THE GIST
- High blood oxygen levels entering the brain and low blood oxygen levels leaving the brain suggest the brains of ME/CFS patients are sucking up all the energy (oxygen) they can to keep going.
- Why this is happening isn’t clear. It could be due to low blood flows to the brain, but since the patients were tested while lying down, that seems less likely, at least in the prone position. More likely causes are problems with energy production and/or widespread inflammation.
- As researchers dig up more and more evidence that neuroinflammation is taking place, much of this blog will focus on inflammation, and who better to find out about that than Jarred Younger (“the neuroinflammation man” :)) who runs the Neuroinflammation, Pain, and Fatigue lab at the University of Alabama at Birmingham.
- Younger believes that immune cells in the brain called microglial cells are primed to spew out inflammatory factors at the slightest stress in ME/CFS: a bit of exercise, a psychological stressor, an infection. He found, for instance, that it took just a small dose of a bacterial toxin to inflame the brains of fibromyalgia patients (but not healthy controls).
- Younger is ready to close the door on the neuroinflammation or brain inflammation question in ME/CFS. He believes that the evidence is incontrovertible and that over the next year, there will be consensus on that question.
- In a nice fit with the first presentation, Younger found increased lactate – the substance the brain turns to when it can’t produce enough energy using the oxygen it’s getting – in ME/CFS patients’ brains.
- Younger is doing an “arterial spin labeling” study to assess, in more detail than has been done before, whether reduced blood flows to the brain are contributing to the neuroinflammation he’s finding.
- Younger has spent about 10 years trying to figure out how to determine whether immune cells from the body are getting into the brain. Because these powerful immune cells can cause all sorts of damage, only in the most dire circumstances will the brain call them in.
- With Younger’s new approach (created with two collaborators at UAB) ready to go, he expects to start testing ME/CFS patients over the next month or so. Finding that immune cells are getting into the brains of people ME/CFS could explain a lot and open the door to new treatments.
- In a recent YouTube video, Younger noted that the extensive testing he does uncovers, in about 35% of the cases, problems like diabetes, thyroid issues, and anemia that have been undetected. Since he believes that inflammation is a huge driver of illness in ME/CFS/FM, he does lots of inflammatory tests and came up with 8 tests that people with these diseases can pretty readily get done to assess their inflammation levels. See the blog for more.
- Michelle James Neuroimaging Research and Discovery lab at Stanford is on the cutting edge of imaging technique technology. Besides studying Alzheimer’s disease, multiple sclerosis and stroke, she’s also studying ME/CFS.
- Like Younger she believes that chronic inflammation is driving the fatigue, pain, PEM, etc. in ME/CFS.
- Recently, she’s been doing the first whole-body PET imaging ever done in ME/CFS (and perhaps in other diseases). Besides indications of inflammation in the brain, she’s also found it in the salivary glands (site of intense mast cell activity), the muscles, and, interestingly, the bone marrow (where many immune cells are produced). (Check out the images in the blog).
- The brain session, then, pointed to more evidence of neuroinflammation – and inflammation elsewhere (the muscles!) – as well of energy systems gone awry.
- We should learn a lot more over the next year as a host of Younger’s studies get published about the immune-brain interface.
Brain Metabolites

Xiang Xu is working with Benjamin Natelson at Mt. Sinai.
Xiang Xu of Mt. Sinai gave one of the young investigator talks. She’s working with Dr. Benjamin Natelson – a longtime ME/CFS/FM investigator – on a long-term ME/CFS subset study.
Her structural MRI didn’t find any alterations and that’s not a surprise. While we’ve seen evidence of spinal problems we’ve seen little evidence of overt structural changes inside the brains of ME/CFS/FM patients. That’s a good thing given how difficult it is to turn that kind of damage around.
The physiological MRI was another deal. While it found abnormalities in the thymus and other areas, the big finding from her study involved one of our favorite subject – oxygen and energy production. First, she found reduced oxygen levels in the venous circulation in ME/CFS-like long-COVID patients. (The venous circulation involves the blood that’s leaving the brain after the brain has sucked up all the nutrients and oxygen it can from it.)
Interestingly, arterial oxygenation taken at the finger was significantly higher in the long-COVID group. That brought up an interesting question – if higher oxygen levels are present in the blood-feeding the brain – why would lower oxygen levels be found in the blood leaving the brain? In other words, why were the brain cells of the ME/CFS-like long-COVID patients eating up so much oxygen? (Oxygen is the substance that generates ATP (energy) for the brain.)
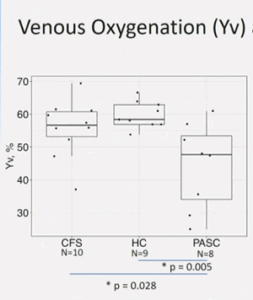
Dramatically reduced high arterial oxygen, reduced venous oxygen, and increased brain oxygen consumption suggested the brains of the long-COVID patients were snatching up all the oxygen they could find.
