It seemed ever so clear. The reduction in the number of small nerves in the skin and eyes of roughly forty percent or so of the people with fibromyalgia (FM) and chronic fatigue syndrome (ME/CFS) must bring the body – as opposed to the brain – into play. How could nerve damage in the body, after all, be explained by central sensitization in the brain? Something separate must be going on in the body.
The only problem with that notion is that, thus far, researchers have been unable to track down that something. Given the small nerve “burnout” found, the immune system was immediately fingered, but immune studies, including a recent study that looked for an autoimmune cause, have been unrewarding.
Plus, the oddness of the small fiber neuropathy (SFN) occurring in FM had to be taken into account. The SFN in FM appears to result in smaller nerves as well as reduced nerve density – an unusual combination. Functionally, the nerve problems appear to be different as well. One study suggests that the nerve conduction velocity in FM slows as nerve activity increases. (That is reminiscent of a similar pattern in ME/CFS of pooping out under the stress of an increased workload.)
Plus, the SFN symptoms in FM are a bit different. The traditional stocking presentation seen in diabetes, where the symptoms first start in the feet and moves upwards, often doesn’t occur in FM. Nor is the symptom presentation quite as expected. One study suggested that the SFN in FM is not increasing pain levels but is associated with pins and needles (paresthesias) sensations, and interestingly, autonomic nervous system symptoms.
A Brain-Caused Small Fiber Neuropathy?
To our knowledge, this is the first demonstration of an entirely “top-down” pathogenic mechanism by which increased CNS excitatory tone not only increased pain behavior but also altered the density and length of peripheral nerve fibers. Harte et al. 2017
Perhaps we are ready for a different interpretation of the SFN in FM. That new interpretation first showed up in a 2017 study by Stephen Harte and Christopher Watson. Harte and Watson noted that brain imaging scans indicated that increased activity exists in the connections between the two brain regions (insula and anterior cingulate cortex (ACC)) involved in producing pain sensations. (Both of these regions have been implicated in ME/CFS as well.)
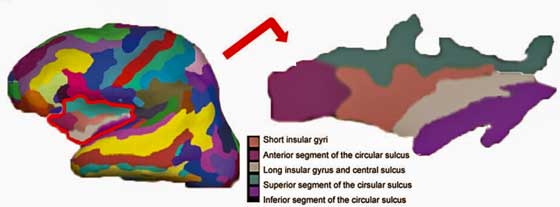
A part of the limbic system – the insula – regulates basic processes such as pain levels, the autonomic nervous system, and the emotions. Problems with the insula have been implicated in both ME/CFS and FM.
Several studies have produced a proximate cause – increased levels of the excitatory neurotransmitter, glutamate, and reduced levels of an inhibitory neurotransmitter, GABA – in the insula. Studies indicate that when Lyrica works, it works by rebalancing those levels.
Harte and Watson’s novel idea proposed a very strong brain/body connection existed indeed. They proposed that hyperactivity in the insula is producing such strong pain sensations that the body was effectively burning out the nerves in the periphery in an attempt to stop them.
Increasing the glutamate levels in the insulas of rats resulted in increased sensitivity to heat and pressure and – a reduction in the nerve fiber density in the rodents’ hind paws. The authors presented two possibilities: the peripheral nervous system “reorganization” to stop the flow of sensory stimuli to an already overheated brain; and the possibility that the alterations in the insula – a major autonomic nervous system regulator – could be impacting the small autonomic nervous system fibers found in conjunction with the sensory nerve fibers in the skin.
Six years ago Marco proposed that glutamate could be causing the problems in ME/CFS/FM – including small fiber neuropathy.
The study also offered some hope for FM patients. Despite increasing the insula’s glutamate levels high enough to produce dramatically increased pain and reduce nerve density in the rodents’ feet, no nerve damage was found in the insula. It had been thought that glutamate-induced excitotoxity could be damaging neurons in FM – a more difficult situation. This study, though, found that both the insula and the anterior cingulate cruciate were fully intact; they were simply being supercharged. Neuron repair, then, in FM might not be needed.
The Chicken or the Egg? The Brain and Pain Sensitization in the Skin
A recent Brazilian study, “Potency of descending pain modulatory system is linked with peripheral sensory dysfunction in fibromyalgia: An exploratory study“, expands on the brain-body / body-brain connection in FM. The study, which used humans this time, determined whether a factor associated with reduced pain inhibition in the brain called BDNF was associated with problems with skin sensitization as measured by NPS (numerical pain scale) and QST (quantitative sensory testing) testing. This testing was used as an analogue for small nerve fiber problems.
If increased sensitization in the skin was purely a peripheral (or body) phenomenon, it should not be related to problems in the brain. If, on the other hand, increased pain levels at the skin were associated with weaker brain inhibitory pain pathways – the two are likely related.
The Gist
Reduced small nerve fiber densities in the skin or the eyes has been found in about 45% of people with fibromyalgia patients.
The reductions in never density have been presumed due to a process occurring in the body – as opposed to the brain
An experiement to increase glutamate levels in the insula of rats suggested that idea may be wrong. Increased activity in that key pain producing area of the brain resulted in reduced nerve fiber levels in the rats feet.
The authors proposed that body may be reducing nerve fiber levels in an attempt to reduce the levels of stimuli reaching an already over-heated area of the brain.
Another study indirectly associated problems with nerve fibers in the skin with reductions in the pain producing pathways in the brain.
