot going for it.
A couple of months ago, Health Rising reported on a metformin long-COVID study. In this blog, we take a deeper look at metformin and the study itself in light of recent results.
This 1,000-person-plus trial, “Outpatient treatment of Covid-19 and the development of Long Covid over 10 months: A 2 multi-center, quadruple-blind, parallel group randomized phase 3 trial“, assessed what happened when overweight but healthy people were given metformin (500 mg – 1 day, 2 500 mg for 4 days, 500 mg in the morning and 1,000 mg in the afternoon for 9 days) or placebo as they came down with COVID-19. Note how short the time period was – just two weeks. (The trial also assessed the effectiveness of ivermectin and fluvoxamine and did not find them helpful.)
They were followed monthly with a final assessment 9 months after receiving metformin. The patients were deemed to have long COVID if a physician had diagnosed them with it – suggesting that the incidence of long COVID may have been understated. While we don’t know what type of long COVID they had, studies that ask about the incidence of post-exertional malaise in long COVID find that a majority of long-COVID patients have it.
The study found that people receiving metformin early in the infection had a 42% reduction in the incidence of long COVID. Interestingly, during the acute stages of COVID-19, metformin was also found to prevent about 40% of emergency department visits, hospitalizations, and death. The drug was most effective when taken earlier in the infection (<4 days since symptom onset).
In his recent Ground Truths blog, Eric Topol was very high on the finding, calling it a “breakthrough” stating that given metformin’s cheapness and safety profile, if he came down with a coronavirus infection, he’d take the metformin at the doses recommended in the study.
The authors asserted that the fact that the drug was most effective when taken earlier in the infection (<4 days after symptoms) suggested that its antiviral properties may have played a major role in preventing long COVID.
They also suggested that metformin be tried in combination with drugs like Paxlovid. The possibility of using multiple drugs together to synergize their effects on viruses is welcome indeed given the mediocre drugs that have been used in ME/CFS to fight off the herpes viruses.
While Skip Pridgen has been trialing a two antiviral/anti-inflammatory drug combination in fibromyalgia, the antiviral avenue has hardly been touched in clinical trials of ME/CFS. Comparing this thousand-plus person trial to the one very small placebo-controlled antiviral trial that the ME/CFS field managed to get produced, despite years of interest in the Epstein-Barr virus, shows what a different world we’re in now.
Now for a deeper look at metformin and the ability of other drugs and supplements to impact a crucial energy regulator called AMPK.
Metformin, AMPK Activation/mTORC1 Inhibition
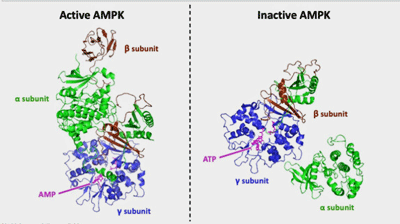
The goal – get AMPK into its active state. (Image by LBattist- Wikimedia Commons)
Metformin is an AMPK activator. Described as a cellular energy sensor and metabolic regulator, AMPK activation switches the body from being in an energy-consuming to an energy-producing state. AMPK is activated to respond to cellular conditions that reduce energy, and ensures that proper ATP levels are present in our cells. As ATP declines during say, exercise, AMPK triggers more ATP production. AMPK should, therefore, be activated in the muscles during exercise. Interestingly, given the recent findings of impaired fatty acid oxidation in ME/CFS, AMPK also promotes fatty acid oxidation.
In 2015, Julia Newton found greatly impaired production of AMPK and a cytokine (IL-6) involved in AMPK activation in ME/CFS, but when Newton whacked the muscle cells from ME/CFS patients with exercise, she found no evidence of AMPK activation. She proposed that the lack of AMPK activation impaired muscle contractions during exercise in ME/CFS, leading to exercise intolerance.
Newton followed up her 2015 study with a 2018 study that showed that metformin increased AMPK activation in cultured ME/CFS muscle cells. Unfortunately, that’s as far as we’ve gotten with metformin in ME/CFS.
AMPK activation and autophagy (see below) is inhibited by the coronavirus as well. In fact, many viruses, including herpesvirus and Coxsackie, modulate AMPK activity in order to impact autophagy, fat and lipid metabolism, glucose metabolism, etc., and facilitate viral replication.