A reduction in blood flows to the brain could do it. If not enough blood is getting to the brain, then what blood made it to it would surely get stripped of all the oxygen possible.
To determine whether that was happening, she took MRIs of the blood vessels leading to the brain and calculated the “metabolic rate of oxygen”. That refers to the amount of oxygen being used by the brain. It’s considered “a direct index of energy homeostasis and brain health” and is usually maintained at a constant rate in healthy people.
While lying down, the ME/CFS patients exhibited normal blood flows to the brain but higher rates of oxygen consumption in the brain. ((Note that reduced blood flows to the brains of people with ME/CFS have been found when they’re upright (sitting, standing) and this study was done while they were lying down.) With that finding, what was happening became clear. The reduced venous oxygen levels found in the ME/CFS patients resulted from abnormally active brains that were stripping out just as much energy (oxygen) as they could from the blood.
That’s potentially a big deal. When we’re at rest, the brain consumes over 20% of the body’s total energy. Because the brain doesn’t have a lot of “storage capacity”, it needs to get constantly fed properly to function normally. Because increased oxygen consumption has also been found in various nasty neurological diseases, this finding again suggests that ME/CFS, at least in part, is a neurological disease.
Because the low venous oxygen levels were positively correlated with the physical fatigue scores; i.e. the lower the venous oxygen levels, the higher the physical fatigue, it’s possible the physical fatigue could result from the long-COVID patients’ brains’ voracious appetite for energy (oxygen).
Or is an overactive immune system sucking the energy from the rest of the body, as Mark Davis suggested. Again, we see a similar theme emerging – overactive systems in the brain, the immune system, and the autonomic nervous system that could be putting a big hurt on energy production.
That set the stage nicely for Jarred Younger’s talk.
Jarred Younger: “Brain inflammation in ME/CFS – how we measure it and how we treat it.”
Younger noted that he does “high-risk” experimental studies (high risk / high reward, that is) and boy, is he right about that. He’s been right out there on the skinny branches developing new techniques to study brain inflammation and immune infiltration in the brain. Plus, unlike most other research scientists, Younger has had a strong interest in assessing treatments. It was Younger’s small studies that birthed the interest in low-dose naltrexone in fibromyalgia and ME/CFS.
Younger noted that in their primed state, microglia can look relatively normal but explode into action at the slightest trigger. Many possible microglial triggers exist: lots of fat cells, any peripheral inflammatory state, an altered gut microbiome, normal aging, reduced oxygen in the brain, chronic stress, and viral/bacterial infections – particularly when a series of them occurs together.
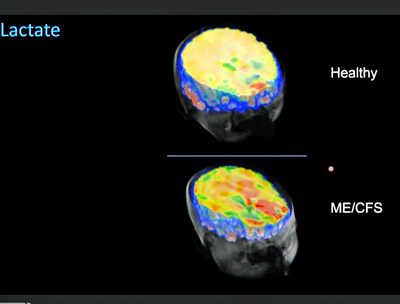
The high lactate levels (red – ME/CFS patients at bottom) suggested their brain cells had switched to an alternative energy source.
Once those microglial cells are activated in ME/CFS, Younger believes that even something as small as a bit of exercise can activate them. The goal is to get the microglia back into their resting state.
Younger showed how on edge the microglia are in fibromyalgia. Injecting fibromyalgia patients with a vanishingly small amount of bacterial toxins caused the microglial cells to act up across much of their brain. They also showed a huge increase in a microglia activator called leptin, and a reduction in a microglial inhibitor called fractalkine. Since about half of the FM patients met the Fukuda criteria for ME/CFS, Younger felt there was a good chance the same is occurring in ME/CFS.
He’s about half done with an NINDS-funded study in ME/CFS that will, if positive, he said, constitute “very powerful evidence” that the microglia are in an inflamed and agitated state in ME/CFS patients’ brains; i.e. that neuroinflammation is present.
Younger noted that many studies, including his, have found elevated lactate throughout the brain in a subset of ME/CFS patients and he has found very high elevations in lactate in a subgroup of patients. Lactate is what the brain turns to to produce energy when it runs out of oxygen; i.e. it’s what we would expect the brain to do given the results of the first study cited above.
Combining the PET and MRS studies done so far gives Younger great confidence that neuroinflammation is present in ME/CFS. A few more data points – which he expects to come this year – and that finding will be incontrovertible.
Instead of a brain-centered problem, the inflammation could also, as noted above, be due to problems with orthostatic intolerance and/or blood vessel issues that prevent enough oxygen from getting to the brain. Younger is testing for that with his arterial spin labeling study. Because his study is being done with the participants lying down, if it does not show hypoperfusion, he’ll test the participants while lying supine, then get them upright, and put them back into the scanner.