Another possibility is that autonomic nervous system issues are driving the small nerve fiber problems in FM.
Lastly, a just published study suggests that the small nerve issues are associated with increased pain levels and disease burden in FM.
The study measured a factor called BDNF (brain-derived-neurotrophic-factor) which has been found increased in FM. BDNF strengthens the excitatory and pain-triggering nerve pathways in the brain and weakens the inhibitory and pain reducing pathways. In fact, one study suggests this bad boy may even be able to turn pain inhibiting pathways into pain producing pathways (!).
This study confirmed our hypothesis that the peripheral sensory dysfunction is associated with the disengagement of the descending pain modularity system (DPMS) in FM. The authors
Unfortunately, the study did not directly measure small fiber density. Nor did it directly measure the strength of brain inhibitory pathways. Its findings did indirectly suggest, though, that weakened brain pain inhibitory pathways are directly associated with increased pain sensitization in the skin.
Next Steps
The evidence is pretty preliminary, but it suggests that nerve problems in the skin and altered pain pathways in the brain may be linked in FM. If this is true, four possibilities have been raised: strange signal transmissions from the small nerve fibers in the skin are causing the pain inhibitory pathways in the brain to go bananas (turn themselves off). Or, as Harte suggested, a weakened pain inhibiting system is causing the body to wreak havoc on the small nerve fibers in an attempt to protect itself. A third possibility is that both are happening.

Which comes first? The small fiber neuropathy or the brain dysfunction? Or do they both arise together? (Image-by-Noupload-from-Pixabay)
Another possibility exists – a possibility that takes in findings from Parkinson’s disease as well – that autonomic nervous system problems are driving the small nerve problems in FM and ME/CFS. That’s an interesting possibility given that autonomic nervous system symptoms were associated with reduced small nerve fiber density in FM in one study.
It’s also possible that something as yet unidentified in the skin is damaging the small nerve fibers there. Plenty of evidence – from increased levels of oxidative stress, mitochondrial problems, inflammation, pain mediators to sympathetic nervous system alterations – suggests problems exist in the skin of FM patients.
A just published study put an extra pin in the possible importance of SFN in FM and possibly ME/CFS. This large study suggests that reduced small nerve fiber density is strongly associated with increased pain levels and disease burden in FM. That casts doubt on Harte’s and Clauw’s claim that SFN is a kind of secondary factor in FM and suggests that finding out its cause may be more critical than ever.
If the brain is driving the small nerve fiber problems, then we need to find out how to cool down the hyperactive pain-producing pathways and buff up the pain-inhibiting pathways. If the small fiber problems are helping drive the problems with the pain-producing and inhibiting pathways in the brain, then we need figure how to stop whatever whatever is hammering the small nerves in the body.
Learn more about small fiber neuropathy from Health Rising.






Glad the research is ongoing. I’ve thought for years that glutamate was one of the contributing factors in HyperPOTS over sympathetic state. I found this out the hard way. When Aspartame gum, went into glutamate and caused a severe mast cell response to my heart and very pronounced POTS issues. Spent a night in ICU over a container of gum. For some reason my pathways don’t work properly and what should go down a GABA pathway, instead appears (by symptoms) to go into glutamate. Glutamate is highly excitatory and an activate a sympathetic response. Whereas GABA is supposed to create a parasympathetic response. But even if I take GABA, it goes down wrong pathway with me. I think it’s a dysfunction in Kyrenine/Quinn pathways.
But…..that being said. I have recently been working on upping my BDNF with supplements and using natural opioid type herbal. Not only do I feel like my brain function is better, but so is my pain and neuropathy. I’m really jazzed. I also have increased energy and sense of wellbeing. I’m also starting to feel my feet/toes and the pain of walking on my feet is so much better.
We (my Functional doc and I) are also working on possible viral and fungal issues. I was in a bad slump and felt like I was going down for the 3rd time. But feeling much better now.
So I think there is definitely something to this.
Issie
Herbal (opiate like) is California Poppy. Can be gotten at any health food store.
Wild Lettuce works for some but was too strong for me. (Made me feel dizzy and spacey, but helps pain.) Also at health stores.
What are the natural opioid and supplements you are using?
See above post Laura. The California Poppy seems to be milder and Wild Lettuce more potent. Both help with pain.
Sorry, meant that last comment for Cindy……but maybe will help Laura too. 🙂
Sometimes if i am having a very very bad flare a small amount of the supplement kratom, which also binds to opioid receptors ,calms the flare and takes the edge off. I cant take too much at all, just a few sips or it has a terrible reverse affect on me. This has always been a mystery. Now im starting to think of maybe a glutamate/ and or/ immune modulation of sorts, maybe a little how ldn might work for some? Trying the stablon/ tieneptine mentioned in marcos glutamate article soon… be interesting to see
Wow – the GABA pathway turns into the glutamate pathway = talk about trouble! As the article noted the same thing has been found before – a pathway switch of the worst kind – ouch!The kynurenine pathway issue possibly could relate to the Metabolic Trap hypothesis, I think.
Yeah complex and complicated! Any time my sister her son or I take any GABA or something that could turn into GABA we have a paradox reaction. So there must be some pathway issues with us and it may be genetic. And as dejurgen noted……if there is a conversion issue and/or it goes down wrong pathway ….you got a problem.