The AMPK/mTORC1 Connection
By inhibiting mTORC1, AMPK also provides other benefits including reducing lactate production, increasing ATP levels, improving cognition, and inducing autophagy – a process the cell uses to clean defective mitochondria up – and which a recent Simmaron study suggests may be impaired in ME/CFS. If defective mitochondria are not cleaned up, the cells they are embedded in can become senescent, start deteriorating and begin secreting pro-inflammatory cytokines.
Autophagy is also induced by the muscle contractions that occur during exercise and is necessary for maintaining muscle activity. Mice deficient in stress-induced autophagy exhibit less endurance during exercise.
Metformin and Chronic Pain
mTORC1 also regulates the activity of sensory neurons in the periphery and central nervous system, including the A-fiber neurons that have been shown to be damaged in FM, ME/CFS, and long-COVID patients with small fiber neuropathy. Given the many, many symptoms – including sensitivity to light, sound, and touch as well as the odd, hard-to-describe symptoms – reported in FM, ME/CFS, and long COVID, it would seem odd indeed if the sensory neurons were not affected in some way.
That brings us to the question of whether metformin and similar AMPK-activating / mTORC1 inactivating compounds might help with pain.
A rather meaty 2020 review, “Metformin: A Prospective Alternative for the Treatment of Chronic Pain“, proposed, in fact, that metformin could be a new pain drug and might even be helpful with anxiety, cognition, and sleep as well.
The review indicated that researchers have been studying the effects of metformin on pain in the lab and animal models for over ten years. One animal study, which found that metformin suppressed “aberrant translation pathways”, and neuronal excitability, and inhibited pain, made it seem like metformin might be just the thing for overstimulated and tweaked pain pathways in FM.
Another study found that metformin was able to reverse allodynia (extreme sensitivity to touch) – a nightmarish treatment-resistant problem. Further studies suggested metformin may be able to diminish a form of allodynia associated with fructose (chronic fructose-induced tactile allodynia).
Plus, metformin’s apparent ability to impact the dorsal root ganglia – a primary sensory processing center and the putative source of “chronic inflammatory pain”, as well as a common hiding place for herpesviruses – adds another possible dimension to this interesting drug, given its antiviral properties.
Finally, metformin has positive effects on the gut, including increasing the levels of butyrate and short-chain fatty acids and bulking up the integrity of the gut wall. A recent article proposes repurposing metformin to reduce inflammation and oxidative stress, and enhance gut barrier integrity and the gut microbiome in inflammatory bowel disease. Despite this it can produce some negative symptoms in some people.
Despite a decade of successful animal and laboratory studies, few human studies have assessed metformin’s ability to reduce pain.
Metformin and other AMPK Activators and mTORC1 Inhibitors in Fibromyalgia (FM) and Chronic Fatigue Syndrome (ME/CFS)
Given the mitochondrial aberrations seen in fibromyalgia (diminished AMPK activity, decreased mitochondrial production, reduced oxygen consumption), metformin’s ability to enhance mitochondrial activity and reduce oxidative stress would make it seem like a good possibility for FM.
Indeed, a 2015 FM study found that the reduced AMPK activation found in FM tissues (fibroblasts) was reversed by metformin in a laboratory study. (Note that AMPK activation = mTORC1 inhibition = possibly increased ATP production, reduced lactate, reduced activation of pain nerves). The authors stated, “these results suggest that AMPK plays an essential role in FM pathophysiology and could represent the basis for a valuable new therapeutic target/strategy.”
That was followed by a 2016 study that again found “deficient AMPK activation” in FM. In a small treatment study, metformin treatment (200 mg/daily) “increased AMPK activation, restored all biochemical alterations… and significantly improved clinical symptoms, such as pain, fatigue, depression, disturbed sleep, and tender points” in six people with FM.
In the 2020 paper, “Is insulin resistance the cause of fibromyalgia? A preliminary report“, the authors reported that metformin completely resolved the pain of half (8/16) of the FM patients and that some responded only to metformin. (Others were getting drugs like amitriptyline and gabapentin). Plus, the metformin treatment effects seemed to stick over time. (This study was retracted by PLOS ONE because it had not received a proper ethics review. PLOS ONE was also concerned that the control group was not sufficient.)
Other AMPK activators/mTORC1 Inhibitors
Some evidence suggests that other AMPK activators/mTORC1 inhibitors might help in diseases like FM and ME/FS. Rapamycin – which returned one person with ME/CFS to health – inhibits mTORC1 as well and has been shown to reduce pain hypersensitivity in animal studies.
