

The title of this metabolomic study “Evidence for Peroxisomal Dysfunction and Dysregulation of the CDP-Choline Pathway in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome” made it clear that something new was in the works. Peroxisomal dysfunction had hardly popped up at all in the mitochondrial and metabolomic studies done to date – and yet it was headlining the outcome of a big, meaty NIH-funded study.
The study with the strange title came out of Ian Lipkin’s Center for Solutions for ME/CFS and featured a metabolomic analysis of 888 metabolic analytes in 106 ME/CFS cases and 91 frequency-matched healthy controls.
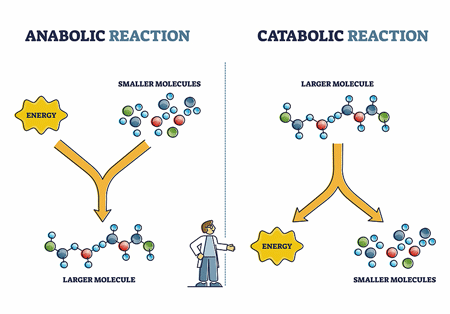
Metabolism involves reactions that break down compounds (catabolic) and reactions that build them up (anabolic).
Metabolomics studies assess the levels of metabolites. A metabolite is the end product of metabolism which refers to the sum total of chemical reactions that keep us alive. We break down molecules (catabolic) or build up (anabolic) compounds to keep the energy flowing, the organs operating correctly, the body repair processes working, etc. Since we know the metabolic pathways in detail, we can use altered levels of metabolites to determine where breakdowns are occurring. Since energy – either the use of it or creation of it – plays a key role in metabolism, metabolomic studies are particularly good at ferreting out problems that occur when energy production is lacking.
The metabolomic studies are particularly interesting because of the ground they cover. If you throw, as this study did, nearly 900 metabolites into the mix, there’s no telling what might pop up. If studies of that breadth show a consistent signature – you’ve probably got something.
The Lipkin team noted that since Naviaux’s effort in 2016, at least five large-scale metabolomics studies have been done – and they do show a common thread: decreased levels of phospholipids and metabolic dysregulation. Phospholipids are complex compounds found in our cellular membranes which play critical roles in ATP generation, and protection of the cell membranes and the myelin sheath lining the nerves.
Results
The results of this latest study were encouragingly consistent with those of past studies but seemed more refined, and more targeted – which is what we want. We want to get past the general “metabolic dysfunction” stage and pinpoint exactly where the problem is. This study seemed like it was a step on the road to doing that, and in doing so, it showcased a part of the cell that has received almost no attention in chronic fatigue syndrome (ME/CFS).
Small Organelles Hit the Big Time
Decreased levels of the plasmalogens that protect the phospholipids in the cellular membranes from damage suggested that membranes of the ME/CFS patients’ cells were taking a beating – probably from oxidative stress.
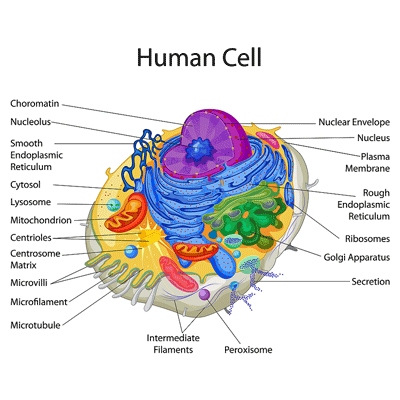
Small organelles show up in a big way. The peroxisome is the little purple circle at the bottom of the cell.
The low plasmalogen levels implicated small organelles in the cell called peroxisomes. These organelles, which have not been highlighted in ME/CFS before, are the site of plasmologen synthesis. They help maintain the cellular membranes, reduce oxidative stress, and perhaps most importantly, break down very-long-chain fatty acids to metabolic intermediates that the mitochondria can use to produce ATP. Without the peroxisomes breaking down those fatty acids, the mitochondria won’t have the fuel they need to produce ATP.
All three of these functions appear to have been damaged in ME/CFS. Oxidative stress is high, there is evidence of cellular membrane damage, and ATP production appears to be limited.
The authors concluded that a disrupted peroxisome/mitochondrial connection likely contributes to the fatigue and cognitive problems found in ME/CFS. In doing so, they referenced the work of an Australia researcher, Paul Fisher, who recently came to a similar conclusion. Fisher’s findings suggested that reduced levels of the same substrates (metabolized long-chain fatty acids) that Lipkin highlighted in his study were causing reductions in energy production as well.
Carnitines Low – Again
Many studies, including this one, have found evidence of increased oxidative stress in ME/CFS.
The Lipkin group also found that people with ME/CFS had reduced levels of carnitine – another key figure in energy production and membrane health. Carnitines play at least 3 major roles in energy production, and low carnitine levels are associated with increased oxidative stress, as well as the reduced health of the cellular and mitochondrial membranes.
