
The Ramsay Awards provide pilot grants for ME/CFS and long-COVID researchers.
The Ramsay Awards provide pilot grants that give researchers the ability to gather the data they need to apply for much larger NIH grants. As it has over the past couple of years, the Solve ME Initiative places a heavy focus on funding clinical trials that could quickly help people with ME/CFS.
I asked Solve M.E. if they were seeing increased interest in the Ramsay Awards since long COVID appeared on the scene. Overall, they were. The fifteen applications this year were less than they received last year but more than in the pre-pandemic period and the quality of the applications – as measured by the grant reviewer scores – is higher than before, as well.
I asked how two of the five grants ended up being for long COVID? They replied that they asked for proposals that could advance chronic fatigue syndrome (ME/CFS) and for long-COVID research that could be applied to ME/CFS. The studies that received the best scores were given funding. They asserted that:
“By being able to study individuals who were infected with SARS-CoV-2 at an early stage of disease, we will inevitably strengthen our understanding of the pathways involved in those with ME/CFS and vice versa.”
The Poop-Plus Study
Immunosenescence and Premature Aging of the Immune System in ME/CFS

Katherine Seton has ME/CFS and did her Ph.D. on ME/CFS
I don’t know if any clinical trials are more needed than fecal microbiota transplant (FMT) trials that have the potential to safely reset ME/CFS patients’ microbiome, thus reducing leaky gut and inflammation, and possibly even improving one’s ability to exercise.
If I read it right, Katherine Seton’s Ramsay award is piggybacking on the 80-person Norwegian Comeback fecal transplant trial. The study will determine what effect the fecal transplants are having, biologically, as they assess immune functioning and premature immune system aging (immunosenescence). That makes sense given the findings of T-cell and NK cell exhaustion. I’m hoping the fact that Dr. Seton is doing this study means that the Comeback team feels the fecal transplants are having a positive effect.
Dr. Seton has quite a story of her own. A person with ME/CFS, her Ph.D. was on ME/CFS (“Investigating immune reactivity to the intestinal microbiome in ME/CFS”). She went above and beyond most studies by choosing to enroll people with severe ME/CFS in her study.
The Gist
- Solve M.E.’s Ramsay Grant Awards provide funding to researchers to produce the data they need for much bigger grants to focus, once again, on new treatment possibilities.
- Katherine Seton’s award will allow her to determine the biological effects – with a special focus on immune functioning – fecal transplants are having on people with ME/CFS.
- The stellate ganglion block award will determine if temporarily blocking a key throughway for sympathetic nervous system signals will reset the autonomic nervous system, thus reducing fight/flight, improving blood flows, reducing pain, and perhaps even improving mitochondrial functioning in ME/CFS.
- The enhanced external counterpulsation (EECP) award will determine if a device that can be used at home will increase blood flows and reduce symptoms in long COVID.
- Rob Wust will compare muscle biopsies before and after exercise to investigate how exercise produces PEM, including the role microclots may play.
- Avik Roy of Simmaron will suppress an autophagy gene in mice to see if doing so will produce PEM in them. If successful, that will buttress a potential link between impaired autophagy, inhibited mitochondrial activity, reduced oxygen consumption, and potentially open new treatment possibilities.
(According to the timeline, the Norwegian fecal transplant study is not expected to end until 2026 (!). That’s when, if the timeline is correct, the last participant is expected to get a fecal transplant. Add data analysis, writeup, and publication, and we’re probably looking at a 9-year trial and a 2028 publication. (It started in 2019). Let’s hope that the timeline is not correct.)
(Producing quick, rigorously produced clinical trials are one place the RECOVER Initiative (for long COVID), with its massive infrastructure, funding, and ready patient populations, should shine. I wouldn’t be surprised to see a fecal transplant trial in the first round of RECOVER clinical trials.)
Autonomic Nervous System Reset?