Similarly, Jarred Younger recently reported that two other AMPK activators – resveratrol (200-600 mg/day; more is better) and curcumin (500 -1,000 mg;) – were helpful in Gulf War Illness.
The Gist
- A couple of months ago, Health Rising published a post on a study that found when given for a short time during the initial coronavirus infection, metformin was able to reduce the incidence of long COVID by about 40%.
- This post goes over that study once again and then dives deeper into the possibility that metformin may be able to reduce pain and increase its ability to turn on a crucial energy regulator called AMPK.
- How metformin is working to reduce the incidence of long COVID is not clear but could be due to its antiviral, anti-inflammatory, blood vessel-protective, and energy-enhancing properties.
- Activating the AMPK enzyme switches the body from being in an energy-consuming to an energy-producing state. As ATP declines during, for instance, exercise, AMPK triggers more ATP production. AMPK should, therefore, be activated in the muscles during exercise.
- When Julia Newton whacked the muscle cells from ME/CFS patients with exercise, though, she found no evidence of AMPK activation. Giving the cells metformin, though, revived AMPK.
- AMPK also inhibits something called mTORC1. By doing so, it can reduce lactate production, increase ATP or energy levels, improve cognition, and induce something autophagy, which a recent Simmaron study suggests may be impaired in ME/CFS.
- Autophagy is a process cells use to clean up defective mitochondria. Problems with autophagy can cause cells to decline and start pumping our pro-inflammatory compounds called cytokines.
- mTORC1 activation on the other hand also appears to increase pain sensitivity. Over the past ten years, laboratory studies suggest metformin may be an effective pain drug.
- Human studies are rare, but a few small fibromyalgia studies in metformin and other AMPK activators suggest they might be helpful. Rapamycin, for instance, returned one ME/CFS patient to health, and Jarred Younger found that two other AMPK activators, resveratrol and cucumin, when used in the right dosages, were helpful in Gulf War Illness (see blog). Oxymatrine, used by Dr. John Chia in the form of Equilibrant, is another AMPK activator that’s proven helpful in ME/CFS.
- Many other possible AMPK activators are present. We don’t know which ones in which doses and in which combinations are most helpful, but much effort is going into finding ways to activate this crucial enzyme in the energy production pathway. It’s something to keep an eye on.
- For ME/CFS/FM and long-COVID patients, a big remaining question is whether the RECOVER Initiative or some other group will produce a metformin trial in long COVID.
Other AMPK Activators
Given the many ways that AMPK inactivation could be impacting diseases like ME/CFS, FM, and long COVID, finding ways to activate AMPK has to be considered. Thankfully, the interest that AMPK activation is sparking in cancer and other diseases has made finding ways to activate AMPK a growth field.
The Self-Hacked website lists an enormous number of possible AMPK-enhancing compounds including berberine (an antiviral), R-Lipoic Acid, Glucosamine, Resveratrol, Curcumin, Gynostemma (often used in AMPK activating supplements), Cod liver/Fish Oil, Anthocyanins, Cannabinoids, Astragalus, Reishi, Creatine, CoQ10, Bitter melon, etc.
Compounds are being tested regularly. Magnolol, for instance, is a plant-derived Chinese compound available as a supplement that was recently found to increase AMPK activation, downregulate MTORC1, and improve autophagy in a Parkinson’s mouse study. Astragalan, another Chinese compound (China is a world center of plant diversity) restored AMPK activity and improved energy metabolism in diabetic mice. A Chinese fungus, Cordyceps sinensis, helped to treat lung injury by activating AMPK. Two yogurt strains, Lactobacillus delbrueckii subsp. bulgaricus 2038 and Streptococcus thermophilus 1131, not only improved gut integrity but increased AMPK activity.
Many attempts, then, are being made to find effective AMPK activators. Which ones might work best in the context of diseases like ME/CFS, FM, and long COVID is unclear, but the growth of the field is encouraging. AMPK activation is something to keep an eye on.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



Appreciate this recap, Cort. Really curious to see further studies into Metformin and it’s efficacy…
Speaking of RECOVER (bottom of The Gist recap), it appears a ME/CFS comparative group has been approved to be incorporated in this initiative, per the recent Newswise feature (3/1/2023) with Dr. Leonard Jason, if I’m reading this correctly:
“He is a vocal advocate and serves on the diagnostics committee of the NIH’s RECOVER initiative, which is studying more than 17,000 people to learn about the long-term effects of COVID-19. Recently, Jason won NIH’s support to include an ME/CFS group as part of the work.”
https://www.newswise.com/coronavirus/researcher-leonard-a-jason-pushes-discovery-on-long-covid-mecfs/?article_id=787890
I saw that OMF shared this development as well: https://twitter.com/OpenMedF/status/1631308894545870849
Do you know any details about this, Cort?