The reduced carnitine levels appear to have contributed to an accumulation of long-chain triglycerides in ME/CFS patients. Because those long-chain fatty acids are ripe for free radical attack, and when attacked can emit substances that then impair mitochondrial functioning, the long-chain triglyceride finding feeds into the oxidative stress/mitochondria inhibition chain. Because the peroxisomes are supposed to step in to compensate for low carnitine levels – and apparently failed to do so in ME/CFS – they’re potentially implicated in the low carnitine levels.
The depleted plasmalogen levels, the high levels of unsaturated triglycerides, and the reduced carnitines all pointed at one logical target – damage to the little peroxisome organelles littering the cell.
The cell membranes may be taking a pounding in ME/CFS.
The Gist
- Ian Lipkin’s NIH-funded research center produced the biggest and most comprehensive metabolomics study yet in ME/CFS. The study contained more people (197) and examined more metabolites (888) than any study done to date.
- The study ended up showcasing a small organelle in our cells that we’ve heard almost nothing about until now – the peroxisome. While tiny, peroxisomes perform several crucial functions: they help maintain the cellular membranes, they reduce oxidative stress, and perhaps most importantly, they break down very-long-chain fatty acids to metabolic intermediates that the mitochondria can use to produce ATP. Without the peroxisomes breaking down those fatty acids, the mitochondria won’t have the fuel they need to produce ATP.
- The authors believe the last issue – that the peroxisomes are failing to deliver the mitochondria the resources they need to produce ATP (energy) – may be a key in ME/CFS.
- Three things implicated the peroxisomes: depleted plasmalogen levels, high levels of unsaturated triglycerides, and reduced carnitines.
- The reduced carnitine levels appear to contribute to the accumulation of long-chain triglycerides in ME/CFS patients which, in the oxidative stress-rich environment found in ME/CFS, are likely emitting substances that impair mitochondrial functioning. This is one of several studies which have found low carnitine levels in ME/CFS.
- Low levels of unsaturated phosphatidylcholines (PCs) are probably also impairing the structural integrity of the cellular membranes, and interrupting the flow of proteins across the mitochondrial membrane – thereby impairing mitochondrial functioning.
- Low choline levels may indirectly impair mitochondrial functioning and affect autonomic nervous system functioning.
- Notice that the authors found reductions in four metabolites all of which impair mitochondrial functioning (and most of which impair membrane integrity and increase oxidative stress.). Plus the study also found increased levels of 3 metabolites all of which impair mitochondrial functioning as well.
- Plus the increased levels of two intermediates in the Krebs or aerobic energy production cycle suggest something has gone awry in the complex process of producing ATP.
- The study with its interlocking findings presented perhaps the most coherent story yet of mitochondrial dysfunction in ME/CFS.
Low Choline Levels – Again
The (almost significantly) low choline levels were potentially illuminating as choline is almost completely devoted to the synthesis of PCs, which as was noted above, is needed for mitochondrial functioning.
The authors pointed out that because choline plays a role in the production of epinephrine, choline deficiency could play a role in the autonomic nervous system problems found in ME/CFS, which may produce reduced blood flows and oxygen supply and ultimately hypoxia (low oxygen levels), and ischemia (blood vessel blockage).
This is the third time low choline levels have been found in ME/CFS.
Hypometabolic State?
A number of factors that are critical to mitochondrial production appear to be low in ME/CFS – suggesting that a hypometabolic state is indeed present:
- Low plasmalogen levels inhibit the breakdown of long-chain fatty acids to compounds the mitochondria needs to produce ATP. (The low plasmalogen levels implicate the peroxisome organelles in the cell).
- Low levels of carnitine may be interfering with ATP production.
- Reduced levels of unsaturated phosphatidylcholines (PCs) may interfere with mitochondrial functioning by impairing the flow of proteins into the cell.
- Low choline levels may indirectly impact mitochondrial functioning by impairing the production of PCs.
Increased Levels of Mitochondrial Antagonists
While people with ME/CFS appear to have low levels of helpful mitochondrial compounds, they appear to have high levels of compounds that can damage the mitochondria.
- High levels of triglycerides plus high levels of oxidative stress suggest the triglycerides may be emitting substances that are impairing ATP production.
- Finding high levels of two intermediate metabolites (α-ketoglutarate (α-KG), succinate) in the electron transport chain where ATP is produced suggested that a blockage or inefficiency had occurred. Increased levels of these substances have been associated with severe metabolic impairments, inflammation, and, once again, increased oxidative stress.
Moderate Predictive Capacity
For all the interesting findings, the authors were unable to use machine-learning techniques to differentiate the ME/CFS patients from the controls. (They could, however, differentiate the female ME/CFS patients from the female healthy controls). Adding another dataset into the mix helped, but the predictive values were not as high as seen in some other studies. (The models correctly predicted 51-74% of entire ME/CFS group and 62-78% of female ME/CFS patients.)
Conclusion

The clearer pathway highlighted in this study was encouraging.
There’s just no substitute for size. Compare the 84 participants and 612 metabolites assessed in Naviaux’s 2016 study, and the 52 participants and 832 metabolites assessed in Hanson’s 2018 study, with the 197 participants and 888 metabolites assessed in the NIH-funded Lipkin study, and you can see why NIH-funded studies are the cat’s meow.
