

This is the second in a series of blogs on the IACFS/ME 2022 International Conference on ME/CFS (and long COVID). The last one focused on David Systrom’s Keynote address. This one focused on an hour-long talk that kicked off the keynote session.
Top Immunologist Featured in Keynote Session

Akiko Iwasaki PhD jumped on long COVID early.
The IACFS/ME really scored when they got Yale immunologist Akiko Iwasaki to start off the keynote session. Iwasaki’s co-leading the Yale COVID-19 Recovery Study, as well as multiple other studies, and is developing animal models for the disease. She was recently featured in a Wired piece on brain fog and long COVID, and has given updates on long-COVID research to the American Medical Association. In short – she’s right in the middle of things – just the person to get us up to date on some exciting new research – which she proceeded to do in her hour-long talk on the “Immunology of long COVID”.
Animal Models
With the reinvigorated Simmaron Research team using an animal model to uncover some potentially fascinating aspects of ME/CFS, animal models are in the air. Given the central role they play in medical research, it’s not surprising at all to see Iwasaki quickly turn to one. Iwasaki and Michele Monje at Stanford produced an animal model to test one of four major immunological possibilities believed to be driving the central nervous system issues in long COVID.
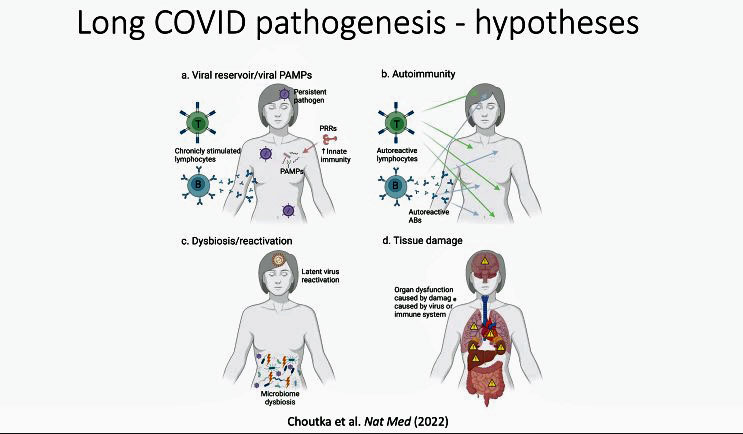
Four long-COVID hypotheses. (from Choutka 2022)
They include an infection in the central nervous system, autoimmunity, tissue damage, inflammation produced in the body by an infection or other means. They tested the last hypothesis by inserting the ACE-II receptor the virus uses to infect cells in humans into the lungs of the mice and then infecting them with the coronavirus. Having the ACE-II receptor be only present in the lungs meant there was no chance of central nervous infection.
Seven days later, an upregulation of the cytokines in the cerebral spinal fluid indicated that the inflammation in the lungs had made it to the central nervous system. Those cytokines remained elevated over the 7 weeks of the study. Interestingly, a chemokine called CCII – which has been found in another long-COVID study – was elevated as well.
Even in mice with mildly COVID-reactivated microglia – long thought to be the source of neuroinflammation in ME/CFS – were found. These findings fit with those from Iwasaki’s long-COVID autopsy study which found increased microglial activity in the white matter surrounding the nerves. The hippocampus – a part of the limbic system involved in encoding memories – was particularly hard hit with a reduction in nerve generation found and a significant loss of the oligodendrocytes that protect the nerves and support the production of the myelin that covers the nerves. A significant reduction in myelinated axons, which would surely impair communication in the brain, was also found.
The conclusion: a direct infection of the central nervous system is not needed for significant damage to occur – even with mild COVID. An infection or inflammation in the periphery (the body) is all that is needed.
The Gist
- The second IACFS/ME Conference blog covers Iwasaki’s immunology presentation, which started off the keynote session. Iwasaki is an accomplished immunologist who, with David Putrino, jumped on long COVID early.
- A mouse study found that a mild COVID infection in mice that does not reach the brain produces significant central nervous system damage including microglial activation, neuroinflammation, and damage to the myelin coverings of the nerve axons, particularly in the hippocampus region of the limbic system. Some evidence of myelin problems and neuroinflammation has been found in ME/CFS.
- She relayed the results of an unpublished study that assessed long-term long-COVID patients who had been sick for an average of 400 days.
- A large study of long-COVID patients who’d been sick for well over a year found numerous immune aberrations including high levels of monocytes, exhausted T-cells, and the presence of double-positive T-cells.
- High levels of anti-coronavirus antibodies indicated that even after all this time, the patients’ immune systems were still responding to the virus – either because it or parts of it were still present, or because an autoimmune or inflammatory response to it remained.
- The coronavirus wasn’t the only virus the immune system was responding to – high antibody levels and markers of activation suggested that herpesviruses, in particular, EBV and varicella-zoster, have become reactivated and were possibly having major effects.
- The factor that REALLY differentiated the long-COVID patients from the healthy controls, though, was low cortisol. Iwasaki called the low cortisol levels “highly predictive” and noted that they alone were enough to determine who had long COVID and who was healthy.
- Low cortisol levels were also predictive of severity: the lower the cortisol levels – the worse off the long-COVID patient was. Normal ACTH levels left the reason for the low cortisol levels a mystery. Autoantibodies that are whacking the hypothalamus/pituitary are a top hypothesis and she is looking for them now.
- The cortisol and EBV findings fit very well with ME/CFS. They also fit well with what we know of overtraining syndrome in which a plethora of endocrine abnormalities including reduced cortisol and hypometabolism have been found. Low cortisol has also been found in fibromyalgia.
- This is the second major long-COVID study to find evidence of low cortisol and EBV reactivation.
- Iwasaki is interested in doing a similar ME/CFS study but doesn’t have funding for it.
Chronic Fatigue Syndrome (ME/CFS) Connection?
We’ve heard a lot about microglial cells in ME/CFS (see Renz-Polster’s paper for a review) and fibromyalgia, but what about the oligodendrocytes that create the myelin sheaths that protect and support the long axonal sheaths that reach from one nerve to the other. Without healthy axons, the nerves can’t communicate with each other. Have problems with oligodendrocytes and reductions in white matter (axons) shown up in ME/CFS/FM?
In “The Pathobiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Case for Neuroglial Failure“, Renz-Polster and friends point out that reduced white matter volume and impaired myelination have been found in ME/CFS, but the findings have been inconsistent. Time will tell with that.
Hugely Predictive Factor Found
That was intriguing – what came next was kind of mind-blowing. When you search back through the history of ME/CFS research, you find two themes that dominated research in the early years – herpesviruses and the HPA axis: both came up big-time in this mammoth study. How right it was that it was at an ME/CFS conference that Iwasaki first shared their results.