The Metabolic Trap Shines During the Symposium on the Molecular Basis of ME/CFS at Stanford – Health Rising
https://www.healthrising.org/blog/2018/10/18/the-metabolic-trap-shines-during-the-symposium-on-the-molecular-basis-of-me-cfs-at-stanford/
Here’s one of Corts articles on this.
Interesting that one of the new things I’ve been doing ups NAD. Ron Davis warns against using Tryptophan as it may create an autoimmune issue. But NAD may not and tweaks the Kynurenine pathway. (There is another article somewhere where I did alot of research on this pathway.)
Neuroinflammation II: the Kynurenine Pathway in Fibromalgia and ME/CFS – Health Rising
https://www.healthrising.org/blog/2015/06/28/neuroinflammation-ii-the-kynurenine-pathway-in-fibromalgia-and-mecfs/
The Metabolic Trap also talks of there being high serotonin. This can also be found in MCAS (mast cell). One of the new herbs I’m trying ups dopamine and may balance this out.
@Issie, @Cort
As Issie mentioned this several times in the past and I thought it needed looking into it, I digged in deeper. This is the best I’ve got so far. First the technical version:
en.wikipedia.org/wiki/Gamma-Aminobutyric_acid
“GABA transaminase enzyme catalyzes the conversion of 4-aminobutanoic acid (GABA) and 2-oxoglutarate (α-ketoglutarate) into succinic semialdehyde and glutamate. Succinic semialdehyde is then oxidized into succinic acid by succinic semialdehyde dehydrogenase and as such enters the citric acid cycle as a usable source of energy.”
Issie seems to report: when she supplements GABA, she gets less GABA increase then expected but plenty of glutamate
Look at picture of en.wikipedia.org/wiki/Citric_acid_cycle and en.wikipedia.org/wiki/File:Citric_acid_cycle_with_aconitate_2.svg
Seems her α-Ketoglutarate dehydrogenase is blocked too. Oxidative stress can do that (this one, aconitase (likely largely blocked too) and some step on the left hand side of the picture); The α-Ketoglutarate dehydrogenase is IIRC easier to block then the one on the left hand side. That would decrease conversion / usage of α-Ketoglutarate. Pilling up more α-ketoglutarate would decrease glutamate consumption.
And above en.wikipedia.org/wiki/Gamma-Aminobutyric_acid step would convert 1 times 4-aminobutanoic acid (GABA) and 2-oxoglutarate (α-ketoglutarate) into 1 times “glutamate plus Succinic semialdehyde is then oxidized into succinic acid”
α-ketoglutarate can be formed from glutamate (and IMO yield 1 NADH or NADPH as bonus with the conversion chosen by the used enzyme).
See table for glutamate in en.wikipedia.org/wiki/Anaplerotic_reactions; IMO there is a NADPH isoform too (better for immune and anti-ox).
The above paragraph would “recycle” the (by converting GABA) produced glutamate and convert much of it into α-ketoglutarate again (that was removed by converting GABA into succinate).
So converting GABA “plus stuff” into succinate “plus other stuff” by itself wouldn’t increase glutamate levels too much. BUT it would decrease the need to convert anything that can be converted into a Krebbs cycle intermediate. So with it it would decrease the need to convert “other, non GABA derived” glutamate and turn it into α-ketoglutarate.
If glutamate was already borderline problematic before supplementing GABA, decreasing the metabolism (need to shuffle it in the mitochondria for energy production) of it may push it too problematic.
Note: this doesn’t mean I speak out on supplementing with a low dose of succinic acid! Effects of that depend on how well the rest of the Krebbs cycle works.
Hey DeJurgen check out this blog on glutamate by Marco – I think you will like it!
https://www.healthrising.org/blog/2013/02/15/glutamate-one-more-piece-in-the-chronic-fatigue-syndrome-mecfs-puzzle-the-neuroinflammatory-series-pt-ii/
Thanks dejurgen. My brain is getting better, but I’m still not where I want to be yet. I appreciate you taxing yourself on my behalf. I need any help I can get to try to figure this out. I know it’s related to the whole picture.
My attempt to translate the above comment in plain speak. Cort may do better if he were up to it ;-).
(And there goes my plan to let my brains rest today… :-(; ME and overdoing it……)
“If you look at the picture in https://en.wikipedia.org/wiki/File:Citric_acid_cycle_with_aconitate_2.svg you’ll see that the Krebbs cycle works on “energy providing chemicals” like citrate, D-isocitrate, alfa-ketorglutarate, succinyl-coa, succinate… that will be broken down for energy production.
Going through these steps in a clockwise fashion can produce useful stuff like NADH, QH2 and not depicted here NADPH. Basically advancing steps in the cycle can produce energy for ATP and some amount of NADPH. NADPH is stuff needed by the immune system and for glutathione recycling.
Progression between these steps in a clockwise manner requires a different enzyme for each consecutive step. Progressing from alfa-ketoglutarate to succinyl-coa requires α-ketoglutarate dehydrogenase as an enzyme. An enzyme is a chemical that is needed to proceed the reaction, but is not consumed by the reaction. So each molecule of enzyme can help in many conversions before it “gets broken”; nothing lasts forever doesn’t it?
However strong oxidative stress can (temporarily) block this enzyme from working. Well, not like going from 100% to 0% but make it far slower in helping the reaction take place. So in plain words: strong oxidative stress makes conversion from alfa-ketoglutarate to succinyl-coa go at a really low speed, almost blocking that conversion and with it the Krebbs cycle.