This present study’s size suggests it’s providing us the most up-to-date assessment of metabolomics to date in ME/CFS. This study’s general findings (decreased phospholipids, problems with ATP generation) did jive with those of past studies, but the specific findings were quite different.
Peroxisomal dysregulation – something that’s hardly been mentioned in ME/CFS before – took center stage in the largest and most comprehensive metabolomic study to date. The nice thing about the peroxisomal dysregulation and other findings in this study was how nicely they fit together.
The reduced plasmalogen levels pointed a finger at the peroxisomal dysregulation, which helped explain the accumulation of the long-chain fatty acids. Besides potentially causing mitochondrial problems, the low carnitine levels may, in part, be due to peroxisomal dysregulation as well. Every one of these problems also potentially increases oxidative stress and impairs membrane integrity.
Reduced levels of unsaturated phosphatidylcholines (PCs) and choline may also impair mitochondrial functioning, plus reduced choline levels could help explain the autonomic nervous system problems in ME/CFS. The fact that both low carnitine and choline levels have been found a number of times in ME/CFS suggests these findings may stick.
Finally, the increased levels of two intermediates in the Krebs or aerobic energy production cycle suggest something has gone awry in the complex process of producing ATP.
Given the size and the increased statistical strength of this study, it was surprising to see that it had a much lower predictive value than Lipkin’s past, smaller study. The fact that merging both metabolomic and metagenomic data points produced a much higher predictive value (.836) than found in this study – with its focus on metabolomic data alone – suggested that a future biomarker for ME/CFS may very well involve multiple systems.
All in all, the study with its interlocking findings presented perhaps the most coherent story yet of mitochondrial dysfunction in ME/CFS.


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!



This sure rings true.
Cory, Thanks for (as usual) another terrific blog.
Thanks. It was good to see all these links to the mitochondrial problems and oxidative stress but I particularly liked that a metabolomics study actually pointed to something really concrete – a part of the cell. Time will tell but that seemed like a step forward.
cort, what type of dr. do we see about this mitochondrial problem?
This is still a tentative finding – it needs to be validated – and I don’t what, if any treatment regimen it might call for. If I was to look for help on a mitochondrial issue, though, I would try to find a functional or alternative health MD.
EMF destroys mitochondria / demylenates nerves. The treatment is habitat free of all such toxins.
After studying hypochlorite ion as explanatory for ASEA water claims and effects … i came to see peroxisomes as intracellular clean-up locations for debris. H2O2, O2, O3, and chlorine gas need ‘bubbles’ to work. Fenofibrate was found in vitro to help CoVid-infected lung cells correct excessive lipid metabolism. It also helps protect dopamine neurons. PPAR has more to show us. “Choose your level of (intracellular) CARWASH for today!
My god thats incredible.
Its just like yes ….yes and yes.
They’ve nailed it.
Can you be more specific about foods to avoid
Very interesting. As I’ve said before I used to feel like an old car, that ran on peculiar, unobtainable fuel. I did take choline supplements for a good while and focused on eating carnitine too. Terry Wahls (Wahl’s Protocol) a long time vegetarian decided she needed to eat meat, for the carnitine. I ate beef liver, meat and eggs. I’ve spent a long time trying to find food that works for me, anti inflammatory as much as possible, as I’m intolerant to so much. For me, there’s been so many areas to pay attention to, like calming my nervous system, sleep etc. I do have more energy now but I just have to keep my life on low revs, otherwise I can still run out – especially my brain.
Tracey Anne (Burgess)
Did you find any difference on choline?
Each person different, for me felt increasing dietary choline felt worse so avoided it ( and beans (lectins?)). As if the choline couldn’t be used properly.
I identify with your phrase “unobtainable fuel.”
(Cort, sorry I missed the auto correct that wrongly changed your name to Cory. You are a one in a billion kind of person, so sorry for getting your name written wrong.)
It was hard to tell Sunie. I definitely noticed a small increase in energy from eating meat. I just put together all the things I thought might help, that I could tolerate. I also took CoQ10.
Another aspect I looked at was helping my digestive system to be able to make use of the food I ate, including proteins. So I started taking betaine hydrochloride. Then I moved onto viridian’s digestive aid, which includes some betaine hydrochloride, which I still take. As we age, our stomach acid lessens apparently and we need that to digest the proteins, I believe.
I was just trying things out that made sense from my researching diets etc., and then sticking to it, unless I thought it wasn’t helping or was making me worse. I think my diet is very nutrient dense and everything is chosen for a reason – though now I can’t remember all the reasons! Cort’s blog reminded me of the choline and carnitine.
I do take a lot of supplements but I try and find the nutrients in food, if I can, as I believe they’re absorbed by the body more easily.
Hey, Sunie, I was interested to read your comment that choline made you feel worse, as though it couldn’t be used properly. When I read, in the above article, that choline helps us to produce epinephrine, I immediately thought, “then maybe the low choline is protective, rather than causal,” because I have an acute sensitivity to epinephrine (which I discovered from a stress test as well as dental procedures). I think we can’t assume that everything that measures “low” in our bodies just needs to be bumped up!