The Effect of Stellate Ganglion Block on ME/CFS Symptoms and Plasma Metabolites
Talk about another timely study. With all the focus placed on the autonomic nervous system and, in particular, the sympathetic nervous system, it seems surprising that the stellate ganglion block, (SGB) technique – which has been used for years in other diseases and appears to be quite safe – somehow didn’t get on the ME/CFS communities’ radar screen until the last year or so.
The procedure seems relatively simple: inject anesthesia near the stellate ganglia to block the sympathetic nervous system signals emanating from the brainstem and reset the fight/flight system to improve immune functioning, blood flows, orthostatic intolerance (ability to stand), gut functioning – even possibly mitochondrial functioning. That’s quite a package!

Dr. Duricka is a neuroscientist with a broad background in academic and industry research.
SGB is another potential gift to ME/CFS from long COVID. It popped up on our radar screens after a case series of its effects on long COVID was published in a scientific journal. The recoveries cited were pretty dramatic, putting SGB high on the list of potential treatments to try.
The study will assess the effects of SGB on orthostatic tolerance, cognitive functioning, symptoms, and plasma metabolites in ME/CFS.
The Squeezer: Improving Blood Flows
Enhanced External Counterpulsation (EECP) to Treat Long Covid Fatigue – A Randomized Controlled Trial
Who is not for increasing blood flows? Given the reduced blood flows in the brain, the low blood volume, the blood pooling in the legs, and the possible problems with the microcirculation, it would be surprising, indeed, if no problems with blood flows existed in ME/CFS and long COVID.
Enter enhanced external counter-pulsation (EECP) – a non-invasive outpatient therapy (you can do it in your home) that has been shown to “improve the functional status and quality of life in some people with impaired blood flows and other blood vessel problems.”
Again, it seems surprising that EECP has not been assessed in either ME/CFS or postural orthostatic tachycardia syndrome (POTS). It may be because it’s often used in people with heart problems that don’t exist in ME/CFS. EECP, though, increases blood flows back to your heart, and a reduction in blood flows to the heart (preload failure) is present in a significant subset of ME/CFS patients.
Several case reports have suggested that EECP may help people with long COVID-induced POTS. One person’s brain fog lifted, her shortness of breath improved, and she ultimately returned to health. Another person showed moderate improvements.
If positive, this study could open up new treatment possibilities for both ME/CFS and POTS. This study, though, is focused on long COVID. It will assess functional status, markers of blood vessel health, and quality of life.
A Deep Dive into Post-Exertional Malaise
Skeletal Muscle Pain and Post-Exertional Malaise in Patients with Long Covid: from Pathophysiology to Treatment
“we will provide a better fundamental understanding of the biological aspects of skeletal muscle adaptations during PEM”
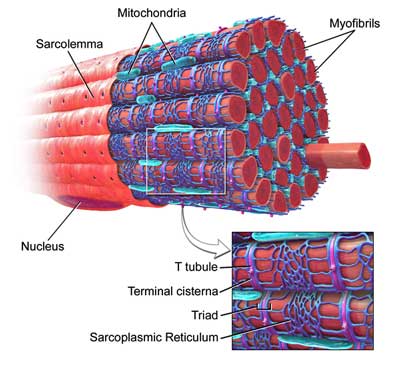
Skeletal muscles – it took a muscle biopsy to uncover carnitine fatty-acid metabolism problems in one study. Carnitine levels in the plasma did not.
This study is beyond overdue. Studies that assess muscle biopsies before and after exercise are pretty rare – if they exist at all. Add in a look at the role microclots may be playing and you’ve got a potentially quite impactful study.
As with blood flows, it would be crazy to think the muscles are not affected in ME/CFS. One review, “Old muscle in young body”, suggested the muscles in ME/CFS were aging more rapidly than normal. The mitochondrial problems revealed by muscle biopsies enabled Systrom’s $8 million mitochondrial trial to take place. Studies suggest that slow-twitch muscle fibers may be being exchanged for fast-twitch ones in both long COVID and ME/CFS. Plus, fibromyalgia muscle studies regularly show problems.