Iwasaki noted that she can Dave Putrino have been collaborating for two years on the Mt Sinai-Yale Long-COVID study; i.e. as soon as long COVID hit – they were on it. Like Iwasaki, Putrino’s been featured in several media articles and podcasts on long COVID.
He was recently featured on Science Friday with Ira Flatow where he underlined how little the message about long COVID has gotten out: People with ME/CFS have been watching with dismay as officials like Anthony Fauci failed to warn the public in a timely manner about the dangers of long COVID.
“I think that from the outset, long COVID has been treated more like a shadow pandemic and less like an actual pandemic. People have not been giving it the level of attention it deserves as a mass disabling event in the United States. And the current sort of switch away from safe practice across the nation should be cause for alarm for everybody.”
“Long COVID is very much a continuing issue that we need to be strongly concerned about. And we still need to strongly get out the message that death is not the only serious outcome of COVID, especially non-hospitalized COVID cases– these so-called mild cases of COVID. There’s nothing mild about an acute case of COVID right now.”
He also underscored the very interesting and troubling finding that people can get through their coronavirus infection without any symptoms at all – and still come down with long COVID.
“We know for a fact that your chance of contracting long COVID is not correlated with the initial severity of your symptoms. So we have certainly seen many individuals who had an asymptomatic course of COVID-19 infection go on to develop long-COVID symptoms, which is incredibly alarming when we consider the fact that it’s hard enough right now to get insurers and other care providers to take long COVID seriously when you have a known case of COVID.”
Back to the Study
Iwasaki and Putrino (and a huge team of collaborators) appear to have thrown just about everything they could (electronic medical records, flow cytometry, antibodies, peptide metabolomics) at their long-COVID patients. They ended up measuring thousands of parameters and assessed hundreds of immune cells for each participant.

T-cell exhaustion has been found in both long COVID and ME/CFS.
These weren’t newbie long -COVID patients they were studying, either. This wasn’t one of those studies focused on patients who had had long-COVID for 1 month or 3 months. With the average time since infection averaging 410 days, long COVID had had ample time to settle in and do its work. These patients came the closest we’ve seen yet to approximating an ME/CFS patient.
Monocyte levels were up (Dr. Patterson would probably like that), dendritic type 1 cells were reduced, and activated B cells were still up.
Reduced levels of CD4 T memory cells suggested that the immune system was having trouble raising the alarm. The presence of “double positive T-cells” which function as both helper and cytotoxic T-cells suggested something strange was indeed going on. The exhausted T-cells suggested that an important part of the immune system had been overwhelmed.
Immune exhaustion has become something of a theme in ME/CFS. First came exhausted NK cells in ME/CFS. Next, there was a glimmer of evidence that the T-cells were exhausted as well. Now – as evidenced by Liisa Selin’s fascinating talk – exhausted T-cells look like they might be a pretty big thing in ME/CFS.
High levels of Anti-SARS IgG antibodies suggested that something somewhere – whether it was the virus itself, a piece of it, or an autoimmune reaction to it, was keeping the immune system activated. On that note, some cytokines (Il-2, IL-4, IL-6) were also elevated.
“Hugely Predictive Factor”

Low cortisol levels were “hugely predictive” for long COVID.
But then came the “hugely predictive” factor – reduced cortisol. Of all the factors, abnormally low cortisol levels were the factor that really differentiated the long-COVID patients from the healthy controls. Iwasaki noted that another long-COVID study found reduced low cortisol after 2-3 months, but their study – done after 18 months – found even more reduced cortisol – suggesting that cortisol levels had continued to drop.
Low cortisol, as well as high chemokines and lectins, were also the factors most associated with increased disease severity. Why the cortisol was so low – about half normal – was a mystery as the levels of its trigger, ACTH, were completely normal.
Machine-learning, an analytic process that Dr. Patterson used with his long-COVID patients, was very accurate at predicting long COVID. It indicated, again, that cortisol was the number one factor distinguishing long-COVID patients from healthy controls. That was followed by alterations in T-cells, EBV antibodies, and reactivity to EBV. Decreased cortisol alone is able to efficiently distinguish long COVID.
Chronic Fatigue Syndrome (ME/CFS) and Fibromyalgia Connection
Interest in the HPA axis – which regulates the production of cortisol – in ME/CFS dates back 30 years. While the cause of the low morning salivary cortisol was never identified, several problems in the HPA axis did pop up over time.
Several factors made the HPA axis and cortisol an obvious early choice. Cortisol’s regulation of the stress response, metabolism, the sleep-wake cycle, and in suppressing inflammation means it has the potential to affect many of the processes at play in ME/CFS and long COVID. While HPA axis work declined over time, in ME/CFS it never really stopped. One intriguing 2018 ME/CFS study linked epigenetic modifications in T-cells to an impaired response to glucocorticoid hormones, such as cortisol, in a subset of patients. Dr. Klimas’s model-generated protocol for Gull War Illness (GWI) and ME/CFS includes both an immune modulator (etanercept) and drug (mifepristone) that she hopes will reset the HPA axis.
Low cortisol has also been found in fibromyalgia as well as prolonged critical care illness – which may be a close cousin to ME/CFS. A 2022 fibromyalgia study found that low salivary cortisol was associated with cognitive issues.
Epstein-Barr Virus (EBV) Pops Up Again
Research into EBV stretches even further into the history of ME/CFS – and continues today, as well, with studies regularly popping up.
The virus ME/CFS has been associated with more than any other has already popped up in a couple of long-COVID studies. One review suggested it might even be knocking off the mitochondria. This study found high elevations of antibodies to EBV, suggesting that it had become reactivated. Iwasaki highlighted one very interesting – to her – correlation: increased EBV reactivity was correlated with a T-cell measure (which I missed). The tight connection suggested EBV reactivation might be playing a strong role in the immune abnormalities seen. Another herpesvirus – varicella-zoster – was also found.
In his Science Friday interview, Putrino echoed what Dave Patterson found – it’s not just EBV – all sorts of past viruses are getting reactivated in long COVID.
“So many people who previously had Lyme disease, but it’s been quite dormant for some time, can experience worsening of Lyme symptoms. Epstein-Barr Virus is another very common virus that people are experiencing reactivations of. And the list goes on. There are many, many previous viruses that can be reactivated by the immune sequelae of long COVID.”
Q&A
A fascinating question concerned whether Iwasaki would consider replicating the work of Goebel et al. which found that putting IgG antibodies from fibromyalgia patients into mice gave them FM. Goebel found the autoantibodies were attacking the dorsal root ganglia – which regulates the sensory signals entering the spinal cord. Iwasaki said she is finding some autoantibodies against neurons and has something similar in the works. Let’s hope she can do it and Goebel can get an ME/CFS study done.