I cannot have any Epinephrine at all for dental work . They have a alternative .
It makes my heart race and my BP OFF THE CHARTS !!!
This rings true for me. In the early days of my CFS, I tried to increase oxygenation when using energy. At some point I realized oxygen had nothing to do with my impairment. It was ATP. On days I ate meat twice a day, I felt better during exercise and recovered quicker. Lately, though, I just feel depleted whatever I eat. But it’s been over 20 years so maybe it’s just aging.
i agree that meat helps for awhile, then stops helping
Totally accurate
Could t you just add in to your dietary lifestyle choline , L carnitine , and acetyle carnitine etc etc ? To buffer ?
I would say it was a blessing from God it this was figured out and we did not have to live like this any more.
It seems to me as a consistent and very promising finding for the first time.
Cort, could this also explain our sticky blood?
Then ME is an acquired metabolic disease. It makes sense.
I hope that other scientists will want to repeat this study. Otherwise nothing will happen for years.
Agatha Andrews
That is incredible re epinephrine, was just at dentist and reminds me i cannot have epinephrine and no lidocaine in the dental ‘freezing’ anaesthetic.
I cannot have epi either. I have passed out in the dentist chair from it. Dentists use it when they use novacaine: because of the effect it has on blood flow, it make the novacaine last longer. I now get another anesthetic, a substitute, and always remind them not to give me epi.
I wonder if this is a common reaction with ME/CFS. It seems it could be (due to ANS issues), and would be helpful for people to know. 3 people above, 4 with me, have a problem with it in this thread. I’ve not seen it mentioned elsewhere.
I would think we could have more confidence in this study’s findings because it was pretty large and was done by the Lipkin group. The fact subsets probably exist in ME/CFS means we need really large and expensive studies – and the NIH is one of the few places that can fund those. Unfortunately, the NIH is still not stepping up to the plate for ME/CFS – so we have to keep working at that.
Large really well-done studies will help the field nail down findings and move forward. Otherwise it kind of drifts around or moves forward very slowly.
Hi Cort… some years ago I followed Healthrising in dealing with my CFS. I’m quite surprised that I haven’t succumbed to my ailments now at 70 years of age.
I just want to throw this out there. A few years ago I developed stomach issues. An endoscopy revealed enlarged veins in my esophagus (varices). Only one issue causes this. LIVER PROBLEMS.
MRI & ultrasound showed advanced cirrhosis (incurable fatty liver disease). I was also dx’d with gastroparesis. So, my liver is no longer breaking down toxins properly. I am dying. Due to my CFS I could never tolerate a liver transplant.
I feel this all ties into the CFS. It may be wise for others to seek medical points of view and possible testing.
I’m glad you’re still hanging in there. Blessings!
Brenda Elliott
hugs
i agree there is something to fatty liver disease being somehow tied to, or caused by, cfs/me
Hi Brenda!
There may be ways to address your health problems – check out Ray Peat!
The medical establishment and doctors tend to rob patients of hope and keep you in a state of learned helplesness.
Godspeed
Does this then provide any insight on how to treat or reverse this process? For example, what reduces or even causes the peroxisomal dysfunction?
I have no idea. It was great to see the FIsher group come to a similar conclusion and I’m going to dig into their paper and see what they say. I would hope this is leading to a treatment. RIght now, though, they’re just trying to figure out what’s going on.
On a promising note, it was good to see Dr. Kaufman check out the results of one metabolomic study and come to the conclusion that oxaloacetate might help and get good results from it. That suggests that there may be treatment possibilities that it would take a doctor to tease out that may be buried in these big metabolomic studies.
Low SPHYNGOMYELIN shouldn’t be forgotten. Without it we don’t get new neurons. Try some dietary supplementation (ovine or bovine brain is the closest available – freeze-dried ‘sphingolin’ or ”grass-fed beef brain”) and you may be surprised.
A very interesting study, indeed! For those who follow the neuroinflammation hypothesis on ME/CFS it may be of note, that peroxisomes are essential for the maintenance of brain health (for reviews, see: https://pubmed.ncbi.nlm.nih.gov/19666893/ and https://pubmed.ncbi.nlm.nih.gov/24120688/ )) Neuroglial cells in the brain contain the highest numbers of peroxisomes in the human body, and these organelles are paramount for the preservation of axonal integrity and for the formation and maintenance of myelin (btw, the myelin membranes of nerve axones are also very susceptible to oxidative stress).
As noted in the above reviews, „all diseases caused by partial or complete peroxisome dysfunction are characterized by myelin and axonal damage as well as the activation of the immune system.“
All this is interesting because recent studies on ME/CFS patients have hinted at abnormally regulated pathways in ME/CFS related to axonal guidance and/or myelination (see Hanson´s proteome study which hightlighted the Eph-Ephrin pathway (https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7931008/) but also Staines et al. which suggests possible Calcium channel abnormalities which may have bearing on myelination – https://www.scirp.org/journal/paperinformation.aspx?paperid=84678). It has also been found that ME/CFS is marked by dysfunctional connectivity within the brain and that this may be due to impaired myelination.