As this would decrease the amounts of energy produced by the mitochondria to *very* slow rates, the body also provides some workarounds to produce another chemical like succinate down the cycle so that from there on other steps can produce energy too. It helps the body from starving for energy.
One such step is using GABA and alfa-ketoglutarate and convert it to succinate plus other stuff, without needing that enzyme that doesn’t work at reasonable rates when there is too much oxidative stress. So, supplementing GABA helps to produce energy when you have too much oxidative stress. However it also produces some extra glutamate on the side.
That last sentence is a strong simplification what I wrote in the previous message but details complicate things. So technically its not accurate, but still supplementing GABA both gives you more succinate (and with it energy) but also very likely decreases glutamate consumption by the mitochondria. If you already have plenty of glutamate, reducing metabolism of it will increase the amount of it in your body.”
And succinate gets very close to fumarate in the Krebbs cycle. More fumarate tends to increase oxidative stress. So with more succinate you’ll likely get more energy AND more oxidative stress. As the latter is bad news, it’s a mixed bag.
Issie, I found your comments about dysfunctional pathways re: glutamate and GABA so relevant to my symptoms. I have genetic variances that slow glutamate conversion to GABA, plus I have significant problems with any meds that are supposed to increase GABA. I get terrible rebound symptoms including worsening pain, anxiety, agitation, and a long list of symptoms that feel like I have too much glutamate and not enough GABA. Doctors just look blankly at me when I try to explain this. Guess I need to explore the Kyrenine pathway you mentioned to see if I can figure this out. Any medication I take that should calm down the nervous system actually just makes things a whole lot worse – meds including benzodiazepenes, sleep meds like Ambien, opioids, neurontin all cause the same type of response.
Worth exploring. I know I’m not the only one who has these issues. Simple fact that my sister and her son react same way let’s me know there must be a connection. Possible genetic involvement here.
What supplements do you find help?
Which BDNF supplements do you find most helpful?
Also found there may be a connection with calcium channels and glutamate. Using my GastroCrom (mast cell medicine that has mild calcium channel blocking properties and mast cell stablizer) along with very low dose Bentyl (muscle relaxer) and low dose Tramadol (works on all neurotransmitters and NMDA (glutamate) and is a mild calcium channel blocker) and of recent considered a mild opiate helps my autonomic response and takes an edge off my pain. I don’t take enough to block all the pain, but it will take an edge off. And the over sympathetic response calms down and I can sleep.
Reading your above 3 comments I came up with this possibility:
From a trusted source 🙂 healthrising.org/blog/2019/06/12/nimodipine-calcium-channel-blocker-fibromyalgia-chronic-fatigue-pt-ii/ :
“Mast cell activation disorder is specifically characterized by high levels of intracellular calcium and reducing this level may help improve allergic symptoms. Calcium channel blockers have also been shown to reduce the symptoms…”
ajnr.org/content/22/10/1813:
The activation of the glutamate receptor then opens the ion channel coupled to the receptor, allowing the passage of extracellular calcium (yellow) into the intracellular cytosol
So, when glutamate is increased body wide, calcium channels are more rapidly activated. When calcium channels are more rapidly activated, mast cells are more easily triggered. Mast cell dump plenty of (neuro)toxic stuff. Together with potential partial hypoxia problems at the skin due to poor blood flow and increased ROS we have a very toxic cocktail for nerves.
Combine this with what I recently wrote:
“Many ME patients have a drop in IgG values. And that links to:
Also mast cell activation is linked to it en.wikipedia.org/wiki/Immunoglobulin_G:
“IgG are also involved in the regulation of allergic reactions… …antigens can cause systemic anaphylaxis in mice through classic pathway by cross-linking IgE bound to the mast cell receptor… …IgG antibodies can prevent IgE mediated anaphylaxis by intercepting a specific antigen before it binds to mast cell–associated IgE.”
So a drop in IgG without a similar drop in IgE values can cause mast cell degranulation increasing susceptibility to all kinds of allergies, maybe chemical sensitivities and mast cell related acid reflux problems.”
If FM patients would see a drop in IgG values too, then they have increased sensitivity to mast cell degranulation problems too, with its consequences.
In this view, immunity, ROS, hypoxia and mast cell degranulation could still be important local (skin) factors.
Yes I have Hypogammaglobulinemia…..extremely low IGG. Mayo said near no immunity to pneumonia. The IGG was low on all levels checked.
Now there is still the issue of:
“Increasing the glutamate levels in the insulas of rats resulted in increased sensitivity to heat and pressure and – a reduction in the nerve fiber density in the rodents’ hind paws.”
One could argue that some of the glutamate added to the insulas increases the glutamate at the rodents paw. That should happen to a small extend, but saying this is the major thing at play here is a long stretch. So we need to be looking for something else.
Somehow the brain must signal something back to the nerves, even if these nerves are considered to report (pain) only one direction. If the brain didn’t signal something back to the end of the nerves, then their density would not have decreased in the rat experiment. It can be a feedback via the nerves or via other things like chemicals delivered through the blood.
My first idea was engineering related. A sensory nerve endpoint (at the skin) is a power source generating an electrical signal. The long nerve between skin and brain is like an electrical wire. The brain’s neurons are an electrical load.