(Who’s to say it’s not both, though? Brains that are battling for all the energy they can get while also getting whipsawed by a further reduction in oxygen levels while upright?)
A Huge Step for ME/CFS? The Immune Cell Invasion Study
Younger has proposed that immune cells from the body (leukocytes) may be making their way into the brain, causing inflammation, brain fog, profound fatigue, anxiety/depression, etc. Only in extraordinary situations (raging infection) would the brain ever call immune cells from the body into it. Whether or not the infection is resolved, damage is going to ensue. It’s like calling missiles in on your own position as it’s about to become overrun.
In multiple sclerosis (MS), T-cells make their way into the brain and, mistaking the myelin sheaths of the neurons for foreign entities, attack them. Nothing in ME/CFS suggests that this kind of damage is present. Instead, ME/CFS, fibromyalgia, and Gulf War Syndrome appear to be diseases of general inflammation. Younger does not lump ME/CFS into the neurodegenerative disease basket (MS, Parkinson’s, etc.) where you find damaged neurons and structurally altered brains. He believes they are a different class of disorders.
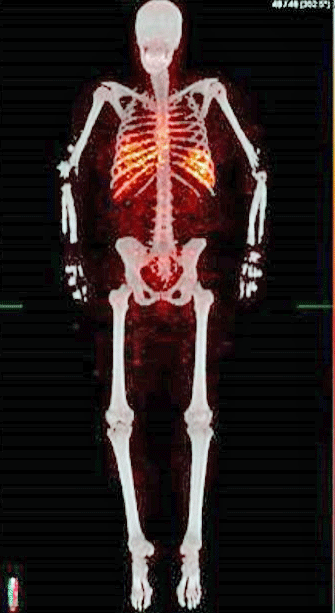
The first human leukocyte tracing image done. Note how the white blood cells (red) are concentrated in the middle of the body around the lungs and the heart, where most of the blood is. Younger will soon start assessing people with ME/CFS.
Younger has spent about ten years trying to figure out how to image white blood cells entering the brain. After digging a lot of dry holes during that time, two of his colleagues at the University of Alabama came up with a new technique: leukocyte tracking using PET scans examining radio-labeled leukocytes.
The leukocytes are taken out of the patient – get labeled with a radiotracer – and then are reinserted back into the patient. A PET scan then determines where the leukocytes went. If immune cells from the body show up in the brain – you’ve got problems. Four healthy controls demonstrated that the technique was safe and effective; i.e. it found that none of immune cells entered the brain.
Younger predicted finding leukocytes in the brains of ME/CFS – if it happens – would be a huge step forward for this disease and point very strongly to new treatments. He has submitted safety data from the healthy controls to the FDA. In February when this video was made, he hoped to get the first patient – an ME/CFS patient – into the study in 2 months – so we should know more in about a month from now. Once Younger had a few patients done, he promised to start spilling the results on his lab’s YouTube channel.
Big props to ME Research UK for stepping in and funding what can only be termed as a high-risk/high-reward study. Their funding allowed Younger and colleagues to develop a technique that could help not just with ME/CFS but with many other diseases as well.
Big Year Coming Up For Younger and Possibly ME/CFS
Younger has gotten some big ME/CFS grants (good day / bad day; neuroinflammation; leukocyte infiltration) funded but has lacked the post-docs needed to complete them. That issue has apparently been solved, as Younger reported this year will be a year of data analysis and writing up papers.
Clinical Trials
Putting on his clinical trials hat, Younger reported that he has a long and growing list of potential treatments – probably around 100 – he would love to get tested in ME/CFS. If one of his studies finds immune cell infiltration into the brain, drugs that knock out those immune cells or that repair the blood-brain barrier could be used. If he finds problems with cerebral perfusion, he mentioned methylene blue – an over-the-counter supplement that calms microglia and helps with cerebral blood flow.
Citing the fact that many of the FM patients in his LDN trial met the criteria for ME/CFS, he called for large low-dose naltrexone (LDN) trials in ME/CFS. He noted, though, that a recent fibromyalgia study of 99 people using 6 mg/day for 12 weeks did not find LDN helped more than placebo. It looked like the study was well done, but a companion commentary asserting that physicians should not give LDN to any new patients went too far given the many ME/CFS/FM patients who have benefited from it.
Plus, 48% of FM patients in the study had a “clinically significant reduction in their symptoms”. Younger proposed that the study may have been too small for its type (parallel study) and asserted that a properly powered trial of LDN is needed. Then he recommended testing a different form of LDN called dextro-naltrexone that could be used in much higher amounts.
Easy Tests For Inflammation
Speaking of his lab’s YouTube channel, Younger has been back releasing videos of late. Two weeks ago, his “Simple blood tests to detect inflammation” YouTube presentation provided tests that will “generally” tell you if your pain/fatigue/symptoms might be caused by inflammation.