Plasmalogens, by the way, constitute 30% of the ethanolamine phospholipids in the brain and are very enriched in myelin (Brites 2004). Many peroxisomal disorders – in which peroxisomal metabolites are shuttled between different brain cell types – have been shown to go along with depletion of plasmalogen levels (and also with abnormal handling of long chain fatty acids).
It may therefore be a good idea to also include the cerebrospinal fluid in these studies because it appears plausible that the metabolomic imprint seen in this study may reflect CNS processes.
Dear Herbert Renz-Polster,
you seem to be interested in deeper research. If you are interested, we are hosting a discord chat server (like a forum) where we discuss experimental ME/CFS treatments that are in research pipelines and also help sourcing those custom synthesis (For example we trialed the Nano Needle Mediactions etc).
A fellow patient with interest in research and with knowledge is always welcome.
If you or other patients are interested to join, please email me at creativemindset@protonmail.com
Cheers
Mitoman (you can also PM me with this username on Phoenix Rising)
new research on this >https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8580522/
I was wondering about the nerves! I didn’t realize, though, that there was a such a close connection! Very interesting. That provides a whole other slant on this possible problem. IT”’s amazing how easily these little organelles which I’d never heard of before fit into a ME/CFS hypothesis. Thanks!
Intuitively, the nerves certainly seem twitchy, irritable, and unsettled 🙂
This is what got me thinking about plasmalogen treatment https://drgoodenowe.com/plasmalogens/. Dr. Goodenowe developed his testing and treatment for ALZ but I think there is a role for this in ME.
That’s very interesting chydorina. I literally pour extra virgin olive oil over my food and eat a small amount of mackerel everyday.
We get excellent remyelination clinically with clemastine 1.34 – 2.68 mg at bedtime. UCSF hiigh dose for MS patients wasn’t necessary. Don’t forget to have some butter in your diet.
Elastin deficiency may not show up in metabolomics since it is a low turnover structural molecule. Low Elastin and collagen stiffness (during and post-viral infection) seems a main driver for low lymphatic movement and extracellular toxicity. I was surprised that nutritional supplementation (‘grassfed beef lung’) worked.
Hi, can someone please recommend supplements that may help this new findings?
Hi Cort, thank you for the work you do to break down the main points of these studies!
It sounds like several readers have tried to change their diets and increase certain nutrients on their own with varying success.
I’m wondering if there are any studies or trials essentially doing what these folks did, trying out diets or added nutrients to help CFS patients ? The drugs available for CFS in my experience haven’t helped or have terrible side effects, with the exception of antidepressants which do help take the edge off my pain. After I read these studies I want to take the suggested supplements, but without knowing how much to change or add. (Like one reader noted, I feel much better eating more meat and did not do well on a vegetarian diet and have trouble digesting beans.)
Cort: I am concerned that the statistics of this study implies that the “conclusions” are actually not meaningful: “For all the interesting findings, the authors were unable to use machine-learning techniques to differentiate the ME/CFS patients from the controls.” Doesn’t this mean that they haven’t found anything
?
Well, I have brain damage from sleeping 23hrs a day for 8 yrs in a coma-like sleep that I litterally couldn’t wake up from.. which has made my research basically going round in circles cos I can’t retain new info, nor connect the dots like I used to. So I rely on other people’s advice to try things. If anyone here can advise me what to try, I’d be most greatful. I’m more than happy to be a guinea pig cos I’ve been rotting away in my bed for 16 yrs and I need to keep trying things or I lose hope. I cannot rely on my own research. Thanks
Hi Sonya,
It sounds like you have been dealing with many levels of CFS. Posted some comments below in response to Courts messages. I know what it is like to be tired and not get out of bed for four years. You can email me if you would like also. Sincerely, Colleen mysticarose3@aol.com
So horrible Sonya to be so unwell and to not be able to retain any information. We’re all so different. And trying new things can come at a price, if it’s not helpful. I do think that there seems to be an increase in research into post infectious onset illnesses and hopefully safe treatments will eventually start appearing. But that’s going to take time and that’s so frustrating for someone who’s been so unwell for so long. I was lucky and found some things that have helped me but I still have issues. Following Cort’s blogs is a good idea, as he picks up on studies and explains them in simpler terms. Wish I could help you. Tracey 🙂
I really feel for you. And I pray for your healing. I, too, tike probably everyone in here, have searched high and low for answers and spent ridiculous amounts of money on supplements only to get worse, not better.
Jesus liften me up from my chronic fatigue, and with time He has led me to Karen Hurd. For 30 years she has helped thousans of people heal from the impossible with the help of soluble fibre from beans and psyllium husk. No supplements. When her family was poisoned and was told to prepare their funeral, she went to the library to study the body’s detox systems, learned about the importance of soluble fibre to mop up all metabolites our body create every day. The end up in bile but 95% gets reabsorbed thus we keep getting sicker.
She explains a lot in her free podcasts on her site. And the green dragon i am about to post was psyllium husk, for an adult 1/2 tsp coarse flakes in water every 20 minutes. The baby was doomed to die in 2 weeks, but is alive and well today 30 years later. Like the rest of the family.