I believe Jason got funding to add a long COVID cohort to his ME/CFS study following college students who came down with infectious mononucleosis. I don’t think we’re in RECOVER yet but this is a nice NIH funded study and it will help us a lot to compare the two.
I just got his memoir by the way and look forward to reading it. 🙂
I believe, two things have transpired, according to this feature…
1) Jason got funding to add a LC cohort to his ME/CFS study (the $842,000 from the NIH to further his research on ME/CFS with Lurie Children’s Hospital, bringing a focus on long COVID into the longitudinal study).
2) Jason, as part of the Commonalities with Other Post Viral Syndromes Task Force (aka diagnostics committee referenced in Newswise) got approval from the Ancillary Studies Committee for a ME/CFS cohort in RECOVER.
I ask since RECOVER could be so vital for these studies (as you mentioned) and if you had any pulse on the development of this ME/CFS cohort / Dr. Jason’s plans.
2) Jason, as part of the Commonalities with Other Post Viral Syndromes Task Force (aka diagnostics committee referenced in Newswise) got approval from the Ancillary Studies Committee for a ME/CFS cohort in RECOVER.
Well, that would be really something! I will try and find out. Let’s hope!
Thanks Cort! I look forward to hearing what you find out.
Approval has been given! A huge step forward…Now we need to get funding.
Thanks for this follow up, Cort!
Clearly, Dr. Jason’s role in RECOVER has been pretty invaluable for the ME/CFS community. Really thankful for his efforts and that RECOVER leadership is respecting Lenny’s voice / experience in this field.
I look forward to future reporting on this and if our community can help in any way.
Yes indeed. I know that Solve ME has been involved in this as well 🙂
I have one clear question about this metformin study. The study was performed on 1125 overweight adults. Now Metformin is a type-2 diabetes drug, particularly used for overweight people. Couldn’t this study just mean that Metformin as such addressed other health issues, making the participants “healthier” and thus reducing their chance of Long-Covid? Whilst it could reveal some mechanisms of Long-Covid, couldn’t it also just mean that it has no effect on previously healthy people? It would lovely to also see a “healthy group without any signs of diabetes” to be included in the next trial.
Yes, they were overweight and that does complicate things. It’s ertainly possible that metformin helped them metabolically and in that way some of them were able ward off long COVID. My understanding is that while they were overweight they were healthy though and were not taking metformin prior to getting COVID. They only took for two weeks after they came down with COVID and the earlier they took it the better – which was why the authors pointed to metformin’s antiviral properties. We don’t know how it helped, though. Too bad they didn’t do more testing!
Totally agree. Perhaps someday we will have more and better studies on antivirals that include immune signatures and all that beautiful stuff. Theoretically antivirals that also reach zones like the gut, eyes and other reservoirs where viruses seem to be able to hide should give us a very good idea on how much viral persistence and reactivation play a role. Hopefully Iwasaki’s Paxlovid starting this month (?) will give us a better idea. I am fearing however that the result won’t be very inspiring.
I disagree with your take on the Paxlovid study. I have had MECFS for 40 years, I got COVID twice in 2022( my wonderful immune system) was able to get Paxlovid when it was first released for COVID19 and it not only prevented me from the harshest symptoms of COVID but it improved many of my MECFS symptoms for a coupl of weeks, it was very hard on my GI system the first time of treatment. The second infection of COVID 10 months later, I got the Paxlovid within 48 hrs. of infection and it was easier on my metabolism it helped with preventing harsh symptoms and again it improved my MECFS symptoms. I am set to try a 1 week trial of Paxlovid while clear of COVID to see what impact it has on MECFS. If it’s positive then a 28 day trial will take place under a doctors care.