He noted that this is not medical advice, and that researchers are often ahead of doctors in this realm and may not conform to what the doctors know of as “best practices”. Younger runs lots of blood tests and about 35% of them detect a problem that the patients didn’t know they had. They find diabetes, thyroid disorders, autoimmune disorders, anemia and others.
Because Younger suspects ME/CFS/FM, Gulf War Syndrome, and long COVID are chronic inflammatory conditions, he does lots of tests of inflammation, many of which are not available at medical labs. Noting that sustained inflammation tends to beget more inflammation, it’s clear that Younger believes tests of inflammation should be part of a yearly package of standardized blood tests and that doctors should be more concerned when they see more moderate levels of inflammation than they are now.
Direct-to-Consumer Medical Testing Labs
I was astonished several years ago to find that some medical labs don’t need a doctor’s order to get tests run. Dr. Younger said he has used these direct-to-consumer labs to good effect. They include Request a Test (directs you to labs), Direct Labs, LabCorp on Demand, Walk-In Labs, Wellness Mama, Everlywell, and others. Some of these send test kits to your home.
Inflammation Tests
The BIG Two – These are readily available.
High sensitivity C-reactive protein – tracks well with inflammatory activity. Want values below 1. If the number reaches as high as 4, you have a “greatly increased risk of having something serious happen”. If it gets above 10, you have something that needs to be figured out.
After the lab opened during the pandemic, he saw lots of healthy controls with 10 values which was a lingering effect of COVID-19 and/or vaccines. They have dropped since then.
Erythrocyte sedimentation rate (ESR) – you want something below 20; 60 = moderately high; above 100 – high inflammation. A bit more consistent than hi-sensitivity C-reactive protein. ESR levels have been a good predictor of who will respond to low dose naltrexone. Fibromyalgia patients with higher ESR levels tend to respond better to LDN. (ESR- 60 mm/hr – 50% reduction in pain; 30 mm/hr – 30% reduction; 15 mm/hr – 20% reduction)
The Next Six
If you have good insurance, can afford them and your doctor will run them, Younger suggests these as well. Most are inexpensive. Some can be ordered labs on your own:
- Serum amyloid A (SAA)
- Plasma viscosity (PV)
- Ferritin
- Fibrinogen
- Interleukin-6 (IL-6)
- Tumor necrosis factor-a (TNF-a)
If all of these tests are low, Younger believes you probably do not have a problem with inflammation.
These are general indicators of inflammation and if they are high, the next step is to talk with your physician and hopefully get more tests run to try to figure out where it is coming from.
The complete blood test (CBC) that is often run can also give some indications if inflammation is present.
- Check out his Neuroinflammation, Pain and Fatigue Lab website and his YouTube channel
A big thanks to Jarred Younger for taking on this field when so few would, and showing that it’s possible to be successful in it.
Michelle James Ph.D – Illuminating Whole Body Immune Responses Using PET

Michelle James Ph.D received an NIH grant to study ME/CFS
Michelle James Ph.D is originally from Sydney, Australia (and sounds like it 🙂 but is now an Assistant Professor at Stanford. The winner of a long list of awards, James now runs the James Neuroimaging Research and Discovery lab which is researching Alzheimer’s disease, multiple sclerosis, myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), and stroke.
The lab’s goal is to develop novel, state-of-the-art medical imaging methods to enable highly accurate and ultra-sensitive detection of these diseases – particularly in their earlier stages. How sensitive is sensitive? PET scans can detect molecular changes at the nanomolar level. James showed a slide of raging inflammation found in mice given a bacterial toxin before they showed any symptoms.
In the last half of her talk, James relayed the wide variety of specialized tracers the lab is developing to allow researchers to see, through their imaging studies, exactly what is going on in these diseases; i.e. what parts of the body, what cells, what immune factors are involved in them. She asked for ME/CFS researchers to send her their tissue samples so she could test them.
The NIH Grant
It was great to see such a high-level approach being used in ME/CFS. In December 2021, James received an R21 (exploratory) grant, “Imaging inflammation in the whole body and brain of ME/CFS patients“, to do the first whole-body PET imaging of ME/CFS from the National Institutes of Neurology and Stroke (NINDS) at the NIH. She noted that “encouraging preliminary data” found evidence of increased neuroinflammation in the brains of people with ME/CFS.
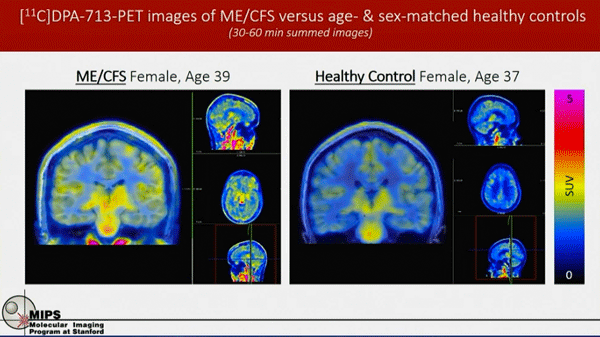
The first PET scans found more signs of activation; i.e. inflammation in the brains of ME/CFS patients.