It’s good to see this researcher, in particular, get involved in long COVID (and hopefully ME/CFS). A Dutch researcher, Rob Wust, has a long track record and has already found muscle fibre atrophy, metabolic alterations, and immune cell infiltration in the muscles of people with long COVID.
Having recently co-authored a paper on “Disorders of mitochondrial long-chain fatty acid oxidation and the carnitine shuttle“, he also appears to be an expert in a mitochondrial problem (long chain fatty acids and carnitine) that’s captured a lot of attention in ME/CFS lately.
Even in the abstract of his long-COVID muscle fiber study – where space is limited and authors must choose their words wisely – he included ME/CFS in stating: “a deeper molecular understanding of skeletal muscle adaptations in patients with long-covid, and ultimately also patients with ME/CFS.”
A Different Mitochondrial Issue in ME/CFS Emerges
Targeting Atg13 in ME/CFS

With Avik Roy Ph.D. , a neuroscientist, and Gunnar Gottschalk, Ph.D. have reinvigorated Simmaron’s research efforts.
The last study from the Simmaron Research Foundation’s Avik Roy Ph.D. is pure ME/CFS and demonstrates how, in some cases, ME/CFS researchers have sprinted ahead of the long-COVID field. It’s the first to assess something called autophagy in both fields. Plus, it’s attempting to create an ME/CFS animal model.
Autophagy doesn’t sound that important – it simply clears out unused proteins and old and damaged mitochondria from the cells, but impaired autophagy results in reduced oxygen consumption, mitochondrial activity, and even problems with exercise. It can also turn cells into pro-inflammatory generators, and lead to clumps of proteins that can damage all sorts of processes.
SImmaron’s work has found indications of reduced autophagy in ME/CFS. This Ramsay study will attempt to duplicate the PEM we see in ME/CFS by altering a specific autophagy gene in mice. A successful result will strengthen the potential link between autophagy, mitochondrial problems, and PEM in ME/CFS and lead to further studies. It would also provide different treatment possibilities than we’ve seen thus far. It’s another example of the Ramsay Awards funding a new and much-needed look at ME/CFS.
As an added bonus, the awards support the work of the home-grown ME/CFS research team at Simmaron that emerged after Gunnar Gottschalk received his Ph.D. and Avik Roy Ph.D. joined the team. Seeing this group engage in novel approaches to ME/CFS is not surprising. The studies principal investigator, Avik Roy, won the Rapid Response Innovation award in Parkinson’s disease from the MJFox foundation, the John E Trufant award for excellence in research by Rush University, and the YIEE award by the American society for neurochemistry. He’s also received multiple patents for his novel nasal drug (KeptideTM) for COVID-19.
Conclusion
Particularly in an underfunded field like chronic fatigue syndrome (ME/CFS) seed grants like the Ramsay Awards play an important role by paving the way for big studies. Thus far, the Ramsay Awards have brought in over $8 million to the ME/CFS research field.






Thanks again Cort 🙂 I’m just coming out of a PEM relapse. Thankfully it was short. Do you think any of these grantees might help discover what a relapse actually is? How does EXERTION trigger such intense miserable fatigue and brain inflammation. What is the mechanism?
It certainly would be good to understand why! In my experience that includes exertion of any kind: mental, emotional, physical—. All of it devours what little energy there is to use, and then— we all know the result: days or weeks or even months required for pain and exhaustion to subside. Until we decide to do something again. Boy, would I like to understand this better! I’ve been dealing with this now for 35 years and I still don’t have it figured out. Just: if you exert energy, there’s a painful, miserable price you pay. Rather than do nothing at all, I do sometimes choose to do something in a restrained way, knowing there will be a penalty, but deciding this particular activity is worth it. I also end up canceling lots of appointments because I simply can’t do them, but I’m always hopeful.