That’s really great and I am very happy to hear that! Of course there is unfortunately no way of knowing whether it really prevented harsher symptoms in your case as there was no control, but the studies on Paxlovid do reveal that it is an awesome drug to prevent a harsher acute illness and has similar effects on Long-Covid prevention as Metformin seemingly does. The far more interesting part for me is that it was able to improve your ME/CFS. Was this an long-lasting improvement? Do you feel that after doing some initial harm it has improved your GI smyptoms in the long run, i.e. it sort of had a reset effect? I hope your Covid improves soon and that you can then again try Paxlovid. I am just very pessimistic about Paxlovid able to do anything about Herpes viruses etc.
COVID definitely reactivated the herpes infections, EBV Simp. 1/2 I had Never had an oral outbreak or a genital outbreak but had been positive for at least 2 decades, but COVID had me in such pain from the oral outbreak I was in tears. Thanks to the awesomeness of Dr Klimas she had her own concoction of Magic Mouthwash that help resolve that! My GI system is a complete mess. But yes I did feel it helped, especially after the second treatment, I had a more normal function of my gut for a couple of weeks. I just had a bizarre GI infection 6 months ago that caused me a colostomy, doctors including surgeon said my insides were a mess, but found no real cause, I just had the reversal surgery 3 weeks ago and am still recovering, but GI functioning thus far. ( Thank God) I am interested in the metaformin! because of the multiple possibilities that it has on the metabolism! I won’t do the Paxlovid until I am at “My” best, to give it the best chance so I can make an appropriate decision!
Doesn’t that just sum up ME/CFS – insides a real mess but no idea why. That’s just it to a T…I hope the gut continues to improve – that would be huge.
So interesting that Paxlovid improved your ME/CFS symptoms
Any updates o. How your trial went?
It should be noted that RECOVER is also following people from the acute illness stage to the long COVID stage so they have ample opportunity to give people who just came down with COVID-19 metformin or Paxlovid and then test the heck out of them to see how they are helping. That would give us a clue how long COVID gets started in the first place.
Definitely would be cool if they did that, but I fear they often test for the wrong things if the researcher involved isn’t someone like Ron Davis. Btw what’s the current status on his salt-nanoelectric-assay biomarker (https://med.stanford.edu/news/all-news/2019/04/biomarker-for-chronic-fatigue-syndrome-identified.html)? My understanding was that they were testing FDA approved drugs for this (or was this just for the itaconate-shunt hypothesis?), but shouldn’t this biomarker still be available even though it’s not clinical.
40% is substantial reduction of risk for the 2-weeks trial though. I’ve heard that every 1 kg/m2 of excess BMI results in 3% higher risk of long COVID, so they could adjust the result for the BMI differential if they don’t want to do the trial again.
Interesting! Yes, they clearly must account for that factor…
True and interesting. But the adjusting doesn’t work like that. Giving “unhealthy” people a drug that is known to improve their health is not equivalent to giving “healthy” people a drug that a priori has no positive health effect on them.
I can see that, unless the drug changed nothing other than the obesity. Even then, reducing the obesity may not be the same as being less obese to begin with.
Yep 👍. As always with ME/CFS questions over questions.
A bit puzzled by this. I take metformin daily for diabetes, as will many others, have done for several years and haven’t noticed any difference to my CFS (although to be fair I don’t know whether I would now feel worse if I wasn’t taking it).
Also on amitryptiline and pregabalin.
Well, right now it’s just a possibility – which apparently hasn’t worked out for you. I wonder if you tried other AMPK activators with it?
This is my concern. Enough people take metformin that you’d have thought we’d hear something anecdotally by now if it was helpful. But here’s hoping!
I’d Like to Ask People and Patients who come here for education and information to support this site financially. Cort Johnson has been keep us all up to date on many related issues to our diseases! We all know how difficult it is to just get from bed to chair, Cort has been a Champion of Information for us all! I donate as I can. I would ask everyone here to do the same if you are able to. His site is my Go To, to stay up to speed! Thank you Cort!
Thanks Bob! 🙂
What an appropriate column for me!
I have just been Dxed with pre-diabetes and I am a skinny girl with no diabetes in my family and I eat low levels of high glycemic foods. I even got tested for autoimmune causes–but I do exhibit some symptoms of PCOS–but don’t formally have that either. To my knowledge I haven’t been infected with Covid, but because I feel crappy so much of the time, perhaps I have, but never noticed. Early PCR tests were negative.