In the grant, she hypothesized, “chronic, unresolved inflammation driven by innate immune cells underlies the central clinical problems of fatigue, pain, and cognitive dysfunction observed in ME/CFS”.
Because they’ll be assessing markers of inflammation and pathogen activity, the oxidative stress study should illuminate many factors in ME/CFS including immune activity (cytokine signatures), herpesvirus activity, the blood-brain barrier disruption, white matter changes in the brain, and disease severity. (That’s a lot – think what she could do with a full R01 grant from the NIH. )
So far they’ve gotten through 4 pairs of ME/CFS patients and healthy controls. They found evidence of increased activity in the thalamus, pons, and other parts of the brain. As other studies have found, the inflammation was more pronounced in the women than the men.
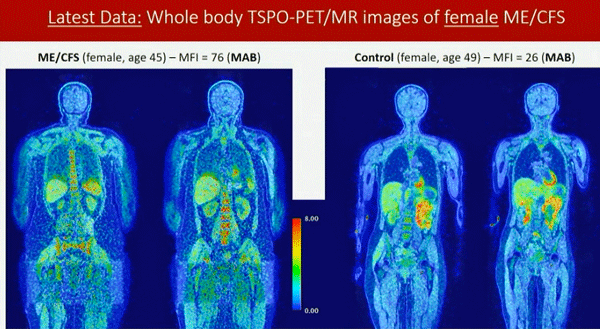
Early full-body PET scans found more activation in the muscles and bone marrow of ME/CFS patients.
One finding, though, intrigued her. After finding increased inflammation in the salivary gland – a site of high mast cell activity – she was eager to do whole-body PET scans. Her colleagues were skeptical they could be done, but she found a way to make it work in ME/CFS and received NINDS funding to do the entire body.
The scan found increased activity in the muscles and bone marrow (in the females). (The bone marrow produces red blood cells, immune cells (white blood cells) and platelets). With a new, more sensitive scanner coming, and with better tracers for the microglia and macrophages on the way, she should be able to dig deeper into ME/CFS physiology.
Besides the whole body approach, she’ll also do in-depth scans of the spleen (immune cell producer, blood filterer), lungs, and gut. Collaborators will use machine learning to assess the results. (Jarred Younger was a named collaborator in her NINDS grant).
Check out the 1st NIH ME/CFS Conference Blog






It’s been over a decade since the Nakatomi paper came out and failed the replication study. I can’t wait for these new studies to get published and confirmed! The immune cell invasion theory sounds like what Mathias has been saying.
There is no evidence of sustained peripheral inflammation in ME/CFS though. Not so far anyway. There have been many studies done on it, and we would’ve known by now if there is a noticeable inflammation. I’d think it’s the hypersensitivity to peripheral inflammation, not peripheral inflammation itself, that’s causing the problem.
You could be right about the sensitivity to peripheral inflammation – that would track perfectly, actually, with what Jarred thinks is happening in the brain with the supersensitive microglial cells going off at the slightest provocation.
Younger does, however, apparently regularly find signs of inflammation in the body – hence his talk on inflammation tests. On that note the results of his Good Day Bad Day study could be revealing.
There is also Goebels fascinating FM study which found evidence of localized inflammation that wouldn’t show up on standard tests.
https://www.healthrising.org/blog/2022/09/30/long-covid-fibromyalgia-autoimmune/
That meant that after all the focus on the central nervous system in FM, Goebel was able to reproduce a fibromyalgia-like condition in mice without ever directly impacting the central nervous system at all. Nor did evidence of systemic inflammation crop up. Whatever the antibodies were doing, they were doing it very locally.
Goebel’s findings are potentially so exciting because they could explain one of the great mysteries in FM and ME/CFS – why we don’t see enormous spikes of inflammation. Goebel’s findings suggest that small amounts of inflammation are probably occurring very close to nerves.
Fascinating. Did Goebel publish the mice study? It should be relatively easy to replicate it since it only involves mice and IgG. I’d think it would be fairly conclusive evidence if it gets replicated.
It is my opinion that there is indeed some very low level inflammation occurring in the periphery of ME/CFS patients, albeit difficult to detect. And I believe that this is being communicated to the brain & in turn the microglia in the brain are hyper-responding to this, creating all the symptoms of ME/CFS like Faigue,Brain Fog, Headaches,Generalized Weakness,etc.
Very exciting and promising discoveries that I hope and pray escalate in solid findings and a more widespread understanding of these maladies we suffer so greatly from ! Thanks Cort ! I was very grateful to God to miraculously be able to focus and actually read and understand the whole article! Not a common ‘event’ my brain and body can do much anymore 🤪😂🙏🏻
I concur! Thank you, Lord, for all these wonderful researchers uncovering these mysteries of the body. And staying with it year after year. And always a Thank you to beautiful Cort!