Now how much power is transmitted from the source (skin nerves) to the load (brain neurons) is not only determined by the strength of the source but also by the “resistance” of the load. A small resistance at the load will cause more current to be drawn from the source. If glutamate in the insula modified the resistance of the brain neurons (by for example triggering faster given a certain signal) up their, then an equally large (amount of voltage) nerve signal would cause a higher current to be drawn, increasing the number of electrons to be drawn from the source. That could “drain” or even depolarize the nerves, and that could increase apoptosis or cell death of the skin nerves. That is, if the complexity of the nerves being no plain copper wire didn’t mess with this idea too much.
Another possibility would be given by science.sciencemag.org/content/361/6407/1112.full, abstract only for free so couldn’t read into it:
“The ion channels then set off a cascade of changes in calcium ion concentration that propagate through the phloem vasculature and through intercellular channels called plasmodesmata. This glutamate-based long-distance signaling is rapid: Within minutes, an undamaged leaf can respond to the fate of a distant leaf.”
It’s research for plants but it does show a potential option for “glutamate modulated” long distance communication.
Of coarse, plain old chemical messaging is still an option. However, when it comes to BDNF, the blog suggests higher levels to be pain increasing, where it contradicts Issie’s observations.
As to glutamate versus GABA: McGregor’s metabolic study didn’t show increased glutamate IIRC. but in an environment of plenty of ROS part of the Krebbs cycle is likely blocked. That should increase the Krebbs cycle turning to using plenty more of glutamate. If the body were to keep glutamate levels high enough despite consuming plenty of it, then it could chose to turn all sources of glutamate to glutamate. As GABA is a source of glutatmate, it could chose to “pillage” GABA for gluatamate production.
Normal amounts of excitatory glutamate but a lot lower anti-excitatory GABA would yield a net excitatory increase and could trigger extra mast cell degranulation on top of the IgG versus IgE thing mentioned in above comment.
I do realise how long all this research takes before it even gets to the translational stages, but oh I wish it would hurry up! I am sat here at my work desk on Lyrica, amitriptylene, codeine and paracetamol (advil) and I am in so much pain I could gnaw my desk.
Some things that initially help pain you may become “immune/non responsive” to. Also, you can have breakthrough if not timed properly and rebound pain that can be worse than initial pain.
Weigh out if the bandaid is worth the end results. We all are working with “bandaids”. But maybe some are better than others.
Very evocative Laura!
Have you tried low dose naltrexone or Cannabis? We have a Cannabis blog coming up shortly. It’s a go to medicine for one pain specialist.
If you haven’t tried LDN for pain check out this LDN resource center:
https://www.healthrising.org/treating-chronic-fatigue-syndrome/drugs/low-dose-naltrexone-ldn-fibromyalgia-chronic-fatigue-syndrom/
Amazing that Tramadol – so often used in FM and ME/CFS – has such diverse effects. Who knows what part of it is actually helping? Glutamate reduction / Calcium channel blocking? Neurotransmitters…?
Tramadol: When one looks in a place like askapatient the comments are ether, the greatest thing ever or take it off the market now.
Another thing to factor in with this is blood flow. The things I’m doing that increase brain function also helps blood flow. Calcium channel blockers slightly dilate veins and could help with that too.
With HyperPOTS and my other issues that go along with it (coagulation issues ( Factor 8, collagen binding – too thick blood, biofilm due to Lyme , CIRS and funneliformis mosseae) more dilation helps me vs vasoconstriction. Very fine line as to how much though. But thinning blood helps.
I wonder if heparin is used too little in these diseases? Here’s a blog on sticky blood and antiphospholipid syndrome – https://www.healthrising.org/blog/2018/08/15/sticky-blood-antiphospholipid-syndrome-pots-chronic-fatigue-syndrome-and-fibromyalgia/
I don’t have to use that. I’m using enzymes 3 times a day. Works great and helps other things. They are finding it’s one of best things to manage issues with biofilm. Keeps my blood thinner and clears biofilm/plaques clogging veins and hindering function of veins.
Brain-derived neurotrophic factor and its clinical implications
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4697050/
BDNF is being talked about alot in circles of autoimmune issues like MS, Parkinson’s, Alzheimers and being said to be helpful with these things. The above article also indicates this.
But the same article does say “BDNF levels not only increased NMDA levels and intracellular calcium concentrations [71] but also relieved Mg2+ block of NMDA receptors [72], promoting long-term changes to synaptic activity.”
It may be more beneficial to have higher BDNF than lower. It moderates glucose, brain function, endothelial function and many other things.
It may however up glutamate. So I’m just thinking out loud…..there has to be a balance. We don’t want to completely block NMDA and glutamate as its necessary to keep our brain function and other things running optimally. But we may have to tone it down as too high levels could possibly cause neuropathy, dementia, vein dysfunction (and therefore blood flow issues).
If the scientists are right and raising BDNF helps correct brain dysfunction (dementia), MS, Parkinson’s, Autism; seems like a more sympathetic response with our autonomic nervous system may again be a compensation and the lessor of two evils. (If, in fact, BDNF activates this response and creates more glutamate and more sympathetic activity. ) I know I don’t need to block my sympathetic response entirely (as some try to do with POTS), I find it more detrimental to me to do that. My higher NE response with standing causes more tachycardia and pumping of my heart to get needed blood and oxygen to it and my head. (Leg movement and assisting with blood flow with muscle contraction helps the heart not have to do all the work.)