She also agreed with Ray Perrin that the glymphatic drainage that clears toxins from the brain could be backed up in long COVID. Her group is now measuring salivary cortisol several times a day in long-COVID patients. She is hearing from a “significant number of people” who reported long-term health problems after vaccination. She wants to study these patients in the future as the immune analyses they use are incredibly sensitive. She agreed that autoantibodes to the hypothalamus or pituitary could be causing the low cortisol and that that was one of the top hypotheses they’re testing right now.
With regard to doing a similar ME/CFS study, she said she was trying to do a similar study but didn’t have funding for it. She said she was extending analyses, though, to ME/CFS with the help of Amy Proal and Michael Van Elzakker.
Conclusions
A mild COVID infection in mice that does not reach the brain produces significant central nervous system damage including microglial activation, neuroinflammation, and damage to the myelin coverings of the nerve axons, particularly in the hippocampus region of the limbic system.
A large study of long-COVID patients who’d been sick for well over a year found numerous immune aberrations including high levels of monocytes, exhausted T-cells, and the presence of double-positive T-cells.
High levels of anti-coronavirus antibodies indicated that even after all this time the patients’ immune systems were still responding to the virus – either because it, or parts of it, were still present or because an autoimmune or inflammatory response to it remained. The coronavirus wasn’t the only virus the immune system was responding to – high antibody levels and markers of activation suggested that herpesviruses, in particular, EBV and varicella-zoster, have become reactivated.
The factor that REALLY differentiated the long-COVID patients from the healthy controls, though, was low cortisol. Iwasaki called the low cortisol levels “highly predictive” and noted that they alone were enough to determine who had long COVID and who was healthy. They were also predictive of severity: the lower the cortisol levels – the worse off the long-COVID patient was. Normal ACTH levels left the reason for the low cortisol levels a mystery. I don’t think anyone thinks low cortisol levels are “it” in long COVID and ME/CFS – these are complicated multisystemic diseases – but it sure showed up big time in this study.
The cortisol and EBV findings fit very well with ME/CFS. They also fit well with what we know of overtraining syndrome in which a plethora of endocrine abnormalities (blunted and late GH (growth hormone), reduced cortisol and prolactin responses to a central stimulation test, reduced testosterone), as well as hypometabolism, have been found.
Encouragingly, they also appear to fit well with Deeks’s massively complex 2021 long-COVID study which also highlighted low cortisol and EBV reactivation\.
Iwasaki warned that more studies need to be done to confirm her findings, but it’s very good to see two such complex studies come to similar conclusions – and to have those conclusions match what we’ve seen in MECFS and fibromyalgia.
Your Contributions Keep the Information Flowing


 Health Rising’s Quickie Summer Donation Drive is On!
Health Rising’s Quickie Summer Donation Drive is On!


Is the nerve damage Iwasaki highlights reversible / treatable? Or is it more a matter of knocking out the autoantibodies in order to stop the inflammatory process from continuing to cause damage?
I don’t know but I would hope that the myelin can be restored – thus allowing the nerve signals to speed along their way better. No one has ever said that the nerve damage found thus far in ME/CFS is not reversible; in other diseases it is – but not, apparently in ME/CFS. We’ll know more as things proceed. We should see a ton of neuroimaging done in long COVID.
Thank you Cort! I like especially hat they are going to look into people who reacted bad on vaccins and booster as i did.
I saw yesterday (you can find the webinar also on facebook from them) the webinar (it is of topic here) from decodeME in the UK. 50.000 ME patients-50.000 controls. criteria: IOM or CCC. All kinds of ME (not only infectious onset), even the verry verry ill ones with help from them, in 2 years the study will be finishd. they already have one gene if i understood well in there 500 patients pre-trial. They would like other countrys did the same, investigaters are welcome, etc So far incredibly good. But then came the hammer…10 years before treatments. So that makes in totall still 12 years. I am to old and to ill to survive that. But it is finally a big study as all studys should be. If i had the energy, i could have cryd. 12 years!!! i write it here in the hope you will watch it and give your opinion or respond to what i wrote now. 12 years is impossible for me. And for many severelly affected, etc Theys said it is all about ME and it is.Not long covid or so. that is what we need, big fat ME research. please, if you want, respond. I feel so down with the 12 years… want your opinion…
Thanks for the tip! Honestly, I would throw out timelines, and let’s just see. The NIH, after all, was given over a billion dollars to spend on long COVID. Money like that can speed things up dramatically – and if studies like this keep popping up – which leave little doubt that whatever the pathogenic trigger – similar things are happening in long COVID and ME/CFS – thing could pop sooner than later in ME/CFS.
really thanks for your comforting words!!! You know, here in Belgium it is still only CFS, GET and CBT. And you can rot in hell. I have been thinking (but am so ill) how to get the decodeME study here in Belgium. Or even in the Netherlands or so. But i break my brain over it. The Uk has at least the nice guidelines. ME action, other ones. Here is nothing that i know. It must move forward faster. Way faster, globally. Also so many tests that excist in the US and not here is incredible. If you have seen it, and if you can and want, please give your critical opinion because i am so severelly ill.
I wonder what Dr Williams Jeffries would’ve said about long Covid.?? And treating Covid with safe levels of cortisol?
This is fascinating! I had EBV meningitis in 1999 at the age of 45. About a year later I was diagnosed with ME/CFS. Over the years, I’ve had multiple reactivations of EBV proven by titer. Around 2002, a doctor thought I was having adrenal problems and did a test. Cortisol did not rise “enough” but he said that it meant my adrenals were fine. In 2010 I was diagnosed with secondary adrenal insufficiency secondary to partial hypopituitary. The endocrinologist I was seeing at the time said that the test in 2002 should have been pursued as it was abnormal. Anyway, I’ve been on hydrocortisone since 2009 and on a fairly stable dose until I got Covid in January 2021. During and after I was having to take massive amounts of HC and prednisone. I never recovered and it seems like I’m getting worse every month, not better. I still have to take 5 to 10 more mg of hydrocortisone a day than I had been taking. I have had a lot of symptoms of EBV but have not been able to get tested where I live now. Anyway, I wonder if low cortisol may have been a factor in my getting ME/CFS and now, even though I’m on replacement therapy, I can’t seem to be able to take enough after COVID.
Interesting! I really wonder if anyone’s uncovered a low cortisol / EBV reactivation link…
Thank you, Cort, for this vivid summary of Michele Monje´s and Akiko Iwasaki´s work! (published in Cell: https://www.sciencedirect.com/science/article/pii/S0092867422007139).