Please contact Karen and ask about your situation. I am sure she can help, healing with real foods. It has helped me so much and I now truly believe I will actually heal from my chronic diseases.
God bless you all. Never give up. Jesus loves us and he wants to carry us when we are to weak to walk.
https://www.karenhurd.com/the-green-dragon.html
My take is this does not mean they haven’t found anything. It means they haven’t found everything. Something remains undiscovered. This does not mean they are on the wrong track, though it also does not mean they are on the right track either. We keep having to explore all such paths until we find the answers we are looking for.
On treatments this shows nothing we can be sure of. Generally treating oxidative stress and getting a good diet and pacing is about the most you can glean from this. We need identifiable critical control points to devise treatment strategies.
One thing I can say is that all the old treatment options have been tried ad nauseum. Some help a few, some help a lot in a very few people, and most do not do much. Some also make things worse in some patients.
I understand the need to go on searching. However its usually wasted money, in a field where there is insufficient money to do the required research. I do not donate much for research but my will has my estate going entirely to research.
I was surprised by this but note that they were able to differentiate the female ME/CFS patients from the female healthy controls pretty well. They only ran into trouble when they added males into the group – so there may be something about men with regard to metabolomic studies that’s different. Since there are always many fewer men than women in studies its harder to find statistically significant results for them.
Since women make up the bulk of ME/CFS research studies the female finding was quite significant.
I absolutely love the work by Lipkin and his team. They carefully follow leads and have provided some incredible insights into this illness.
I wonder if Lipkin’s experience of long covid has also helped? The experience of severe post infectious fatigue is quite unique in a way. For anyone who has experienced this brand of suffering, it so obviously does not extend from psychology and and yet it has such a devastating impact on the mind in terms of frustration, loss of well-being, misery, uncertainty and a whole other raft of unpleasant emotions. I can’t stand the psychologising of this illness. Lipkin’s work is legitimising a biomedical causation, even if it hasn’t managed to fully unravel a complex condition in one study. It’s contributing though and I look forward to seeing where they go next. Many thanks to them.
Agreed. We’re very lucky to have a researcher of Lipkin’s stature working on ME/CFS.
It’s highly possible that this result will lead us to a better understanding of post exertional malaise. For me at least PEM feels like I’ve been poisoned by some substance that starts to build up following exercise and seems to peak about 24 hours later before slowly dissipating over a period of four days. I tried to figure out what that was years ago by getting every test I possibly could and also reading text books about inborn errors of metabolism. The only reproducible result I was able to create was high levels of VLFCA, and the only class of metabolic diseases I have found that trigger symptoms with that long of a delay are lysosome and peroxisomal disorders. The VLFCA pointed to peroxisomal dysfunction but I really couldn’t get any further than that with it. You can’t exactly go to your primary doctor and say I think people with MECFS have peroxisomal dysfunction! So yeah, Super excited to read this. Thanks for the reporting Cort.
Ha! A metabolic disorder with a delayed reaction – that is VERY interesting. The plot thickens!
I’ve thought the same thing – something must build up after exercise…I’ve always thought – put us on a bike and test the heck out of us….Just test EVERYTHING and then do it an hour later and then a day later. The answer is in there.
Cort ~ I think of that so often. It’s the exertion of daily activities and or exercise that create the delayed onset reaction in my body.
The only thing that helps me with the O.I. is a Sea Plamsa thing I drink and it helps me not get nauseas and function, better but still after 3-4 hours of being upright I’m bed-bound and if I push using adrenaline then bed-bound for 6 hours etc…
It’s just in doing errands, chores, even sitting doing emails or bills its the exertion of the brain and or body all using Mitochondria.
My guides told me its Oxidative Stress and Mito damage, but how to fix. I’ve tried so many diets and supplements, programs. It’s been 20 years now and I’m turning 55 this year so now adding aging issues so hard to tell what’s what even though I’m an active CFS/ME and look normal.
For me Pacing and the Sea Plasma and Fulvic Acids are helping me . Stuff helping my detox pathway. A company named Microbes Formula is so helpful for my case of detox issues I feel. The fulvic acid always makes me feel better.
Thank u for this amazing space you have created Cort, you are an old-soul/wise-one.
My ME presents in just the same way as yours. It always lasts exactly 5 days and has been that way for over 30 years. The ‘well’ spaces in between used to be weeks, years ago, now they are a few days. It was a comfort to hear about someone else with the same sort of presentation. I always thought that it must be significant if anyone bothered to follow it up and I’m so impressed with the research that you have done. It’s given me some hope. Thanks so much for posting and thanks Cort for your work.
Brian yes that sounds very similar to my PEM and I’m constantly trying to balance the positive effects (tauter, happier, more flowing me) with the PEM which, if it comes with my moon, is particularly unpleasant (saggier, less happy, disconnected me!).