Cort, thank you so much for keeping us current with what’s happening in research.
I can’t speak for others, but for me Curcumin has been very helpful for me for PEM. Which I think is suggestive that inflammation plays a key role in PEM. But I’m only one person so nothing scientific to back that up.
how much do you take and how often a day if i may ask? the one with black pepper, is that not hard on the stomachh? i have allready stomach issues.
Curcumin is a potent autophagy inducer.
https://web.archive.org/web/20171019063736id_/http://www.tandfonline.com/doi/pdf/10.4161/auto.19471
I don’t think anyone knows but we have some clues. Studies suggest that muscle repair mechanisms do not kick in ME/CFS, that exertion can trigger leaky gut and inflammation, that the sympathetic nervous system may have trouble shutting down, and probably many other things but no one knows exactly what is going on – not yet. It’ll be interesting to find out what Nath found our in his intramural study given that it employed a 2-day exercise stresssor.
I suspect that PEM ( the type that lasts 24 to 48 hours and sometimes builds up like a snowball if don’t recover from previous one ) could be caused by the development of these ‘microclots’ or other such temporary circulation problems, which would make it hard to consistently find in a study unless it takes into account that it only happens during these PEM phases, but then how do we explain that our physical stamina is low though even when we are at our best ?
Thanks for all the updates! I did try a stellate ganglion block. It did nothing for me. However the data for PTSD shows it pretty effective.
Looked up EECP …. Yikes!
What are the side effects of EECP treatment?
Fast or pounding heartbeats, fluttering in your chest. Kidney problems: little or no urination, painful urination, swelling in your feet or ankles, feeling tired or short of breath.
I had the C4/C6 nerve block (both sides) that is used in treating PTSD. I don’t believe it had any overall amazing results either. I’m a moderate case though, not severe.
FMT on the other hand, I bang on about how genuine the results from that are. But there are a lot of considerations. I’ve commented about that here:
https://www.healthrising.org/blog/2021/11/10/bacteria-fatigue-chronic-fatigue-syndrome/
I had very severe ME/CFS in the 1980’s and have recovered only a percentage of my health. Recently I have been slowly restricting my eating to eight hours a day with sixteen hours no food. I also eat early in the evening so I have a long period of fasting before bed. I have found that has helped my energy levels and my brain fog has lessened. It may sound difficult but I would recommend it. Just start slowly increasing the fasting time so your body can adjust. That allows autophagy to happen.
Thanks Esther – I like the idea of slowing increasing the fasting time. I can’t handle long times of going without food now.
I’m still wondering about the impaired autophagy idea. Would an impaired autophagy process result in lower than normal levels of protein in the blood? My Comprehensive Metabolic Panel (CMP) always shows a lower-than-normal Total Protein level, in spite of eating a high-protein diet. Also, is this impaired autophagy process somehow implicated in the ability of the herpes family viruses to hang around in the cells forever and reactivate whenever they feel like it? And what are the implications for the use of mRNA technology for vaccines? After every one of my covid vaccines, I experienced reactivated herpes and/or shingles (both of which had been inactive for years), plus weeks of fatigue. Maybe normal people are able to clean the vaccine particles out of their cells, while those of us with CFS are not so easily able to do that? I had my third vaccine (booster) 6 months ago, and my arm is still slightly sore. Over the past year, I’ve experienced noticeable improvement in my energy level from daily Ubiquinol and NADH supplements – bouts of fatigue are much less frequent, and less severe.
I am also taking NADH + Ubiquinol. At first 2 a day but i felt wired and could not rest anymore during my after- lunch resting time. Now I take one at lunch time with magnesium glycinate.
I also took antihistamines – helped a lot, but they make me gain weight so I stopped them.
what would be the critical symptom constellation difference between ME/CFS and fibromyalgia and on a side note what confounding effect would a schizoid personality disorder have on diagnosis and treatment?