So enter Metformin. I have been taking it for several weeks and my glucose test is still high! No pain reduction what so ever but perhaps it is because my pain is likely from EDS. My ME/CFS fatigue is still the same but lipid profile has improved.
Before the pre-diabetes test I asked my doctor for metformin, just to try. No go. So I did research and found out berberine is just as effective. Titrated up to 1,000mg per day and got horrible gastrointestinal side effects. Then I was granted metformin.
If AMPK is the answer, it is usually activated by exercise, exercise most of us with ME/CFS cannot do. So my next diagnostic hurtle is osteoporosis. It has secondary causes too, but in my case I think it is lack of exercise. When one’s body is out of whack, it seems to turn into chasing all the additional things that go wrong when the body systems are not balanced.
Metformin works to lower your A1C as I understand it. That is a long-range measure and your doctor will send you for blood work at the three-month mark. You wouldn’t see a change in your blood glucose levels after a few weeks.
What dose do you take? I am just diagnosed diabetes 2 and doc determining what treatment, I now will suggest the Metformin!
I totally agree with you about Cort & thank him for all that he has done. However I am not into all the medical jargon & don’t understand a lot that I read so I researched Metformin. I was surprised to see that it is pre scripted for 2 Diabetes & also used for weight loss, etc. In my research I found that Metformin caused gastrointestinal problems & probably I could never use it because of GERD. I continued my search & found an article by Mayo Clinic entitled “Metformin Revisited.” I read it hoping it would say more about FMCFSME that I have, but I did not understand all of it. The doctor was Dr. Nair. I also read that it could help pain & right now my FM pain is worse. So I thought “aha” Metformin is what I need. However, I will wait for the results of the study being done on Covid that we are included in.
Until then I hope & pray they will find something for us!!
Amen! My pain specialist keeps trying new medications. Percocet is what works best, but has GI issues for me. Marijuana makes me more fatigued, using a vape and only one puff? Done ketamine IV and chews, same puts me to sleep. The Metaformin looks promising due to its effectiveness on multiple levels. Also the Oxymatine looks promising for some.
Metformin is interesting in that it does to appear to help with several gut problems that are present in ME/CFS including short-chain fatty acid production and gut wall integrity. On the other hand, it also produces gut symptoms in some people.
Thanks Cort,
I’m going to wait for the outcome of the tests they are doing. Then if it looks promising I will try it, praying it doesn’t affect my GERD. I really need it for my FM pain.
Regarding Metformin gut problems; I have achalasia (no peristalsis so everything I eat or drink just sits in my esophagus for hours). Substances can irritate those tissues so I was worried I’d have big problems with Metformin. My doc prescribed liquid Metformin (a bit pricey) and I am surprised and relieved that I am not experiencing any throat or gut issues.
I am taking 500 mg (5mL) in the evenings with food. Tastes awful even with strawberry flavoring. ;-(
@Amy, I look forward to my next A1C.
Hi Nancy,
Fortunately I have my Gerd under control after many years & medications. I only take Prilosec for the acid reflux & watch what I eat. I’m very sensitive to strong medicine so I stick with the old, tried & true.
I am unfamiliar with Metformin & have decided to wait until the tests are made. Then I will try it to see if it helps my FM pain. Hopefully it won’t affect my gut.
Prayers for you.
Was there any mention of people experiencing hypoglycemia in the long Covid prevention study ?
I used transdermal metformin (100mg/1ml) for a few years – it was excellent for reducing muscular pain and preventing PEM – that could track with muscle cells/insulin resistance theory. Transdermal delivery bypassed our gut sensitivity issue – I found metformin by mouth tanked my sugar levels.
How about that! It may be with metformin as it is with so many things – some people will benefit and others will not. The key will be determining who does and who doesn’t.
Hi..is transdermal metformin prescribed by normal GP and compounded? Thx Kim
Hey Kim. I’m in Australia but both my endocrinologist and GP prescribed it. We did some experimenting with the cream base – 100mg/1ml was the most it would take. Good luck!
I am an n=1, but here goes. I’m a three-year long COVID sufferer with an ME/CFS presentation (in other words, I have ME/CFS plus some other bizarro LC symptoms). I have always been slim and fit, and I eat a Mediterranean diet and have for years. As has been happening with other long haulers, my blood work showed pre-diabetes last fall, and more recently, hyperlipidemia. The remnants of COVID are doing this, because it’s not my diet.