I don’t know whether to laugh or cry that you hypothesised part of this study outcome years ago. We just keep on keeping on 🙄🥴☺️
I always skim these articles with my very limited capacity for screen reading to try and glean ‘actionable intelligence’. Is there any thing I can do with these findings to improve my condition? Does this strengthen the case for choline supplements? Creatine supplements? I can only afford to spend $ on a new supplement if drop a similarly priced supplement I’m already taking.
Hey Geoff- It’s Carnitine or L-Carnitine, not Creatine, that’s different.
That & Choline were the two supplements I gleaned from reading this.
I wish we could say that. There is some evidence that L-carnitine can help with ME/CFS and we’ve been doing a mitochondrial nutrient series. It’s all trial and error at this point. If I was adding one cheap thing it would be to add fermentable foods to my diet (but making them myself) and try and get the butyrate levels up.
Thanks Cort. You know I am a big fan of DIY fermented foods but then recently learned that they may trigger mast cell activation syndrome which is thought to be present in ME/CFS Do you know anything about how likely this is? I sure hope I don’t have to give up my home grown turnip kraut 🙂
Hi Geoff -Turnip kraut! I’m making some turnip/rutabaga/daikon/carrot kimchi right now. I am looking into this and will cover it in a blog on fermenting foods. So far so good for me. I don’t know if I have an MCAS problem, though.
Dear Cort,
would it be helpful to give phosphatidyl Choline I.V? Or other interventions to strengthen the cell membranes?
I did a course of SS-31 / Elamipretide in hopes of strengthening mitochondrial membranes and reducing oxidative stress, but a months course had no impact on my illness (I am severe bedbound)
SS-31 was one of the compounds tested in the Nanoneedle and also proposed by Dr. Prusty to counter mitochodnrial fragmentation due to EBV / HHV
Given what we’ve learned about oxidative stress and the impact it can have on the membranes it certainly makes sense to try and reduce the stress and boost membrane integrity. As to the choline, I really don’t know. I see these long biochemical pathways and then think that a supplement is a pretty blunt instrument. On the other hand, it seems like it’s worth a shot.
How do you think these findings relate to the immune findings? Could all of this be a downstream effect of immune-mediated disease? And if so, how does the immune dysfunction cause mitochondrial dysfunction? I’m hoping someone can connect the two. Anyone know of any theories?
There’s some research going into long covid and mitochondria, hopefully that sheds some light.
For sure the immune system could impact the mitochondrial. You could have an autoimmune process or inflammation that attacks them or inflammation that damages the blood vessels – keeping blood and oxygen from reaching them. I imagine there are a number of scenario’s.
Hello LB,. Cort is right about how the immune system status can trigger a mitochondrial dysfunction process. A suppressed immune system or one that is overactive can trigger a cascade of events allowing chemical proteins, misfiring of signals, and other cellular abnormalities to create havoc to our bodies causing a disruption in the normal pathway of how the autonomic nervous system functions. In some cases, it is ultimately known as autonomic failure and can be termed dysautonomia and or otherwise known as autonomic dysregulation. The disruptions it causes in sleep. Digestion, breathing, bladder control, as well as bowel, vision, vagal nerve function and the cardiac system is mind blowing. There is no doubt carnitine can be helpful. But it can cause a disturbance in how thyroid hormones can be produced or U would be taking it. Supplements such as Ubiquinol, a better form if COQ10, PQQ, NMN, resveratrol, grapeseed extract, ZMg aspartate, NAC, Folic acid and P5P50 with Vit B from PURE ENCAPSULATIONS Support mitochondrial function at different levels. Look also aTt D-RIBOSE and SamBUYTRATE. Indeed, this is beyond complex. I have lived this nightmare for 8 years. There is hope on the horizon. My email is Mysticarose3@aol.com . I am always happy to help. Sincerely, Colleen
Hi LB,
Here’s a theory that helps me understand my crashes that I picked up from what I’ve read over the years. Anything that increases sympathetic tone from overexertion, emotional stress, surgery etc to flight/fright/freeze reactions from physical/emotional traumas may trigger the immune system (in genetically susceptible individuals and/or post virus infection) to create autoantibodies that chew through cell walls, wreak havoc with oxidative stress and further damage organelles like mitochondria and peroxisomes. I pay careful attention to my sympathetic thermostat to prevent crashes and have done well with the exception of unavoidable circumstances such as physical injury/accidents, stress from bullying, racism or misogyny. Superb pacing cannot prevent these circumstances. I help myself recover with mitochondrial support supplements, antioxidants plus others.
Great plan TC 🙂
I agree TC – my immune system and nervous system are interconnected somehow. My current thinking is that the vagus nerve may be involved.
So true
Thank you all. This is all so helpful and interesting.
Hi LB,
For some reason the order of appearance may be reversed: mitochondrial/metabological defects leading to autoimmune diseases; have a look here:
https://factor.niehs.nih.gov/2021/3/papers/autoimmunity/index.htm
I would be interested to see a study of this kind carried out on a cohort of ME/CFS sufferers whose disease specifically began with an infection of Epstein-Barr virus (i.e. the disease sometimes called infectious mononucleosis or glandular fever).