Btw, the key chemokine they identified as marker for Long Covid was CCL11, which seems to influence microglial migration and reactivity. Interestingly, CCL11 was also identified as a key marker of abnormalities in an immune network analysis of cytokines in the cerebrospinal fluid of ME/CFS patients (Hornig et al., 2017).
There are similarities, indeed!
And yes, we definitely need a 1:1 replication of Goebel´s experiment in ME/CFS! Hope someone picks up on this!
Thanks, Herbert. I thought CC11 rang a bell. Thanks for illuminating another potential link 🙂
Sounds like Iwasaki meant by “predictive” that low cortisol is a hallmark symptom. I first thought it was about a predictive risk factor. Maybe she meant that it predicts symptom severity.
I was diagnosed with low cortisol in an annual check-up once. I figured it was related to my CFS and declined to see endocrinologist. But low dose hydrocortisone has been reported effective. (
https://www.healthrising.org/blog/2021/02/15/low-dose-hydrocortisone-fibromyalgia-chronic-fatigue-syndrome/)
As an anecdote, I know I’m less prone to PEM when I’m under some stress. In 2012 when my then fiance was having a psychiatric emergency, I was sure I’d keel over the next day after running around all day. To my surprise, I didn’t. In 2014, our flight was delayed and we had to walk fast for half a mile to catch connecting flight. Again, I didn’t succumb to PEM eventhough fast walk had been a very reliable way to trigger PEM. I’ve been thinking that’s because acute stress is dopaminergic, but it could also be because of elevated cortisol.
Thank you for commenting on this, TK. My question exactly. Is Low Cortisol a result of LONG COVID and/or MECFS or a predictor of who will get LONG COVID from COVID. Anyone have an understanding of this? Cort, what are your thoughts?
And to your comments, TK. I also have had that occur when say a death occurs in the family and mange to attend the funeral. Or when having my 2 kids. In fact, my MECFS specialist wrote a letter/rec to my OBGYNs for both and recommended I get an IV dose of Cortisol immediately following delivery. I felt pretty good after those doses and made it through recovery. They were both such whirlwinds so never thought to look back and think about that specific treatment…
Anyone else have thoughts on this?
Glad to hear. Two anecdotes are twice better than one 🙂
We’ll have to see. I don’t remember exactly who was in the study other than long COVID patients. The fact that it appeared to show up so early in the illness (in the earlier study), though, suggests that it’s an integral part of it rather than some result of chronic stress due to the illness.
We’ll see what the paper says. I took this from notes. I thought of “predictive” as they threw all these measurements – 1000’s of them into a computer program and asked it which ones most distinguished long COVD patients from healthy controls. I hope that’s the one as it seems more powerful than the other interpretation.
Interesting observation about the stress. I wonder if you’re onto something about acute stress being different. I wouldn’t be surprised.
Yeah, that’s probably what she meant: a predictor for the outcome variable in the correlation. Which is probably samething as saying the cortisol level predicts the symptom severity.
Cort, do you mind sharing the link to the study showing a low cortisol level was the predictor by Iwasaki? Thank you in advance. I may have missed the link in your article.
Hi Pei! – We don’t have the paper yet – she was sharing the results prior to publication.
I have had the same response when under stress or even illness. I always get my hopes up that I’m cured but when things settle down I always come back to ME/CFS baseline. 3 anecdotes.
Are there any natural ways, other than acute stress, to raise our cortisol?
Coffee?
Intermittent fasting?
I had some success with Sudafed. I used to take it to relieve the head pressure, but found that it also partially relieved CFS fatigue/ache and let me do a little more. I have no idea why though. It could be cortisol, dopamine, vasoconstriction, anti-inflammation… or it could be just the stimulant effect. A good thing is that a box of Walfed only cost $5, so you don’t lose much if it doesn’t work for you. It’s cheaper than Starbucks coffee.
Hi TK! I was reading info about POTS written by Peter Rowe recently and it said that Sudafed (pseudoephrine) may help some people improve POTS symptoms because it improves vasoconstriction. Here’s the link http://www.dysautonomiainternational.org/pdf/RoweOIsummary.pdf
I also happened upon it working really well for me, a sense of peace, my heart would stop beating like it was going to leap out of my chest.
I don’t think it is as simple as it constricts the vessels etc.
I am weary of using it daily or often.
It was there when I needed to feel peace, at a time when that was hard to come by.
I had read of a doctor in the… 30s? who used it for her Myastenia Gravis.
I tend towards hypertonia, but have episodes of loss of muscle tone. Just like sometimes tachycardia, sometimes bradycardia. Sometimes orthostatic hypotension, sometiems hyoertension. Etc.
Now I am wondering if it can act in some way like an antocholinergic in those who have whatever physiology in them?
Interestingly, low cortisol levels predicted a poor response to CBT!
A2009 paper suggested a rather bizarre way – drain the body of cortisol and allow the system to reset itself!
“A candidate treatment that displays robust properties in the face of significant biological variability and measurement uncertainty requires that cortisol be further suppressed for a short period until adrenocorticotropic hormone levels exceed 30% of baseline.
Treatment may then be discontinued, and the HPA axis will naturally progress to a stable attractor defined by normal hormone levels. Suppression of biologically available cortisol may be achieved through the use of binding proteins such as CBG and certain metabolizing enzymes, thus offering possible avenues for deployment in a clinical setting.”
https://pubmed.ncbi.nlm.nih.gov/19165314/
I have a question about the cortisol levels. Normal cortisol is cyclic being higher around 8:00 a.m. (400-1,000 ng/dL), mid day it dips to 120-300 ng/dL and in the evening it should be 35-100 ng/dL.
In this study was the low cortisol levels across the board or were they discordant with what should be ‘normal’?
Because I was complaining of feeling sluggish and weight gain around my mid-section, my endocrinologist gave me only a midnight salivary cortisol test. I took two tests on consecutive nights and the first was 164 which was high and the second was 26 which was low. Are these wild swings accounted for in Iwasaki’s study? What about folks who don’t follow the normal cortisol rhythms?
I could write a long article about low cortisol, but I’ve got Covid. Helpful book James L Wilson Adrenal Fatigue, and maybe William McK. Jefferies Safe uses of Cortisol. Weak adrenals can be inherited, I can follow it through 4generations. Also Rachel Yehuda wrote an article in a scientific journal about that.
Natural adrenal is easy to come by, aswagandha is good, grapefruit juice andlicorice helps the controlling enzymes.
Test is saliva, AND nights. Short synachen test is USELESS.
In my case the enzymes are malfunctioning and when I lie down my cortisol drops to the floor. My siblings have the same but not as bad.
Hi Cort. Thank you so much for your informative updates. I dont have time or energy to read the papers or watch the symposia so your summaries are an excellent way to stay informed of new findings. I am sure many in this community are grateful for this service you provide so generously.