What would we do without him. Words fail…
🙏🏻🙏🏻🙏🏻
I would like to see Xiang Xu perform brain oxygen usage tests on MECFS (never had covid) patients. And I’d also like to see all of the above testing done before and after separately LDN and Abilify. I believe those are currently the only two medicines MECFS patients are experimenting with that effect the brain? LDN already has a trial coming but LD Abilify is being tried by many patients with no imaging or blood test support.
Hi Cort,
Great article.
What could be the relationship between these fairly definitive
inflammation studies, and the effects on our mitochondria?
Are our mitochondria problems the RESULT of inflammation and over use of oxygen, or could the causality be in the opposite direction, with the mitochondria being primarily causal?
From what I’ve read inflammation could be whacking the mitochondria and/or damaged mitochondria could be producing inflammation (oxidative stress) that ends up damaging them as well.
HHV-6b inflammation theory can explain this: The virus produces molecules who hamper the proper functioning of the mitochondria.
Leading herpes virus researcher Bhupesh Prusty from Germany has found that link in 2020: https://pubmed.ncbi.nlm.nih.gov/32327453/
Thanks for writing this. It gives me hope to see more biomedical evidence, which will hopefully lead to effective treatments.
Have you seen this study?
Showing differences between groups diagnosed with the ICC and the Fukuda criteria? The only way to see the differences in brain structure is using the world’s strongest MRI (7T MRI). The recent NIH study only used the 3T MRI which wouldn’t show much.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9321967/
I wrote about the importance of retiring the Fukuda criteria because it captures people who don’t have ME/CFS as the above study illustrates:
“The use of the outdated Fukuda case definition in the Australian guidelines is concerning as it doesn’t mandate key symptoms like post-exertional malaise (PEM) and neurocognitive signs, potentially leading to over-diagnosis. A recent study demonstrated this by identifying enlarged brainstems in ME/CFS patients meeting the International Consensus Criteria (ICC). The study categorised patients into ICC and Fukuda groups, revealing significant differences in brain structure only in the ICC group, while no such differences were observed in the Fukuda group or healthy controls. This emphasises the necessity of criteria mandating both PEM and neurocognitive signs for research and diagnosis.”
From my recent article: https://medium.com/@amandafrancey/australians-abandoned-by-healthcare-sentenced-to-living-death-2d0097184091
Wow. Thanks so much. That is really something – and here I was thinking that the definitions don’t matter THAT much – but they clearly do.
What an impactful article on Medium.
What the heck happened to Andrew Lloyd of the mighty Dubbo studies? I wrote one of my very, very few negative articles on an ME/CFS researcher on him in 2015.
https://www.healthrising.org/blog/2015/05/16/the-decline-and-fall-of-an-mecfs-researcher-the-case-of-andrew-lloyd/
Much vilified and he will be proven correct – that an immune event hits the brain, and that is where the problems lie in terms of perpetuation of the illness. As Younger is finding.
He got a lot of unjustified abuse from people in the ME/CFS community. There’s still plenty of people who think ‘problems in the brain’ = psychosomatic, which is simply wrong!
He probably got sick of the abuse. Or maybe he turned to something else. Or both
It’s interesting that Younger is finding that lactate might be causing brain inflammation. I wonder if he is also testing for ammonia in the brain?……(also a byproduct of anaerobic energy production) that would cross the blood-brain barrier? Both substances would cause brain inflammation presumably?
Nicotine can help stop immune cells getting into the brain 🙂
See this:
https://mscanada.ca/ms-research/latest-research/ms-society-funded-study-shows-that-nicotine-reduces-the-invasion-of
There are quite lot indication,that the Immunsystem play a big role in MC/CFS.
I am looking for immunhistologicale investigations of brain -, muscle- and gut- tissue.
If in these perivascular cells are deposit of IgA1, C3 and fibrinogen, that would mine an activation of
the alternative Complement-System has happened.
A step forward to understand the pathomechanism of ME/CFS.
If You have done or know literature (authors) of these immunhistological investagation,
please inform or send it to me.
Thank You for helping
Sincerely
D.Günter Schuhfried
3340 Waidhofen an der Ybbs
Oberer Stadtplatz 34
Austria
Thanks for this article, very interesting. I’ve suspected an immune problem in my case for a while, since my sister has MS, and there are many other auto-immune diseases in the family. The good news is that LDN 4mg made a huge impact for me, so i’m also grateful for that. Kind regards
What i find counterintuitive is that the CNS is using all this O2 when it is presumed that me/cfs hallmark is anaerobic glycolysis that does not proceed to oxidative phosphorylation.
Interesting article, as always!