Hi Tracey,
Yes, the vagus nerve is definitely involved. HRP mentioned above that axonal myelin is particularly damaged by oxidative stress. Dr. Stephen Porges’ vagus nerve research discovered that about 30% of the ventral branch of the vagus nerve is myelinated for speedier conduction of signals to brake the flight/fright response of the sympathetic system. With a weakened ventral vagus the dorsal branch (which is unmyelinated) dominates and puts us into freeze reaction with stress and also lets the sympathetic system go unchecked. Hence the vicious circle that is so difficult to break out. Oxidative stress causing stress causing more oxidative stress. I have learnt over many years to look after my vagus nerve as it helps prevent and support recovery from crashes. Also this component of my self-care is free compared to the expensive supplements I mentioned above that I take.
Someone mentioned Ray Peat … strong advocate of saturated fatty acids.
As for me it has drastically worsened my general condition, while with the use of extra virgin olive oil and Omega 3 from mackerel, organ meat, I had a slight remission on some symptoms of the disease (let’s always remember that each of us has different causes).
PUFAs are seen as the “evil” but the literature tells us that they are the only fats to be agonists of PPAR ligands (there are three, organ specific).
Haidut, a commercial user who creates products for research (Idealabs) has two products for mitochondrial membranes … but they are mostly based on saturated fat as they believe monounsaturated and PUFA are harmful.
Have you tried methylene blue?
Now that in the news an Epstein Barr infection is a ‘precursor’ to MS (giant military personnel study) the following statement found at the link given by Chydorina is very interesting.
“ Since then, research evidence has expanded to show that plasmalogens are part of the root cause of neurodegeneration that leads to Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis.”
Because this ties plasmalogins to Epstein Barr, in a round-about way.
Me too!
The is almost word for word the explanation an integrated doc gave for my sons symptoms last year. The phosphatidylcholine tastes so bad it has to be good for you. Thanks for the summary Cort.
Did the drug help your so ?
Interestingly PC tastes good to me. A functional doc I was seeing ages ago said that was really telling.
Until the research community puts their genius into discovering the cause of ME/CFS, studies that point to low levels of this or high levels of that tell us nothing. Maybe the levels are where they need to be to keep us alive.
How many people have said they felt “poisoned” after exercise? We live in an increasingly toxic environment and many of these toxins can be stored in our bodies. Exercise is one way to release these toxins into the bloodstream; so is stress; rapid weight loss; too much heat and sun exposure.
I have served for 20 years as a co-chair of the Public Interest Partners, a group sponsored by the National Institute of Environmental Health Sciences. Partners represent national health groups where the environment may play a part in the origin of the illnesses.
One of my responsibilities each month is to send out research reports from the institute on the studies that are being done.
Why is none of this research ever considered in ME/CFS studies? Its not like we have all lived in a vacuum without any toxic exposure.
Here is the CDC’s latest report on toxins found in blood and urine samples of Americans.
https://www.cdc.gov/exposurereport/index.html
All of these topics and studies can be the result of one cause active viral infections affecting the cells of the body. PCR DNA test is the only way to test for active viral infection.
When I first became ill I went to see a fancy doctor and amongs other things (new dietary regiment, supplements and orally taken Nystatin – a treatment for Candida), I was given phosphatidylcholine/PC to try (the fancy liquid BodyBio stuff). After a few days of taking PC I felt totally normal again (it was a miracle), only to fall back into sickness about a week later (never to recover). As per NHS scheduling I ended up in Professor Peter White’s M.E surgery in London a 2 years later (yes, that Professor White) and informed him of my total, but temporary, recovery after taking PC. This helped to confirm to him in 2016 that I indeed had M.E, because the initial drastically postiive reaction ot PC was an indication. Ive taken PC on and off every since because i’ve read it’s great brain food, but it has not done anything to aleviate M.E symptoms.
Hi Kaz,
Did you take the Nystatin at the same time as the PC, or was that before and not effective?
Dear Cort: I have had ME/CFS symptoms since mid 80s with increasing severity since 1998. It prob started with Bell’s Palsy (severe) on left side of face 1988 followed by same palsy on right side in 1998. Mid 80s I tested positive for EBV as I acquired significant fatigue, brain fog etc symptoms. Also, in 1996 I was diagnosed with obstructive sleep apnea and use CPAP since. Is there a source you know that lists helpful supplements so I can check? I use l-acetyl carnitine, alpha lipoic acid, extra virgin olive oil, virgin coconut oil, butter, astaxanthin, resveratrol , ester-c, omega 3s, and multivitamin. Am functioning about 40% if good day. Thank you for your work which is a God send!
I just wanted to point out to the author that an analysis performed via machine learning and artificial intelligence methods , has identified both Choline deficiency and peroxisomal dysfunction as early as 2017. Please see below (second figure) :
http://algogenomics.blogspot.com/2017/05/machine-learning-nlp-and-network.html
You can see in this graph nodes related to peroxisomes (pparalpha, ppargamma and the term peroxisomes) as well as a node called “Choline_deficiency”
Of interest : Since 2017 no one decided to use this technology systematcally to help accelerate research on MECFS.
I also very much agree with a comment I saw related to Liver function.