I was surprised to read about the low cortisol. Both my kids (19yr old son and 21 yr old daughter) have Cfs/me for 10yrs and 7 years resp. Both have high cortisol. GPs explained this as likely to be caused by long-term SSRI (sertraline) use. As antidepressants are so commonly prescribed for Cfs/me I wondered if you have come across this before?
Thanks Romeny, I didn’t know of the antidepressant connection but high or low cortisol – both are clearly problems. I saw one fibromyalgia study which showed low cortisol and one which showed high cortisol levels (!).
hip hip hooray!
feels like research is getting somewhere and more pieces of puzzle are dropping in to place.
be interesting if they take whatever Ron Davis found is whacking cfs/me samples and introduce it to animal models. wonder if sensitizing scids mice to EBV first would show more significant results.
kudos Cort on keeping us in the know– it wouldn’t happen otherwise
EBV causing myelin depletion also relates to the cause of Multiple sclorosis.
Interesting. Thanks Cort.
I’ve struggled on and off with ME/CFS Fibro and crohns (all in varying times lines) for the past 20 years. I actually got a full hormone and cortisol (salvia 4 x day) panel a month before I got infected with Covid in Feb 2020. It showed basically NO cortisol and very little estrogen/androgen hormones, basically nothing that gave my body resiliency. It was affirming as to why life was extra hard. Compete HPA axis burn out.
Fast forward 2.5 years, I’m a bedbound longhauler with brain damage and all associated issues like POTS MCAS etc. It’s bad, sickest I’ve ever been. My naturopathic doctor put me on low dose hydrocortisone and I’ve been on it for well over a year now. It definitely helps the death feels and pain associated with Long Covid. I tried to reduce my dose to see and it wasn’t good. You need cortisol to fight infections, so much so that people with adrenal insufficiency have to double their dose of cortisone when sick to help their bodies ward off the illness. I’m the perfect anecdotal case to prove the science above. Not sure if it’s the chicken or the egg in terms of having low cortisol before or after infection, but from my experience it was before Covid got me and not having any means to fire up the “fight” of the virus in my body. I never produced antibodies/seroconverted either, so just a complete flop on the immune system front. I obviously came into the pandemic with a myriad of chronic health issues, I’d be interested to see what these levels would be like in the “healthy” people who became long haulers. Perhaps this is why the reactivation of EBV and herpes is being seen so much because that “fight” is getting weakened by low cortisol and viral reservoir of Covid. It’s definitely a confusing web of issues…
Either way, scientists please keep researching!!
Adrenal exhaustion can occur in stages (up to 7 if I recall correctly). I studied this for years, but it has been years since I have studied it. If my memory serves me correctly, any form of stress (including viral infections) will increase cortisol output to cope, but the adrenals cannot sustain a high output indefinitely. Generally, in response to chronic stress (such as from chronic immune activation) cortisol levels will first be high, will then go through a period of unstable output levels, and will finally collapse into exhaustion with generally low cortisol production. This may explain both high and low cortisol outputs in response to the same disease/stressor.
My personal daily cortisol rhythms are both generally low and reversed, being lowest in the morning and highest at night. My ACTH levels also do not respond adequately to exercise, so any and all components of the HPA axis may need to be studied to identify which stage of this auto-adjusting axis might be primary to the deregulation of cortisol.
Another note: I would suggest being careful with cortisol supplementation, as it will very probably reduce some symptoms in the short run, but long term use can cause adrenal atrophy and dependency, and many problems.
Last point: I had my first exposure to herpes virus about 6 months before my CFS/fibro symptoms first got severe at age 30. However, I was also working in a building with ever increasing levels of black mold. I suspect both as possible triggers.
My guess is that hormone deregulation is implicated, but that the root of the problem is much more complex. Still, I welcome research of any deregulation caused by our disease(s) – which could be instrumental to fill in the still missing pieces of the puzzle.
Thanks again for the great synopsis Cort.
Do you have the scientific references for the low cortisol and long covid please
Court, I would like to see the article linked to low cortisol as well. Thank you
As always no funding for ME/CFS )-: The cortisol and EBV theory makes me think about 20 years ago…. I had at a stressmoment high cortisol in hospital. I also had high titers of EBV then.
Cort, isn’t true that HIV patiënts also have problems with EBV and other herpesviruses when they don’t use their medicine.
Hasn’t Professor Scheibenbogen proven that ME/CFS patients have a smoldering EBV infection?
A longer comment on low cortisol (my opinion as a patient not “medical advice”):
1) Specificity to ME/CFS:
As far a I know, low cortisol and low DHEA (both are produced in the adrenal glands from pregnenolone) can also occur in other chronic disease. Low cortisol also occurs in the decendants of Holocaust survivors (probably epigenetically “inherited”). Although low cortisol may thus not be specific to ME/CFS, it may represent an important field of disregulation present in ME/CFS.
2) Model of “Phases of adrenal fatigue” in response to continuous presence of stressors, or following singular high stress event:
To any patient or researcher interested in this, I recommend taking a look at the “phases of adrenal fatigue” concept. It describes progressive imbalances in the production of adrenal hormones (Cortisol, DHEA etc.) in the presence of continuous body stressors (which can include a similar range of stressors as are known to trigger ME/CFS), or also following singular high stress events.
Adrenal glands secrete important stress hormones such as cortisol and adrenaline. They also produce many other important hormones such androgens and precursors of sexual hormones https://en.wikipedia.org/wiki/Adrenal_gland .
As far as I understand it, adrenal fatigue a disease “in its own right”, but rather an important secondary disregulation with an endpoint of gland exhaustion that results from continuous presence of stressors such as inflammation, chronic infection, food allergies etc. This will in turn affect production of other hormones.
The various phases can be characterized by characteristic combinations of high or low cortisol and DHEA levels. Phases can also be skipped going directly to a later phase of the model.
Although the concept has not yet found its way into standard medicine, in my opinion it makes a lot of sense.
Recommended reading is:
– Chapter 7 on Adrenal Fatigue in Dr Kharrazians Thyroid book, which provides an overview of 7 phases of adrenal fatigue. (Dr Kharrazian is a progressive functional medicine clinician who is well-known in the thyroid community; his thyroid book is the only one I know that actually differentiated the metabolic mechanisms behind various forms of hypothyreosis).
– This graph providing an overview of adrenal hormone conversion chains (adrenal steroidogenesis): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6857441/figure/fig0005/
To give you an impression of what this is about, I am copying here a description of the model from another website: https://drjockers.com/7-phases-adrenal-fatigue/
“Functional medicine experts such as Dr Datis Kharrazian and Dr Michael Lam have created an even more detailed way of understanding the level of adrenal fatigue with a 7 phase model. Here is how this works:
Phase 1: Initial Alarm Reaction:
This has elevated cortisol and DHEA. This is the normal acute response to stress and is often seen in hardworking individuals and athletes who are overtraining.