I’ve been updating the Wikipedia article on ME/CFS. The Wikipedia article currently states there are structural changes in the brains of people with ME/CFS, partially based on this review: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7466519/, which says multiple studies have found structural abnormalities in the brain stem: “Five studies from two groups reported structural abnormalities in the brain stem in ME/CFS using MRI”. Would you say that is controversial or incorrect then?
I did say there was little evidence of “overt” or large structural changes in the brain but as you point out microstructural changes have been found in the brainstem (and I believe in the prefrontal cortex). Except for the brainstem they’ve received that much attention; instead the focus has been more on neurovascular coupling and functional connectivity. Thanks.😊
I wonder how/if perispinal etanercept treament would help ME/CFS patients with their brain inflammation. Would it deactivate (normalize) the highly triggered microglia?
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4110406/
https://www.sciencedirect.com/science/article/abs/pii/S1359644608003735
Dr. Edward Tobinick pioneered it and has a clinic in florida doing it. Has anyone gone/done it?
https://www.sfgate.com/health/article/How-etanercept-is-aiding-stroke-victims-4128887.php
Onset of MECFS can be variable in nature: infectious, gulf war syndrome, ptsd, physical trauma, surgery, anesthesia, CCI, etc, the list goes on. We must ask ourselves, how do they all connect? Blood flow changes to the brain is what I feel could connect us all.
Finding what is causing the blood flow changes to the brain may be unique in each individual. For those that have signs of inflammation, those may be people who have poorer blood flow than others. Then you have can have someone having inflammation AND physical obstructions of blood flow like the hyperclotting folks of Long Covid and other post-acute viruses (ie measles and potentially others). Then you have mechanical obstructions of blood flow like CCI, etc. I would think a mitochondrial disorder or injury could slow blood flow as well as everything would work a bit slower.
If the blood flow to our brains were affected, I would assume that is a major alarm and I bet our body would be pushed into an altered state. If blood flow is our connection, we could connect the immune system easily with the adrenal glands and their connections with the baroreceptors and RAAS.
The baroreceptors (responsible for relaying information derived from blood pressure within the ANS) could be detecting these chronic changes, these subpar O2 deliveries to our body parts and brain. The baroreceptors have effects on the immune system down the line. By alerting the Renin Angiotensin Aldosterone System (RAAS), the RAAS then alerts the malfunctions to the Adrenal glands. Per google AI: the adrenal glands produce hormones that regulate the immune system, blood pressure, metabolism, and the stress response. The adrenal glands also produce secretory products that regulate cytokine expression, immune cell activation, and bacterial proliferation. THIS is how chronic blood flow issues can alter the immune system, hormones, and metabolism. Find the reason for the altered blood flow (different in each patient), and the bodies baroreceptors will stabilize the immune system. 🙂
All these events probably hamper the immune system for some time and then some HHV-6b reactivation can happen. The virus replicates in immune cells and in then in the brain. When you can’t stop it the chronic inflammation damages the immune system more and the inflammation gets worse and worse.
https://www.brunel.ac.uk/research/projects/reactivation-of-herpesviruses-in-chronic-fatigue-syndrome
Can you please tell me the abnormalities found in the thymus please. I had to have my thymus removed and know of another person whith ME who had an abnormality. So this is an interesting find.
Cort, thanks again very much for all that you do.
Regarding the Erythrocyte Sedimentation Rate (ESR) test, here’s the web link to one of your Heath Rising blogs that also addresses ESR.
https://www.healthrising.org/blog/2019/11/06/clumper-slider-esr-chronic-fatigue-fibromyalgia-poll/
Best wishes to all.
Sorry random question time! Has anyone, especially women, tried the herb dong quai? And if so, did it help with fatigue etc?
The recent study from the UK on long covid showed a link between levels of hepcidin and fatigue in long covid. Dong quai (angelica sinensis) can modify hepcidin
Surely an inflammatory response in the brain can be the result of reduced blood flow? This results in repair work and possibly an infiltration of white blood cells and macrophages. Shouldn’t these also be visible in the spinal fluid?
And is this examination performed lying down and sitting? This may be an important factor or after an exercise test where most of the blood is drawn to the muscles.
Interesting study, I’m curious. But if it is positive and finds white blood cells in the brain, a lot of research still needs to be done into the cause, etc… that could all take a very long time. We wait again.
I have talked about this research many times over many years. Block CCL2! And nicotine can do that
https://medicalxpress.com/news/2009-02-inflammatory-disease-fatigue.amp
MCP-1 increased in ME/CFS patients. MCP-1 is a different term for CCL2.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8474618/
I don’t understand about CRP. I’ve never had high CRP so far (might want to do one again). Also CRP is pretty standard in German lab tests, and so many people have ME/CFS but normal standard lab values.
Renz-Polster and Prusty will be happy about the microglia and bone marrow connection mentioned in the Gist, connectin to their own theories on reactive microglia and herpes viruses ((?) in the bone marrow, respectively.