IDK, the more I learn the less I realize I know. And there is still so much to learn.
Hi Issie, I missed your comment but if you read my comment I’ve mentioned that SSRI medications are known to boost BDNF causing dendritic growth of neurones. in fact boosting BDNF maybe SSRIs primary role as the serotonin hypothesis doesn’t seem to fit, especially because even though they lift serotonin up to normal levels in 6 hours from the first dose, it still takes 3 weeks to relieve depression in depressed people (not saying ME is depression)
Interesting it also takes that same 3 weeks for dendrites to grow, aided by BDNF that was boosted with an SSRI. That maybe why ME patients get a small amount of relief from some SSRIs, i.e. pain relief, anxiety relief and an ever so slight energy lift. But unfortunately still no cure of the disease.
@Brendan Rob,
I’m doing different things to boost BDNF. I didn’t find antidepressants to be of any help to me in past trials.
The latest alternative im using is NR2 (nicotinamide riboside – ups NAD) and RG3 (Red ginsing is a source). There is actually a compounded script for these things (Synapsin). But I’m compounding my own. Since I’m working on severe brain fog issues, spraying it into my nose gets it closer to the brain. I’m also using another product that has herbals in it that helps increase blood flow to the brain and helps with reperfusion issues. The one I use is called Clari-T (if you want to see ingredients). And I’m also not upping serotonin but dopamine with another herb (velvet bean). I swear……I’m getting my brain back. I have a better sense of well being and more energy.
There are other things we are doing for the pathogens and possible retrovirus. But something is helping. I’m on several new things and I’m not sure which one or the combination is key.
Issie
Yes – the body is soooo complicated. One of the papers I read noted that there is confusion about the role BDNF plays – bad or good – in acute pain. I gathered from that it’s role is pretty established in chronic pain. However, we all are different – pathways scrambled as your earlier comment noted – and who knows how things work. I think its amazing that some substances work well at very low doses and have entirely different effects at higher but still moderate doses.
I’d estimate that the following plays a nice role in both ME/FM ncbi.nlm.nih.gov/pmc/articles/PMC3956206/:
“The neuroprotective roles of BDNF in hypoxic ischemic brain injury”
Doesn’t sound too bad when having moderate increases (for most people)…
True, also true on supplements and medications we take. I keep stressing how low a dose I’m on of Tramadol. That amount is a huge help for me. It has so many properties. But, when it stops the function I need…..I go off it for a few weeks and back on at an even lower dose. Then it starts working again. Then when I’m back up to 1 and if really bad days 2 pills a day, I cycle off again. I’ve done this for years and manage my POTS pretty well – despite Mayo telling me I was the hardest subset type to treat. I don’t do well on “traditional ” things given for POTS. I found it to hurt me much more rather than help.
It may take a very small amount of something to get the body to swing into right function.
What enzymes do you use to help blood flow and thin blood?
I was interested to see that GABA is effected, as that would help explain some of the anxiety issues in both patient groups (That and poor blood flow in the brain).
Ativan (aka Lorazepam) is the very drug (benzodiazepine) that Ron Davis’s son has. It works on the GABA receptor. I have similar benefits with anotherbenzodiazepine Clonazepam.
However I’m more interested to see BDNF (Brain Derived Neurotropic Factor) mentioned. BDNF is like a growth aid to help ‘dendrites’ (bunches of neurones) grow. SSRI anti depressants that were initially thought to work on just serotonin, actually also work by somehow boosting BDNF. Which in turn encourages much needed neuronal dendrite growth in depressed people. And I presume we ME sufferers lack too due to all the brain fog.
Some of us ME patients notice a slight energy increase on an SSRI medication, it’s hardly a cure as I’m still very unwell. However the 3 major benefits of the SSRI paroxetine I take is a massive anxiety reduction and a reasonable reduction in pain sensitivity, and that small energy boost.
I don’t recommend paroxetine if you value your already limited short term memory though. However it’s done wonders for those mentioned symptoms. Unfortunately ME is so complex an SSRI is only a bandaid of slight overall relief.
Note: SSRI’s can be libido killers for approx 60% of people. When I was more healthy and able to exert more, I’d not take my normal dose on the day of ‘hanky-panky’. and a reduced dose the day before that. (Doctors call it “Weekending”)
It’s not something I worry about much anymore because I rarely have enough energy for sex due to the illness worsening. Plus it often results with a week bedridden with PEM flu like sensations.
Anyway the SSRI Citalopram is much better for cognition and memory, but slightly less effective as an anti anxiety compared to paroxetine. It’s still a reasonable pain reducer too.
Some people say Citalopram is a very good anti anxiety for them. And it is less destructive on the libido, but still can be an issue for some people. Prozac (aka fluoxetine) doesn’t reduce pain.
Also be aware that SSRI’s are hard to come off and a recent review in the UK found they take many times longer to wean off that previously recommended.
Low doses of Tricyclic antidepressants are more effective pain relievers and good for sleeping, easier to come off too. But I found them (at higher levels) no good for anxiety and an energy zapper.
Re Citalopram. It took me 18 months to come of this drug. And I continued to experience brain zaps for a further 3 moths. I did try taking paroxatine, but sadly within a few days of taking them the brain zaps came back with a vengeance
I think this points out a very good point – that many probably most of the substances we talk about – like BDNF, pro-inflammatory cytokines, even oxidants – play an important and beneficial role in the body in the right amounts. Ramp them too much, though, and they turn pathological. If high BDNF levels are strengthening the dendrites or cause more of them to grow on pain producing neurons we’ve got a real problem. On the other hand if BDNF is causing more dendrites to grown on the right neurons – then it’s all good!