Phase 2: Deep Alarm Reaction:
At this point the cortisol remains high but the DHEA drops down to normal. This indicates a progressive state of the alarm reaction and the body is no longer under acute stress. The reason that DHEA drops is that it is being used to make cortisol. This is where we begin to notice symptoms.
Phase 3A: Resistance Stage:
This stage is marked by high cortisol and low DHEA as pregnenolone steal is in place. With low DHEA, it sets the stage for major hormonal imbalances in the body. This stage is typically marked by insulin resistance.
Phase 3B: Deep Resistance Stage:
In this stage cortisol is low and DHEA is low. This indicates the progression of resistance into exhaustion stage. Now the adrenals are so overworked they are losing the ability to have any control over stress.
Phase 3C: Non-Adapted Adrenal Exhaustion:
At this stage cortisol is low and DHEA is normal. This is where pregnenolone steal has failed to shift all the resources into cortisol production. There is still some level of DHEA but cortisol levels are failing. This phenomenon could be due to insulin resistance which should be evaluated.
Phase 3D: Inappropriate DHEA:
This is where Cortisol is low but DHEA is high. The individual may be supplementing with DHEA or they may have Polycystic ovary syndrome (PCOS). Insulin resistance may also be in play here.
Phase 4: Adrenal Fatigue in Full Exhaustion:
Cortisol and DHEA are both very low. In this state the adrenals have completely lost the ability to control cortisol and adapt to stress. Frequent blood sugar imbalances and crashes are common in this state.”
3) Implications for treatment:
Both Dr. Kharrazian’s thyroid book and Dr. Wilson’s book on adrenal fatigue stress the importance of finding and adressing ongoing stressors that continuously put the adrenal glands under strain.
Additionally, there is the possibility of supporting adrenal glands with supplements that either support adrenal gland function, or so-to-say relieving the glands of part of their work load and trying to correct imbalances by supplementing some of the hormone precursors (such as DHEA, pregnenolone) that the adrenal glands produce:
Most of Dr. Kharrazian’s recommendations for herbal supplements are notoriously inapplicable for self-treatment because they contain no information on dosages or product names.
DHEA, pregnenolone, or animal adrenal gland s are being used by patients to support adrenal function. Recommendations on safe dosage of DHEA are found in Chapter 8 of Dr. Kharrazians book. Dr Wilson’s book on adrenal fatigue also provides information on dosage of supplements such as DHEA, as well as general information on treating adrenal fatigue such as specific nutrition. (I remember correcting abérrant DHEA levels was mentioned as part of one of your recovery story protocols.) Recently, there appears to be also 7-keto-DHEA supplements, which seems to be DHEA metabolite that supplements only DHEA effects specifically, without being converted into sexual hormones like DHEA does.]
Notably!, hydrocortisone treatment (which would be the go-to in conventional medicine with true adrenal failure i.e. addison’s disease) is usually not recommended as it causes too much of a dampening feedback loop on overall hormone production; hormone precursors such as DHEA or pregnenolone are deemed safer in that regard.
4) Considerations for my own case, and possible implications for ME/CFS pathology: Among the only lab parameters that are “off” in my case are low DHEA and low androgens such as testosterone (which are converted from DHEA in the body). Cortisol is not always low for me with ME/CFS. Looking at the phases of adrenal exhaustion model, there are stages where cortisol is generally on the low side, but can still be high in stress situations – Actually, that is a bit what my life feels like with moderate ME/CFS bordering on severe: It seems like I have only 2 gears: Either I am “low”. Or when I push myself, I enter a state of high nervous arousal that it is actually difficult to get out of again (I assume cortisol could be high during the pushing).
Reading the Dr Wilson book on what he assumes to be adrenal fatigue, it strikes me that both triggers and treatment have many things in common with ME/CFS (such as avoiding stress, treating ongoing stressors such as ongoing infection). There is also a similarity that according to Dr. Wilson, “adrenal fatigue” can persistently exascerbate following singular stress events. However, the book also notes that aerobic exercise is one way of relieving the adrenal glands by lowering the stress reaction, which would mostly not be possible with ME/CFS.
I draw 2 conclusions from this:
1.) I think “adrenal fatigue” (i.e. low adrenal gland function/imbalances resulting from the presence of continuous or strong singular stressors) is likely to be a secondary pathology present in ME/CFS and other chronic disease. In that regard, looking at cortisol only is likely too narrow; adrenal function should be considered more comprehensively including DHEA status. In my ME/CFS case, cortisol is not continuously low, but DHEA is. Importantly, DHEA also relates to energy. The status of DHEA and cortisol are relatively easy to diagnose (in Germany it is even covered by insurance when ordered as a blood status by an endocrinologist). In my opinion it is worth a try to supplement e.g. DHEA to try and relieve one layer of fatigue in ME/CFS. I will try this and plan to report on this blog.
2.) I still believe that the “pushing part” of PEM is underresearched. PEM happens when pushing oneself beyond limits; I experience pushing to involve a high nervous system arousal that I expect to very likely involve the adrenal glands (as they secrete adrenaline and cortisol). In the same manner, I also observe stress to produce a temporary drenaline-fueled higher functionality that results in a crash later on.
– Therefore, given that the pushing-crashing process itself likely involves adrenal function, and given that adrenal fatigue as described in the Wilson book shares some similarities with ME/CFS regarding triggers and treatment, I suggest that the “phases of adrenal fatigue” model and adrenal gland function (beyond just cortisol) could be further researched within a model of ME/CFS pathology.
Typo corrected: “DHEA, pregnenolone, or animal adrenal gland extracts are being used by patients to support adrenal function.”
Thanks JR. Great information.
I can also relate to only having a couple of gears. My body felt more elastic in the first couple of decades with this illness than it does in my third.
Now I’m not particularly elastic at all. If I am subject to stress my heart and nervous system get jammed on overdrive for a number of days. Then I seem to flip to nervous exhaustion with wipeout, body wide inflammation and brain fog. Then eventually I return to a fairly comfortable but highly limited status quo.
I can definitely relate to your comment.
debsw, overdrive is a great word :-)!
Slight correction/additions to my post, after having started to re-read Dr. Wilson’s book and googled some more:
– On hydrocortisone treatment: Dr, Wilson’s book states that synthetic corticosteroids as well as natural hydrocortisone supress adrenal function, an effect that can linger for example after synthetic corticosteroid therapy. However, in more severe cases of low cortisol he does consider temporary supplementation of natural hydrocortisone as one therapy option, which can also be combined with animal adrenal extracts. (The goal of hydrocortisone supplementation would be to allow adrenal glands some time for recovery by relieving them of the need to produce it themselves).