Im surprised no one mentions glucose is the brains primary food after oxygen.
if i eat sugar all day, i feel pretty great.
yeah, i know, its not a long term treatment, but it’s backed by science.
I also believe that inflammation is an important part of ME/CFS. Whenever I take an Advil, I feel better. But, Advil taken too often makes my right kidney hurt.
The bottom line question, however, is what is causing the neuroinflammation. In my case, I believe this was kicked off by a combination of a virus HHV-6A and exposure to Dursban used for flea control in our home where I had my office at the time.
Dursban was widely used in the first Gulf War where nearly one-third of the soldiers developed Gulf War Syndrome. After Dursban was taken off the approved list for military use, there have been no more cases of GW Syndrome that we are aware of.
I have a body burden of xylene, a solvent used in Dursban. I believe that when this is released into my bloodstream, it causes a constant toxic (poisoned) feeling and suppresses my immune system allowing the rise in quiescent herpes infections like HHV6 A & B, EBV and others as well as bacteria. These infections further suppress immunity leading to a never ending cycle of illness.
All of these events could lead to constant inflammation.
Xylene and other toxicants can be stored in body fat. One of the most vulnerable body systems for toxin storage is the brain where every cell has a fatty sheath.
I am not at all sure that everyone who reads Cort’s blog has the same thing I do with the same origin.
One small test could be: can you wear cologne or perfume now? Could you before you got sick?
Interesting. Seems unlikely to be a common exposure or cause, and I have not been tested, but I did have years of inhalation and direct skin exposure to a sealant laden with plenty of xylene and toluene. We were told it was safe and the company never even used gloves, or any type of ventilation (granted I dont know of any of my ~10 former coworkers who got sick); we would finish jobs with our hands covered in the stuff and it took days to ‘wear off’ (I finally smartened up and bought and wore my own gloves).
Now, thats just one of many long-term chemical exposures Ive had, so who knows (cropduster-glyphosate-soaked childhood on a farm, automotive fuels and solvents, heavy-duty Swedish wood floor finishing sealant, regular ‘pest spraying’ of the whole house when I lived in the buggy South, etc etc).
Not to mention several other *potentially* contributing factors such as a TBI, cPTSD, chronic bladder infections and at some point EBV and CMV (and now C19x3), decades of very high stress and ‘push-thru till ya drop’ workaholic mentality, possible genetics and/or Agent Orange effects through my Father’s Vietnam service exposure just a couple months before I was conceived (half-sister also has major issues such as MS and Vascular EDS with spontaneous bowel perforations). Hard to parse all that out, and likely the combination of all contributed to making me more susceptible to ME. But I honestly never gave much thought to the xylene exposure.
What kind of test would reveal if any of those chemicals are still ‘in my system’? Is there a way to get them out?
Oh, and I definitely have bad chemical sensitivities now, most anything scented makes me horribly ill -definitely perfume. And laundry stuff, I have to stay inside and close the windows when any neighbor is doing laundry.
Dear Never Give Up, I believe my test for xylene was done at Great Smokies Lab in Asheville which has now become Genova. I will check with my doctor when I see him next week to make sure, but Genova offers many interesting tests including a detoxification profile which helps determine if you are able to break down various toxins and drugs efficiently. You mentioned that most of the workers exposed to the same chemicals did not get sick. They may not have been susceptible or they may develop other illnesses like cancer and autoimmune diseases that take longer to identify.
I was interested to learn that you were the child of a Vietnam veteran. I am the director of Birth Defect Research for Children which sponsors the National Birth Defect Registry. Over 500 children of Vietnam veterans in the registry have ME/CFS.
Bi-partisan legislation has just been sponsored by Senators Tessler and Rubio to do further testing of the reproductive effects of toxins like Agent Orange and Gulf War exposures that veterans have been exposed to during their military service.
If you would like to know more about this, please visit our website at http://www.birthdefects.org and go to the Veterans Research Section.
As far as detoxification goes. That is very tricky. If your body can’t process toxins, you don’t want to suddenly move them from your body fat to your blood stream where they can settle in places like your kidney and heart.
We have read of one successful detox program done by the medical officers at Allied Chemical Company in the 70’s. The workers making a chlorinated chemical called Kepone began to display neurological problems and infertility. They were treated with cholestyramine, an anti-cholesterol drug that released the chemical from body fat; bound to the toxin and carried it safely out of the body.
The two caveats here are the cholestyramine is not without its own problems and whatever agent like this that is used must be able to bind to the chemical of concern so the toxin isn’t released throughout the body where it can do more damage.
This, however, is where research should be funded so that we can safely detoxify from many toxic chemicals in our modern environment.
Further reading on venous blood being de-oxygenated:
https://mylongcoviddiaries.medium.com/
https://www.reddit.com/r/cfs/comments/poz2d0/amazing_thread_with_a_bunch_of_info_about_pots/