It more and more seems to be all about restoring homeostasis – and that may require a more personalized medicine approach.
Agreed and we are all different and we have to find a balance. So there will be no way one pill will work for the mass lot of us. It will be us discovering where our dysfunction is and WHY. Being in tune and having awareness of what IS the problem is powerful. Then we can work on what to do about it. I have so many pieces of my puzzle and have alot figured out. But science is always advancing. There is so much more to learn.
You are also correct in there needing to be a moderation in different areas of the body. Things play a part for different functions. Upregulations and downregulations……its so complex.
Thx Cort for putting the info together as always
I’m interested in knowing if any of your readers have tried the ASEA redox signalling molecule water?
Perhaps this isn’t the spot to pose that ? but their reps claim people with ‘neurological disorders’ are experiencing complete turn-around results
I’m skeptical of course but am also leery to completely discount it if there are FM and/or ME/CFS folks who’ve had some success with it 🙂
Fine to point that out – we are certainly talking about the nervous system. 🙂
Began following a Ketogenic Diet with Intermittent Fasting 2 years ago… every symptom of my Fibro AND CFS and IBS have virtually disappeared( that and the side effect of losing weight, brain fog is gone and increased vision with added energy) … I was diagnosed in 1996… let that sink in for a moment… a few months back I had a cheat weekend… birthday cake, junk food…processed goodies, hamburger…and Soda took me 2 weeks to get pain free again that my IBS came back full force… back on track and will never have a cheat weekend again…there is something to this way of eating…
Wow…I’ve been on one for two months! I like the diet and will stay on it but I haven’t seen a turnaround like that. How long were you on it when it started working?
Thanks for passing that on and congratulations – you definitely found your subset! 🙂
Fasting has been found to also up BDNF and improve brain function and other good things. I and my hubby fast for 16 hours a day with an 8 hour eating window. I can tell it has made improvements for both of us.
Before having dinner (even when having had a modest breakfast) I often feel clearer in the head and have less trouble doing a small walk. I used to think it has to do with less energy spend to digestion and/or some increase in ketones. When being borderline close to fasting people on a non-keto diet produces some ketones too.
But sometimes this increased energy turns into a “hollow head” like when you inhale too much oxygen and I get more anxious then energized. Maybe it’s my body pushing up adrenaline in order to convert more glycogen into glucose?
It could be bdnf too. Haven’t figured this thing out yet, meaning I can’t get it working predictably.
Possibly a type of awakening. Like how autistic people do when coming more alert. Maybe your body has been so low on energy, when you have some it’s like “OMG what is this and what do I do with it”. Or it could be another mechanism that is yet to be discovered……forever in research. Aren’t we?
Its not the food, its the microbes you feed and the toxins they produce, get your microbiome tested.
I like hearing personal experiences so I’ll add my own. I have me/cfs but not fibromyalgia. I don’t experience a lot of pain with with my illness as long as I stay within my activity limits.
I had burning feet every evening for several months and have no idea why. I also had and still have occasional shooting nerve pains in arms and fingers, triggered almost undoubtedly by sugar and MCT oil.
I’m not prediabetic or anything, I’ve even checked my blood sugar just after this type of pain and it’s in the normal range.
As someone with severe neuropathic pain in the skin I have recently (on another blog) raised the work of Anne Oaklander at Harvard/ Massachusetts General Hospital wrt SFPN and syndromic disease. Has anyone ever tried to link her up with researchers looking at ME specifically? It might be useful.
The findings as presented above interest me considerably but tbh many of us needed answers ‘last decade’ and we’re running out of time. A more joined-up, concerted effort would be appreciated.
See: https://www.youtube.com/watch?v=s66LvWQ5Qso&t=33s
Thanks for mentioning her. I’ve done blogs on several of her papers. I think she’s going to be VERY important. She actually presented at the NIH ME/CFS conference and she’s been working with David Systrom – the pulmonologist. She’s probably the most important in the SFN FM field right now.
Her drug study on SFN patients suggested an autoimmune process might be responsible
https://www.healthrising.org/blog/2017/12/13/small-nerves-fibromyalgia-chronic-fatigue-ivig-autoimmunity/
Cort, check out David Andersson’s recent discovery that autoantibodies in the periphery cause the pain in FM. His presentation describing his innovative and rigorous study at IIMEC14 in May 2019 caused many attending researchers to take note among them Dr. Fluge. http://www.investinme.org/IIMEC14.shtml
Thanks David! Looking forward to that 🙂
Thanks Cort. If her interest can be maintained and improved it would be useful. I just wish we had someone of her stature in the UK who can perform and interpret a punch biopsy properly… I have tried to find out but to no avail. Oaklander’s secretary kindly linked me to the website where most of the info for public perusal is posted including a drop down menu of consultants around the world who are cognisant of her work but the solitary offering in the UK has disappeared from that list. If anyone knows of a neurologist who is up to the job I’d be very interested.
I’ll look at the other links..thanks…
Haven’t finished watching this yet……but very interesting. Thanks for positing.
Neuropathy runs in my family for 3 generations. This subject is of much interest to me.
Thanks for post, very interesting!