– On adrenal gland extracts & postviral sequelae: A cornerstone of Dr. Wilson’s “adrenal fatigue” treatment approach are also pork or bovine adrenal extracts to help rebuild adrenal glands. His book also offers an interesting historical perspective of how adrenal extracts were used widely until the introduction of synthetic corticosteroids. Notably, the chapter on adrenal gland extracts mentions that during the 1918 flu epidemic, gland extracts taken by flu patients resulted in a fast recovery instead of the long post-infectious recovery period usually observed in that epidemic – to me, another clue that adrenal function might be involved in postviral fatigue/sequelae.
More resources on “adrenal fatigue” (for those interested):
– Dr. Wilson, Dr. Lam, Dr. Kharrazian are clinicians who focus on adrenal fatigue and have studied adrenal research. Both Dr. Wilson and Dr. Lam have both published patient-oriented books on the topic; and Dr. Kharrazian’s thyroid contains a chapter on it.
– 2017 presentation by Dr. Wilson discussing adrenal fatigue in relation to CFS and Fibromyalgia, and outlining treatment approach https://acainfo.org/wp-content/uploads/2017/04/Adrenal-Fatigue-Presentation-Wilson.pdf
– 2017 Dr. Wilson Webinar “Immunity and Adrenals”, description: “In this hour long webinar, Dr. Wilson discusses the basic interactions between steroid hormones and immune function.” https://www.youtube.com/watch?v=U-AWei4QRvE
-2018 Dr. Wilson Webinar discussing variations of HPA axis in various disorders including “adrenal fatigue” https://www.power2practice.com/wp-content/uploads/2018/09/hpa-axis-cortisol-depressionn-presentation-slides.pdf
-2020 presentation by Dr. Kharrazian: “https://www.facebook.com/datiskharrazianpage/videos/adrenal-fatigue-is-it-real/881852425977868/”
As far as I know (from Dr. Wilson’s book and my endocrinologist), established medicine only regards total adrenal failure (corresponding to Addison’s) as pathological, but not adrenal hypofunction/dysfunction. That does not make a lot of sense to me, as for other important glands such as the thyroid, hypofunction is well established. So, although the concept of adrenal “fatigue”/hypofunction doesn’t seem to be regarded as serious by established medicine, it seems to me something worth looking into – in particular in view of the links between adrenal function/cortisol and immune system/inflammation (e.g. this blog seems to explain them in simple terms – don’t know enough to judge its accuracy though https://marcellepick.com/how-cortisol-affects-the-immune-system/ ).
Would love to hear an opinion from Herbert Renz-Polster if there would be any links between adrenal function and glia cell reactivity?
So, the finding mentioned in Dr. Wilson’s book that during the 1918 flu epidemic, adrenal gland extracts (plus a small amount of thyroid and sex glands extract) taken by flu patients resulted in a fast recovery instead of the long post-infectious recovery period usually observed in that epidemic may imply that:
“adrenal fatigue” could not only be a kind of secondary dysregulation within postviral conditions, but that adrenals could also be involved in the initial genesis of postacute sequelae,
maybe sth. like: infection putting a greater demand on adrenal glands then they can meet – with consequences for the overall system?
The book also mentions that autopsies of 1918 flu deaths showed pathological changes of adrenals attributed to the flu infection.
(The 1918 flu epidemic publication referred to should be Lucke (1919): Lucke, B. et al.: Archives of Internal Medicine, 154, XXIV; August 1919; from sources annex of Dr. Wilson’s book).
Thank you for this interesting research update. My question is a bit off topic, but I remember hearing a while ago that scientists thought the mRNA technology might expedite treatment of fibromyalgia. Have you heard anything about this?
Cort, thanks as always for fanning the flame of hope for all of us
Thanks! And I really appreciate everyone who’s allowed me to keep digging into these subjects.
It is notable that there seems to be no memory at all that the syndrome before CFS was called “Chronic Epstein-Barr Virus Syndrome”
The CEBV Syndrome was abandoned by the CDC in 1986 after Dr Paul Cheney proved to the CDC that the “Lake Tahoe Mystery Disease” fit the parameters of the CEBV syndrome in all respects except for a total lack of EBV.
“Chronic Fatigue Syndrome” was the replacement.
btw. The rationale for replacement is written in the Holmes 1988 Chronic Fatigue Syndrome definition, should anyone care to look.
But Erik – there’s no need to dig into that group. We have far better technology and people are still getting sick. Better to bring the big technology guns to people who are getting sick now.
This is spot on with everything I’ve been through with covid. Tanked tcells, EBV reactivation, low cortisol, low dhea, high and low sugar crashes, platelets, and much more, and also other lab work off! The symptoms have been eye opening! Thank you for this research! It will change lives, and it will save lives! God Bless! -Norman, Ok
The adrenal gland itself can undergo changes, with the cortex ‘thinning’ out, and less cortisol production, aldosterone, etc.
The way docs/researches
turn to autoimmunity when
they can’t find an explanation
that fits their framework of physiology
reminds me of
when doctors turn to
” it’s a psychological problem”
when they can’t find anything ‘wrong’ in blood test
. . .
The adrenal gland can and does regenerate. My cortisol 8:00 am cortisol trended low and lower fo ryears, and now it’s raging back too high.
Now, how to get the adrenal MEDULLA to shrink… or to get the appropriate medulla/cortex proportion – that seems to take significantly longer, for some more than others.
My guess DHEA, testosterone and prgesterone trending lower too. I wonder if they are being tracked too?
Can confirm in my case (ME/CFS) low DHEA & low testosterone; progesterone within normal range though.
About your comment: “The adrenal gland itself can undergo changes”: This reminded me of Dr. Wilson’s adrenal book mentioning that these changes were discovered in rat animal experiments by the “father of stress research, Hans Selye”. Your comment made me google it and I found this interesting 2017 publication summarising Hans Selye’s work: https://www.researchgate.net/publication/317849217_Stress_is_80_Years_Old_From_Hans_Selye_Original_Paper_in_1936_to_Recent_Advances_in_GI_Ulceration
A quote from the publication is: “Autopsy of stressed rats revealed three major,
grossly visible changes: hyperemia and enlargement of the adrenals, atrophy of the thymus and lymph nodes as well as hemorrhagic gastric erosions/ulcers (the “stress triad”, and it includes a photograph of these organ changes.