Endocrinologist started me on metformin in October and at the three month mark, my A1C did go down a few tenths. The hyperlipidemia was caught more recently and I was out on pravastatin.
I have not noticed any improvement in my symptoms as a result of being on metformin. My fatigue is just as bad. My small fiber neuropathy has recently gotten worse, spreading to my legs after being limited prior to my arms and temples. I’ve been on metformin now for six months, and I guess I’ll be on it for a while, so we’ll see if any of this changes.
Incidentally years ago when I was going through infertility treatments, at some point my doc added metformin to the cocktail of injections and pills. And presto, I got pregnant that month. But i subsequently had a ectopic pregnancy and was rushed into surgery to remove it. The same cocktail never worked again after that, and today we are cat parents only :). Point is, metformin is a pretty versatile drug. It’s great we’re keeping our eye on it.
@Amy, you may be N=2! Your profile sounds so similar to mine (excluding the achalasia). I’m curious (snoopy actually), do you also have Ehlers-Danlos? I also had/have PCOS symptoms like oily skin, some hirsutism, thin head hair, acne etc. but my endocrinologist can’t call it PCOS because I don’t fit the full criteria. Having lipid disturbances is also part of that profile.
The berberine totally brought down my hyperlipidemia–rapidly–but I couldn’t tolerate it–at least at that dose. Not sure what the Metformin will do.
And I too have a household of cats!
Hi Nancy – no, I don’t have EDS or PCOS, though I do have mast cell activation syndrome and attending symptoms such as eczema, as well as Epstein Barr viral reactivation and small fiber neuropathy. Interesting about the berberine. I’m going to get my cholesterol retested in a few weeks and I’m hoping I can cut down on my statin dose. I have a feeling I’ll be on the metformin for a while.
I’ve been taking metformin for a long time to fight aging and control weight gain, and I haven’t seen any improvement in my chronic pain.
At I understand, metformin trial was about prevention, not a treatment, of long COVID. So, what it does to help exercise intolerance may not be germane in the case.
Since metformin does not reduce the symptom severity, my guess is that it’s the anti inflammatory property, not antiviral one, that is helping to prevent long COVID. The body could be having hard time getting rid of the virus that it has never seen before and therefore suffer prolonged inflammation, even in asymptomatic cases. That prolonged inflammation could be resulting in the hypersensitivity to peripheral inflammation which leads to neuroinflammation after exercise.
I’d have to say it plays a more important role in limiting damage from insulin resistance which I believe is taken to an extreme level during active infection and subsequent timelines
Aspirin is also AMPK activator. Can it be used?
Metformin don’t hold any promise. Many people with CFS have diabetes and use metformin without any improvement in CFS symptoms.
Aspirin is reported by many Long covid patients to be helpful. Maybe it’s not aspirin AMPK thing but blood coagulation effect.
You make a good point. I think we would know if it was a game-changer. However it does appear to be doing some things we would like to see happen – increase AMPK activation, improve gut functioning, reduce inflammation – and a small FM study found it did help. One person also reported that it helped – so there may be a subset of patients that it helps.
What if a non diabetic take metformin?
Does that cause hypoglycemia and serious problems with low sugar?
Or is doesn’t affect sugar of people with normal blood sugar (non diabetic) ?
I have no medical or scientific training, but as someone with a normal BMI / no signs of diabetes, my PCP was fine with my taking metformin to reduce the risk of long COVID. He just suggested I avoid alcohol while I am taking it, as that can cause issues with low blood sugar while on metformin.
Anecdotally I can say that in the 5 days I’ve been taking it so far, I’m pretty sure I have not had hypoglycemia.
I will say that while my diet is fairly healthy, it is not very low carb — I am kind of curious whether someone on a very low carb diet would want to avoid metformin because their blood sugar is likely already quite low!
So now Metfrormin is the new panacee drug -:)
Doesn’t make any sense at all
Very interesting finding. Fasting and carbohydrate restricted (ketogenic) diets also upregulate AMPK, which may be why some people with ME/CFS (like my daughter) experience improvement in their symptoms on these diets.
A wealth of good info here, thanks Cort!
Interesting article, but can I get journal references to share with my Dr?
Just clicks on the links – they should all be there.
I have to say that AMPK activator from Life Extension has really helped my energy. I have CFS. I just came across the term “AMPK activator” about a month ago. It is such a big piece of the puzzle – probably the biggest for me.