What if there is a source of body-wide real pain that is the trigger for all this brain-controlled sabotaging of the nervous system? This explains why the hypersensitivity to probing even though the nerves are being “shut down”. The nerves are sensing real pain, lots of it, all along. The researchers looking for the start of the vicious cycle, “in the brain”, are looking in the wrong place. What’s happening in the brain is somewhere halfway through the cascade of effects.
You are probably right Phillip. A very intelligent friend and I, are still digging into this. My friend has uncovered some very interesting info based off what we know of my genetic mutations and dysfunctions caused by them in connection to mitrocondria, the Krebs cycle and my response to certain things. I feel I’m closer to knowing WHY to several answers I’ve been trying to sort for years now. It is physical, genetic and may explain alot. I know I can’t be only one with this. As it seems to fit many. Stay tuned…….it also explains why my new protocol is pulling me out of this slump, in the last week, and I’m doing better. (Just hope it doesn’t fizzle out or backfire. )
While this research says “the brain has part in the origin of FM” it also says another thing:
mathematical approximation:
sensed pain = (real physical pain per nerve) * (amount of nerve sensors) * (some signal loss between skin nerve and brain) * (sensitivity of brain neurons at nerves ending) * (amplification or dampening of pain sensation by brain processing).
This research hence says two things:
1) feeding glutamate in mice’ insula reduces amount of nerve sensors; likely to receptors being oversensitive; this process originates in the brain
2) if the brain is actively “weeding out” 40% of skin nerves, then it reduces the total amount of pain signals by 40% or this part of the chain acts by weakening the signal rather then amplifying it.
This research actually also points to part of the brains working reducing the sensation of pain (next to the parts that likely increase it)
IMO that points to pain sensation being both: real physical pain that can be either mechanical or cellular damage or sensing too much undesired chemicals like ROS and several forms of pain modulation by the brain. The latter will likely be a mix of both amplification and dampening in most people as this research suggests. This opens the possibility that there are other dampening paths too. Most FM patients IMO seem to have a net strengthening of pain sensation due to brain amplification, but that doesn’t says the problem is in the brain:
even more simplified: pain felt = physical pain * brain modulation.
If that is for example: enormous amounts of pain = physical pain * amplification by 5 times
then total body wide “physical pain” and with it physical or biological problems is still “very high”.
Very possible – in fact I would learn towards this hypothesis. The fact that increasing glutamate in rodents brains can result in small fiber losses in their feet is very interesting though!
If you watch the video listed with Dr. Anderson talk, he said they put IGG of a person with FMS into rats and it gave them symptoms. If they put IGG of well people it didn’t. So they are thinking there is an autoimmune connection. They have not tested this out with ME/CFS, but he feels it will be the same.
So another possible issue. But with the connection to IGG and glutamate dysfunction ….there still may be a connection.
Glutamate is an amino-acid. Maybe balancing amino-acids (supplementing with particular ones while avoiding other ones) could balance glutamate levels and help ameliorate pain and/or other symptoms?
Good results so far with
-avoiding glutamate /glutamine/ BCAA’s/ threonine/ arginine
-supplementing with serine/ glycine/ methionine (as SAMe)/ tyrosine/ tryptophan (as 5HTP)/bphenylalanine. -On the fence about aspartate and lysine.
I had glycine go down wrong pathway too. And GABA you’d think would go down the GABA pathway – but doesn’t with me. So, if your cycle is working properly, I’d so for sure supplements may help. But if that cycle is all wonky…..you might create disaster.
Hi Issie, it’s about combining the aminoacids. Glycine without SAMe used to make me cranky and later on sleepy. Tyrosine without phenylalanine initially made me feel wired and backfired a day later with extra fatigue. 5HTP on its own caused nausea. Serine was the only one I could tolerate on it’s own. However combining these supplements prevented them ‘going down the wrong pathways’ and is working wonders for me. It makes sense from a biochemistry perspective.
@Moira, interesting……good to know there may be a way to tweak them. Glad you figured it out for yourself and it’s working.
Someone just commented on an old blog that we talked about glutamate. Even though two of us had a bit of debate on it, it has good info.
Genes Highlight Inflammation and Mast Cells in Fibromyalgia | Health Rising’s Chronic Fatigue Syndrome (ME/CFS) and Fibromyalgia Forums
https://www.healthrising.org/forums/threads/genes-highlight-inflammation-and-mast-cells-in-fibromyalgia.4627/
I wonder if some myelopathic induced SFN could be possible as well…
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4844812/
First time poster here, I would suggest (If this information already has not been shared or brought to anyones attention) all involved on this forum consider ruling out conditions such as Jawbone Osteonecrosis (Cavitations) as it related to the removal of Wisdom Teeth, Amalgam fillings, etc.
https://iaomt.org/
And also researching the DNA Appliance for Underdeveloped Jaws/Asymmetrical TMJ Joints, where the Underdeveloped/Crooked jaw significantly undermines the lymphatic system in the Trigeminal/Neck area.
Also, im sure everyone is already aware of “Monosodium Glutamate,” But its many, many, many derivatives, like “yeast extract,” “dextrose,” “maltodextrin.” and several more. The work of Russell Blaylock, M.D. is obviously integral in regards to these issues.
Best,
Dan
In addition, with the Underdeveloped Jaw/Asymmetrical TMJ Joints, it contributes to all kinds of systemic health issues depending on the individual. It is not solely an issue of the Lymphatic System