This seems to be the original 1936 Hans Selye paper “A Syndrome produced by Diverse Nocuous Agents”: https://www.nature.com/articles/138032a0
How interesting that Selye’s original abstract states that the body shows the same stress response to various kinds of damaging agents, his list being reminiscent of the wide range of known triggers for ME/CFS:
“Experiments on rats show that if the organism is severely damaged by acute non-specific nocuous agents such as exposure to cold, surgical injury, production of spinal shock (transcision of the cord), excessive muscular exercise, or intoxications with sublethal doses of diverse drugs (adrenaline, atropine, morphine, formaldehyde, etc.), a typical syndrome appears, the symptoms of which are independent of the nature of the damaging agent or the pharmacological type of the drug employed, and represent rather a response to damage as such.”
You might like reading Ray Peat
I’ve been reading up a bit on case studies on adrenal hyperplasia. Cort has a post on someone who had it, and it got me thinking.
I like reading up the oldee literature, the waY they described symptoms made for a richer experience, not so reliant on blood test values exclusively.
I don’t know what to make of the use of blood test values… The whole issue with creatinine secretion being off, and varying throughout the day, depending on energy expenditure and lack of resources to meet it – makes it not the ideal candidate to normalize urine metabolites concentration against, which is what is the standard.
Plus this whole thing of water leaking out of veins into tissues/ extracellular space – how does that influence test results? I definitely have more edema in summer than winter. My blood test would look diff. In summer vs winter? Plus what does it matter if your levels are fine per the range, if you have LESS blood volume to begin with? It means your total amount of whatever was measured is lower. Etc etc.
And what does it even mean that something is in the blood?
Where is it going to?
where is it coming from?
Even the TILT test where they measure catecholamines in supine and then again in standing… if water is leaking out of blood when you come to standing, how can this then… I’m still wrapping my head around this.
Cort also has a post on someone who measured 24 hour cortisol, I’m assuming urine. They found that in ME/CFSers, it was usually low(er).
24 hour shouldn’t be standardized to creatinine. I have seen labs where they don’t in the States, or they give you both the original value and the unadjusted one.
Wher I live, they just give you the adjusted one, so I consider it useless, unless it is dramatically low/high.
There’s adrenal hyperplasia, hypertrophy and then there are changes in the relationship of cortex to medulla.
I think this is why congenital adrenal hyperplasia can look similar too, and the RXCC theory is really just the effects of prolongued stress, and how the body adapts. You don’t need genes for it.
Stress in the Selye sense of the word. Anything that requires more energy than the organism can produce/give.
M, wow, you know a lot about adrenals! What’s RXCC? Thank you for your suggestions!
Real time cortisol tracking seems to be in the works! It would be so interesting to track Cortisol throughout a push-crash-cycle!
https://www.sciencedirect.com/science/article/pii/S2666053921000126
https://www.future-science.com/doi/10.2144/fsoa-2019-0061
https://www.technologynetworks.com/proteomics/news/prototype-sensor-tracks-cortisol-hormone-in-real-time-347689
https://newsroom.ucla.edu/releases/cortisol-sensing-smartwatch
Wasn’t Ron Davis also working on a sweat sensor for something?
Cort, YOU have been a one man wonder in pushing modern medicine to take us seriously!!!! Thank you.! This edition was the first “practical” one that offered more than blind hope. It even points the way to something that WE can do. My doctor is oblivious and seems contemptuous of anything having to do with this disease, so help never seems forthcoming. Thank you for this article. It was right on!!!!!!
Our model could explain these results. The problem is not the virus, but the cells with EBV latency that cause the immune alteration. Patients who develop EBV-associated diseases were previously completely healthy. All it would take is for the patient to have one of the “ancestral” MHC-II haplotypes weak against EBV and become infected with EBV, as the gp42/MHC-II interaction influences virus entry and forms a new gp42-MHC-II complex that alters antigenic presentation to CD4 T cells. This results in an acquired immunodeficiency that allows any inflammatory stimulus in any tissue (such as other infections) to lead to the formation of EBV-infected ectopic lymphoid aggregates. This, together with the altered immunosurveillance caused by the evasion mechanisms of these EBV latent cells (increased Th2 and Treg), would lead to increased proliferation of these cells, which would increase the risk of neoplastic transformation or autoimmune disease in these tissues. Because these CD4 T cells cannot recognize and eliminate EBV latency I cells, these latency cells would proliferate and increase the other latency types (II and III) and also lytic phase cells. Latency II and III can be recognized by latent protein-specific T cells and NKs. And lytic phase cells can also be recognized by lytic protein-specific T cells. All this leads to an exhaustion of CD4 and CD8 T cells caused by chronic exposure to these viral antigens, because CD4 T cells are unable to control latency I cells. This ultimately leads to the development of an acquired immunodeficiency that results in reactivation of this and other viruses.
In the case of Long COVID two things can happen. That patients have other “weak” HLA alleles against that virus or that they are the same weak haplotypes against EBV allowing SarsCov2 infection to cause recruitment of EBV latency B cells and these lymphoid aggregates to form. As for treatments. Antivirals can help to prevent worsening of the disease by decreasing replication but they do not eliminate the latent cells that are really the problem. In the article we propose some methods to eliminate these latent cells.
Based on the hypothesis that the problem is EBV latency cells, there are few effective treatments. If you restore cortisol levels with corticosteroids you can further immunosuppress the Th1 response and therefore further proliferation of these latency cells. If you give antivirals against EBV you prevent viral replication but you do not eliminate the latent cells. If you only give antivirals against SarsCoV 2 you do not eliminate the ectopic lymphoid aggregates with EBV latency that have formed and therefore the problem will continue. And if you try to reduce the proinflammatory cytokines with some immunomodulatory therapy, the immunodeficiency caused by the gp42/MHC-II interaction is not eliminated, so the CD4 T cells will not recognize well the cells with EBV latency I. The treatment that would be useful would be a combination of antivirals and some treatment that would make the cells with latency move to the lytic phase or move to another type of latency that is recognized by the T cells. So treatment against these EBV latency cells would help eliminate all EBV-associated diseases.
Recently published article with our hypothesis: https://www.mdpi.com/2076-0817/11/8/831
In this other article based on the same hypothesis we describe how ME/CFS can develop after EBV infection: https://www.frontiersin.org/articles/10.3389/fimmu.2021.656797/full
Add that the rise of antibodies against EBV is common in all EBV-associated diseases due to evasion mechanisms of these ectopic lymphoid aggregates with EBV latency that promote specific Th2 humoral responses to viral antigens (Figure 1 of the Pathogens article).
Thanks so much for chiming in, Manuel. It was so striking to see EBV pop up so quickly in long COVID. I look forward to reading your paper.
Thanks to you for making these studies available to ME/CFS patients. We are currently conducting the study in patients to see if the hypothesis is fulfilled. Let’s hope for good results.
Best regards,
Manuel.